Abstract
Objective
Alpha-amylase is a crucial hydrolase enzyme that breaks down complex carbohydrates, specifically hydrolyzing starch into sugars (maltose and glucose) and has wide applications in detergent, food, paper, textile, and pharmaceutical industries. This study involved the isolation, characterization and optimization of alpha-amylase-producing bacteria (APB) from Bishoftu lakes, Ethiopia.
Methodology
Isolation and characterization of bacteria from water and soil samples were conducted using conventional methods. Amylase-producing isolates were screened by starch-hydrolysis test. The best amylase producing bacterial isolates were optimized using different physiological parameters. The crude enzyme activity was further optimized against various enzymological factors, and its activity was measured under the standard enzyme assay protocols. Finally, partial purification of the amylase was carried out using ammonium sulfate precipitation, and the purity was checked using SDS-PAGE.
Results
A total of 60 APB were isolated; among them, 3 potential isolates were selected and identified as
Conclusion
α-Amylase extracted from
1. Introduction
Today, more than half of the carbohydrates consumed by humans comes from starch, which is the most prevalent dietary source. 1 Starch comprises two types of polysaccharides: amylopectin (branched), and amylose (linear). 2 Amylose is composed of α-1,4- glycosidic linkages and contains up to 6000 glucose units. It consists of alpha-D-glucose molecules linked by α -1, 4- glycosidic bonds to form covalent chains. 3 Amylopectin is made up of short linear chains with α-1,4 linkages composed of 10-60 glucose units, and side chains with α-1,6 links containing approximately 15-45 glucose units. 4
The proportions of amylose and amylopectin are influenced by the starch source; typically, 20-30% amylose is present in most common starches such as barley, corn, and potatoes. 5 To be used in soluble form, starch must be broken down into smaller molecules through processes called liquefaction and saccharification. The efficient breakdown of starch into its constituent glucose molecules requires the action of amylase. 6 Plants, animals and microorganisms are capable of synthesizing amylase in nature. 7 Among these, amylases produced by microorganisms are more advantageous due to low costs, suitability for bulk production, and the ease of manipulation to produce enzymes with desired properties. 8
Despite the global extraction of alpha-amylase-producing microbe by numerous researchers, identifying more effective enzymes for practical applications in Ethiopia remains essential. 9 Current industrial alpha-amylase suffer from thermal and pH instability, calcium independency, poor oxidant stability, and other optimization challenges.10,11 This is because most industrial sectors employ alpha-amylase under harsh conditions, including extreme temperatures and pH levels. Therefore, it is critical to identify and technologically characterize amylase-producing bacteria to overcome the challenges faced by the starch processes industry. 10 To this end, the objective of this study was to isolate, characterize, and optimize alpha-amylase-producing bacteria (APB) from soil and water samples collected from Bishoftu lakes, Ethiopia.
2. Materials and Methods
2.1. Description of the Study Area and Sample Collection
Environmental Data of the Study Area

2.2. Isolation and Screening
One gram of soil or 1 mL of water was blended with 9 mL of distilled water to create a 10-1 dilution, followed by further dilutions up to 10-7 (soil) or 10-4 (water). Aliquots of 0.1 mL were transferred onto nutrient agar plate and incubated at 37°C for 24 h. Distinct colonies from plates containing 30 - 300 colonies were aseptically transferred into nutrient broth, and pure cultures were obtained through repeated sub culturing using the streak plate technique. Each pure isolate was grown on 1% starch medium at 37 °C for 24 h. After incubation, the plates were flooded with a 1 % iodine solution; a clear zone around the colonies indicated amylase production.
12
The amylolytic index (AI) was used to screen the amylolytic isolates using the following formula:
2.3. Submerged Fermentation and Crude Enzyme Activity
To identify the most efficient amylase producers among 60 amylase-positive isolates, a modified submerged fermentation method was employed. 13 The fermentation media contained 10 g/L of starch, 10 g/L of peptone and 20 g/L yeast extract, along with essential minerals (g/L): 0.02 MgSO4, 0.01 FeSO4, 0.05 CaSO4, and 0.05 K2HPO4. Each 1000 mL flask containing 500 mL of fermentation medium was inoculated and placed in a rotary shaker at 37 °C, operating at 150 rpm, and incubated for 24 h. Following incubation, the culture broth was centrifuged for 10 min in 4000 rpm, and the supernatant was collected as crude enzyme.
2.4. Enzyme Assay
Enzyme activity was measured by the dinitrosalicylic cacid (DNS) method using maltose to generate a standard curve. The reaction mixture, containing 1 mL of crude amylase and 1 mL of 1% starch, was incubated for 15 min at 37°C. Following incubation, l mL of DNS reagent was added, and the reaction was terminated by boiling the mixture in water bath for 10 min. Absorbance was measured at 540 nm using a spectrophotometer.
14
One unit (U) of enzyme activity was defined as the amount of amylase required to liberates 1 µmol of reducing sugar (maltose equivalent) per minute under the specified assay conditions.
2.5. Characterization and Identification of Amylolytic Isolates
2.5.1. Morphological and Cultural Characterization
The morphological features such as shape, color, and texture on agar plates, were observed using a colony counter. These isolates were further described by their microscopic features under a compound microscope. The bacterial cells were stained and observed under oil immersion (100×) objective lens. 15
2.5.2. Biochemical Characterization
Biochemical tests, such as Gram staining, methyl red, Voges-Proskauer, citrate, urease and catalase tests, were performed using 24 h bacterial cultures inoculated into the appropriate media. 16
2.5.3. Molecular Identification
Molecular identification was conducted by sequencing the 16S rRNA gene. The full-length 16S rRNA gene was amplified using the primer pair 27 F: 5′-AGAGTTTGATCCTGGCTCAG-3′ and 1492 R: 5′ GGTTACCTTGTTACGACTT-3’. The PCR conditions consisted of an initial denaturation at 94 °C for 10 min, followed by 32 cycles of 94 °C for 30 s, 55°C for 20 s, and 72 °C for 55 s; with a final extension at 72 °C for 5 min in a total reaction volume of 25 μL. The amplified PCR products were sequenced, and analyzed using the BLAST algorithm against the NCBI GenBank database for identification. Sequence alignment was performed using ClustalW in MEGA 12 software. A phylogenetic tree was constructed using the neighbor-joining method with 1,000 bootstrap replications. Evolutionary distance were computed using Kimura 2-parameter model, as previously described. 17
2.6. Growth Optimization for Amylase Production
Various growth parameters for amylase production were optimized using a one factor at time approach. The optimal temperature was determined by incubating the isolate at 20, 30, 40, and 50°C; the resulting reducing sugars were measured by the DNS method. 18 To determine the effect of pH, microbes were cultured in fermentation media adjusted to pH 5, 6, 7, 8, and 9, with enzyme assays conducted after 24 h of incubation at 40°C. The impact of incubation time was evaluated at intervals of 12, 24, 48, and 72 h, 19 and the optimum period was identified by measuring the amylase activity at each interval. 20
To identify the best carbon source, 1% (w/v) of glucose, maltose, sucrose, lactose, or fructose was added to fermentation media. 21 The effects of organic nitrogen sources (urea, tryptone, beef extract) and inorganic source (ammonium chloride, and ammonia) were evaluated after 24 h incubation at 40°C and pH 7. 22
The influence of inoculum size was tested by inoculating the bacterial isolate at concentrations of 0.5, 1, 1.5 and 2 mL, followed by enzyme activity measurements after 24 h of incubation at 40°C and pH 7. 21 Additionally, the effects of metal ions (0.002% w/v) were examined by adding potassium chloride, sodium chloride, zinc sulphate, or manganese sulphate to the basal media. Finally, the concentration of starch and NaCl were optimized between 0.5% to 2%, and the activity was measured after 24 h of incubation at 40°C and pH 7. 23
2.7. Characterization of Crude Enzyme
To determine the optimal temperature for alpha-amylase activity, the enzyme was assayed in ranges of temperatures from 35 to 50°C. For pH optimization, a 1% starch medium was prepared in phosphate buffer, with the adjusted pH to values ranging from 5 to 9 using NaOH and HCl. 24 The effect of divalent and monovalent ions (Ca2+, Mg2+, Mn2+, Zn2+, Na+, and K+) were evaluated at concentration of 5 mM. 25 The effect of the reaction time was determined by incubating the enzyme with substrate for 15, 30, 45, and 60 min. 26 Finally, the thermostability of the alpha-amylase was determined by pre-incubating the enzyme at 45, 50, 60, and 65°C for 30 and 60 min. The activity of the untreated enzyme was considered 100%. 27
2.8. Purification and SDS-PAGE Analysis
2.9. Specific Activity
The protein concentration of the crude and partially purified enzymes were determined using the Bradford method. Samples were incubated at room temperature for 10 min, and optical density was measured at 595 nm. A standard curve was generated from these absorbance measurements to calculate protein concentration. Crude and partially purified enzyme samples were prepared and the absorbance was checked at 595 nm as standard. Finally, the specific activity was calculated using the following formula.
29

2.10. Application of Enzyme
The effectiveness of the extracted enzyme was evaluated through detergent preparation. Two soap variants were laboratory-formulated using sodium hydroxide, coconut oil, sodium chloride, and distilled water. The first variant served as a control (without enzyme), while the second was supplemented with 3 mL of crude α-amylase. Unform pieces of white cloth, pre-contaminated with potato waste, were treated with 20g of either the enzyme-free soap or the enzyme-enriched soap. Following treatment, the cloths were rinsed with distilled water, and their cleanliness was compared against a clean control cloth. 30 This experiment was conducted in duplicates.
2.11. Statistical Analysis
Origin Lab 2024 software was employed for graphic development. To ensure reliability, all analyses were conducted in duplicate. The data are presented as mean ± standard deviation.
3. Results
3.1. Microbial Isolation and Selection for Amylase Production
The results showed that 60 isolates tested positive for amylase activity (data not shown). Clear zone forming around colonies on starch media indicated starch degradation (Supplementary Figure 1). These 60 isolates were subjected to submerged fermentation, after which, enzyme activity was assayed. The three best performing isolates, B1W1, G2W1, and G3W1, that exhibited activities of 1.28 ± 0.05, 1.96 ± 0.04, and 1.40 ± 0.02 U/mL, respectively, were selected for further analysis (Supplementary Table 1).
3.2. Conventional and Molecular Identification
Morphologically, B1W1 produced transparent colored colony, G3W1 produced white colonies, and G2W1 produced yellowish colonies. The B1W1 isolate was dry in texture, while G2W1 and G3W1 were rounded and mucoid, respectively. Microscopically, all the three isolates were rod-shaped. B1W1 was Gram-positive, while G2W1 and G3W1 were Gram-negative.
Regarding motility, G3W1was non-motile, whereas B1W1 and G2W1 were motile. Based on 16S rRNA gene sequence analysis, the three isolates were identified as Phylogenetic tree constructed based on 16S rRNA gene sequencing
3.3. Optimization of Amylase Production by One Factor at a Time (OFAT) Approach
The optimum temperature for all three isolates was 40°C, with The effect of temperature (A), pH (B), and incubation time (C) on the strains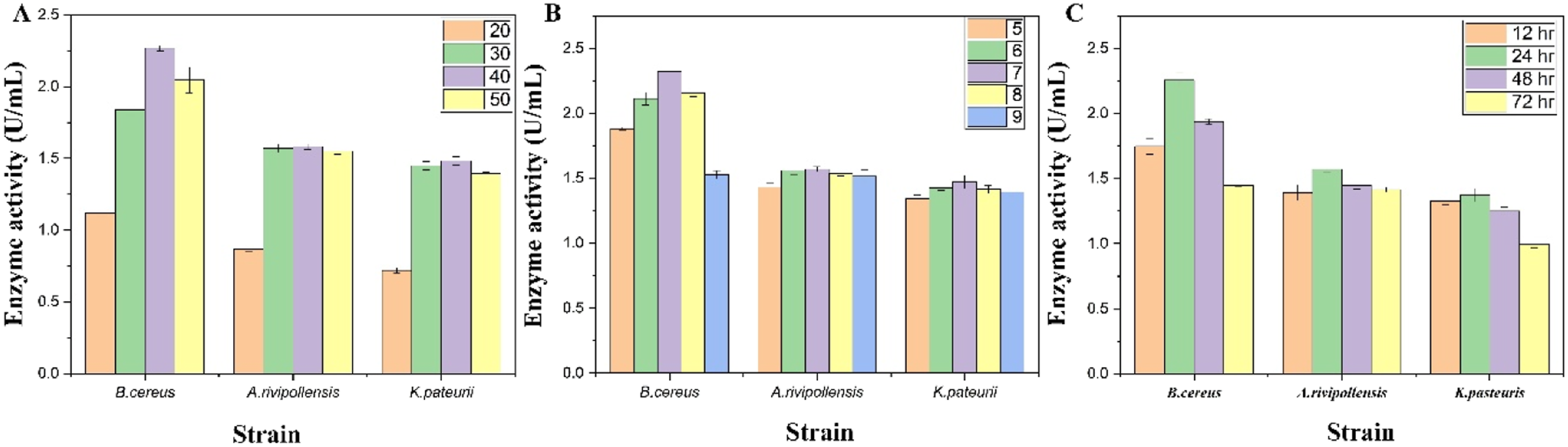
All the three strains produced high levels of amylase activity when glucose was used as carbon source. The maximum activity was recorded for Carbon source (A), nitrogen source (B), metal ions (C), inoculum size (D), starch concentration (E), NaCl concentration (F) on the strains
Regarding metal ions,
At 1.5 mL of inoculum concentration, the highest amylase activity (3.3 U/mL) was observed for both
3.4. Characterization of Crude Enzyme
The alpha-amylase extracted from the three strains showed high enzymes activity at 45°C, which is slightly higher than the strains’ optimum temperature for the enzyme production (Figure 4A). The optimum pH for alpha-amylase was pH 7, matching the optimum pH for the strains (Figure 4B). The enzyme exhibited high activity in the presence of Ca2+, Mg2+, Mn2+, Na+, K+, and Zn2+, respectively. Among these, Ca2+ was the most effective ion for enhancing enzyme activity across all strains (Figure 4C). Regarding reaction time, amylase activity decreased as the time increased, with the optimal reaction time identified as 15 min (Figure 4D). Enzymological characterization of the crude enzymes extracted from three bacterial strains. (A) The effect of temperature, (b) the effect of pH, (C) the effect of metal ions, (D) the effect of reaction time
In terms of thermal stability, the residual activity of alpha-amylase decreased as temperature and incubation time increased. At 45°C, the residual activity remained above 90% after 30 min and ≥ 86% after 60 min (Figure 5A). At 55°C, activity remined ≥ 80% for 30 min but dropped below 50% after 60 min (Figure 5B). At 65°C, the enzyme retained over 50% of its activity when treated for 30 min; however, after that point, activity fell to < 40% of the original activity (Figure 5C). These findings suggest that while the enzymes possess good thermal stability at moderate temperatures, its stability is significantly compromised at higher temperatures. Thermal stability of treated enzyme at different temperature and time. (A) Thermal stability of the treated enzyme at 45°C for 30 and 60 min, (B) Thermal stability of the treated enzyme for 55°C for 30 and 60 min, (C) Thermal stability of the treated enzyme for 65°C for 30 and 60 min
3.5. Partial Purification and Specific Activity
The
Crude and Partial Purified Specific Activity of Alpha Amylase Enzyme

3.6. Biotechnological Application of Enzyme
The effectiveness of the extracted alpha amylase was checked for detergent application. The results showed that soap containing crude alpha-amylase showed better potato waste removal capability than soap without enzyme. After washing, the piece of cloth treated with amylase-containing soap has removed the potato waste roughly up to 50% (Supplementary Figure 3), indicating successful degradation of the starch in the potato waste.
4. Discussion
The ability of
We employed the one factor at a time approach to determine the optimal conditions for alpha-amylase production, allowing the systematic examination of several parameters. Previous research demonstrated that different carbon sources exert varying effects on amylase production; for instance, Vijayabaskar et al, 21 found that glucose increases yield. In contrast, sucrose was identified as the optimal carbon source in the present study. These findings show that differences in production potential and optimal conditions among these isolates contribute to the understanding of bacterial amylase and its suitability for industrial applications. Such variations highlight the metabolic diversity among the bacterial isolates and their specific requirements for alpha-amylase production.
Ammonium chloride and beef extract were the best inorganic and organic nitrogen supplement for alpha-amylase production from
Salinity is the key factor impacting the growth and enzyme production of these microorganisms, as it maintains osmotic balance with their environment. Our study found that a NaCl concentration of 1.5% (w/v) was optimal for maximal amylase production across all the isolates. Beyond this concentration, enzyme activity gradually decreased. This reduction is attributed to the hypertonic condition, where a saltier environment leads to plasmolysis, which subsequently inhibits both growth and enzyme production. 41 These results demonstrated that maintaining an optimal saline environment is essential for cell integrity and maximizing enzyme yield.
We observed that increasing the starch concentration enriched the growth of isolates, reaching an optimal level up to 1.5%. This indicates that 1.5% starch provides sufficient substrate for optimal amylase production by the identified strains under our assay conditions. However, increasing the starch concentration beyond this optimum led to a decrease in enzyme production. Elevated starch concentrations increased the viscosity of culture broth, which hindered O2 transport and limited the dissolved O2 required for microbial growth. 42 In addition to production levels, the properties of enzymes were evaluated through enzymological characterization. The alpha-amylase produced by the three species function optimally at 45°C and pH 7, making them suitable for industrial application that operate under these specific conditions.
Three of our isolates showed higher enzymatic activity at moderate temperatures (40-50°C) than in the lower temperature like 35 or below (Figure 4A). This is attributed to the strain’s ability to evolve specific structural adaptations that provide increased thermal stability, preventing them from denaturing in hotter environments. In contrast, at lower temperatures, these enzymes exhibit reduced activity due to lower molecular kinetic energy and increased structural rigidity, which results in fewer successful enzyme-substrate collisions. 43 Furthermore, amylase activity was dependent on the availability of calcium and other divalent cations. 44 At higher temperatures, calcium ion participate in achieving the correct configuration of the enzyme’s thermostable form, function for which they are irreplaceable; however, at lower temperatures, calcium acts as an non-specific cofactor. 44
The strain
Partial purification of the enzyme from
Relatively, our purification procedure confirms the presence of protein in the sample and the band is aligned with alpha-amylase. 47 Biotechnological application was tested by formulating a soap using the crude enzyme. The enzyme-enriched soap successfully broke down the starch on the cloth contaminated with potato waste. Although further investigations are required to elucidate the enzyme zymogram or N-terminal sequence to validate the SDS-PAGE band as α-amylase and the applications in the wider areas, the current experimental finding supports the cleaning efficiency of this identified amylase and demonstrates its potential for use in laundry detergents to remove starch stains.
While this study followed the one factor at time approach to characterize the alpha-amylase extracted from the three strains, future studies should utilize Response Surface Methodology (RMS) to analyze the interactions of several factors simultaneously. The authors also recommended further purification of the enzyme via high-throughput purification techniques and wider sample collection across different seasons for future research to fully capture the potential of the study area.
5. Conclusion
The current research indicates the possibility of isolating amylase-producing bacteria from the soil and water of the Bishoftu lakes. The isolate produced an efficient yield of amylase during submerged fermentation. The study also confirmed that
Supplemental Material
Supplemental Material - The α-Amylase Producing Bacteria From Bishoftu Lakes: Extraction, Purification and Application
Supplemental Material for The α-Amylase Producing Bacteria From Bishoftu Lakes: Extraction, Purification and Application by Misgana Anbessa, Asnake Desalegn, Feng-Yan Bai, and Fitsum Tigu in Natural Product Communications.
Footnotes
Acknowledgments
We thank the Department of Microbial Sciences and Genetics, Addis Ababa University, and The Chinese Academy of Science, President’s International Fellowship Initiative (PIFI) (No. 2024PVB0012) for their technical and financial supports.
Ethical Considerations
IRB is not applicable since human test subject were not involved in the study.
Author Contributions
Conceptualization: FT, AD; Data curation: MA; Formal Analysis: MA, FT; Investigation: MA, FT, AD; Methodology: MA, FT, AD; Resources: FT, AD, FYB; Supervision: FT, AD; Writing-original draft: MA, FT; Writing-review & editing: FT, AD, FYB.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Addis Ababa University 10th round Thematic Research Fund (Grant no. RD/LT-308/2022).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
