Abstract
Introduction
Since its earliest days, nanoscience has demonstrated its value to humanity in several fields, including agriculture, wastewater treatment, cosmetics, drug delivery, etc 1 Nano sizes (1 nm equals 1 × 10−9 m), thermal conductivity, and large surface-to-volume ratio are only a few of the distinctive characteristics of nanoparticles (NPs).2,3 These NPs were created using chemical, biological, and physical techniques. Even though physical and chemical processes may yield nanoparticles with specific sizes, many drawbacks have emerged, including the use of toxic materials, high heat and pressure requirements, unavailability, and high cost. As a result, the focus has shifted to more accessible and safe techniques.2,4,5
Bio-synthesized NPs have an advantage over those produced chemically or physically since they are very stable, affordable, and readily available, with no toxic by-products.4,5 Compounds produced naturally from plants or microbes are used in the biogenic reduction of the metal to nanoscale particles. The primary source of medicinal metabolites is plants. A remarkable ability to reduce metal ions to NPs and coat the produced nanomaterials to enhance their stability has been demonstrated by plant metabolites, such as proteins, flavonoids, alkaloids, carbs, and polysaccharides.1,5 Plenty plant-derived parts, extracts, metabolites, and other derivatives were successfully and effectually applied for biogenic syntheses of diverse metals’ nanoforms, with supported and augmented bioactivities as anticancer, antimicrobial, anti-inflammatory, antidiabetic, and immune stimulant biomaterials.6–10 The employment of plant-derived molecules for biogenic synthesis of nanometals provided outstanding advantages, eg bioactivity enhancement, biosafety warranting, facile synthesis and homogenous products, compared to physic-chemical approaches.8–10 A widely used material for metal NPs production is “Plant-originated mucilage”. Mucilage is a naturally occurring polysaccharide produced by most plants from the seed coat. It consists primarily of highly branched carbohydrates, composed of D-galactose, D-xylose, L-arabinose, L-rhamnose, and galacturonic acid monomer units. 11
Purslane “Portulaceae oleracea L.” is a widely distributed herbaceous plant from the Portulaceae family. Purslane is a promising pharmacological agent since the WHO “World Health Organization” lists it as one of the most used therapeutic plants.12,13 Purslane vegetable nutritional importance based on its richness in (omega-3) fatty acids, diverse vitamins types (eg Vitamin A, B complex, C …), beneficial minerals (eg calcium, magnesium, iron, and potassium) and carotenoids.8,9 Furthermore, the purslane extracts exhibited potential bioactivities including antimutagenic and antioxidant actions due to their (eg the alkaloid betalain pigments) that are key phytoconstituents detected in the purslane extracts.14,15 Purslane's polysaccharides have been shown to possess strong antibacterial, wound-healing, antifungal, and anti-inflammatory activities in both in vitro and in vivo investigations. These polysaccharides have been utilized in various applications since studies have shown them to be safe and non-toxic.9,13–15
Numerous metal and metal-oxide NPs, such as Au, Ag, CuO, ZnO, and MgO, have been synthesized since the emergence of nanoscience. Among these metals, silver's antimicrobial properties have long been recognized, as it has a strong capacity to suppress infections.2,16 Additionally, as silver is transformed into its nano form (AgNPs), its effectiveness in eliminating pathogenic microbes increases due to the increase in surface area to volume, increasing the reactive sites. 4 All studies conducted on AgNPs have demonstrated an ideal effect as an antibacterial (against both positive and negative Gram bacteria, such as P. aeruginosa, E. coli, and S. aureus), antifungal (against S. cerevisiae, A. fumigatus, and C. tropicalis), and antiviral (hepatitis B and herpes simplex), besides their anti-inflammatory qualities.8,17,18
Furthermore, given the severity of this issue, the ability of AgNPs to combat antibiotic-resistant microbes has attracted the attention of many researchers. AgNPs exhibit biocidal activity through the gradual release of Ag+ and mechanisms that would undoubtedly result in cell destruction, including protein and enzyme interaction, restriction of protein synthesis, suppression of DNA replication, and oxidative stress generation. These mechanisms make it more challenging for microbes to develop resistant strains.4,17
A positively charged carrier is essential for delivering antimicrobial NPs to the microbe since negative-charged metal NPs repel negative-charged proteins and lipids that make up the microbial cell's outer layer. For this task, nothing works better than chitosan.19,20 Deacetylation of crustaceans chitin produces chitosan, a naturally occurring polymer. Apart from its positive charge, chitosan also has antifungal and antibacterial properties, in addition to its distinctive features which include bio-activity, eco-friendly, zero toxicity, bio-compatibility, and bio-degradability. The nano form of chitosan (NCt) has the same unique properties as bulk chitosan, in addition to its larger surface area and greater efficiency against microbes.19,21
Chitosan-metal nanocomposites have been extensively employed in numerous medicinal applications since chitosan has been shown to boost the antibacterial, antifungal, anti-inflammatory, antiviral, anticarcinogenic, and antioxidant effects of metal NPs. 22 The effectiveness of these nanocomposites in eliminating microbes was greater than that of either metal NPs or NCt used alone. A broad spectrum of activity was demonstrated by NCt loaded with gold, silver, or copper against both positive and negative Gram bacteria. 15 The most studied metal/chitosan complexes are silver-based nanocomposites, which were discovered to give the nano-silver more efficacy against microorganisms and less toxicity against biological tissues.20,23
Human candidiasis is a fungal infection caused by the yeast species Candida. In humans, Candida can naturally occur on the skin or in the mouth, vagina, or rectum with no obvious harmful effects. Candida has the potential to turn pathogenic under certain conditions, leading to dangerous infections which can be responsible for over 70% of global fatalities. 24 Candida is an “opportunistic organism” that multiplies rapidly when the immune defenses are weakened or when antibiotics are used excessively, eliminating beneficial microbes and allowing Candida to multiply rapidly. 25 Candida's risk is found in its ability to cause health problems in the infected areas only or its ability to spread throughout the bloodstream and cause more adverse consequences when it reaches vital organs like the heart and kidneys and even death. 26
In response to the emergence of Candida resistant to antibiotics, the lack of effectiveness of antifungals, and their negative side effects, this study was carried out to test a combination of green-synthesized AgNPs using purslane with NCt particles and determine their effectiveness as a natural and safe antibiotic against Candida albicans.
Experimental Materials and Methodology
Reagents
Every chemical, media, and reagent used, unless otherwise noted, was acquired from “Sigma-Aldrich Co.” (United States). Seeds of Portulaca oleracea (purslane) were purchased from the ARC “Agricultural Research Centre” located in Giza, Egypt. MIRCEN “Microbiological Resources Centre” at Ain Shams University in Egypt provided the tested strains of C. albicans.
Extraction of Purslane Seeds Mucilage (PSM)
De-ionized water (DIW) was used to thoroughly cleanse purslane seeds to get rid of any undesirable contaminants. The seeds were air-dried, and then finely ground, twice as much DIW was added, and they were stirred continuously at the ambient temperature. The purslane seeds aqueous extract was then properly filtered through a Whatman no. 1 filter disc and any leftover material was disposed of. The extract was mixed with a comparable volume of absolute ethyl alcohol, which was then kept at 4 °C for a while. The bottom-formed mucilage is next separated and dried in a vacuum drying furnace at 40 °C.
Plant-mediated Silver Nanoparticles (AgNPs) Synthesis
To formulate AgNPs using PSM, 1.7 g of silver nitrate was dissolved in 100 ml of DIW to make an aqueous solution of silver nitrate (AgNO3). Equal quantities of silver nitrate and mucilage solutions (1% w/v) were amalgamated under shaking conditions until the color turned from light brown to burnt brown and finally black, which validated the bio-reduction of AgNO3 to Ag-NPs. Following their collection via centrifugation at 10 000 rpm for five minutes, the nanoparticles underwent multiple rinsing with ethanol and DIW before being dried.
Construction of PSM-AgNPs/NCt Nanocomposite
In the diluted form of acetic acid, 0.1 g of chitosan powder was dissolved and agitated until an impurity-free chitosan solution was formed. Drop by drop, a 2:1 mixture of PSM-AgNPs: chitosan was progressively mixed to incorporate PSM-AgNPs with chitosan particles.
Nanocomposites Characterization
Various methods were employed to characterize nanoparticle composites to validate their size, shape, morphology, superficial charges, and the alterations of bio-chemical bonds brought about by the molecular combination and interaction.
Using an FTIR spectroscopy “Thermo Fisher, Nicolete IS10, Waltham, MA”, samples of NCt, PSM, PSM-AgNPs, and PSM-AgNPs/NCt were mixed individually with 1% Potassium Bromide which serves as a carrier with 100% transmittance, and were examined to identify the unique biochemical interactions resulting by molecules combining. 4000 cm−1 to 500 cm−1 wavenumber range was obtained. The resulting peaks were plotted on a Y-axis representing transmittance (%) and an X-axis representing wavenumber (cm−1).
AgNPs production was verified via “UV–visible spectroscopy”, which identified the SPR “surface plasmon resonance” of particles. Using a spectrophotometer “model UV-2450, Shimadzu, Japan” the AgNPs’ spectra in the 250-800 nm wavelength range were examined.
Transmission and Scanning electron microscope imaging were taken to assess the particle size, morphology apparent shape, and arrangement of the produced nanoparticles using “SEM; IT100, JEOL, Tokyo, Japan” and “TEM; Leica-Leo 0430; Cambridge, UK”. Different concentrations of NCt: PSM/AgNPs were prepared, (2:1), (1:1), and (1:2), to measure their size as well.
After dissolving in DIW and sonicating the generated nanomaterials, their size and zeta potentiality (ζ) were recorded between +150 and −150 mV at 30 °C using a zetasizer “Malvern, Worcestershire, UK”.
Evaluation of Anticandidal Activity
Tested Strains
To evaluate the anticandidal effect of PSM-AgNPs/NCt, a standard strain (A) Candida albicans ATCC 10231 and (B) isolated and identified strain (EMCC −105) from a human source were employed. The strains were cultivated and kept alive at 37 °C on SDA “Sabouraud Dextrose Agar”. The collected cell mass was diluted in 0.85% NaCl solution to prepare the inoculums for further testing. The cell concentration was set to 1 × 104 cells per milliliter using a hemocytometer. The isolated strain, which was obtained from Kafrelsheikh University Hospital, was characterized using molecular biology techniques.
Qualitative and Quantitative Evaluation
The effect of PSM-AgNPs/NCt on Candida was evaluated qualitatively through “agar diffusion examination”. On 20 ml SDA plates, 100 µL of Candida were planted using a sterile cotton swab. After that, 0.6 cm spherical sterile discs containing 20 µL of the item to be tested were loaded onto the Candida dishes, and incubated aerobically for 12-24 h at 37 °C. NCT, PSM, PSM/AgNPs, NCt: PSM/AgNPs (2:1), (1:1), and (1:2) were tested separately on both strains A and B in comparison with a standard anticandidal agent (Fluconazole). Three iterations of the experiment were carried out. The ZOI “Zone of inhibition” diameters were measured, and the mean was calculated.24,25
Using MFC “Minimum fungicidal concentration”, the nanomaterials were quantitatively examined to identify the lowest concentration that eradicated Candida. Substances NCT, PSM, PSM/AgNPs, NCt: PSM/AgNPs (2:1), (1:1), and (1:2) were gradually diluted to achieve concentrations of 5 −100 μg/mL. One milliliter of each concentration of the substances to be examined was then added to broth media tubes. Subsequently, Candida was injected into the test tubes and incubated at 37 °C for 24 h. Afterward, the MFC required to prevent Candidal growth was identified. Under the same conditions, the Fluconazole's MFC was also examined.24,25
Morphological Alterations
Scanning microscope “SEM; IT100, JEOL, Tokyo, Japan” was utilized to assess PSM-AgNPs/NCt's effect on Candida's morphology. Samples were prepared using the “Marrie and Costerton” protocol. Candida incubated for 24 h, were exposed to PSM-AgNPs/NCt, and incubated again for 0, 6, and 12 h. After being centrifuged for 10 min at 6000 rpm, cells were rinsed with PBS “phosphate-buffered saline; pH 7.4” and fixed with 1% osmium tetroxide (OsO4) and 2% glutaraldehyde. Once the samples were fixed, they were washed in PBS and then dehydrated in an increasing sequence of ethanol concentration. Following drying, the candidal cells were covered with a thin coat of gold. The samples were then examined using SEM.
Statistical Analysis
The SPSS software “V 17.0, SPSS Inc., Chicago, IL” is employed in statistical computing. Three duplicates of each experiment were conducted, and the results were presented as means SD. Using “one-way ANOVA” the results’ significance at P < .05 was determined.
Results
Nanocomposites Characterization
FTIR analysis made a precise assessment of changes in biochemical groups and bonds. NCt was analyzed first, then PSM, PSM-AgNPs, and finally PSM-AgNPs/NCt, as shown in [Figure 1]. The most notable peaks of the NCt spectrum were at: 3415 cm−1 and 2931 cm−1 (O−H and aliphatic C−H stretching vibration), 2881 cm−1 (C–H stretching), 1948cm−1, 1699 cm−1 (C = O stretching of amide I), 1616 cm−1 (N–H bend), 1500 cm−1 (C = O stretching),26–29 1415 cm−1 (–CH2 vibrations), 1315 cm−1, 1149 cm−1 (CN (NH2) stretching vibration), 1033 cm−1 (C6−OH stretching vibration), and 649 cm−1 (−OH vibrations).30–34
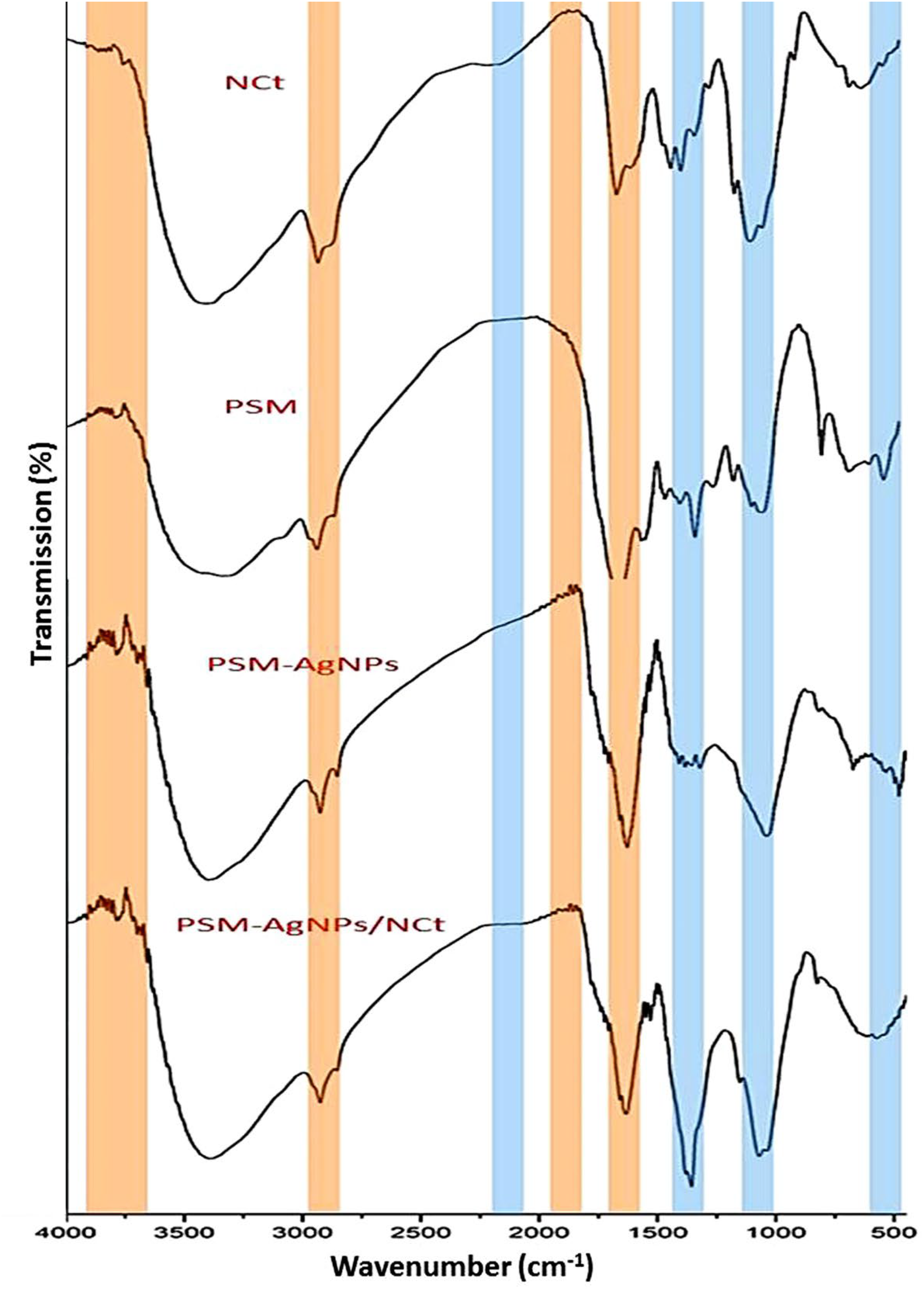
FTIR spectra of NCt, PSM, PSM-AgNPs, and PSM-AgNPs/NCt. The PSM-AgNPs/NCt and NCt shared similarities shown by blue lines, whereas the PSM-AgNPs/NCt and PSM-AgNPs shared similarities represented by red lines.
In the PSM spectrum, peaks at 3415 cm−1 (−OH vibrations), 2931 cm−1, 1632 cm−1, 1533 cm−1 (C = C stretching), 1448 cm−1 (C-H Bending), 1033 cm−1 (C-O stretching vibrations), and 566 cm−1 (C-H Bending) were found.35–40 As demonstrated in earlier research, PSM exhibits significant promise in linking, reducing, and stabilizing AgNPs.1,3,41,42 The 1632 cm−1 peak in the PSM spectrum is linked to overlapping the polysaccharide's stretching N–H vibration group and the C = O group. 38 The methyl group's C–H vibration stretching was identified as the cause of the peaks at 2931 cm−1 in the same spectrum. 37
In the PSM-AgNPs spectrum, the organic molecules function as stabilizing/capping agents during the production of AgNPs are indicated by the peak at 2931 cm−1. Moreover, bands detected at 1649 and 1632 cm−1 result from the amide-II bonds’ N–H, which connect the protein's amino acids.
The AgNPs biogenic synthesis was proved via visual assessments [Figure 2]. The maximal SPR of the generated color was determined by absorbance measuring in the 250-800 nm range. The result in [Figure 2B] showed a strong absorption peak at 422 nm indicating

A: TEM image of the biogenic AgNPs using PSM. B: UV-Vis spectrum of PSM/AgNPs.
The PSM-AgNPs/NCt exterior shape, size, and molecular arrangement were ascertained by TEM [Figure 2A] and SEM [Figure 3]. Figure 2A displayed uniform particle dispersion and spherical PSM-AgNPs morphologies with comparable diameters. The concentration (2:1) of NCt: PSM/AgNPs [Figure 3A] revealed particles with sizes ranging from 56-422 nm. The concentration (1:1) [Figure 3B] includes particles ranging from 44 to 360 nm, while the greatest particle sizes have been observed in NCt: PSM/AgNPs (1:2) [Figure 3C]. The particles’ morphologies were homogeneous, showing little aggregates.

SEM micrographs of the different concentrations of NCt: PSM/AgNPs: A-(2:1), B-(1:1), and C-(1:2).
Zeta potential (ζ) analysis was utilized to assess the particle size and exterior electrical charge of nanocomposites to validate the SEM image results. The diameter range and mean diameter were ascertained. [Table 1] displays the findings. The mean diameters of NCt and PSM/AgNPs were 232.1 and 11.9. NCt: PSM/AgNPs (1:1) was the concentration with the smallest mean diameter out of all the others.
Particles’ Size and zeta Potential Analysis of Screened Materials/Composites.

*ND, not determined.
The NCt, PSM, and PSM/AgNPs charges were measured. According to the data in [Table 1], NCt's ζ potential was significantly positive (+34.65), whereas PSM and PSM/AgNPs’ ζ potentials were −23.52 and −26.12, respectively.
Evaluation of Anticandidal Activity
Qualitative and Quantitative Evaluation
PSM-AgNPs/NCt was evaluated to assess its effect on the selected strains and the minimal dose necessary to eradicate Candida. The effect was compared with Fluconazole, the standard antibiotic against Candida. All tested substances demonstrated a good anti-candidal effect. The best values of ZOI and MFC were determined based on [Table 2] results. According to the data displayed, it was observed that the broadest zone free of Candida (both A and B) was at the concentration (1:1), followed by concentration (2:1) and then (1:2). NCT and PSM were less effective against strains A and B than PSM/AgNPs. The best MFC value was 5.0 mg/L for NCt: PSM/AgNPs (1:1) against Candida A, and 4.5 mg/L against Candida B for the same concentration. Other concentrations required larger amounts to eliminate candida. In both analyses ZOI and MFC, the NCt: PSM/AgNPs (1:1) values against Candida A and B were generally better than that of the standard antibiotic Fluconazole.
Anticandidal Activity* of Synthesized Agents Against Candida A and B.
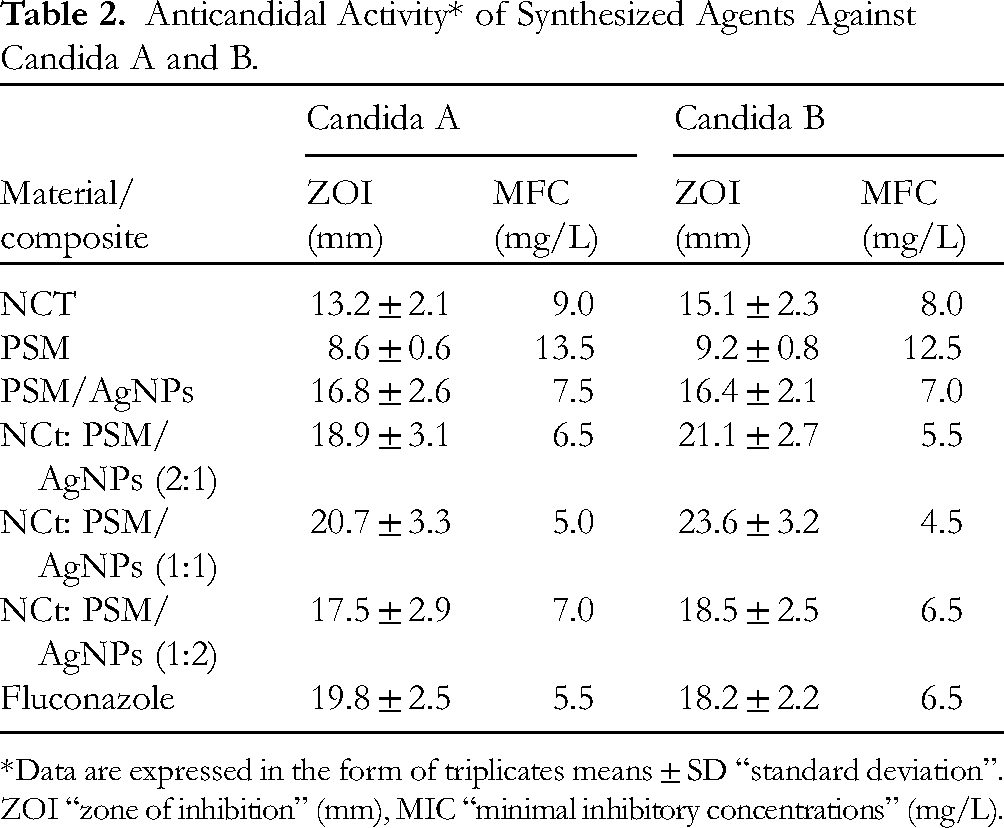
*Data are expressed in the form of triplicates means ± SD “standard deviation”. ZOI “zone of inhibition” (mm), MIC “minimal inhibitory concentrations” (mg/L).
Morphological Alterations
The morphological mutations of Candida treated with PSM-AgNPs/NCt were observed using SEM after exposure for 0, 6, and 12 h. The typical spherical form of Candida is illustrated in [Figure 4A], along with a few tiny spheres on its surface reflecting the nanomaterials that were applied for 0 h (shortly after exposure). The compound's attachment to the negatively charged Candida cells demonstrates the influence of PSM-AgNPs/NCt's positive charge, as this is the initial stage in the destruction process. [Figure 4B] illustrates the second stage in the destruction process.

SEM images of treated Candida after exposure for 0 h, 6 h, and 12 h.
The rough, uneven surface of the Candida indicates the start of cell wall destruction, as nanoparticles adhere to the wall and begin to disintegrate it to reach the internal contents of the cell. [Figure 4C] represents the final stage of destruction “cell death”.
Discussion
FTIR is a basic analysis for investigating chemical changes in the composition of compounds, as it works to identify each component's functional groups and determine the change in them after the compound reacts with another.22,23 As chitosan “poly β-(1,4)-2-amino-2-deoxy-d-glucose” is chitin in its deacetylated form with potent antimicrobial properties, the main detected bonds in its FTIR spectrum reflected these functional groups.16,24 NCt possesses the primary characteristics of bulk chitosan.30,31 The broad band at 3415 cm−1 was associated with the O–H and N–H strained intramolecular bonding of hydrogen, while the bands at 2931 and 2881 cm−1 are common polysaccharide bands. 17 The C-N stretching vibration visible at 1315 cm−1 revealed the existence of an acetyl group. 28 In the PSM-AgNPs spectrum, the organic molecules function as stabilizing/capping agents during the production of AgNPs are indicated by the peak at 2931 cm−1. Moreover, bands detected at 1649 and 1632 cm−1 result from the amide-II bonds’ N–H, which connect the protein's amino acids.30–33
Compared with AgNPs synthesized using other biological materials, those biosynthesized from plant extracts showed more activity. This is because the surface of the synthesized AgNPs was coated with plant metabolites, which prevented them from aggregating and increased their stability.1,33 Numerous compositional alterations were noted following the PSM's reaction with Ag to generate the nanoparticles. When PSM and PSM-AgNPs spectrums are compared, new peaks, such as 3948 cm−1, 3881 cm−1, 3732 cm−1, 3566 cm−1, 1782cm−1, 1749cm−1, 1715cm−1, and 815 cm−1 have emerged indicating the formation of new chemical bonds between the Ag and PSM. A few peaks were relocated, peaks at 1398 cm−1, 666 cm−1, and 516 cm−1 in the PSM spectrum were shifted to the left to be located at 1415 cm−1, 657cm−1, and 549 cm−1 respectively in PSM-AgNPs spectrum. Some peaks, such as 3049 cm−1, 1481 cm−1, 1249 cm−1, 1166 cm−1, 1083 cm−1, and 782 cm−1 vanished. These changes are conclusive evidence of the reduction of the AgNO3 with PSM and the creation of desired nanoparticles.1,18,33
Findings revealed that the PSM-AgNPs/NCt spectrum shared functional groups with the NCt and PSM-AgNPs spectrums. The similarities between the PSM-AgNPs/NCt and NCt were shown with blue lines, while the similarities between the PSM-AgNPs/NCt and PSM-AgNPs were represented with red lines in [Figure 1]. These similarities demonstrated how NCt and PSM-AgNPs were combined to create the final compound PSM-AgNPs/NCt. Numerous investigations reported matching findings in further nanocomposites using NCt as the key component for synthsesis.34- 38
A visible sign that the silver has been reduced to AgNPs by PSM is the color shift to burnt brown and then black.1,2,39 AgNPs formation was further verified via UV analysis as well. The λmax findings of biogenic AgNPs at 422 nm support earlier studies that found that AgNPs have a localized SPR in the 400-460 nm region, indicating the success of synthesizing homogeneous-sized nanoparticles using PSM.3,40,41 The AgNPs biogenic synthesis with plant extracts was verified in numerous former studies,7–10 from the suggested biosynthesis pathways are involving the reduction of Ag ions from the ionic atom (Ag+ or Ag2+) into the ground state atom (Ag0), using the reducing powers of active plant molecules (eg polyphenols, enzymes, proteins, essential oils, steroids, … etc).6–10,33,41 These phytochemicals frequently implement the aqueous metals’ ions bio-reduction to generate the relevant zerovalent metals, which could be claimed in AgNPs reduction here using PSM biomolecules. However, more specifications of whether biogenic PSM/AgNPs are in ground (zerovalent, 0) state atom or ionic (Ag+ or Ag2+) atoms are suggested to stipulate the role of AgNPs oxidative state in their anticandidal action.6–10,33,41 One of the key elements influencing a nanoparticle's ability to be effective as an antimicrobial is its size. Silver's antimicrobial properties are enhanced when reduced to nanoparticle form because microorganisms are exposed to more of the particle's surface area. 4 The DLS and electron microscopy findings align well with the previously discussed size analyses results; PSM/AgNPs with a diameter range of 1.3-24.6 nm are considered smaller than the nanoparticle sizes of 20-25 nm, 344-446 nm, 117.4 nm, and 5-40 nm, synthesized in earlier studies.36,37,39 The stability of nanoparticles is another key element influencing a nanoparticle's efficacy as an antimicrobial. ζ potential value is used to determine the stability of the particles, NPs with ζ values larger than +30 mV or less than −30 mV exhibit very high stability, and ±20-30 mV is regarded as having medium to high stability.33,42
This negative charges of screened materials could be attributed to capping agents, including the flavonoid and polyphenolic compounds found in plant extract.1,33 The charge of various NCt: PSM/AgNPs concentrations was also determined. Since NCt has a high positivity, the charge in the PSM-AgNPs/NCt compound increased as the amount of NCt increased. ζ potential of the NCt: PSM/AgNPs (2:1), (1:1), and (1:2) were +30.63, +24.13, and −11.72, in that order, which were formerly reported in earlier studies investigated NCt-based nanocomposites.43-47
The superiority of NCt: PSM/AgNPs (1:1) as an anti-Candidal over other substances is due to the small size of its molecules and its positive surface charge, as the positive charge of chitosan attracts the NPs to Candida cells and adheres to their surface, then the small molecules penetrate the cell surface to start attacking the cell's vital systems. Following cell penetration, AgNPs can break down the sterol found in Candida's cell walls by preventing the formation of the ergosterol pathway, which causes the cells to die. The primary sterol found in Candida is ergosterol, which is produced in the endoplasmic reticulum and is in charge of the plasma membrane's rigidity, integrity, homogeneity, and fluidity.2–5
Conclusion
This study used purslane plants to successfully green-synthesize silver nanoparticles, and FTIR and UV-visible spectroscopic techniques were employed to validate the synthesis. Besides its antibacterial qualities, NCt was added to serve as a positively charged carrier for the molecules. FTIR was used to verify the compounds’ integration, while SEM, TEM, and zetasizer were used to characterize the compounds’ sizes, distribution, and external charge. PSM/AgNPs have a diameter range of 1.3-24.6 nm, making them ideal for usage as antibiotics. The compound's efficacy against Candida was subsequently demonstrated using qualitative and quantitative techniques (ZOI and MFC). The concentration (1:1) NCt: PSM/AgNPs was the most suitable choice. SEM microscope was used to assess the compound's effect on Candida visually. PSM-AgNPs/NCt has demonstrated success in eliminating Candida and outperforms the currently used anticandidal Fluconazole. The main suggested limitations to be prospectively addressed are the examination of nanocomposites biosafety attributes using animal models, the elucidation of exact anticandidal actions of nanocomposites using molecular approaches and the optimization of biosynthesis process using factorial design programs.
Footnotes
Acknowledgements
The authors declare their appreciation and foremost gratefulness to ALLAH for the merciful and generous guiding throughout this work.
Authors’ Contributions
AAT contributed in the conception, investigation, interpretation of data, supervision, work drafting and submission; AASA contributed in the conception, design of the work, investigation and analysis and work drafting; MMZ contributed in the conception, interpretation of data, work drafting, and revising; MOIG contributed in the design of the work, investigation and analysis, interpretation of data, and work drafting; MAA contributed in the conception, interpretation of data, work drafting, revising and supervision; AEO contributed in interpretation of data, resources, supervision and work drafting. All authors read and approved the final manuscript.
Data Availability
“All data generated or analyzed during this study are included in this published article”.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
“Ethical Approval is not applicable for this article.”
Statement of Human and Animal Rights
“This article does not contain any studies with human or animal subjects”
Statement of Informed Consent
“There are no human subjects in this article and informed consent is not applicable.”
