Abstract
Objectives
Chronic heart failure (CHF) is a prevalent clinical syndrome with high mortality rate. This study evaluates the therapeutic effects of total secondary ginsenosides (TSG) on CHF in rat and dog models compared to Xinbao pills.
Methods
CHF models were established via left anterior descending coronary artery ligation in rats and via inferior vena cava constriction in dogs. Following the administration of TSG at 50-200 mg/kg/d for rats and 15-60 mg/kg/d for dogs, cardiac function, left ventricular configuration, hemodynamics, urinary excretion, and pulmonary edema were assessed, together with the echocardiography, hemodynamic parameter measurements, urinary excretion analysis, cardiac hypertrophy analysis, cardiomyocyte apoptosis, vasoconstrictor factor levels, and lung water content.
Results
TSG treatment at low, medium and high dosage all significantly enhanced the myocardial contractility of CHF rats and dogs with improved the left ventricular ejection fraction (LVEF), fractional shortening (FS), and the maximum rate of left ventricular pressure (±LVdp/dtmax). In comparison to the pre-treatment, the cardiac load was significantly reduced following TSG treatment as the left ventricular end-diastolic pressure (LVEDP) was decreased. In addition, cardiac remodeling was ameliorated and pulmonary edema was alleviated following TSG treatment. Interestingly, TSG showed superior effects than Xinbao pills on increasing -LVdp/dtmax, correcting myocardial hypertrophy, and promoting urinary output and sodium excretion.
Conclusion
TSG demonstrated superior therapeutic efficacy than Xinbao pills in CHF models by improving cardiac function, reducing cardiac load, and ameliorating cardiac remodeling. The study provides preclinical evidence supporting the use of TSG in CHF treatment, warranting further clinical investigation.
Keywords
Introduction
Chronic heart failure (CHF) is a common clinical syndrome and the final stage of many cardiovascular diseases, resulting in specific disease symptoms such as loss of physical strength and dyspnea.1,2 The statistical data shows that there are about 64 million patients with CHF worldwide, whose 5- and 10-year survival rates are 56.7% and 34.9%, respectively.3,4 Due to population growth and aging, the total number population suffering from heart failure continues to grow at a rate of 2 million per year. 5 Notably, 2%-17% of CHF patients died on their first admission, 17%-45% of CHF patients died within 1 year of admission and 50% within 5 years. 6 Thus, CHF is a serious threat to human life, and is one of the important cardiovascular diseases that need to be addressed urgently.
The core for CHF treatment is to inhibit myocardial remodeling and restore cardiac function. 7 Conventional Western treatment such as diuretics, renin-angiotensin-aldosterone system (RAAS) inhibitors, and beta blockers, which are commonly used for the clinical treatment of CHF, can stabilize the disease faster, but the prognosis improvement for most patients is still not satisfactory.8–12 In recent years, many advances have been made in drug therapies of CHF. For example, angiotensin receptor-neprilysin inhibitors (ARNIs) suppressed both the RAAS and the catabolism of endogenous natriuretic peptides, thereby reducing hospitalization and mortality. 13 However, ARNIs were not indicated in patients with hypotension, and the long-term effects were unsatisfactory. In addition, trimetazidine, an adjunct to conventional treatments, improves heart failure by modulating energy metabolism and alleviating cardiomyocyte apoptosis, fibrosis, and inflammation. 14 Nevertheless, it has been discontinued clinically due to dyskinesia side effects. More recently, other potential drugs have also been explored, such as the sodium-glucose cotransporter-2 inhibitors (SGLT2is) which targets to improve blood glucose control, as well as the soluble guanylate cyclase (sGC) stimulators modifying the nitric oxide-sGC-cyclic guanosine monophosphate cascade, a pathophysiological pathway that has been targeted with limited success in heart failure with reduced ejection fraction previously.15–17 Additionally, Menasche et al conducted the first-in-man use of a cardiovascular cell-derived secretome in heart failure, which revealed the potential therapeutic value of the cell-derived secretome in patients with CHF. 18 Although advances have been observed in therapeutic strategies for CHF, it still cannot meet the clinical needs, and remains a highly lethal disease with an increasing hospitalization burden and high healthcare expenditures.19,20 Hence, it is important to develop effective drugs for the prevention and treatment of CHF.
Ginseng, known as the “king of all herbs”, is one of the traditional Chinese medicines with the most widely used and oldest history in China and Southeast Asia. 21 Ginsenosides are a group of important pharmacological active ingredients in ginseng. 22 The studies have shown that ginsenosides have potential in the prevention and treatment of cardiovascular and related diseases. 21 In a randomized controlled trial, ginseng treatment was demonstrated to significantly enhance the myocardial contractility, and improve cardiac function of CHF patients. 23 Moreover, Calcium ions (Ca2+) play an essential role in regulating contraction and intracellular signaling of the cardiovascular system, which are crucial for cardiac function. 24 The studies have revealed that ginsenosides, the active ingredients of ginseng can suppress Ca2+ entry, improving cardiac functions.25,26 However, most of the active ingredients effective against cardiovascular diseases are total secondary ginsenosides (TSG, in vivo metabolites of ginsenosides), regrettably, TSG are found in extremely low levels in plants.21,22 More importantly, their native ginsenosides could not show obvious pharmacological action due to too low conversion rate for body absorption. 27
After many years of research and development, the researchers realized the industrialized production of TSG for which they patented the technology (Patent No. CN1723912A). The TSG extracted by this technique has many active ingredients, such as, ginsenoside Rg3 was shown to have a positive effect on the abnormal Ca2+ cycle in the cardiomyocytes of mice with heart failure 28 ; and it could attenuate myocardial fibrosis to ameliorate heart failure through increasing aminoacylase-1 expression and inhibiting TGF-β1/Smad3 pathway 29 ; ginsenoside Rg2 was found to improve cardiac function and inhibit collagen deposition in mice with myocardial infarction. 30 More importantly, the phase I clinical trial verified the safety and tolerability of TSG orally disintegrating tablets in 30 healthy cases. 31 However, the therapeutic efficacy of TSG on CHF has not been clarified.
In this study, we evaluated the effects of TSG on cardiac function, left ventricular configuration, hemodynamics, urinary excretion and pulmonary edema of the rat and dog CHF models, aiming to reveal the efficiency and provide favorable preclinical data for the treatment of CHF with TSG.
Materials and Methods
Drugs and Animals
The test drug TSG (Lot number 20170712) was obtained from Shanghai Huiyu Biomedical Technology Co., Ltd (Shanghai, China). TSG content (weight percent) of this batch was 68%, with the major secondary ginsenoside Rg3 17.3%, Rg2 7.8% and Rh1 3.9% (chemical structures and molecular weights as in Figure 1). The positive control drug Xinbao pills (Lot number 20170305) were purchased from Guangdong Xinbao Pharmaceutical and Technology Co., Ltd (Guangdong, China), and enalapril maleate tablets (Lot number 15120711) were purchased from Jiangsu Pharmaceutical Co., Ltd (Yangtze River Pharmaceutical Group, China).

Chemical structure of the main components in TSG. (A) Chemical structure of ginsenoside Rg3. (B) Chemical structure of ginsenoside Rg2. (C) Chemical structure of ginsenoside Rh1.
Male Sprague-Dawley rats (7-8 weeks old) were obtained from Beijing Vital River Laboratory Animal Technology Co., Ltd (Beijing, China; permit number SCXK(Jing) 2016-0011). The beagle dogs (the ratio of male to female is 1:1; 10-12 months old), were obtained from Shenyang Kangping County Experimental Animal Institute (Liaoning, China; permit number SCXK(Liao) 2014-0005). All experiments were approved by the Institutional Animal Ethical Committee. The animals lived under specific pathogen free (SPF) conditions at an ambient temperature of 22-26 °C and 40%-70% relative humidity with a 12 h light-dark cycle, and were allowed ad libitum access to water and laboratory chows. The animals were accommodated for 1 week before proceeding to experiments.
Model Construction and Treatment for Rats and Dogs
All rats were randomly divided into the control group and the model group in a 1:6 ratio. The rats of the model group underwent ligation of the left anterior descending coronary artery to establish the CHF model. 32 More specifically, in a SPF environment, after anesthesia by intraperitoneal injection of 3% sodium pentobarbital (60 mg/kg), the rats were connected to a rodent ventilator, left thoracotomy and pericardiotomy were performed to expose the heart, then the left anterior descending coronary artery was ligated with 6/0 silk suture. Subsequently, the thoracic cavity was closed to empty the internal air, and the muscle layer and skin were sutured. The rats of the control group were subjected to sham operation (defined as sham group). They were only threaded under the left anterior descending coronary artery without ligation, and other surgical operations were kept the same as in the model group. After 4 weeks of operation, cardiac functions were determined using a Doppler ultrasound (Vevo 770TM-120, Visual Sonics Inc, Canada). The rat CHF model was considered successfully constructed as the left ventricular ejection fraction (LVEF) decreased by more than 30% in comparison with that of the pre-operative period.33,34 Animals that did not satisfy the above criteria indicated modeling failure and were excluded from the experiment. Thereafter, the rats in the model group were randomly divided into model control group (Model), TSG low-dose group (TSG-L), TSG medium-dose group (TSG-M), TSG high-dose group (TSG-H), Xinbao pill group (XBP) and enalapril group (ELP) (n = 10/group). According to the effective concentrations obtained in the pre-experiment (Tables I and II in Supplemental Material 2), except for the model control group, the remaining rats were given 50 mg/kg/d TSG in TSG-L group, 100 mg/kg/d TSG in TSG-M group, 200 mg/kg/d TSG in TSG-H group, 75 mg/kg/d Xinbao pills in XBP group, and 2 mg/kg/d enalapril power in ELP group, respectively. The drugs were ground and dissolved in 0.5% sodium carboxymethyl cellulose (CMC) (10 mL/kg) to prepare a suspension given by intragastric administration. Meanwhile, the sham group and the model control group were given only the equivalent volume of 0.5% CMC (10 mL/kg). All rats were administered one time per day for a continuous treatment period of 30 days.
On the other hand, all beagles were randomly divided into the control group and the model group in a 1:5 ratio. The dogs in the model group were subjected to inferior vena cava constriction to establish the CHF model. 35 To recap, after anesthesia, the dogs were connected to a ventilator and underwent thoracotomy to isolate the inferior vena cava, which was narrowed by approximately half depending on its outer diameter. In the sham group, the dogs were only threaded under the inferior vena cava. Postoperatively, the animals were housed in a single cage and underwent wound care for approximately 2 weeks. Echocardiograms were performed periodically in the awake state from the time when ascites became evident (approximately 30 days after surgery). The CHF model was successfully constructed once the LVEF decreased by more than 30% from the pre-operative level. 33 The dogs in the model group were randomized into model control group (Model), TSG low-dose group (TSG-L), TSG medium-dose group (TSG-M), TSG high-dose group (TSG-H) and Xinbao pill group (XBP) (n = 6/group). Except for the model control group, the remaining four groups were given 15 mg/kg/d TSG in TSG-L group, 30 mg/kg/d TSG in TSG-M group, 60 mg/kg/d TSG in TSG-H group, and 25 mg/kg/d Xinbao pills in XBP group by oral administration based on the conversion of equivalent dose administered to rats. The method of drug preparation was kept the same as that of rats. Meanwhile, the sham group and the model control group were given only the equivalent volume of 0.5% CMC. All dogs were administered one time per day for a continuous treatment period of 28 days.
Detection of Echocardiography and Hemodynamic Parameters in Rats and Dogs
Echocardiograms were performed on experimental animals in pre-operative as well as before and after drug administration using Doppler ultrasound system. Experimental rats were tested before operation, before drug administration, and on days 0, 10, 20, and 30 after drug administration; and experimental dogs were tested before operation, before drug administration, and on days 0, 7, 14, 21, and 28 after drug administration. M-mode echocardiography were acquired and stored. The interventricular septum in diastole and systole (IVSd and IVSs), left ventricular internal diameter in diastole and systole (LVIDd and LVIDs), left ventricular posterior wall in diastole and systole (LVPWd and LVPWs), left ventricular volume in diastole and systole (LVVd and LVVs), LVEF and fractional shortening (FS) were determined by offline workstation software.
After 12 h at the end of the last drug administration, the experiment animals in each group were intubated with a pressure catheter to the left ventricle, and the left ventricular end-diastolic pressure (LVEDP), left ventricular systolic pressure (LVSP), the maximum rate of rise of left ventricular pressure (+LVdP/dtmax), and the maximum rate of fall of left ventricular pressure (-LVdP/dtmax) were determined. Also, systemic arterial blood pressure, including systolic arterial pressure (SAP), diastolic arterial pressure (DAP), mean arterial pressure (MAP), and heart rate (HR) were measured. All parameters were acquired and recorded with the multi-channel physiological signal recording and analysis system (MP150, BIOPAC Inc, United States), and data measurements were performed with AcqKnowledge software (version 3.9.1).
Urinary Excretion Analysis in Rats
After 25 days of drug administration, rats in each group were water-loaded by subcutaneous injection with normal saline [2 mL/100 g body weight (BW)] and assisted in urine emptying. Subsequently, 0-2 h and 2-24 h urine volumes were collected and accurately measured. The excretion rate of urinary was expressed as urine volume (mL)/100 g BW. The sodium assay kit and potassium assay kit (Nanjing Jiancheng Bioengineering Institute, China) were used to measure the Na+ and K+ concentrations in the urine from 0-2 h and 2-24 h, respectively. Then, the excretion rates of Na+ and K+ in the urine were calculated.
Analysis of Cardiac Hypertrophy in Rats
At the end of the in vivo experiment, hearts of rats were taken and blood was removed from the surface. The whole heart, left ventricle to tibia length ratio (mg/mm) was determined and expressed as the degree of myocardial hypertrophy.
Analysis of Tumor Necrosis Factor α (TNF-α) Levels in Rats
At the end of the in vivo experiment, about 50 mg of tissue from the risk zone of the heart was taken and prepared as a tissue homogenate in 0.9% saline. The homogenate was collected and centrifuged at 10 000×g for 10 min at 4 °C. Then the supernatant was collected and immediately assayed. TNF-α levels were determined by Rat TNF-α ELISA Kit (Bio-Swamp, China).
Analysis of Cardiomyocyte Apoptosis in Rats
The hearts of rats were cut in a coronal position, fixed, wrapped with paraffin and sectioned (5μm thickness). Subsequently, the tissue slices were used for hematoxylin and eosin (H&E) staining to visualize histopathological changes, and stained with an in-situ cell death detection kit (Roche Ltd, Switzerland) following the operator's manual to detect cardiomyocyte apoptosis. The cardiomyocyte apoptosis rate was expressed as the ratio of the number of apoptotic cells to the total cell number. In addition, about 50 mg of tissue from the risk zone of the heart was taken and prepared as a tissue homogenate in 0.9% saline. The supernatant tissue fluid was taken by centrifugation at 3000 rpm with 4 °C for 10 min. After protein quantification, caspase-3, Bcl-2, and Bax protein expression were determined by Elisa assay (Bio-Swamp, China).
Analysis of Vasoconstrictor Factor Levels in Dogs
For experiment dogs, the blood samples were taken by arterial cannulation and centrifuged at 4000 rpm for 10 min. The endothelin-1 (ET-1), angiotensin II (Ang-II) and nitric oxide (NO) levels in serum were measured using the dog ET-1 ELISA kit (Cusabio Technology LLC, United States), the dog Ang-II ELISA kit (Cusabio Technology LLC, United States) and NO assay kit (Nanjing Jiancheng Bioengineering Institute, China), respectively.
Heart Weight Analysis in Dogs
At the end of the experiment, the dogs were anesthetized for blood sampling and then executed. The hearts were removed, and the chambers were washed of residual blood and weighed. At the same time, the tibia length of the left hind limb was measured and the index of each chamber was calculated. Chamber weight index (g/cm) = Chamber weight (g) / tibia length (cm).
Detection of Lung Water Content in Rats and Dogs
At the end of the in vivo experiments, the lung tissues of the experimental animals were autopsied out, removed the residual blood with filter paper, and weighed to be recorded as wet weight (ww). Then, the lung tissues were dried at 50 °C and weighed as dry weight (dw). The lung water content (%) was expressed as (ww-dw)/ww × 100.
Statistical Analysis
All data were expressed as the mean ± standard deviation (SD). GraphPad Prism software (La Jolla, CA) was used for statistical analysis. The data with normal distribution and uniform variance were analyzed by one-way analysis of variance (ANOVA) followed by the least significant difference (LSD) test. The data with uneven variance or non-normal distribution were tested by the Kruskal-Wallis and Dunn's non-parametric test. P < .05 was considered statistically significant.
Results
Therapeutic Efficacy of TSG in CHF Rats
Analysis of Cardiac Function in Rats
Compared with the sham group, LVEF and FS exhibited a significant decrease in the model group (P < .001), which remained relatively stable for 30 days after drug administration (Table 1). No adverse effects were observed in CHF rats during the trial. Administration of TSG-L, TSG-M, TSG-H, XBP, and ELP for 30 days led to an increase in LVEF compared to before administration by 16.5% (P > .05), 20.9% (P < .05), 27.9% (P < .01), 18.8% (P < .05), and 21.9% (P < .01), respectively; and an increase in FS by 21.4% (P > .05), 31.6% (P < .05), 36.2% (P < .01), 23.7% (P < .05), and 29.1% (P < .01), respectively (Table 1). Furthermore, in comparison with the sham group, the model group showed a significant reduction in both +LVdp/dtmax and -LVdp/dtmax by 28.1% (P < .001) and 31.9% (P < .001), respectively, indicating a marked decline in left ventricular systolic and diastolic functions (Table 2). After 30 days of treatment, compared to the model group, TSG-L, TSG-M, TSG-H, XBP, and ELP groups increased +LVdp/dtmax by 12.6% (P > .05), 48.6% (P < .05), 74.4% (P < .001), 43.6% (P < .05), and 81.5% (P < .001), respectively; and -LVdp/dtmax increased by 2.1% (P > .05), 30.3% (P > .05), 55.4% (P < .05), 9.4% (P > .05), and 71.7% (P < .01), respectively. Notably, compared to the XBP group, the -LVdp/dtmax in the TSG-H group was significantly improved (55.4% vs 9.4%, P < .05) (Table 2).
The LVEF and FS in Different Groups of Rats (n = 10/Group).
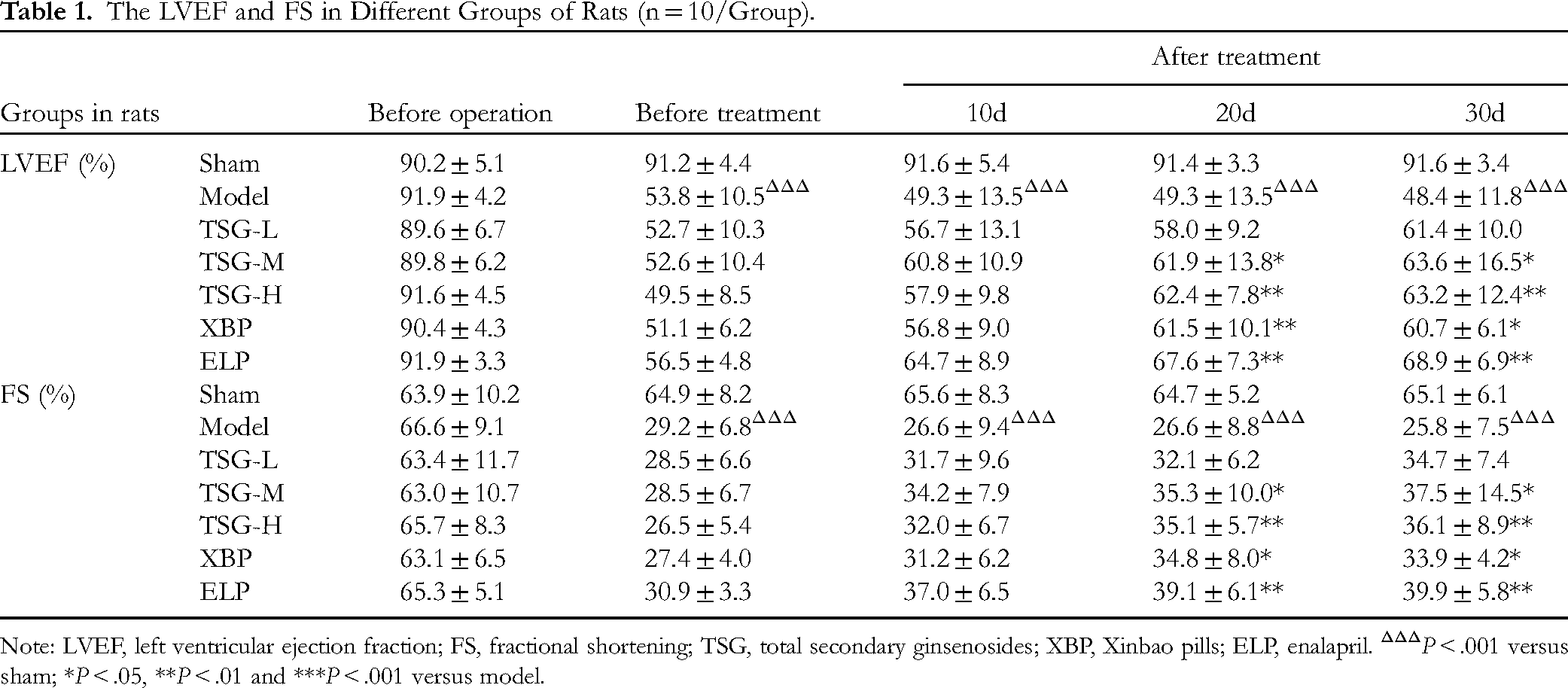
Note: LVEF, left ventricular ejection fraction; FS, fractional shortening; TSG, total secondary ginsenosides; XBP, Xinbao pills; ELP, enalapril. ΔΔΔP < .001 versus sham; *P < .05, **P < .01 and ***P < .001 versus model.
The Hemodynamic Parameters in in Different Groups of CHF Rats (n = 10/Group).
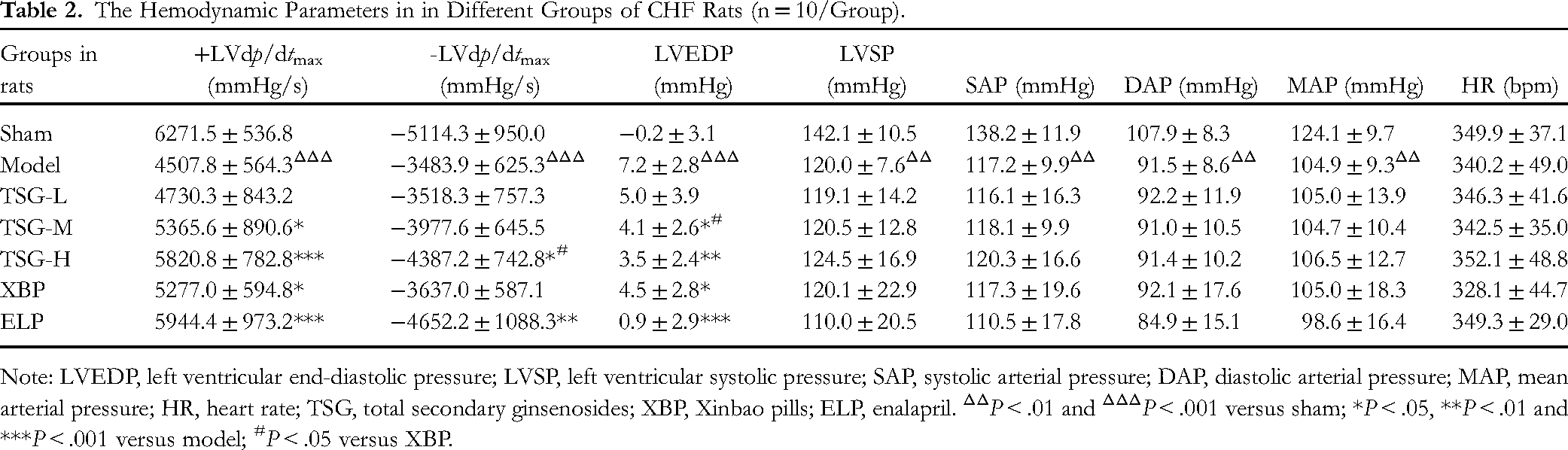
Note: LVEDP, left ventricular end-diastolic pressure; LVSP, left ventricular systolic pressure; SAP, systolic arterial pressure; DAP, diastolic arterial pressure; MAP, mean arterial pressure; HR, heart rate; TSG, total secondary ginsenosides; XBP, Xinbao pills; ELP, enalapril. ΔΔP < .01 and ΔΔΔP < .001 versus sham; *P < .05, **P < .01 and ***P < .001 versus model; #P < .05 versus XBP.
Analysis of Left Ventricular Configuration in Rats
Doppler ultrasound analysis of cardiac configuration revealed no significant effects of TSG and the positive drug on IVS and LVPW (Figure S1, Tables S1 and S2). Compared with the sham group, the model group showed a significant increase in LVID and LVV (P < .001). After 30 days of treatment with TSG-L, TSG-M, TSG-H, XBP, and ELP, LVIDd was reduced by 2.8% (P > .05), 8.7% (P = .055), 16.9% (P < .001), 6.8% (P > .05), and 21.4% (P < .001), respectively (Table S3); LVIDs was reduced by 5.8% (P > .05), 18.5% (P < .001), 25.2% (P < .001), 15.1% (P < .001), and 22.2% (P < .001), respectively (Table S3); LVVd was reduced by 6.3% (P > .05), 17.9% (P = .055), 32.1% (P < .001), 15.5% (P > .05), and 41.9% (P < .001), respectively (Table S4); and LVVs was reduced by 22.6% (P < .01), 37.1% (P < .001), 46.7% (P < .001), 30.5% (P < .001), and 58.4% (P < .001), respectively (Table S4). It is evident that TSG-H outperformed the XBP group in improving LVIDd and LVVd.
Analysis of Hemodynamics in Rats
Compared with the sham group, the LVEDP in the model group was significant increased, indicating a marked increase in cardiac preload. After 30 days of treatment with TSG-L, TSG-M, TSG-H, XBP, and ELP, the elevated LVEDP was reduced by 29.7% (P > .05), 41.9% (P < .05), 50.0% (P < .01), 36.5% (P < .05), and 85.1% (P < .001), respectively (Table 2). Additionally, TSG, XBP, and ELP had no significant effects on LVSP, SAP, DAP, and MAP (Table 2).
Analysis of Myocardial Hypertrophy in Rats
The ratio of heart weight to tibia length in the model group significant increased in comparison with the sham group (P < .001), indicating myocardial hypertrophy in CHF rats. After treatment with TSG-L, TSG-M, TSG-H, XBP, and ELP, the increased LV/Tibia ratio was reduced by 44.0% (P > .05), 68.0% (P < .01), 84.0% (P < .01), 60.0% (P < .05), and 88.0% (P < .001), respectively (Figure 2A); and the increased Heart/Tibia ratio was reduced by 22.6% (P > .05), 54.8% (P > .05), 64.5% (P < .05), 41.9% (P > .05), and 96.8% (P < .001), respectively (Figure 2B).
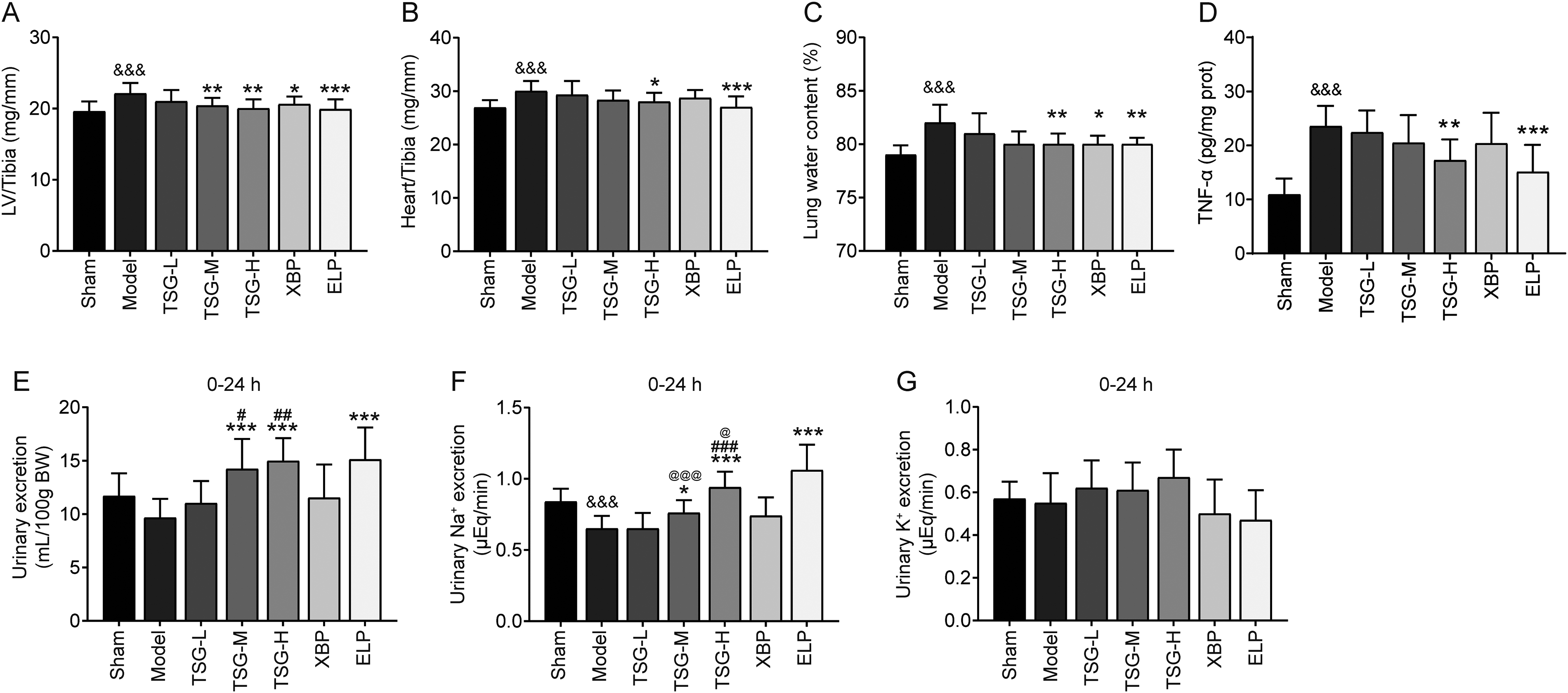
Effect of TSG on myocardial hypertrophy, pulmonary edema, urine volume and urinary electrolytes in CHF rats. (A) The LV/Tibia ratio in rats after treatment; (B) The Heart/Tibia ratio in rats after treatment; (C) Lung water volume in rats after treatment; (D) TNF-α levels in rats after treatment; (E) Effect of TSG on urine volume with 0-24 h in CHF rats; (F) Effect of TSG on urinary Na+ excretion with 0-24 h in CHF rats; (G) Effect of TSG on urinary K+ excretion with 0-24 h in CHF rats. Each group n = 10. &&&P < .001 versus sham; *P < .05, **P < .01 and ***P < .001 versus model; #P < .05, ##P < .01 and ###P < .001 versus XBP; @P < .05, and @@@P < .001 versus ELP.
Analysis of Pulmonary Edema in Rats
Compared with the sham group, the lung water content in the model group showed a significant increase (P < .001), indicating pulmonary edema in CHF rats. After treatment with TSG-H, XBP, and ELP, the increased lung water content was reduced by 77.1% (P < .01), 72.7% (P < .05), and 82.3% (P < .01), respectively (Figure 2C). It is evident that TSG-H, XBP, and ELP have an ameliorative effect on pulmonary edema.
Analysis of TNF-α Levels in Rats
Compared with the sham group, the TNF-α level of myocardial tissue was significantly higher in the CHF model group (P < .001). After treatment with TSG-H and ELP, the increased TNF-α level was reduced by 49.8% (P < .01) and 66.9% (P < .001) (Figure 2D). However, the treatment of XBP could decrease the raised TNF-α by 25.3%, but there was no statistical difference (P > .05) (Figure 2D).
Analysis of Urine Output in Rats
The model control group showed a decrease in urine output and urinary Na+ excretion rate (Figure 2E-F and Figure S2), while urinary K+ excretion rate remained unchanged (Figure 2G and Figure S2), indicating water and sodium retention in CHF rats. Compared to the XBP group, TSG-M had a stronger effect on the 0-24 h urine output/100BW ratio (47.3% vs 19.4%, P < .05) (Figure 2E); TSG-H had a stronger effect on both the 0-24 h urine output/100BW ratio (55.0% vs 19.4%, P < .01, in Figure 2E) and urinary Na+ excretion rate (44.6% vs 13.8%, P < .001, in Figure 2F).
Analysis of Histopathology and Myocardial Cell Apoptosis in Rats
In the sham group, myocardial fibers and cytoplasm were uniformly stained, cells were neatly arranged, tissue stroma was free of inflammatory cell infiltration, and no myocardial necrosis lesions appeared; in the model group, the extracellular space was widened, cardiomyocyte cell volume was increased with the phenomenon of vacuolization, interstitial and cellular edema was obvious, nuclear fragmentation was found, myocardial fibers were arranged in a loss of the normal structure with an obvious wave-like pattern, and there was inflammatory infiltration (Figure 3A). TSG, XBP and ELP treatments reduced these pathological findings in model group, with increasing the number of normal cells, decreasing the number of degenerated and necrotic cells, and reducing the pericellular space (Figure 3A). In addition, the CHF model control group showed a significant increase in the percentage of myocardial cell apoptosis, as well as the expression of Caspase-3 and Bax/Bcl-2 (Figure 3B-F). TSG-M and TSG-H groups were able to reduce the myocardial apoptosis rate compared to the model group by 30.8% (P < .05) and 35.1% (P < .01), respectively (Figure 3B), and also reduced the Bax/Bcl-2 ratio in cardiomyocytes (Figure 3F), indicating a regulatory effect of TSG on apoptosis. XBP and ELP groups were also found to have similar effects.

Histopathologic analysis of myocardial tissue in CHF rats. (A) Representative images of HE staining of myocardial tissue (scale bar 20μm); (B) Representative images of cardiomyocyte apoptosis (scale bar 20μm) and the statistics of cardiomyocyte apoptosis; (C) The expression of Caspase3 protein in different test groups; (D) The expression of Bax protein in different test groups; (E) The expression of Bcl2 protein in different test groups; (F) The expression of Bax/Bcl2 protein in different test groups. Each group n = 10.&&&P < .001 versus sham; *P < .05, **P < .01 and ***P < .001 versus model; @P < .05, @@P < .01 and @@@P < .001 versus ELP.
Summary of the Therapeutic Effect of TSG in CHF Rats
TSG treatment at low, medium and high dosage all significantly improved myocardial contractility and cardiac systolic/diastolic functions, as evidenced by increased LVEF, FS, and ± LVdp/dtmax in CHF rats (Tables 1 and 2). TSG also significantly reduced the cardiac load, as evidenced by decreased LVEDP, which may be related to the increased urine excretion and reduced water and sodium retention (Table 2 and Figure 2D-F). Meanwhile, TSG significantly improved cardiac remodeling, as demonstrated by reduced cardiac dilation, alleviated myocardial pathological injury, corrected myocardial hypertrophy, regulated the balance of apoptotic proteins Bax/Bcl-2, and inhibited myocardial cell apoptosis (Figures 2A, B and 3). Additionally, TSG alleviated pulmonary edema (Figure 2C). Most importantly, compared to XBP, the advantages of TSG in treating CHF rats are evident: TSG-H significantly increased -LVdp/dtmax (Table 2); TSG-M and TSG-H significantly reduced LVIDd and LVVd (Tables S3 and S4); TSG-H significantly reduced the heart/Tibia ratio (Figure 2B); TSG-M and TSG-H significantly increased the 0-24 h urine output/100BW ratio and urinary Na+ excretion rate (Figure 2D-E). It is evident that oral administration of TSG was effective in treating CHF rats with an effective dose range of 100-200 mg/kg.
Therapeutic Efficacy of TSG in CHF Dogs
Analysis of Cardiac Function in Dogs
Following the successful modeling of CHF in dogs, LVEF and FS significantly decreased and remained relatively stable over 28 days. After administration, there were no detectable symptoms of discomfort in dogs. After 28 days of drug administration, compared to the model group, TSG-L, TSG-M, TSG-H, and XBP could respectively increase LVEF by 24.8% (P < .01), 45.7% (P < .001), 48.6% (P < .001), and 45.4% (P < .001) compared to before administration; and increase FS by 36.5% (P < .01), 68.8% (P < .001), 76.4% (P < .001), and 69.7% (P < .001), respectively (Table 3). Moreover, the continuous administration of TSG-L, TSG-M, and TSG-H for 28 days showed a dose-dependent increase in the reduction of +LVdp/dtmax and -LVdp/dtmax caused by CHF, enhancing myocardial contraction. Compared to the model group, +LVdp/dtmax increased by 25.1% (P < .05), 34.5% (P < .01), and 52.6% (P < .01), respectively; -LVdp/dtmax increased by 47.4% (P > .05), 61.8% (P < .05), and 66.6% (P < .05), respectively (Table 4). After 28 days of XBP administration, +LVdp/dtmax and -LVdp/dtmax increased by 39.8% (P < .01) and 52.3% (P > .05), respectively, compared to the model group (Table 4).
The LVEF and FS in Different Groups of Dogs (n = 6/Group).

Note: LVEF, left ventricular ejection fraction; FS, fractional shortening; TSG, total secondary ginsenosides; XBP, Xinbao pills. ΔΔΔP < .001 versus sham; *P < .05, **P < .01 and ***P < .001 versus model.
The Hemodynamic Parameters in in Different Groups of CHF Dogs (n = 6/Group).

Note: LVEDP, left ventricular end-diastolic pressure; LVSP, left ventricular systolic pressure; SAP, systolic arterial pressure; DAP, diastolic arterial pressure; MAP, mean arterial pressure; HR, heart rate; TSG, total secondary ginsenosides; XBP, Xinbao pills. ΔΔP < .01 and ΔΔΔP < .001 versus sham; *P < .05 and **P < .01 versus model.
Analysis of Left Ventricular Configuration in Dogs
After the formation of CHF, the thickness of the interventricular septum and the posterior wall of the left ventricle significantly thinned. After 28 days of administration, TSG-H had a significantly stronger effect on IVSd and IVSs than XBP (P < .05) (Table S5). The TSG-H group significantly inhibited the trend of LVPWs reduction, with a 44.3% decrease compared to before treatment, and a significant effect compared to the model group (P < .05); whereas XBP had no significant effect on LVPW (Table S6). Additionally, after 28 days of administration, compared to before treatment, TSG-M, TSG-H, and XBP groups could respectively reduce LVIDs by 24.4% (P < .05), 31.0% (P < .01), and 26.7% (P < .05) (Table S7); LVVs decreased by 50.2% (P < .05), 60.2% (P < .01), and 56.4% (P < .05), respectively (Table S8).
Analysis of Hemodynamics in Dogs
After continuous administration of TSG and XBP for 28 days, there was a significant inhibition of the CHF-induced increase in LVEDP and decrease in LVSP (Table 4). Compared to the model group, TSG-M, TSG-H, and XBP could reduce LVEDP by 62.1% (P < .01), 71.9% (P < .01), and 46.5% (P > .05), respectively; LVSP increased by 32.7% (P < .05), 32.1% (P < .01), and 30.1% (P < .01), respectively (Table 4). Furthermore, after continuous administration of TSG-H and XBP for 28 days, there was a significant inhibition of the blood pressure decrease caused by CHF. Compared to the model group, TSG-H could increase SAP, DAP, and MAP by 39.7% (P < .05), 51.9% (P < .01), and 44.4% (P < .01), respectively; XBP could increase by 40.3% (P < .01), 52.0% (P < .05), and 44.7% (P < .01), respectively (Table 4).
Analysis of Myocardial Hypertrophy in Dogs
Compared to the sham group, the indices of cardiac chambers in the model group were reduced (Figure 4A-D). After continuous administration for 28 days, compared to the model group, TSG-H significantly inhibited the decrease in the ventricular indices caused by CHF, with the left ventricular/tibia increasing by 22.9% (P < .05) and right ventricular/tibia increasing by 25.8% (P < .05), respectively (Figure 4C-D). Comparatively, XBP had no significant effect on the indices of cardiac chamber weights (P > .05).

Effect of TSG on heart weight, pulmonary edema and levels of vasomotor factors in CHF dogs. (A) The Left atrium/Tibia ratio in dogs after treatment; (B) The Right atrium/Tibia ratio in dogs after treatment; (C) The Left ventricle/Tibia ratio in dogs after treatment; (D) The Right ventricle/Tibia ratio in dogs after treatment; (E) Lung water volume in dogs after treatment; (F) ET-1 levels in the different groups of dogs; (G) NO levels in the different groups of dogs; (H) Ang-II levels in the different groups of dogs. Each group n = 6. &P < .05 and &&P < .01 versus sham; *P < .05 versus model.
Analysis of Pulmonary Edema in Dogs
Compared to the sham group, dogs in the model group showed a significant increase in lung water content (P < .01), indicating the presence of pulmonary edema in CHF dogs (Figure 4E). TSG-H and XBP groups could reduce the increased lung water content by 55.9% (P < .05) and 44.1% (P > .05), respectively.
Analysis of Vascular Factors in Dogs
Compared to the model group, the TSG-H significantly inhibited the increase expression of ET-1 (P < .05, Figure 4F) and Ang-II (P < .05, Figure 4H). After TSG administration, the NO expression level tended to increase, but the difference was not statistically significant (P > .05, Figure 4G). Comparatively, XBP had no significant effect on the aforementioned biochemical indicators (Figure 4F-H).
Summary of the Therapeutic Effect of TSG in CHF Dogs
15-60 mg/kg TSG significantly improved myocardial contractility and cardiac systolic/diastolic functions in CHF dogs, as evidenced by increased LVEF, FS and ± LVdp/dtmax (Tables 3 and 4). TSG significantly ameliorated the abnormal changes in cardiac configuration, as shown by narrowing of LVIDs and LVVs, inhibition of thinning of LVPWs, IVSd and IVSs, and correcting of left ventricular remodeling (Tables S5-S8 and Figure 4C-D). Meanwhile, TSG-H could reduce the preload and afterload of the heart, as evidenced by the inhibition of the increase in LVEDP, which may be related to the reduction of vasoconstrictive substances (ET-1 and Ang-II) (Table 4 and Figure 4F-H). Additionally, TSG-H significantly alleviated pulmonary edema (Figure 4E). More importantly, compared to XBP, TSG has advantages in treating CHF dogs manifested in: TSG-M and TSG-H significantly inhibited the increase of LVEDP, as well as enhanced -LVdp/dtmax (Table 4); TSG-H significantly inhibited the thinning of LVPWs, IVSd and IVSs (Tables S5 and S6), increased the ventricular indices (Figure 4A-D), reduced lung water content (Figure 4E), and inhibited the increase expression of ET-1 and Ang-II (Figure. 4F and H), while the clinically equivalent dose of XBP had no significant effect on the aforementioned actions. It was shown that oral TSG was effective in the treatment of CHF dogs at an effective dose of 15-60 mg/kg.
Discussion
The present study investigated the therapeutic effects of TSG on CHF in rat and dog models, providing a comparative analysis with XBP treatment. Our findings demonstrated that TSG significantly improved cardiac function, ameliorated cardiac remodeling, and reduced pulmonary edema in CHF models, suggesting its potential as an effective treatment for CHF.
LVEF was of great importance in the diagnosis, treatment, prognosis, and risk stratification of heart failure.36–38 Heart failure could be categorized based on LVEF measurements: heart failure with reduced ejection fraction (HFrEF), with mildly reduced ejection fraction (HFmrEF), with preserved ejection fraction (HFpEF). 39 Recent data have suggested that the incidence of heart failure was essentially flat or declining as a result of better management of cardiovascular disease, but might be increasing due to the lack of effective treatments for HFpEF.12,37 This highlighted the essential differences in treating different heart failure phenotypes and intensified research into HFmrEF and HFpEF.37,38,40 Additionally, the investigators found that increasing LVEF might improve left ventricular systolic function and attenuate stress-induced remodeling in CHF rats. 41 The combination preparation (Shenfu decoction) containing ginseng components was effective in increasing LVEF in the treatment of heart failure rats. 42 In the present study, after construction of CHF models in rats and dogs, the LVEF of the model groups was 53.8 ± 10.5% and 52.7 ± 1.4%. This made our study correspond to therapeutic studies of HFpEF. Excitingly, HFpEF was effectively rescued after TSG treatment, and the treatment group was more convergent to the sham group (normal control) as the administered dose increased. In the rat CHF model, LVEF was increased by 16.5% (TSG-L), 20.9% (TSG-M), and 27.9% (TSG-H), respectively, compared to the pre-administration level by a dose-dependent increase in TSG; similarly, in the dog CHF model, LVEF was TSG dose-dependently increased by 24.8% (TSG-L), 45.7% (TSG-M), and 48.6% (TSG-H), respectively. Once again, the therapeutic effect of TSG on CHF as well as HFpEF was illustrated.
The potential mechanisms by which the TSG exerts its therapeutic effects in CHF can be attributed to several factors. First, TSG enhanced myocardial contractility as manifested by an increase in ± LVdp/dtmax (increased 55.4-74.4% by TSG-H treatment in rats; increased 52.6-66.6% by TSG-H treatment in dogs), which could be related to the modulation of intracellular Ca2+ handling and improvement of cardiomyocyte function.43–45 Secondly, TSG reduced cardiac load, as demonstrated by a decrease in LVEDP (reduced 41.9% and 50.0% by TSG-M and TSG-H treatment in rats; reduced 62.1% and 71.9% by TSG-M and TSG-H treatment in dogs), which may be related to its diuretic effect and reduction of sodium retention.46,47 Thirdly, TSG improved cardiac remodeling as evidenced by reduced cardiac dilatation and attenuated myocardial hypertrophy, which could involve inhibition of fibrosis through the TGF-β1/Smad3 pathway, as in the previous studies.45,48,49 Meanwhile, TSG treatment caused a significant reduction in TNF-α expression (reduced 49.8% by TSG-H treatment in rats), suggesting a potential inhibitory effect of TSG on inflammation and fibrosis. Finally, the role of TSG in preventing cardiomyocyte loss, a key factor in the progression of CHF, was demonstrated by downregulating the Bax/Bcl-2 ratio and decreasing the rate of cardiomyocyte apoptosis (reduced 30.8% by TSG-M treatment and reduced 35.1% by TSG-H treatment in CHF rats).50–52
The commonly used methods for constructing animal models of CHF are mainly categorized into myocardial ischemia method, cardiac blood volume loading method, cardiac pressure overload method and drug-induced method. 35 Compared with the previous two methods, the cardiac pressure overload method takes a long time to be modeled and the modeling process is more complicated. 53 Besides, drug induction may affect the assessment of other drug treatment effect. 53 Thus, the myocardial ischemia method and cardiac volume loading method were mainly used to construct CHF animal models in this study. In rats, myocardial ischemia was induced by ligating the anterior descending branch of the left coronary artery to trigger heart failure. 32 This method is close to the pathophysiologic evolution of congestive heart failure in humans and helps to study the role of TSG on heart failure. In dogs, the cardiac blood volume loading method was used to construct the heart failure model by narrowing the inferior vena cava, resulting in obstruction of venous return, which allows for a decrease in return blood flow to the heart. 35 This model simulates the clinical pathology of reduced ventricular volume loading due to poor venous return, which continues to develop resulting in heart failure. This pathophysiological process is similar to clinical heart failure and can stably simulate the state of heart failure, providing a reliable research object for drug development and clinical treatment. 35 Notably, rats might have a more rapid progression of heart failure and a more pronounced inflammatory response due to their smaller body size, which might affect the observed therapeutic effects of TSG. In contrast, while individual differences might exist in dogs, their larger body size and longer lifespan might allow for a more gradual and subtle manifestation of CHF, potentially providing insight into the efficacy and safety of long-term therapy. Hence, the construction of the two models contributed to a comprehensive understanding of the therapeutic efficacy of TSG for CHF.
Despite the promising results, our study has several limitations. Only male rats and different-sex beagle dogs were used in this study model, and did not find sex-specific differences in the therapeutic effects of TSG during the experiments, but this might affect the generalizability of our findings given the relatively small sample size of the animal models. Thus, further studies are needed to determine whether the TSG treatment has sex-specific differences. Meanwhile, the focus on differences in treatment time proposed by Crnko et al also needs to be further investigated. 54 Besides, while we have demonstrated the efficacy of TSG in animal models, the translation of these results to human patients requires further investigation, including pharmacokinetics and clinical trials. Future research should focus on translating the preclinical findings into clinical settings. This includes conducting randomized controlled trials to assess the safety, tolerability, and efficacy of TSG in human CHF patients.
Conclusion
In CHF animal models, TSG was found to improve cardiac systolic/diastolic function and conformation by increasing LVEF, FS, and ± LVdp/dtmax, attenuating myocardial pathological injury, correcting hypertrophy, and reducing LVEDP to lower cardiac load. Noticeably, TSG exhibited unique effects compared to XBP, such as on improving the -LVdp/dtmax, myocardial hypertrophy, and the rate of urinary output and urinary sodium excretion XBP. Overall, the oral administration of TSG was effective for the treatment of CHF at doses ranging from 100 to 200 mg/kg in rats and 15 to 60 mg/kg in dogs, respectively. This study provided compelling preclinical evidence for the application of TSG in the treatment of CHF. Further, the related clinical trial is underway, aiming to confirm its safety and efficacy in CHF patients.
Supplemental Material
sj-docx-2-npx-10.1177_1934578X251316651 - Supplemental material for Therapeutic Effects of Total Secondary Ginsenosides on Chronic Heart Failure in Rats and Dogs: A Comparative Study with Xinbao Pills
Supplemental material, sj-docx-2-npx-10.1177_1934578X251316651 for Therapeutic Effects of Total Secondary Ginsenosides on Chronic Heart Failure in Rats and Dogs: A Comparative Study with Xinbao Pills by Xiangwei Xu, Yuhui Zhang, Yue Xiao, Aiqun Zhang, Shangxian Gao, Zhiquan Di and Jian Zhang in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X251316651 - Supplemental material for Therapeutic Effects of Total Secondary Ginsenosides on Chronic Heart Failure in Rats and Dogs: A Comparative Study with Xinbao Pills
Supplemental material, sj-docx-3-npx-10.1177_1934578X251316651 for Therapeutic Effects of Total Secondary Ginsenosides on Chronic Heart Failure in Rats and Dogs: A Comparative Study with Xinbao Pills by Xiangwei Xu, Yuhui Zhang, Yue Xiao, Aiqun Zhang, Shangxian Gao, Zhiquan Di and Jian Zhang in Natural Product Communications
Supplemental Material
sj-docx-4-npx-10.1177_1934578X251316651 - Supplemental material for Therapeutic Effects of Total Secondary Ginsenosides on Chronic Heart Failure in Rats and Dogs: A Comparative Study with Xinbao Pills
Supplemental material, sj-docx-4-npx-10.1177_1934578X251316651 for Therapeutic Effects of Total Secondary Ginsenosides on Chronic Heart Failure in Rats and Dogs: A Comparative Study with Xinbao Pills by Xiangwei Xu, Yuhui Zhang, Yue Xiao, Aiqun Zhang, Shangxian Gao, Zhiquan Di and Jian Zhang in Natural Product Communications
Supplemental Material
sj-docx-5-npx-10.1177_1934578X251316651 - Supplemental material for Therapeutic Effects of Total Secondary Ginsenosides on Chronic Heart Failure in Rats and Dogs: A Comparative Study with Xinbao Pills
Supplemental material, sj-docx-5-npx-10.1177_1934578X251316651 for Therapeutic Effects of Total Secondary Ginsenosides on Chronic Heart Failure in Rats and Dogs: A Comparative Study with Xinbao Pills by Xiangwei Xu, Yuhui Zhang, Yue Xiao, Aiqun Zhang, Shangxian Gao, Zhiquan Di and Jian Zhang in Natural Product Communications
Footnotes
Acknowledgements
Not applicable.
Author Contributions
Design of the work: Shangxian Gao, Zhiquan Di and Jian Zhang. Acquisition and analysis: Xiangwei Xu, Yuhui Zhang, Yue Xiao and Aiqun Zhang. Interpretation of data: All authors. Drafted the manuscript: Xiangwei Xu, Yuhui Zhang and Yue Xiao. All authors contributed to the article and approved the submitted version.
Consent for Publication
Not applicable.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study was approved by the Experimental Animal Ethics Committee of Tianjin Tiancheng New Drug Evaluation Co., Ltd.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by CAMS Innovation Fund for Medical Sciences (2019-I2M-5-020).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with Experimental Animal Ethics Committee of Tianjin Tiancheng New Drug Evaluation Co., Ltd (IACUC: 2022051701) approved protocols, and followed ARRIVE guidelines.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
