Abstract
Background
6-hydroxykynurenic acid (6-HKA), isolated from Ginkgo biloba leaves, has demonstrated neurorestorative properties against cerebral ischemia in middle cerebral artery occlusion (MCAO) mice models. However, our previous studies showed that 6-HKA was difficult to penetrate the blood brain barrier (BBB) to exert its pharmacological effects in rats. Therefore, this study aims to investigate the pharmacokinetics and cerebral cortex distribution of 6-HKA in MCAO mice, to determine whether the ability of 6-HKA to cross the BBB is contingent upon barrier integrity.
Methods
This study established and validated the ultra-performance liquid chromatography tandem mass spectrometric (UPLC-MS/MS) methods for the quantification of 6-HKA in mouse plasma and brain, and investigated the pharmacokinetics and cerebral cortex distribution of 6-HKA in MCAO mice and sham-operated mice.
Results
This study demonstrates that the mean residence time (MRT0−t) of 6-HKA in the MCAO group was significantly prolonged compared to the sham-operated group, and there was a higher distribution of 6-HKA in the ischemic cerebral cortex than in the contralateral normal cerebral cortex.
Conclusion
This study illustrated that in MCAO mice, the prolonged MRT0−t of 6-HKA and the increased permeability of BBB made the accumulation of 6-HKA in the ischemic cerebral cortex, thereby enabling 6-HKA to exert neurorestorative effects.
Introduction
Ischemic stroke typically results from the focal occlusion or narrowing of an artery in the brain. 1 It can lead to cellular bioenergetic failure, followed by a series of physiological and pathological events, including excitotoxicity, oxidative stress, inflammatory responses, ion channels damage, blood-brain barrier dysfunction, microvascular injury, and ultimately the loss of brain function, posing a serious health risk and economic burden to patients and their families.2–5
Ginkgo biloba leaf extract (EGb) has been used for treating pulmonary and hepatic symptoms since ancient China.6,7 Recently, attention has shifted to its neuroprotective properties. Significant efforts have been devoted to elucidating the neuroprotective bio-functions of its primary bioactive constituents, including terpene trilactones (ginkgolides A, B, and C, as well as bilobalide) and flavonoids.8–10 However, while EGb has been shown to be effective in preclinical experimental stroke models, the results of clinical studies have heterogeneous.11–13 One possible explanation for this variability is the diverse compositions of EGb, as most extraction methods focus on terpene trilactones and flavonoids, while organic acids are often overlooked.
One important organic acid found in EGb is 6-hydroxykynurenic acid (6-HKA, Figure 1), which has attracted increasing attention in recent years. 14 As reported, 6-HKA has been proved to be an antagonist of Alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors (AMPARs), a family of glutamate receptors. 15 The excessive release of glutamate and the subsequent overactivation of glutamate receptors cause excitotoxicity, which is considered the main mechanism of neuronal dysfunction and cell death in ischemic stroke. 16 Therefore, 6-HKA is expected to exert neuroprotective effects by competitively antagonizing AMPARs. Additionally, 6-HKA has demonstrated significant scavenging activity against authentic ONOO-, which is associated with oxidative stress. 17 Furthermore, a study has shown that 6-HKA can mitigate neurological impairments, reduce average infarct size, and decrease the levels of malondialdehyde and superoxide dismutase in rats with severe ischemia/reperfusion injury, further supporting the neurorestorative effect of 6-HKA in vivo. 18
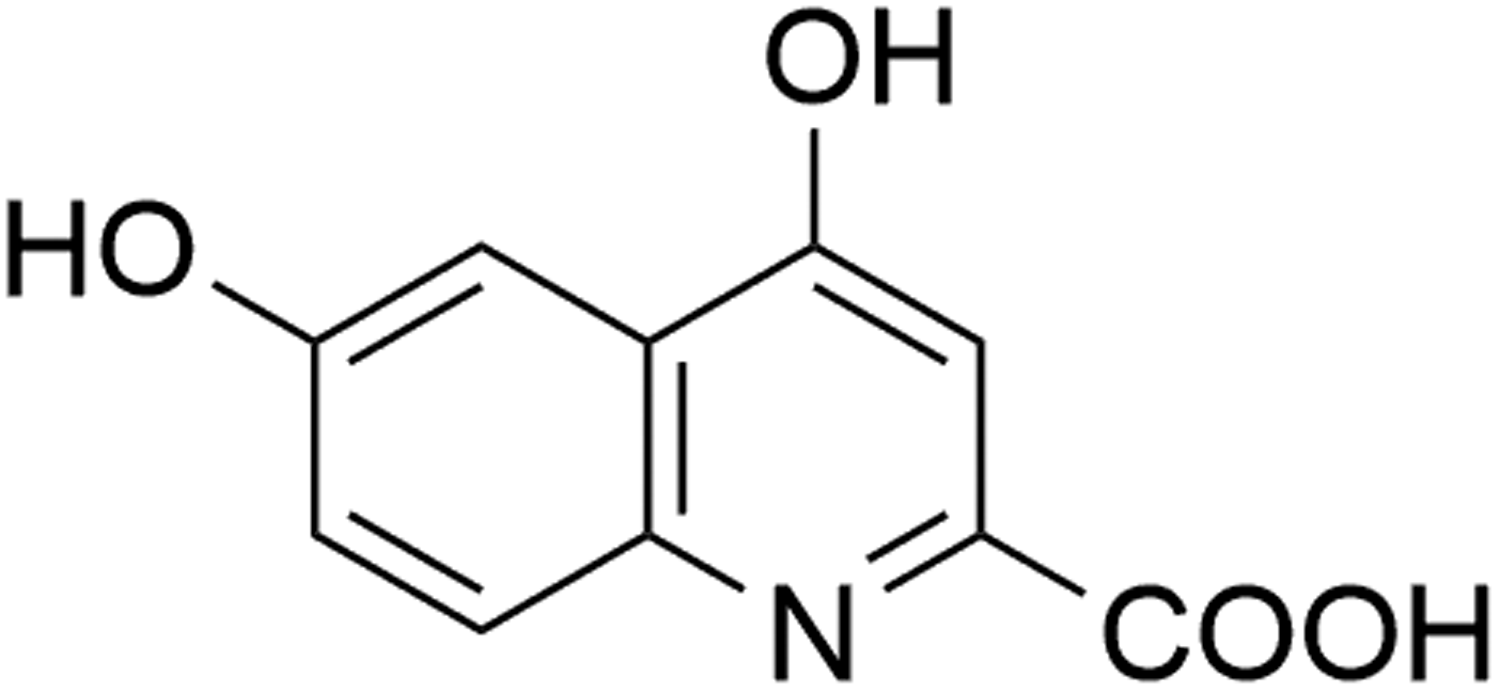
The structure of 6-HKA.
The efficacy of a drug is closely linked to its distribution within the target tissue. However, the blood-brain barrier (BBB) significantly hinders the entry of xenobiotic compounds into the brain, especially drugs with low lipophilicity. Unfortunately, 6-HKA is precisely such a low-lipophilicity compound. Our previous studies on the apparent permeability and brain distribution of 6-HKA showed that it had poor ability to penetrate the BBB, resulting in minimal distributions within the brains of healthy rats.19–21 Interestingly, the extremely low brain distribution of 6-HKA seems to be inconsistent with its good neurorestorative effect in vivo. Therefore, it is hypothesized that under conditions that the integrity of the BBB is compromised, such as focal cerebral ischemia induced by middle cerebral artery occlusion (MCAO) operation, the permeability of BBB will increase, which facilitates 6-HKA to entry into the brain and exert neurorestorative effects.
In this study, the established ultra-performance liquid chromatography tandem mass spectrometric (UPLC-MS/MS) methods for the quantification of 6-HKA in rat plasma and brain were re-validated to determine the concentration of 6-HKA in mouse plasma and brain, and the pharmacokinetics and cerebral cortex distribution of 6-HKA in MCAO mouse were investigated. This study discussed whether focal cerebral ischemia injury will increase the distribution of 6-HKA in ischemic cerebral cortex, which may provide a theoretical basis for further development of 6-HKA.
Materials and Methods
Reagent and Chemicals
6-HKA (>99% purity) was provided by Zhejiang Conba Pharmaceutical. Tolbutamide (>99% purity, the internal standard, IS) and alcohol were purchased from Aladdin. Tween-80 was purchased from Amresco. Pentobarbital sodium, dimethyl sulfoxide (DMSO), ammonium formate, sodium chloride and sodium bicarbonate were purchased from Sinopharm. HPLC-grade methanol was purchased from Merck.
Animals
C57BL/6 mice (8-10 weeks, male) were provided by Shanghai Linchang biotechnology Co., Ltd The mice were maintained under pathogen-free conditions and were housed with no more than five animals per cage. The experiments were performed according to the guidelines by the animal ethics committee of Zhejiang University (Ethics Code: ZJU20200147).
Solutions and Samples Preparation
Solutions and samples were prepared according to the previous study.18,21
Primary stock solutions of 6-HKA (0.2 mg/mL) and IS (5.0 mg/mL) were prepared in DMSO and stored at −20 °C. Working solution of 6-HKA was diluted with methanol/water (1:1, v/v), while IS working solution (300 ng/mL) was prepared using methanol. Quality control (QC) solutions were prepared by diluting another primary stock solution of 6-HKA with methanol/water (1:1, v/v).
For the 5.0 mg/mL dosage form, weighed an appropriate amount of 6-HKA and thoroughly grinded it in a mortar. Then, sequentially added Tween-80, DMSO, and saline while continuing to grind. Finally, added saturated sodium bicarbonate solution to adjust the pH and ensure complete dissolution of 6-HKA.
Brain homogenate was acquired by adding 30% methanol–water (v/v) to weighed brain tissue at a 6:1 (v/m) ratio and homogenizing with a tissue grinder (60 Hz, 2 min). 50 μL of plasma or brain homogenate was precipitated by IS working solution. The mixture was vortexed for 3 min and then centrifuged at 15,000 rpm and 4 °C for 15 min. After centrifugation, 100 μL of the supernatant was transferred to a new Eppendorf tube and mixed with 100 μL of water. Finally, 100 μL of this mixture was transferred to an HPLC vial for detection.
UPLC-MS/MS Method
The concentrations of 6-HKA were detected by an UPLC-MS/MS system, which comprised of Agilent 1290 Infinity Series liquid Chromatograph coupled to Agilent 6460 Series Triple Quadrupole mass spectrometer (Agilent, Santa Clara, CA, USA). Chromatographic separation was conducted on a ZORBAX Eclipse Plus C 18Rapid Resolution HD column (2.1 mm × 100 mm, 1.8 μm, Agilent). The mobile phase A and B were 2 mM ammonium formate in water and methanol, respectively. For plasma, the gradient elution was starting at 90% A for 2 min, decreasing linearly to 10% A at 2.6 min, holding 10% A until 3.6 min, returning to 90% A at 3.61 min, and re-equilibrating to 5 min. The flow rate was 0.25 mL/min, and the injection volume was 2 μL. For brain homogenate, the gradient was optimized by extending the holding time at 90% B from 3.6 to 3.9 min. The injection volume was increased to 3 μL.
Analytes were quantified under multiple-reaction monitoring (MRM) mode. The optimized mass spectrometric as follows: The ion mode was positive for 6-HKA and negative for IS. The transitions (precursor to product) monitored were m/z 206.1→160.0 for 6-HKA and 269.0→170.0 for IS, respectively. The collision energy and fragmentor voltage were 15 eV and 100 V for 6-HKA, 10 eV and 130 V for IS.
Validation of Analytical Method
The UPLC-MS/MS analysis methods for detecting 6-HKA in rat plasma and brain have been fully validated before.19,21 But in this study, the matrixes have been changed to mouse plasma and brain homogenate. Therefore, according to the “Bioanalytical Method Validation, Guidance for Industry” developed by the U.S. Food and Drug Administration's (FDA), partly validation is required, including specificity, calibration curve, intra-day accuracy and precision. 22
The specificity of the method was evaluated by comparing chromatograms of blank plasma or brain homogenate from six different sources, blank plasma or brain homogenate spiked with 6-HKA and IS, and plasma or brain homogenate obtained from mice after administration of 6-HKA, to investigate the potential interferences at the retention times of 6-HKA and IS.
The calibration curve was constructed by plotting the peak area ratio (y) of 6-HKA to IS against the nominal concentration of 6-HKA (x), using a weighted 1/x² linear regression. Calibration samples were prepared by spiking 47.5 μL blank plasma or brain homogenate with 2.5 μL working solution of 6-HKA.
The intra-day accuracy and precision were evaluated by the determination of QC samples in five replicates for each concentration.
Drug Administration and MCAO Mouse Model Preparation
To address the contradiction mentioned above, the drug dosage and administration route were determined based on Yao's pharmacodynamic study of 6-HKA. 18 In addition, considering the lower limit of quantification (LLOQ) of 6-HKA in brain tissue, the dosage was adjusted upwards in line with its solubility to ensure adequate concentrations. In the experiment, eighty mice received intragastric administration of 50 mg/kg of 6-HKA per day for three consecutive days before surgery. On fourth day, all the mice were randomly divided into two groups: MCAO group and sham operation group. Two groups were anesthetized and operated immediately after intragastric administration of 50 mg/kg of 6-HKA, respectively.
MCAO operation was carried out as described before.23,24 In brief, mice were anesthetized by intraperitoneal injection of with 1% pentobarbital (60 mg/kg). Cerebral blood flow (CBF) was monitored in the middle cerebral artery (MCA) by laser Doppler flowmetry, with a probe attached to the skull over the cortex supplied by the right MCA (2-mm caudal to bregma and 6-mm lateral to midline). The 6/0 nylon suture with rounded tip was inserted into the internal carotid to occlude the origin of the MCA. A reduction of more than 80% of CBF should be observed to ensure successful occlusion of the MCA. The sham operation was carried out the same way, without inserting the 6/0 nylon suture. During the surgery, mice were warmed using a heat mat with thermostatic control, and the rectal temperature was maintained at 37 ± 0.5 °C. MCAO operation in our study can lead to focal cerebral ischemia in the right cerebral cortex. 23
Blood samples and cerebral cortex of both left and right brains were collected at 20 min, 1 h, 6 h, and 24 h (N = 10 per time point of each group) after 6-HKA administration. Plasma was obtained by centrifuging blood samples at 5000 rpm for 10 min, and then stored at −80 °C until analysis. Cerebral cortex was washed by saline, and stored at −80 °C until analysis.
Statistical Analysis
Pharmacokinetic parameters were calculated by non-compartmental analysis (NCA) in WinNonLin pharmacokinetic software. Data was presented in mean ± SEM. Wilcoxon matched-pairs signed-rank test was used to evaluate differences between the ischemic cerebral cortex and the contralateral normal cerebral cortex in the MCAO group, including brain concentrations and brain-to-blood ratios. Mann-Whitney test was used to evaluate the differences between the MCAO group and the sham-operated group, including plasma concentration, distribution in the right cerebral cortex and the brain-to-blood ratio of the right cerebral cortex. P < 0.05 was considered statistically significant.
Results
Validation of Analytical Method
The UPLC-MS/MS analysis methods for detecting 6-HKA in rat plasma and brain have been fully validated before.19,21 But in this study, the matrixes have been changed to mouse plasma and brain homogenate. Therefore, according to the guidance developed by FDA, 22 the specificity, calibration curve, intra-day accuracy and precision of the UPLC/MS-MS methods for assaying 6-HKA in mouse plasma and brain homogenate were validated. The typical chromatograms of blank matrix (mouse plasma or brain), blank matrix spiked with 6-HKA and IS, mouse plasma or brain samples from mice after administration of 6-HKA (Figure 2 and Figure 3) showed that no interfering peak was observed at the retention time of 6-HKA or IS, suggesting that the methods were specific for the analyte determination. The typical calibration curves and linear ranges of 6-HKA in mouse plasma and brain were presented in Table 1. The linear range for plasma and brain were 2.5–250 ng/mL and 1.0–100 ng/mL. A summary of intra-day precision and accuracy was presented in Table 2. The intra-day accuracy and precision were evaluated by the determination of quality control samples in five replicates for each concentration. The intra-day accuracy of 6-HKA ranged from −6.8% to 13.7% with the precision < 10.0%, which were within the acceptance criteria of FDA. Those results suggesting that those methods were suitable and reliable for assaying 6-HKA in mouse plasma and brain. Therefore, we further applied those methods for the quantification of 6-HKA in mice that received the MCAO or sham operation.

Typical chromatograms of mouse blank plasma (A, E), mouse blank plasma spiked with 6-HKA (2.5 ng/mL, LLOQ) (B, retention time: 2.617 min) and IS (F, retention time: 4.555 min), mouse blank plasma spiked with 6-HKA (200 ng/mL) (C, retention time: 2.593 min) and IS (G, retention time: 4.558 min), mouse plasma sample from mice after administration of 6-HKA (D, 6-HKA retention time: 2.721 min; H, IS retention time: 4.531 min).

Typical chromatograms of mouse blank brain (A, E), mouse blank brain spiked with 6-HKA (1.0 ng/mL, LLOQ) (B, retention time: 2.556 min) and IS (F, retention time: 4.133 min), mouse blank brain spiked with 6-HKA (80 ng/mL) (C, retention time: 2.547 min) and IS (G, retention time: 4.125 min), mouse brain sample from mice after administration of 6-HKA (D, 6-HKA retention time: 2.560 min; H, IS retention time: 4.120 min).
Typical Calibration Curves and Linear Ranges of 6-HKA in Plasma and Brain of Mice.

Intra-day Accuracy and Precision for the Determination of 6-HKA in Plasma and Brain of Mice.

Cerebral Ischemia Increased the Distribution and Residence Time of 6-HKA
Before the formal experiment, we investigated the plasma concentration and cerebral cortex distribution of 6-HKA in MCAO mice and sham-operated mice at 5 min, 15 min, 30 min, 1 h, 2 h, 4 h, 6 h, 10 h and 24 h after 6-HKA administration through a pre-experiment. The results (Figure S1) showed that the time to reach the maximum concentration (Tmax) of 6-HKA of the MCAO group and the sham-operated group were 15 min and 1 h, respectively (Figure S1 (A)).
In combination with the pre-experiment results, taking into account the time required for the surgery (15-20 min), we selected 20 min (near the Tmax of MCAO group) and 1 h (the Tmax of sham-operated group) as the sampling time points in the formal experiment. In addition, as reported, the significant change of brain permeability was usually observed at 6 h after the onset of ischemic stroke,25–27 so 6 h was selected as another sampling time point. At 24 h, 6-HKA was undetectable in both groups (Figure S1 (B)), so 24 h was chosen as the final sampling time point. Therefore, 20 min, 1 h, 6 h and 24 h were finally selected as the observation time points for the formal experiment. Considering that the experiment accuracy in the intra luminal suture model was easily compromised with the temperature during surgery, surgical precision, body weight and so on,28,29 ten mice were arranged in each group at each time point.
In the formal experiment, we determined the concentrations of 6-HKA in plasma and bilateral cerebral cortex of mice at 20 min, 1 h, 6 h and 24 h after 6-HKA administration on the operation day, the concentrations of plasma and brain and the pharmacokinetic parameters and were shown in Figure 4 and Table 3.

Plasma concentration and cerebral cortex distribution and of 6-HKA in the MCAO group and the sham-operated group after 50 mg/kg oral administration of 6-HKA. (A) The plasma concentration of 6-HKA. (B) The distribution of 6-HKA in cerebral cortex. (C) The ratio of brain-to-blood of 6-HKA in cerebral cortex. *P < 0.05 ***P < 0.001. N was ranged from 6 to 10 at each time point of each group since some mice died during the experiment. Data was expressed as Mean ± SEM.
Pharmacokinetic Parameters of 6-HKA in the MCAO Group and the Sham-Operated Group Following Oral Administration of 6-HKA (50 mg/kg) (N = 10).

*Cmax, the maximum plasma concentration; Tmax, time to Cmax; AUC0−t, area under the time-concentration curve from t = 0 to last measurable concentration; MRT0−t, mean residence time from t = 0 to last measurable concentration.
According to Figure 4 (A), the mean plasma concentration of the MCAO group didn’t differ significantly from the sham-operated group at 20 min and 6 h after 6-HKA administration, but it was lower than that of the sham-operated group at 1 h after administration, while the opposite was observed at 24 h after administration. The pharmacokinetic parameters of 6-HKA listed in Table 3 also summarized the characteristics of the 6-HKA plasma concentration-time profile. The Tmax of 6-HKA was different between the two groups, being 20 min and 1.0 h, respectively. Although the mean Cmax of the MCAO group was slightly lower than that of the sham-operated group, and the mean AUC0−t of the MCAO group was slightly higher than that of the sham-operated group, but these differences were not statistically significant. However, the MRT0−t of the MCAO group was 2.4-fold higher than that of the sham-operated group.
As shown in Figure 4 (B) and (C), with changes in plasma concentration of 6-HKA, the mean concentration of 6-HKA in the right cerebral cortex (the ischemic cerebral cortex) of the MCAO group was significantly lower and higher than that of the sham-operated group at 1 h and 24 h after 6-HKA administration, respectively. However, the mean concentration of 6-HKA in the ischemic cerebral cortex of the MCAO group showed no significant difference from that in its contralateral cerebral cortex, and there was no significant difference in the mean brain-to-blood ratios of 6-HKA in the right cerebral cortex between the two groups.
In addition, there was no significant difference in cerebral cortex distribution or ratio of brain-to-blood of 6-HKA between the left and right cerebral cortex within each group at 20 min, 1 h and 24 h after 6-HKA administration. However, at 6 h after 6-HKA administration, the mean concentration and the mean ratio of brain-to-blood of 6-HKA in the ischemic cerebral cortex of the MCAO group were significantly higher than those in the contralateral normal cerebral cortex of the MCAO group, while there was no significant difference in the mean concentration and the mean ratio of brain-to-blood of 6-HKA between the left and right cerebral cortex of the sham-operated group.
Discussion
BBB is a crucial physiological structure composed of brain capillary endothelial cells linked by tight junctions, alongside active efflux transporters and the major facilitator superfamily domain-containing protein 2A (MFSD2A). This barrier meticulously regulates the brain's microenvironment and modulates neural activity by selectively controlling the transport of substances into and out of the cerebral tissue. However, while the BBB is essential for maintaining brain homeostasis and protecting the central nervous system from harmful agents, it also significantly impedes the delivery of therapeutic drugs to the brain, posing a substantial challenge for the treatment of neurological disorders. Strategies such as temporarily opening tight junctions, inhibiting efflux transporter activity, and enhancing transcytosis, show great promise for increasing BBB permeability. 30 It is worth noting that BBB permeability can also be altered under certain pathological conditions, potentially impacting drug delivery and therapeutic outcomes.
In our previous studies, the apparent permeability coefficients (Papps) of 6-HKA in human brain microvascular endothelial cells (HBMECs) and Madin-Darby canine kidney cells transfected with the human MDR1 gene (MDCK-MDR1) was low, and the brain distribution of 6-HKA in healthy rats after 20 mg/kg administration was hard to be detected, indicating that 6-HKA was hard to exert neuroprotective effect in healthy animals.19–21 However, the neuroprotective effect of 6-HKA has been proved in vivo recently. 18 Since drug efficacy is closely related to its distribution of the action target, the extremely low distribution of 6-HKA in brain seems to be inconsistent with the good neurorestorative effect of 6-HKA in vivo. Therefore, this study aimed to investigate the pharmacokinetic basis of the neuroprotective properties of 6-HKA under MCAO pathological conditions.
To analyse plasma and brain homogenate samples of mice, we partly verified the UPLC/MS-MS methods for assaying 6-HKA we developed before, and proved that the methods were still sensitive, specific and stable after changing the matrix from rat to mouse. The methods were suitable for the study of cerebral cortex distribution, as the concentrations of 6-HKA in both plasma and cerebral cortex samples were generally higher than the LLOQ.
The ischemic symptoms induced by the MCAO operation in our study were relatively mild, as only the right cervical cerebral artery was occluded. Additionally, CBF was maintained through the ipsilateral external carotid artery and the circle of Willis.24,31 Within 24 h post-operation, seventy-two experimental mice survived, with no fewer than six mice in each subgroup. The high modeling success rate ensured that each subgroup had a sufficient number of samples, thereby making the data more reliable.
The formal experimental results showed that 20 min after 6-HKA administration, there were no significant differences in plasma concentration, cerebral cortex distribution or ratio of brain-to-blood between the two groups or between the left and right cerebral cortex. According to the experimental method, it can be inferred that the right cerebral cortex had not experienced ischemia and the BBB was intact before the operation was completed, leaving comparable physiological conditions between the two groups at that time. At 1 h after 6-HKA administration, unlike the sham-operated group, the plasma concentration of 6-HKA in the MCAO group did not continue to rise. As reported, ischemic stress can lead to the release of vasoactive substances, which may induce gastrointestinal mucosal ischemia, hypoxia and reperfusion injury, and further influence the peristalsis of the gastrointestinal tract by lowering its contraction and transmission ability, resulting in gastrointestinal motility disorders. 31 Based on this, it is hypothesized that gastrointestinal mucosal injury may contribute to a reduced absorption rate of 6-HKA in the MCAO group, potentially explaining the observed differences in Tmax and Cmax.
In addition, the significantly higher MRT₀₋ₜ in the MCAO group suggests prolonged systemic exposure to the drug, likely due to altered clearance or distribution under ischemic conditions, potentially enhancing its ability to exert therapeutic effects. However, this increase in MRT₀₋ₜ was not accompanied by a significant change in AUC₀₋ₜ, indicating that the total drug exposure over time remained relatively consistent between groups. Due to practical constraints, the sampling time points in the formal experiment were relatively sparse, which may affect the accuracy of pharmacokinetic parameter predictions, despite the selection of these time points being based on pre-experiment. Therefore, whether MCAO operation affects the pharmacokinetic characteristics of 6-HKA, still requires further research with more intensive sampling time points. Considering that only 1–2 blood samples can be collected from each mouse, using MCAO rats for pharmacokinetic studies may be a better choice, although species differences may exist.
According to the literature, changes in the BBB following ischemic stroke occur as a gradual and progressive process. Various cellular and molecular mechanisms regulate BBB permeability after ischemia stroke. These include the involvement of oxidative and nitrosative stress, cytokines, chemokines, matrix metalloproteinases, adhesion molecules and vascular endothelial growth factor. Additionally, the roles of different cell types, such as endothelial cells, astrocytes, and microglia, are critical in modulating BBB permeability during ischemic stroke. 32 As reported, the significant change of brain permeability was usually observed at 6 h after the onset of ischemic stroke.25–27 As cerebral ischemia progresses, pathological changes may also occur in the brain areas remote from the initial ischemic regions, including Evans blue extravasation, degenerated pericytes displaying mitochondria with cristae disruption, vacuolated endothelial cells containing large autophagosomes, degenerated astrocytes and perivascular edema, and appearance of parenchymal astrogliosis. 33 The changes in the distribution and brain-to-blood ratio of 6-HKA in cerebral cortex observed in this experiment also corroborated previous research findings. At 1 h after 6-HKA administration, the content of 6-HKA entering the cerebral cortex of the two groups was closely related to the plasma concentration of 6-HKA, and there was no significant difference in the mean concentration of 6-HKA between the left and right cerebral cortex of the MCAO group, which was consistent with that the BBB has not been disrupted in the initial stages of cerebral ischemia. 25 At 6 h after 6-HKA administration, with the increase in the permeability of the BBB, 6-HKA could enter the ischemic cerebral cortex more easily, leading to an increase in both the distribution and the ratio of brain-to-blood of 6-HKA in the ischemic cerebral cortex. At 24 h after 6-HKA administration, there was no significant difference in the distribution of 6-HKA between the left and right cerebral cortex in the MCAO group, indicating compromised BBB integrity in remote brain areas. 33
Despite the significant findings of this study, several limitations must be acknowledged. Firstly, slight differences in the duration of the surgery and the individual variability among the mice could both impact the results. Secondly, variability could arise from differences in anesthesia injection, surgical procedures, and post-surgical care, especially since anesthesia can reduce, delay, or alter drug absorption through its indirect effects on gastric motility and emptying. 34 Although we eliminated this interference by setting up a sham-operated group, we cannot simulate the pharmacokinetics and brain tissue distribution of 6-HKA in real stroke situations (without anesthesia impact). Therefore, further experiments are needed to avoid this impact in the future. In addition, although we have previously studied the distribution of 6-HKA in brain tissue of healthy rats, adding a healthy mice group as another control group may strengthen the analysis by providing a baseline comparison of 6-HKA pharmacokinetics.
In previous study, we found that 6-HKA may be a substrate for organic anion transporter 3 (OAT3) but not for organic cation transporter 2 (OCT2), organic cation/carnitine transporter 2 (OCTN2), or multidrug resistance protein 1 (MDR1). 20 Since OAT3 is primarily expressed on the apical side rather than the basolateral side of the BBB, we speculate that MCAO might increase the BBB permeability of 6-HKA by disrupting the tight junctions of brain endothelial cells. However, further investigation is still needed to determine whether endocytosis in cerebral endothelial cells changes following the procedure of MCAO, as well as to uncover the deeper mechanisms underlying the increased permeability of 6-HKA.
Ischemic injury to the cerebral cortex leads to neuropathological changes such as brain edema, neuroinflammation, and neural cell death, ultimately resulting in severe neurological deficits. Research has advanced our understanding of the pathogenesis and mechanisms of stroke, which include cellular excitotoxicity, mitochondrial dysfunction, neuroinflammation, BBB impairment, and cell death. 35 Various signaling pathways are activated during these pathological processes, and targeting these pathways could offer promising neurorestorative effects. AMPARs are a family of glutamate receptors, which can mediate neurotoxicity when excessively stimulated. 16 As reported, 6-HKA exhibited a high affinity to AMPARs in CA1 hippocampal pyramidal cells, indicating its potential neurorestorative effects. 15 The deeper mechanisms underlying the neuroprotective effects of 6-HKA remain to be further investigated.
All in all, from this study, it is evident that although our previous research found that 6-HKA had difficulty penetrating the BBB of healthy rats, it can more easily penetrate the compromised BBB following ischemia. In addition, 6-HKA also exhibits a longer MRT0−t in the MCAO mice models, which contributes to its accumulation and pharmacological effects in the ischemic cerebral cortex. Furthermore, the differences in pharmacokinetic characteristics between disease and health models also provide insights for pharmacokinetic research. When necessary, specific disease models should be selected to more accurately reflect the pharmacokinetic characteristics of drugs.
Conclusion
In this study, we established and validated the sensitive and specific UPLC-MS/MS methods for the quantification of 6-HKA in mouse plasma and brain, and investigated the pharmacokinetics and the cerebral cortex distribution characteristics of 6-HKA in MCAO and sham-operated mice. The results demonstrated that the MRT0−t of 6-HKA in the MCAO group was longer than that in the sham-operated group, and the distribution of 6-HKA in ischemic cerebral was more than that in the normal cerebral, indicating that the compromised BBB in MCAO mice could allow 6-HKA to enter the brain despite its low lipophilicity. These findings suggest that when the BBB is compromised, the penetration of 6-HKA and other low lipophilicity drugs may become easier, highlighting their potential use in treating brain disorders. In conclusion, this study provides valuable insights for the advancement of 6-HKA and the development of similar compounds, emphasizing the potential significance of BBB permeability in therapeutic strategies targeting neurological disorders.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251315082 - Supplemental material for Cerebral Ischemia Increases the Distribution and Residence Time of 6-Hydroxykynurenic Acid in the Ischemic Cerebral Cortex of Mice
Supplemental material, sj-docx-1-npx-10.1177_1934578X251315082 for Cerebral Ischemia Increases the Distribution and Residence Time of 6-Hydroxykynurenic Acid in the Ischemic Cerebral Cortex of Mice by Zhuowei Shen, Yanhong Chen, Kui Zeng, Haihong Hu, Jie Pan, Mingcheng Xu, Kaifeng He, Fengting Ou, Lushan Yu, Su Zeng and Lu Chen in Natural Product Communications
Footnotes
Acknowledgments
We thank Dr Xiangnan Zhang (College of Pharmaceutical Science, Zhejiang University) for helping to prepare MCAO mice models.
Author Contributions
In this study, Lu Chen, Su Zeng and Lushan Yu formulated the idea of the paper and supervised the research. Zhuowei Shen performed the research, collected data and drew the figures. Kui Zeng, Haihong Hu, Jie Pan, Mingcheng Xu, Kaifeng He and Fengting Ou performed the experiments. Zhuowei Shen and Yanhong Chen wrote the paper. Su Zeng and Lu Chen revised the paper. All authors reviewed the manuscript.
Data Availability
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Key Research and Development Program of China (2017YFC0908600) and the Zhejiang Provincial Medical and Health Technology Project (2022485016).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
