Abstract
Aims
Acute alcohol intoxication (AAI) caused by the intake of excessive alcohol is a harmful clinical condition and potentially life threatening. Therefore, there has been increasing attention on the development of anti-alcoholism products. In this study, we evaluated the therapeutic benefits of an anti-alcoholism product (Tengcha–Gegen Plant Solid Beverage, which contains Ampelopsis grossedentata, Pueraria Lobata, Hovenia dulcis Thunb, and turmeric) in an AAI mouse model.
Methods
The AAI mouse model was established through alcohol gavage. Male Kunming mice were first given three doses of Tengcha–Gegen Plant Solid Beverage for 30 min and then gavaged with 52% (V/V) liquor (0.15 mL liquor/10 g body weight). The impact of Tengcha–Gegen Plant Solid Beverage on AAI was evaluated by the sober-up time, the plasma alcohol content, and the plasma and liver superoxide dismutase (SOD), glutathione (GSH) and malondialdehyde (MDA) contents or activities.
Results
The Tengcha–Gegen Plant Solid Beverage could shorten the sober-up time and decrease plasma alcohol levels at 0.2 and 0.4 g/kg body weight. The mechanistic studies showed that Tengcha–Gegen Plant Solid Beverage increased the plasma SOD and liver GSH levels but decreased the plasma MDA levels at the indicated doses.
Conclusions
This study confirmed that Tengcha–Gegen Plant Solid Beverage might have anti-alcoholism therapeutic potential and protect the liver against high oxidative stress induced by acute alcohol exposure in AAI mice.
Short Summary
In this study, we showed that the Tengcha–Gegen Plant Solid Beverage at 0.2 and 0.4 g/kg body weight could shorten the sober-up time and plasma alcohol level, increase the plasma SOD level and liver GSH, but decrease the plasma MDA level. This indicates that the Tengcha–Gegen Plant Solid Beverage might have a specific anti-alcoholism effect in the acute alcohol intoxication mouse model.
Keywords
Introduction
With the continuous development of society, there has been a rapid increase in the demand for alcoholic beverages. However, this surge in alcohol consumption has led to increasingly severe health and social problems. 1 According to WHO statistics, excessive drinking leads to 3 million deaths per year and 7.2% of premature deaths in people under 69 years old.2,3 Acute alcohol intoxication (AAI) caused by the intake of excessive alcohol is a harmful clinical condition that is potentially life threatening. Heavy one-time drinking causes AAI, which includes acidosis, heart failure, respiratory depression, cardiac arrest, and even death as a result of autonomic nerve and brain dysfunction.4,5 Long-term drinking is related to liver injury, cancer, and diseases of central nervous system, heart, endocrine system, and other organs. 6 With the increasing alcohol consumption and the attention on a healthy lifestyle, there is increasing attention to developing anti-alcoholism products.
China has an enduring tradition of a drinking culture. 7 Traditional Chinese medicine (TCM) has a long history of preventing and treating alcoholism and hangovers. Some traditional unilateral anti-alcoholism drugs, such as Radix puerariae and Radix angelicae dahuricae, Gehua jieke decoction, Zhigeyin, and Wuling powder, have shown specific anti-alcoholism effects. In addition, some natural foods, such as ginseng, mung bean, radishes, and dandelion, are used to treat and relieve alcoholism. 8 Because of the wide range of pharmacological activity and low toxicity, the development of anti-alcoholism products based on TCM has advantages.
In this study, we evaluated the anti-alcoholism impact of Tengcha–Gegen Plant Solid Beverage (hereafter referred to as the Tengcha–Gegen product, which contains Ampelopsis grossedentata, Pueraria Lobata, Hovenia dulcis Thunb, and turmeric) on AAI mice by investigating the sober-up time, the plasma levels of alcohol, and the plasma and liver contents/activities of superoxide dismutase (SOD), glutathione (GSH), and malondialdehyde (MDA).
Materials and Methods
Drugs and Reagents
The experimental Ping An Tsumura brand Tengcha–Gegen Plant Solid Beverage (standard number GB/T29602) product was supplied by China Medico Corporation (Tianjin, China). Haiwang brand Jinzun tablets (standard number Q/JLHW0040S, Neptune Health Technology Group Co., Ltd, Shenzhen, China) were from the drugstore. The 52% (V/V) Niulanshan Erguotou liquor (standard number GB/T20822, Beijing Niulanshan Erguotou Group Co., Ltd, Beijing, China) was purchased from a supermarket. Disposable EDTA anticoagulant blood collection tubes (367861) were purchased from BD Company (USA). The plasma alcohol content detection kit (E036-1-1) was obtained from Nanjing Jiancheng Biotechnology Company (Nanjing, China). The plasma SOD detection kit was purchased from Beijing Zhongsheng Beikong Biotechnology Company (Beijing, China). Commercial kits for detecting SOD (S0101S, Beyotime Biotechnology, Inc., China) in the liver, GSH (#BC1175, Beijing Solarbio Science & Technology Co., Ltd, China) in the liver and plasma, and MDA (#BC0025, Beijing Solarbio Science & Technology Co., Ltd) in the liver and plasma were used. The BCA protein concentration detection kit (A55866) was purchased from PierceTM, Thermo Scientific, Inc. (Waltham, MA, USA).
AAI Mice Model
All animal care and experiments in this study were approved and carried out according to the guidelines on animal rights and treatment of the Animal Care and Use Committee of the Institute of Medicinal Biotechnology, Chinese Academy of Medical Sciences (Beijing, China, Approval Number: IMB-20220320D2). Specific pathogen-free (SPF) male Kunming (KM) mice weighing approximately 25 g were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd (Beijing, China). All mice were housed in an SPF environment with free access to food and water at 23 °C with a 12/12 h light/dark cycle.
Then, we established the AAI mice model using gavage. 9 The mice were randomly divided into six groups, including the negative control group (control group, n = 5), model group (n = 10), low-dose Tengcha–Gegen product group (low-dose group, n = 10), medium-dose Tengcha–Gegen product group (medium-dose group, n = 10), high-dose Tengcha–Gegen product group (high-dose group, n = 10), and positive drug Haiwang brand Jinzun tablets group (positive group, n = 10). For the positive group, the tablets were dissolved in 0.9% normal saline and given to KM mice at 0.4 g/kg body weight. For the low-dose, medium-dose, and high-dose groups, Tengcha–Gegen products were dissolved in 0.9% normal saline and administered to KM mice at 0.2, 0.4, and 0.6 g/kg body weight, respectively. All mice were fasted overnight. Each group was administered it once via gavage, and the control group and model group were administered an equal volume of 0.9% normal saline. After 30 min, mice in the model, low-dose, medium-dose, high-dose, and positive groups were administered 52% (V/V) Niulanshan Erguotou wine via gavage at 0.15 mL/10 g body weight, and mice in the control group were administered 0.9% normal saline via gavage at 0.15 mL/10 g body weight. After 8 h of alcohol administration, the mice were anesthetized with isoflurane. Blood was collected from the fundus vein using an anticoagulant blood collection tube. The plasma was separated via centrifugation at 3000 rpm for 15 min by centrifuge (LD5-2A, Jingli Centrifuge Co., Ltd, Beijing, China) and then stored at −80 °C. The livers from each group of mice were quickly collected, immediately frozen using liquid nitrogen, and then stored at −80 °C.
Evaluation of the Alcohol-Induced Loss of the Righting Reflex
After acute alcohol exposure, the number of drunken and dead mice were closely observed and recorded. The time points of alcohol-induced loss and recovery of the righting reflex in all mice were recorded, and the sober-up times were calculated.
Detection of Plasma Alcohol Content
The plasma alcohol concentration was determined according to the instructions of the plasma alcohol content detection kit using a Multilabel Reader (EnVision 2105, PerkinElmer, Fremont, CA, USA).
Contents/Activities of SOD, GSH, and MDA in Plasma
The plasma levels of SOD were detected using the commercial test kits according to the instructions on an automatic biochemical analyzer (7100, HITACHI, Japan). The contents/activities of GSH and MDA were detected using the commercial test kits according to the instructions on a Multilabel Reader (EnVision 2105, PerkinElmer, Fremont, CA, USA).
Contents/Activities of SOD, GSH, and MDA in the Liver
The contents/activities of SOD, GSH, and MDA were detected using commercial test kits on a Multilabel Reader (EnVision 2105, PerkinElmer, Fremont, CA, USA) according to the instructions. Briefly, the livers in all groups of mice were homogenated using the lysis buffer from the commercial test kits, and the protein levels were detected using a PierceTM BCA protein assay. The contents/activities of SOD, GSH, and MDA in the liver were normalized to the protein concentration.
Statistical Analysis
The data are expressed as the means ± SEMs and plotted using GraphPad 8.0. One-way ANOVA analysis was used to compare the differences in three or more of the three groups, where *P < 0.05, **P < 0.01, and ***P < 0.001.
Results
Effect of the Tengcha–Gegen Product on AAI
The drunken states of the mice in the model and administration groups were recorded, and the non-intoxicated mice and dead mice in each group were removed. The sober-up time in AAI mice was calculated. As shown in Figure 1, compared with the model group gavaged with 52% (V/V) liquor, the low-dose and medium-dose groups with the Tengcha–Gegen product had significantly shortened sober-up times (P < 0.01), and the high-dose group also had a shortened sober-up time but without a significant difference (P = 0.071). In addition, there were no significant differences in the sober-up time between the positive (Haiwang brand Jinzun tablets group) and model groups (Figure 1). The above results revealed that the Tengcha–Gegen product at low and medium doses significantly shortened the sober-up time and showed a good anti-alcohol intoxication effect.
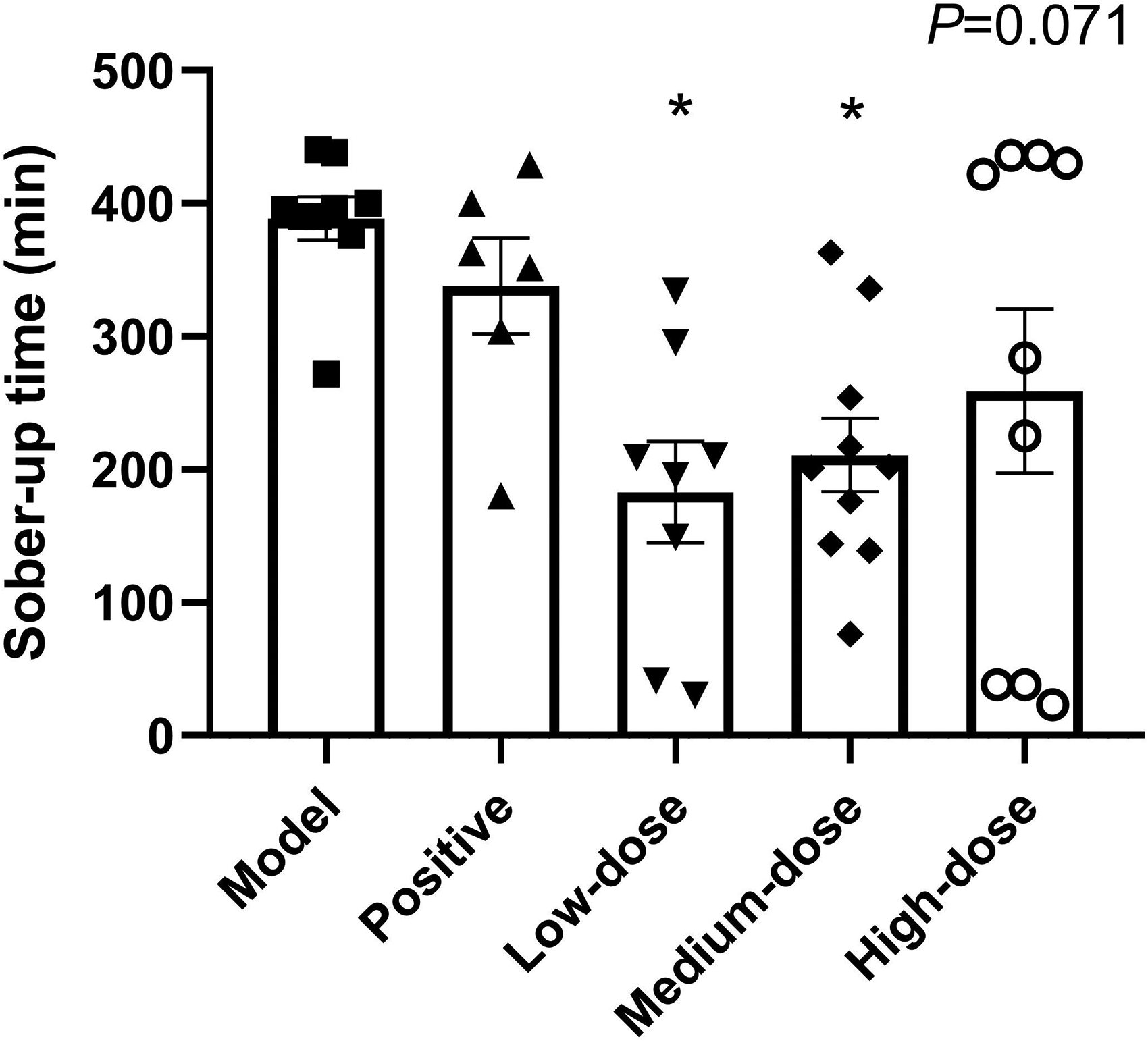
Sober-up times with the Tengcha–Gegen product in acute alcohol intoxication (AAI) mice. The data are expressed as the means ± SEMs. One-way ANOVA was used for analysis. **P < 0.01 versus model group. The P values between 0.05 and 0.20 are labeled.
Effect of the Tengcha–Gegen Product on Plasma Alcohol Level
Ethanol enters the digestive tract through the mouth, and most is absorbed by the gastrointestinal tract, which increases the concentration of alcohol (ethanol) in the blood. 10 To further examine the effect of the Tengcha–Gegen product on blood alcohol content, we determined the plasma alcohol levels. As shown in Figure 2, compared with the control group (6.30 ± 1.71 mmol/L), the plasma alcohol concentration in the model group (42.15 ± 1.62 mmol/L) significantly increased (P < 0.001), indicating that the alcohol concentration in the plasma of the mice significantly increased after a single gavage. Compared with the model group, the plasma alcohol concentrations in the mice in the positive group (33.01 ± 3.50 mmol/L, P < 0.05), low-dose group (30.59 ± 1.75 mmol/L, P < 0.01), medium-dose group (31.93 ± 1.35 mmol/L, P < 0.01), and high-dose group (32.69 ± 2.28 mmol/L, P < 0.05) significantly decreased (Figure 2), indicating that both Tengcha–Gegen products and the positive drug Haiwang brand Jinzun tablets could significantly decrease the plasma alcohol concentrations and have an anti-alcoholism effect in AAI mice.
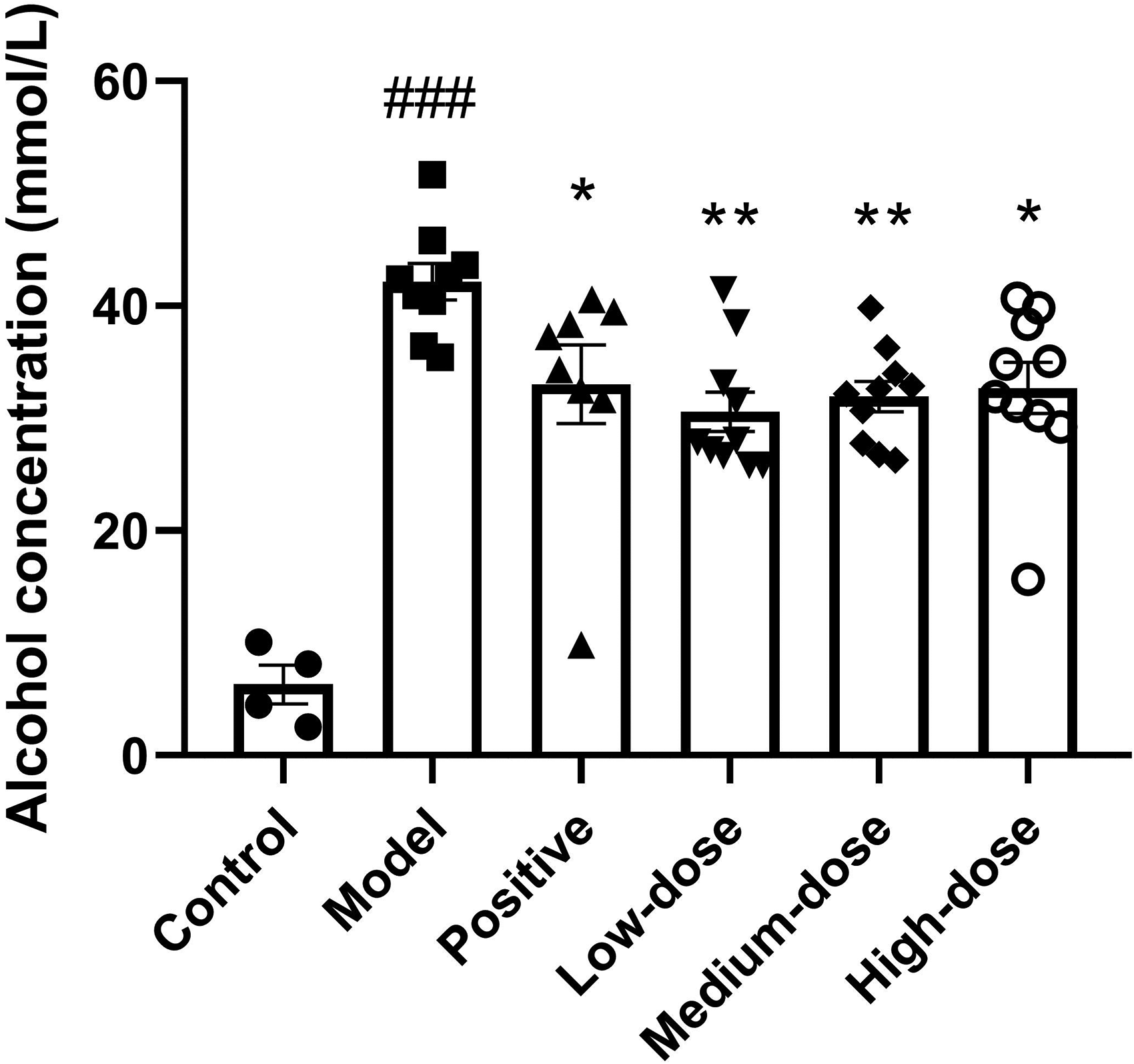
The effect of the Tengcha–Gegen product on the plasma alcohol concentration in AAI mice. The data are expressed as the means ± SEMs. One-way ANOVA was used for analysis. ###P < 0.001 versus control group. *P < 0.05, **P < 0.01.
Effect of the Tengcha–Gegen Product on Acute Alcohol-Induced Oxidative Stress
Reactive oxygen species (ROS) produced via ethanol metabolism are considered one of the leading causes of alcoholic liver injury by producing oxidation stress, and ethanol or its metabolites might decrease the antioxidant levels in the body. 11 SOD is an important antioxidant enzyme that can remove ROS through its antioxidant activity, and thus it plays a protective role in the liver. 12 GSH serves as a powerful antioxidant and has a crucial role in the detoxification of alcohol and many other toxic substances in the body.13,14 MDA, one of the leading products of liver lipid peroxidation, mainly reflects the overall oxidation level and can lead to extensive cellular death. 15 Therefore, the effects of the Tengcha–Gegen product on SOD, GSH, and MDA were examined in the plasma and liver of each group of mice.
As shown in Figure 3, the plasma SOD (P < 0.001) and plasma GSH (P < 0.01) levels in the model group were significantly lower than in the control group. By contrast, plasma MDA (P < 0.05) and liver MDA (P = 0.068) levels increased after acute alcohol exposure. Compared with the model group, the Tengcha–Gegen product increased the plasma SOD activity (P < 0.001 for the low-dose group, P < 0.05 for the medium-dose group, and P = 0.064 for the high-dose group), and the Tengcha–Gegen product at the three doses significantly decreased the plasma MDA levels (P < 0.01 for the low-dose group and medium-dose group, and P < 0.001 for the high-dose group) but not liver MDA levels. In addition, the Tengcha–Gegen product at medium and high doses significantly increased liver GSH levels (P < 0.05) but not plasma GSH levels. The positive group significantly increased the plasma SOD activity (P < 0.05) and significantly decreased the plasma MDA levels (P < 0.01). Thus, the Tengcha–Gegen product was effective in regulating SOD, GSH, and MDA contents/activities, revealing the potential protective liver effect of the Tengcha–Gegen product against high oxidative stress induced by acute alcohol exposure in AAI mice.

Effect of the Tengcha–Gegen product on superoxide dismutase (SOD), glutathione (GSH), and malondialdehyde (MDA) activity in the plasma (A–C) and liver (D–F) of AAI mice. The data are expressed as the means ± SEMs. One-way ANOVA was used for analysis. #P < 0.05, ###P < 0.01, ###P < 0.001 versus control group. *P < 0.05, **P < 0.01, ***P < 0.001 versus model group. The P values between 0.05 and 0.20 are labeled.
Discussion
Before the main experiment, we explored the alcohol consumption of the AAI mouse model. The righting reflex of male KM mice given 52% (V/V) Niulanshan Erguotou wine via gavage did not disappear at a dose of 0.10 mL/10 g body weight, while the righting reflex of male KM mice given 52% (V/V) Niulanshan Erguotou wine via gavage disappeared at a dose of 0.15 mL/10 g body weight. In addition, there was no death at the doses of 0.10 mL/10 g body weight and 0.15 mL/10 g body weight. Therefore, for this study, we chose the dose of 0.15 mL/10 g for the main experiment.
The anti-alcoholism effects of the Tengcha–Gegen product on AAI were investigated in the AAI mice model. Low and medium doses of the Tengcha–Gegen product considerably shortened the sober-up time in the AAI mice, indicating that it had an anti-alcoholism effect. Because the influence of alcohol (ethanol) on various tissues depends on its concentration in the blood, 16 the plasma alcohol concentration was recorded in this study. The results showed that the low-, medium-, and high-dose Tengcha–Gegen products considerably decreased the plasma alcohol concentration. Overall, this study confirms that in low and medium doses, Tengcha–Gegen has a good anti-alcoholism effect and therapeutic potential in AAI mice.
After alcohol consumption, it is thought that most of the alcohol metabolism in the liver is mediated by metabolic enzymes such as alcohol dehydrogenase (ADH) and aldehyde dehydrogenase (ALDH).15,17 Our results showed that the Tengcha–Gegen product has no apparent effect on these two enzymes (data not shown). It is known that excessive drinking will decrease the antioxidant level but increase oxidative stress in the liver and gastric mucosa. 18 ROS produced by ethanol metabolism is a main cause of alcoholic liver injury by producing oxidative stress. SOD and GSH have antioxidant activities and thus play a protective role in the liver.12,14,19 This study showed that the low-dose and medium-dose Tengcha–Gegen products significantly increased the plasma SOD level. Moreover, the medium-dose and high-dose Tengcha–Gegen products significantly increased the liver GSH levels. However, the Tengcha–Gegen product at the three doses notably decreased the plasma MDA levels. This suggested the potential protective liver effect of the Tengcha–Gegen product against high oxidative stress induced by acute alcohol exposure, which might be related to its anti-alcoholism effect in the AAI mouse model.
Because the anti-alcoholism results of Tengcha–Gegen in AAI mice did not show a considerable dose-dependent effect, this may be related to the multiple targets of TCM, which requires in-depth efficacy and mechanistic studies under various doses and frequencies to validate the therapeutic potential. Although the findings yielded promising effects on AAI, which the one-time intake of excessive alcohol might cause, further animals’ studies need to be conducted to validate the efficacy and safety of the Tengcha–Gegen product on long-term drinking. It is also important to conduct future clinical studies to examine the efficacy and safety of the Tengcha–Gegen product on alcoholism.
Conclusion
In this study, we showed that the Tengcha–Gegen product at 0.2 and 0.4 g/kg body weight could considerably shorten the sober-up time, decrease the plasma alcohol level, and alleviate oxidative damage caused by acute alcohol exposure, indicating that the Tengcha–Gegen product has a certain anti-alcoholism effect on AAI.
Footnotes
Acknowledgments
Data Availability
The authors confirm that the data supporting the findings of this article are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by Ping An Tsumura Pharma, Inc., Tianjin, China.
Ethics and Patient Consent
We confirm that ethical committee approval was sought where necessary and is acknowledged within the text of the submitted manuscript. No patient-related studies were performed in this study, so patient consent is irrelevant to our manuscript. We confirm that guidelines on animal rights and treatment have been met, and any details of approval obtained are indicated within the text of the submitted manuscript.
