Abstract
Objective
Naringin was known to be a flavonoid compound with many biological activities and was present in pomelo peel. Duong La Cam pomelo was a speciality grapefruit in Tan Trieu commune, Vinh Cuu district, Dong Nai province. This study evaluated Duong La Cam pomelo extract's naringin content and antioxidant and antibacterial activities.
Method
Duong La Cam pomelo extract was extracted by ultrasonic method. Naringin content was determined based on the naringin standard curve. Biological activities were evaluated by DPPH, ABTS, Agar disk-diffusion method and The minimum inhibitory concentration (MIC).
Results
The research results of enriching the Naringin component in the Duong La Cam Pomelo extract were recorded with a content of 114 mg/gDW. Duong La Cam Pomelo extract was extracted by ultrasonic method with solvent and raw material ratio 1 g/10 mL, time 60 min, temperature 70 °C and using 80% Ethanol solvent. The results showed antioxidant capacity with IC50 values of 0.127 mg/mL (ABTS); 14 511 mg/mL (DPPH). Besides, the antibacterial ability of the sample was also recorded at the minimum inhibitory concentration of Escherichia coli of 2.5 mg/mL and Staphylococcus aureus of 5 mg/mL.
Conclusions
The study showed that the ultrasonic extraction method saves time but still ensures high naringin content and good activity in extraction. It can be seen that naringin extract from Duong La Cam grapefruit has great potential for application in the medical field.
Introduction
In Vietnam, pomelo is widely grown. There are many popular grapefruit varieties such as Tan Trieu pomelo (Dong Nai), Da Xanh pomelo (Ben Tre), Doan Hung pomelo (Phu Tho), Nam Roi pomelo (Can Tho), Phuc Trach pomelo (Ha Tinh). 1 Among them, Tan Trieu grapefruit is a pomelo variety grown in Tan Trieu village with pomelo varieties such as Duong La Cam pomelo, Guava pomelo, Duong Da Lang pomelo, Thanh Tra pomelo, Duong Hong pomelo, Da Xanh pomelo. Among them, Duong La Cam pomelo and Guava pomelo are two pomelo varieties with the most unique quality, are very popular with consumers and are granted geographical indication protection. In 2012, the Intellectual Property Office Of Vietnam (IP Viet Nam) issued Decision No. 2837/QD-SHTT on granting Geographical Indication Registration Certificate No. 00031 for two types of pomelo in Tan Trieu: Duong La Cam pomelo and Guava pomelo. Along with the development of grapefruit products, a large amount of pomelo peel was wasted. Meanwhile, Vietnam every year still had to import many drugs containing naringin at high prices, while the reserves of pomelo peel in Dong Nai provinces were very large. If this source of raw materials is exploited well, it will solve the shortage and cost of drugs containing naringin.
Naringin is a natural flavonoid found in pomelo and many other citrus fruits.2–5 Naringin is a flavanone glycoside found in high concentrations in pomelo peel.6,7 The distribution of naringin is assessed through the parts of the pomelo, with naringin content decreasing in order: white peel (43.28 mg/g), green peel (3.91 mg/g), fruit flesh (0.22 mg/g), and seeds (0.028 mg/g).2,8,9 The biological properties of naringin have been widely studied, from its strong antioxidant capacity to its ability to reduce cholesterol, prevent high blood pressure, reduce cardiovascular complications, restore stomach ulcers, and support the prevention of cardiovascular diseases prevent cancer.5,9–12 The Ghanbari-Movahed group has synthesized the inhibitory abilities of naringin on human malignant tumors. Research has shown that cancer cells begin to be inhibited after at least 24 h. This shows naringin's potential to inhibit cancer cells. In addition, naringin also has cancer-preventing and anti-cancer effects in different models of oral, breast, colon, liver, lung and ovarian cancer. 5 In Kanokorn Sudto's research, naringin was isolated using the solvent methanol and water at a ratio of 1:6 and crystallized with Dichloromethane for 3-4 days. Research results showed that naringin extraction efficiency was 15% with a recorded content of 24 mg/gDW with purity >98%. 6 Ioannou's group studied the influence of extraction methods on the naringin extraction process. Commonly used methods (CSE), microwave-assisted extraction (MAE), ultrasonic-assisted extraction (UAE), supercritical CO2 extraction (SCE) and high-pressure extraction (HPE). Research results show that the UAE method gives the highest naringin absorption content (1 g/kg DW). 13 Author Jolita Stabrauskiene's group used ultrasound combined with magnesium aluminometasilicate to extract naringin in pomelo peel. The results showed that the naringin content in pomelo peel extracted with 50% ethanol solvent reached 25.05 mg/g dry matter and 70% ethanol reached 35 mg/g. Shows that the ultrasound method has good naringin extraction ability. 7 Therefore, this study utilized the pomelo peel of Duong La Cam to extract the naringin-containing extract by ultrasound method and evaluated the antioxidant and antibacterial properties of the extract.
Experimental
Pretreatment and Chemicals
DPPH (2,2-Diphenyl-1-picrylhydrazyl, ≥95%), ABTS (2,2′-Azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt, ≥98%), L-Ascorbic acid (Vitamin C, ≥99%), Amoxicillin, Naringin were provided by Sigma-Aldrich, America. Sodium Carbonate (Na2CO3), Methanol (CH3OH), Potassium peroxydisulfate (K2S2O8), Sodium Chloride (NaCl) were provided by Xilong, China. Peptone powder; Yeast Extract Powder; Agar powder were provided by Himedia, India. Bacterial strains including Staphylococcus aureus (S. aureus) ATCC 6538, Escherichia coli (E. coli) ATCC 8739 were preserved on Luria - Bertani (LB) agar blocks at 4 °C. Duong La Cam Pomelo (DLCP) was collected in Tan Trieu, Dong Nai province. The DLCP peel was stripped of damaged parts and washed with water several times to remove dirt. The DLCP peel was separated into two parts, mesocarp and exocarp. The DLCP mesocarp was separated and cut to size 10-15 mm wide, and 20-30 mm long. DLCP mesocarp was dried at 65 °C for 24 h using a Faithful Forced Convection Oven. DLCP mesocarp was ground into fine powder and stored in a desiccator at room temperature.
Ultrasonic Extraction Method
1 g of DLCP powder was added to a 100 mL Erlenmeyer flask containing 10 mL of ethanol solution 80% (v/v). The solution mixture was sonicated in an ASONIC PRO 60S–SLOVENIA ultrasonic bath at a frequency of 40 Khz and a temperature of 70 °C for 90 min. The mixture was filtered to obtain the extract using a Rocker Vacuum Filter with filter paper of pore size 15 ∼ 20 µm. Samples after filtration were stored in a refrigerator at 10 °C. 14
Method for Determining Naringin
Naringin content was determined using a Metash V-5100 UV-Vis Spectrophotometer. The device can detect wavelengths between 190 and 1100 nm with high resolution and accuracy (±0.1 nm). The choice of medium is made on the basis of Naringin solubility. Phosphate buffer solution (PBS) pH 7.4 was chosen to prepare the calibration curve. 10 mg of Naringin was dissolved in an aliquot of PBS, pH 7.4, and finally diluted to 10 mL in a volumetric flask to obtain a concentration of 100 μg/mL. The results were scanned for λmax in the ultraviolet range of 200-400 nm using an ultraviolet-visible spectrophotometer. The standard curve was prepared by diluting the stock solution to create solutions with concentrations ranging from 4-20 µg/mL and measured with a wavelength of 283 nm. 15
Method for Determining Total Polyphenol and Flavonoid Content
Total phenolic content was determined through the Folin – Ciocalteu method. 16 0.5 mL of extract added to 2.5 mL of Folin – Ciocalteu (10%) and shake well. After 4 min, 2 mL of Na2CO3 solution was added to the mixture, shaken well and incubated for 2 h at room temperature.
Total flavonoid content was determined based on the Sitthichai method. 16 0.5 mL of the sample solution (diluted to the appropriate concentration) was added to 0.1 mL of 10% AlCl3 solution. 0.1 mL of 1 M CH3COOK solution and 4.3 mL of distilled water were added to the mixture and shaken well. Leave the solution at room temperature for 30 min.
The absorbance of the solution after reaction was measured at 760 nm (polyphenol) and 415 nm (flavonoid) on a UV-Vis spectrophotometer. Gallic acid was used as a reference standard and the results were equivalent to milligrams of gallic acid/1 gram of material sample ((mgGAE).(gDW)−1). Quercetin was used as a reference standard and the results were equivalent to milligrams of Quercetin /1 gram of material sample.
Evaluate Antibacterial Ability
DPPH Free Radical Scavenging Ability
In vitro antioxidant activity was evaluated using the ascorbic acid standard by the 1, 1-diphenyl-2-picrylhydrazyl radical scavenging method (DPPH captures the hydrogen groups of the antioxidant)
16
iefly, 1 mL of DPPH methanol solution (0.6 mM) will be thoroughly mixed with 1 mL of DLCP solutions of different concentrations (12.5, 25, 50, 100 μg/mL) and incubated under conditions dark for 60 min. The absorbance will then be recorded at 517 nm in a UV-VIS spectrophotometer and the IC50 value will be calculated from the ascorbic acid standard curve. DPPH* free radical scavenging activity (OR %) was determined based on the formula:
ABTS•+ Free Radical Scavenging Ability
10 μL of DLCP extract was placed into eppendorfs pre-filled with 990 μL of ABTS•+ solution. The sample tube was sealed to protect from light, resulting in a final volume of 1 mL. The reaction mixture was incubated at room temperature for 6 min, after which the optical density was measured at 734 nm. The results of the antioxidant activity of the extracts are expressed as IC50 values. The free radical resistance percentage of a substance is calculated according to the formula:
Statistical Analyses
Each experiment was repeated three times. Excel 2021 for Microsoft Office was used to process experimental data. Statistical analysis was performed using MS.Excel 2021. All the data are presented as the means ± SDs. All charts were drawn using origin Pro 8.5 software.
Results and Discussion
Evaluation of Raw DLCP Powder
DLCP peels were purchased and determined from manual factories in Tan Trieu commune, Vinh Cuu district, Dong Nai province (belonging to Tan Trieu Grapefruit village). DLCP peels were sent to the Ginseng and Medicinal Center of Ho Chi Minh City for plant identification. The results shown in Figure 1 show the characteristics of the DLCP variety with the scientific name Citrus maxima (Burm) Merr, belonging to the genus Citrus, family Rutaceae. Compared to other types of pomelos, DLCP has small, oblong leaves and small fruit with thinner skin. The peel of DLCP was 15 ± 0.017 mm thick, thinner than the peel of Da Xanh pomelo (17 ± 0.021 mm) or Nam Roi pomelo (20 ± 0.015 mm).

Sample image of DLCP tree in Tan Trieu commune, Vinh Cuu district, Dong Nai province.
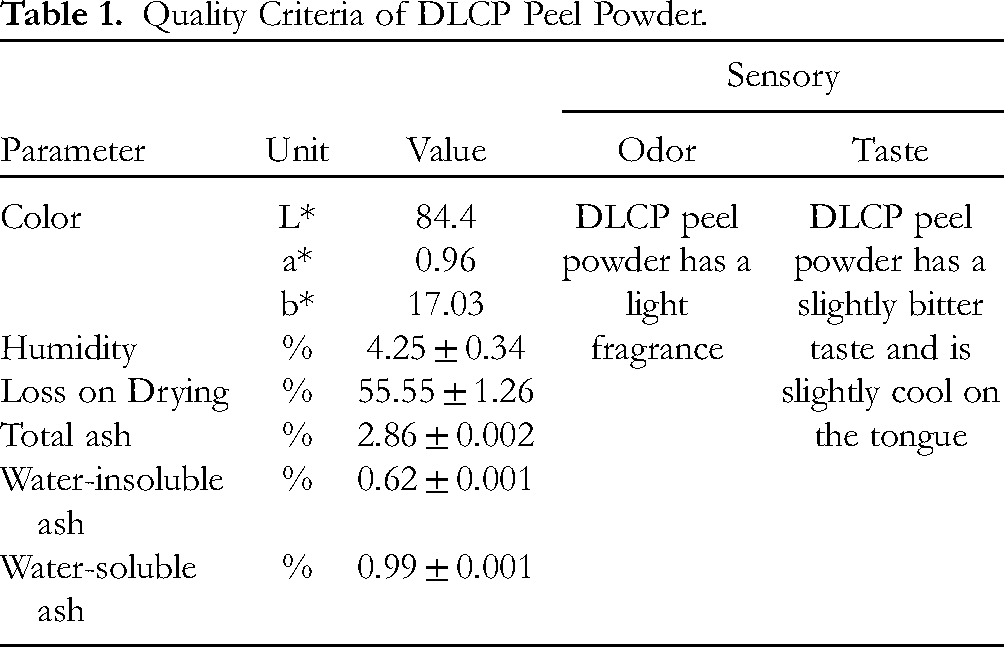
DLCP peels, after being purchased, processed and dried, yield DLCP peel powder. Color values and basic quality parameters of raw DLCP peel are shown in Table 1. The quality of raw DLCP powders was evaluated based on Vietnam Pharmacopoeia V- appendix 12.2-page PL271. Raw powder was determined to be raw powder when the dry ingredient is ground and more than 95% of the particles pass through the No. 2000 sieve. Raw powder was determined based on the powder and rail size standards in the Vietnam Pharmacopoeia V- appendix Appendix 3.5-page PL119. The color of DLCP peel powder is evaluated through the L*a*b* value. The color space value (The Commission Internationale de l’éclairage Lab color space – CIELAB color space) was recorded as L* (84.4), a* (0.96), b* (17.03). Based on the color space and L*a*b* value, the DLCP powder was determined to be light brown (Dutch white). The Moisture value of the DLCP peels was recorded with a value of 4.25%. This shows that the DLCP peel powder was completely dry with a low moisture value of <10%. In addition, the values of total ash, soluble ash, and insoluble ash were also calculated with values of 2.86%, 0.99%, and 0.62%, respectively. The results show that the insoluble ash content of the raw powder was low. This shows that the material's content of organic compounds that can dissolve in water and some other solvents was quite high. In this study, Naringin was the main compound extracted from DLCP peels. Therefore, the process of evaluating factors affecting Naringin in DLCP peels was conducted.
Quality Criteria of DLCP Peel Powder.
Evaluate Factors Affecting the Naringin Extraction Process
Factors in the extraction process that affect the absorption of naringin from grapefruit peel. Therefore, influencing factors such as ethanol concentration, solvent ratio, and extraction time were evaluated. In Jolita Stabrauskiene's research, the ultrasound method was evaluated as the best method to extract naringin from citrus fruit peels.
7
Therefore, the ultrasound method was used to evaluate naringin extraction in this reseach. The effect of ethanol concentration on Naringin content was shown in Figure 2. The results show that the naringin content obtained at different ethanol concentrations was 30.74 ± 0.012 mg/gDW (60% Ethanol); 43.287 ± 0.018 mg/gDW (Ethanol 80%); 34.261 ± 0.005 mg/gDW (Ethanol 100%). It can be seen that at 80% ethanol ratio, the most naringin is obtained. ANOVA analysis results showed that ethanol:water ratio had a significant effect on naringin content with 95% confidence level (

Effect of ethanol:water ratio to naringin.
The solvent ratio has a great influence on the process of obtaining naringin from DLCP peel. The effect of solvent ratio on Naringin content is shown in Figure 3. The results showed that the naringin content obtained at different ratios was 28.52 ± 0.330 mg/gDW (5 mL); 53.95 ± 1.274 mg/gDW (10 mL); 46.507 ± 0.515 mg/gDW (15 mL); 41.278 ± 0.703 mg/gDW (20 mL). The results showed that the naringin content obtained was in the range of 40-50 mg/gDW. It can be seen that the naringin content the most yields at the ratio of 1 g/10 mL. ANOVA analysis results showed that solvent ratio had a significant effect on naringin content with 95% confidence level (
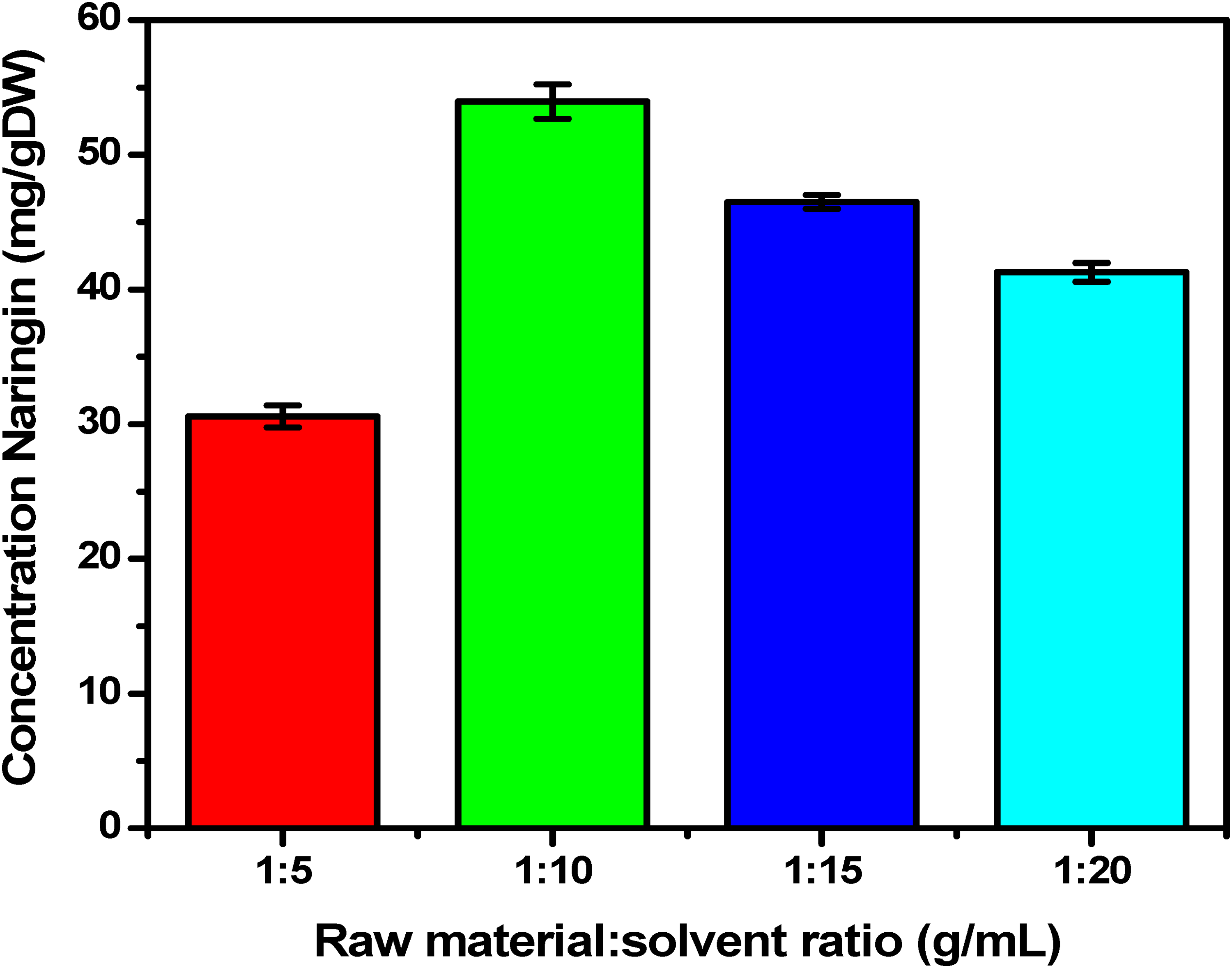
Effect of solvent ratio to naringin.
The effect of extraction time was performed at intervals of 30 min, 60 min, and 90 min and shown in Figure 4. The results showed that the naringin content obtained at different periods was 82.126 ± 0.011 mg/gDW (30 min); 117.198 ± 0.035 mg/gDW (60 min); 114.095 ± 0.093 mg/gDW (90 min). The results showed that naringin content increased rapidly when increasing the time from 30 min to 60 min and balanced when increasing the time to 90 min. It can be seen that after 60 min, almost all of the naringin content has been extracted. ANOVA analysis results showed that time had a significant effect on naringin content with 95% confidence level (

Effect of extraction time to naringin.
Evaluation of Antioxidant Capacity
After studying the Naringin content of DLCP, the next step was to investigate and evaluate the antioxidant activity. These findings would be useful in evaluating and standardizing the quality of naringin from DLCP. Antioxidant capacity was easily affected by factors during the extraction process. In Xiaoju Tian's study, the free radical scavenging ability of naringin from grapefruit peel by the ABTS method (84.03%) was performed and was better than that of the DPPH method (21.88%). In addition, Xiaoju Tian also evaluated the antioxidant capacity using the FRAP method. The results showed that the reducing ability of the method was 0.995 mmol Fe(II)/gDW. 19 In Rahman's study, the FRAP assay was found to have a poor correlation with many antioxidants analyzed previously. 20 The results showed that the FRAP method was not suitable for evaluating the antioxidant capacity of naringin. This suggests that the naringin compound has a high antioxidant capacity and is best performed by the ABTS and DPPH method. Therefore, the antioxidant capacity was present in grapefruit using ABTS and DPPH methods. Antioxidant activity was demonstrated by the antioxidant donating a hydrogen atom to reduce the purple DPPH free radical to yellow DPPH-H, determined by spectrophotometry at 517 nm. 21 The ability to reduce DPPH free radicals gradually increased with concentration as shown by the results of evaluating the antioxidant capacity of vitamin C and naringin. The antioxidant capacity of vitamin C was rangning from 12.43% to 88.79%. The DPPH free radical removal efficiency of vitamin C was lowest (12.43 ± 2.68%) at a concentration of 0.0004 mg/mL and the removal efficiency reached over 50% at a concentration of 0.0035 mg/mL of 60.72 ± 7.48%. The results of building a standard curve showing the correlation between vitamin C concentration and antioxidant capacity were shown in Figure 5.

Graph of % inhibition of vitamin C according to DPPH.
The antioxidant capacity of vitamin C was ranging from 16.93% to 99.85%. The ABTS free radical removal efficiency of vitamin C was lowest (16.93 ± 2.04%) at a concentration of 0.005 mg/mL and the reduction efficiency was over 50% at a concentration of 0.015 mg/mL, 55.87 ± 0.65%. The results of building a standard curve showing the correlation between vitamin C concentration and antioxidant capacity were shown in Figure 6.

Graph of % inhibition of vitamin C according to ABTS.
The antioxidant effect of naringin was determined based on its DPPH free radical neutralization efficiency. The antioxidant results were shown in Table 2. At 80% ethanol concentration, the IC50 value was 8.056 mg/mL (ABTS); 8053 mg/mL (DPPH). At a solvent ratio of 10 mL, the IC50 values were 0.999 mg/mL (ABTS); 16 356 mg/mL (DPPH). At 60 min, the IC50 value was 0.127 mg/mL (ABTS); 14 511 mg/mL (DPPH). It cloud be seen that the minimum 50% inhibitory concentration of the extract from the DPPH method had a higher concentration value than the ABTS method, corresponding to lower activity. This was explained in Melanie Platzer's research on the ability of organic compounds to interact with the DPPH and ABTS methods. Naringin was a flavanone compound, so it interacted with ABTS than DPPH through the sequential proton loss electron transfer method (SPLET). 22 In the study of Nesrine Benkhaira and Anna Floegel, the VCEAC value was expressed as the DPPH free radical scavenging activity of the sample. Higher VCEAC means higher DPPH free radical scavenging activity.23,24 The results showed that the VCEAC value at 90 min was the highest, close to the VCEAC value of Vitamin C. The Naringin concentration was 117 mg/gDW at 60 min and 114 mg/gDW at 90 min. The Naringin concentration didn't change too much, but the inhibitory activity of the extract was improved.
IC50 Value of DPPH and ABTS Free Radical Scavenging Methods.

Evaluation of Antibacterial Ability
Naringin was a flavanone compound, so antibacterial ability to be an important value in evaluating naringin's activity. Antibacterial ability was performed based on qualitative methods (Agar disk-diffusion method) and quantitative methods MIC. The results of the antibacterial ring and MIC were shown in Figure 7 and Table 3. The DLCP extracted was dilution into different concentrations from 2 to 8 mg/mL. The results of the antibacterial ring showed that for the
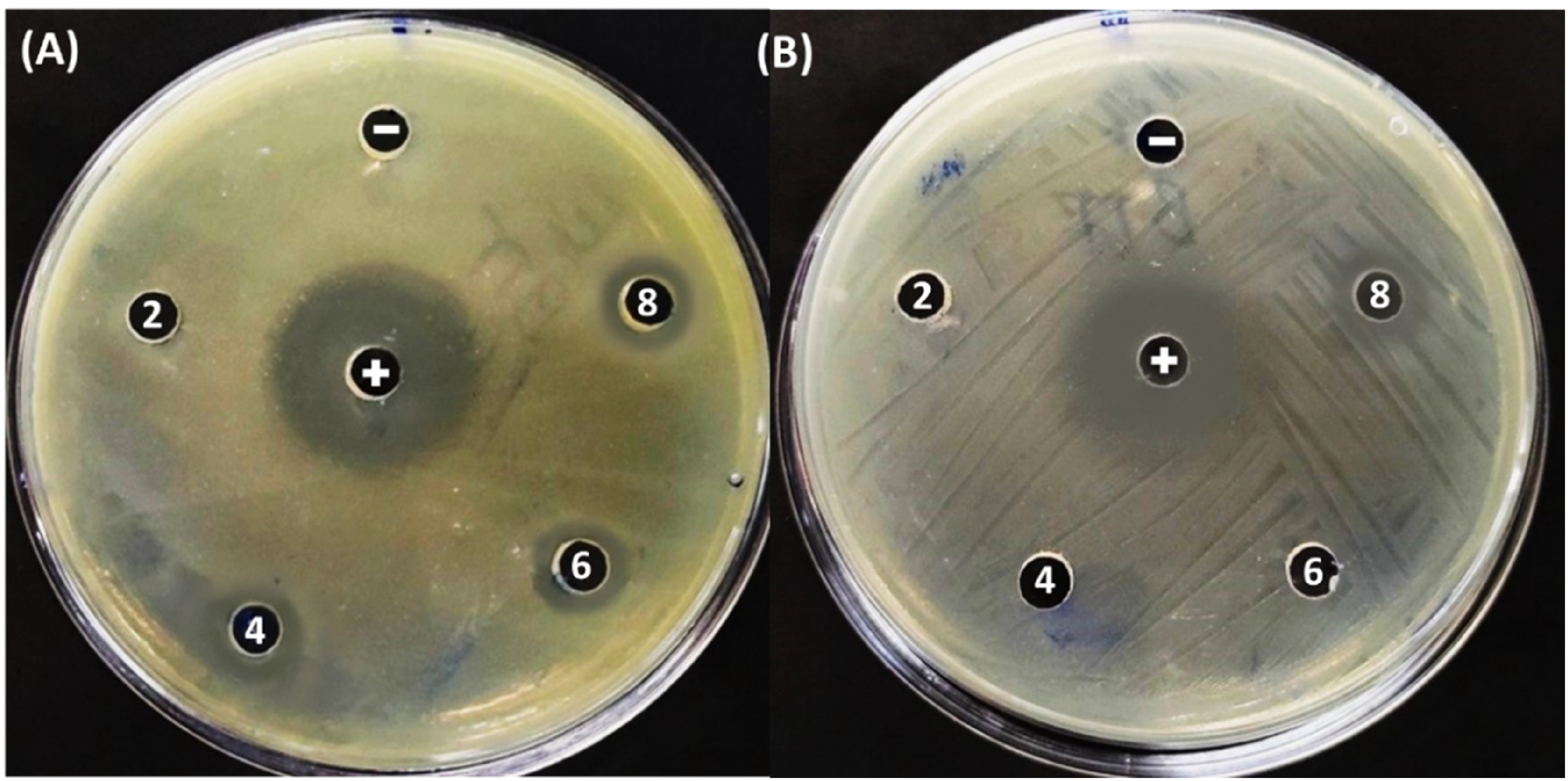
Antibacterial activity of DLCP extract with Escherichia coli (A) and Staphylococcus aureus (B) at different concentrations.
Evaluation of the MIC of Escherichia coli and Staphylococcus aureus bacteria of DLCP Peel Extract.
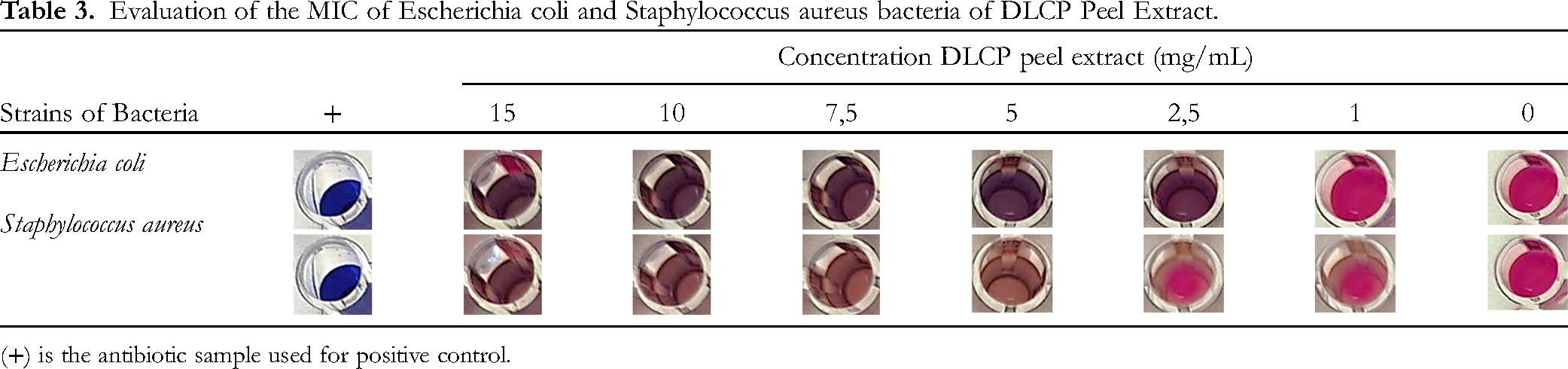
(+) is the antibiotic sample used for positive control.
The MIC was evaluated and shown in Table 3. The sample was evaluated on two bacterial strains,
Comparative Evaluation with Other Extraction Methods
For an overview and comparison between the methods, the immersion and soxhlet methods were used for evaluation and the conditions used to extract naringin from DLCP peel were based on the study of Ly Thi Thuy Duyen. 14 The results of naringin content from different extraction methods were shown in Table 4. In which the naringin content from immersion, soxhlet and ultrasound method were 13.86 mg/g, 54.88 mg/g, 117 198 mg/g. The results show that using the ultrasound method was more effective in extracting naringin than the above two methods. The ethanol concentration shows that at a rate of 80%, the naringin content was completely extracted. Analysis shows that using less water during the extraction process would be more convenient in removing the solvent, so extraction with a ratio of ethanol and water of 80%:20% was the most optimal. Because naringin dissolves well in polar solvent ethanol/water, the water ratio of 20% helps raise the temperature of the entire dissolved solvent to make the extraction efficient. In addition, naringin content wasn’t improved when using ultrasound combined with enzymes in the study of Hung and Nguyen.29,30 It can be seen that ethanol played an important role in the ultrasonic extraction process. The ratio of solvent used for extraction was also lower. This would help save time and money when extracting naringin from DLCP peel. For Soxhlet extraction method, the narigin content was recorded higher when ethanol concentration was at 80% and the time and solvent ratio increased. It could be seen that Soxhlet method was quite time-consuming for extraction. For the immersion method, it can be seen that the presence of water in the solvent at a ratio of 20% helps the naringin content to be dissolved better. Besides, the extraction time is one of the important factors of the immersion method. The longer the time, the more naringin is obtained. This shows that the immersion method depends a lot on the soaking time. The extraction time is too long, which will increase the operating cost. In addition, the long-term extraction process at high temperature caused some flavanoid and polyphenol compounds to decompose during the extraction process.12,31,32 This also reduced the naringin content obtained after the extraction process. The immersion and soxhlet methods were no longer effective in extracting naringin from grapefruit peel. Isha Gupta's study also showed the superiority of the ultrasonic method in extracting naringin using ethanol solvent. The naringin content in ethanol solvent was doubled compared to other solvents such as DMSO, DMF, acetone. 33
The Naringin Content Was Obtained by Different Extraction Methods.

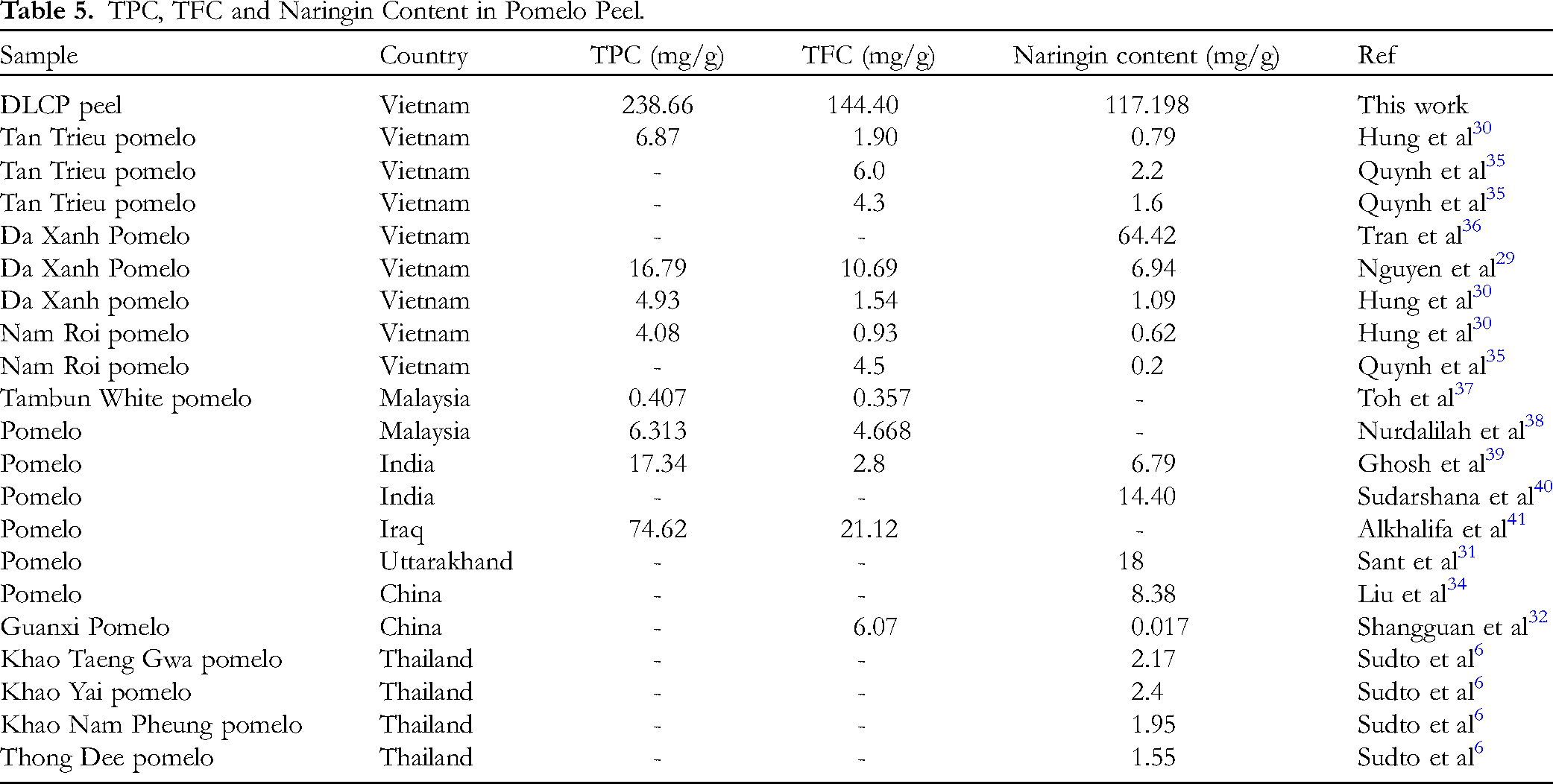
In addition, the naringin content from different types of grapefruit peels is also compared in Table 5. Depending on the different types of grapefruit peels, the polyphenol, flavonoid and naringin content is recorded differently. The results show that in Vietnam, there are many studies on naringin from grapefruit peels, grapefruit varieties have been used to serve the process. The naringin content is also recorded from 0.2-0.6 mg/g for Nam Roi grapefruit, Da Xanh from 1-64 mg/g, Tan Trieu 0.7 to 2.2 mg/g by different extraction methods. In which, the orange leaf grapefruit in this study was recorded as the highest at 117 mg/g by ultrasonic extraction method. For grapefruit varieties in other countries, the naringin content was recorded as high at 18 mg/g in Uttarakhand, 14.4 mg/g in India, 8.38 mg/g in China, respectively. Grapefruit varieties in Thailand showed low naringin content, possibly due to inappropriate extraction methods. This is a highlight for researchers to pay attention to improving naringin extraction methods.
TPC, TFC and Naringin Content in Pomelo Peel.
Conclusion
DLCP was collected and dried and evaluated for color and quality based on The Commission Internationale de l'éclairage Lab color space and Vietnamese Pharmacopoeia V - appendix 12.2 - page PL271. The DLCP powder was determined to be light brown (Dutch white) with a light aroma, slightly bitter taste, and a slightly cool sensation on the tip of the tongue. The moisture, total ash, soluble ash, and insoluble ash of the powder were recorded at values of 4.25%, 2.86%, 0.99%, and 0.62%. The influencing factors were evaluated and selected with the ratio of solvent and DLCP powder (1 g/10 mL), time (60 min), temperature (70 °C), and Ethanol solvent (80%) by ultrasonic method. The results recorded Naringin content as 114 mg/gDW. The results showed antioxidant capacity with IC50 values of 0.127 mg/mL (ABTS); 14 511 mg/mL (DPPH). Besides, the antibacterial ability of the sample was also recorded at the minimum inhibitory concentration of Escherichia coli of 2.5 mg/mL and Staphylococcus aureus of 5 mg/mL. Evaluation of the parameters in the extraction process to optimize the naringin production process on an industrial scale, making the most of grapefruit peel by-products in Vietnam. It can be seen that naringin extract from grapefruit has great potential for application in the fields of medicine, cosmetics, food, and smart packaging. However, the limitation of the study was that it didn't evaluate the activities that naringin brings, the small sample size, and the lack of in vivo studies. This was the premise for in-depth research as well as research on the application of naringin in practice.
Footnotes
Author Contributions
Author Contributions: Writing-original draft preparation H.N.B. and H.L.Q; data curation N.V.P. and L.V.T.; methodology N.V.P. and H.L.Q.; writing-review and editing T.T.T. and H.N.B. All authors have assented to the manuscript's published version.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
This research was fund by a grant from the Science and Technology Development Foundation of Nguyen Tat Thanh University with code (SPUD.2024.01.17).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
