Abstract
Background
The recent increase in interest in alternative medicine calls for an investigation into the non-target organ effect. Khaya senegalensis is one of such popular plants used to manage malaria in low-income countries. This study therefore aimed at investigating the effect of stem bark extract of Khaya senegalensis (KSE) on the testes (non-target organ) and its related serum hormones.
Methods
Grouped mature male rats (n = 4) were treated with 100–1000 mg/kg KSE or distilled water for twenty-eight days. Serum hormones were taken immediately after cervical dislocation, prepared appropriately, and assayed for Testosterone, Luteinizing, and Follicle Stimulating Hormone. The hormones levels were determined using ELISA kits The right testis and epididymis from treated animals were harvested and prepared for histological studies.
Results
The result indicated a significant increase (p = 0.045, 0.038) in the epididymis and serum LH in the 1000 mg/kg KSE, but caused no significant increase in serum FSH and Testosterone (p > 0.05), in any of the treatment groups. Histological sections of the testes showed mild to moderate degeneration of the spermatogonia and hyalinization (1000 mg/kg, 500 mg/kg, 100 mg/kg) of the seminiferous tubules with reduced spermatogenesis in a dose-dependent manner.
Conclusion
Khaya senegalensis could have an adverse effect on the morphology of the testis in a dose-dependent manner.
Keywords
Introduction
The interest in herbal medicine continues to increase due to its physical and financial accessibility, especially in less-resourced countries. These herbal materials and their product can cause temporary sterility which is usually on the blind side of its users. The matter of infertility is very significant in African culture. 1 Although females are usually the victims of social stigma attached to infertility and sexual dysfunction males can sometimes be blamed for childlessness. 2 In some societies, couples with no children are scorned and sometimes ostracized from leadership roles. 1 There is, therefore, the need to evaluate the non-target effect of popularly used plants in folkloric practice to ensure the safety of users. Khaya senegalensis is one of the most popular herbal materials found on the Ghanaian market which is used for a number of ailments including malaria.3,4 It is popularly sold on the Ghanaian market as mahogany or ‘kuntunkuni’. A number of studies have been done on its antibacterial, hypoglycemic, and antipyretic effects,5,6 but limited studies with respect to how it affects fertility. Studies that aim to evaluate the efficacy and safety of folklore herbal medicine are of relevance to the scientific and the larger community because it provides us with cheap and easy-to-find alternative to orthodox medicines.
The current study, therefore, sought to find the reproductive effect of the hydro-ethanolic stem bark extract of K. senegalensis on the male wistar rat. This is to assist users of the plant material to make an informed decision concerning their sexual health whenever they are taking the plant.
Materials and Methods
Experimental Animals
Male Wistar rats were procured from the Noguchi Memorial Institute for Medical Research, Accra, Ghana, and housed at the Animal House Facility of the Department of Pharmacology, KNUST- Kumasi, Ghana. The rats were handled in accordance with the guidelines s of the National Research Council's Guide for the Care and Use of Laboratory Animals. They were housed under the required conditions for experimental rats. A total of twenty male Wistar rats with postal natal days between 60–70 days were used. This was to ensure that sexually mature rats were used for the experiment. They were randomly put into a group of 4 with 5 rats each at priori. The four groups consisted of three (3) treatment groups and one control group. Group IV served as the negative control group and received distilled water only while groups I-III received 100 mg/kg, 500 mg/kg, and 1000 mg/kg per os (po) of the ethanolic KSE at priori respectively for 28 days. Dosing was done every morning continuously for 28 days by the method of gastric lavage.
The average weight of the male rats was between 150 g and 250 g.
Housing and Husbandry
Rats were kept under standard temperature and humidity (temperature 25 ± 2° C with a 12 h light-dark cycle) at the Animal House facility of the Department of Pharmacology, Faculty of Pharmacy and Pharmaceutical Sciences, KNUST, and were allowed access to commercial chow, distilled water ad libitum.
Plant Collection and Extraction
The stem bark of Khaya senegalensis was obtained from the Botanical Gardens of KNUST, Kumasi, Ghana. The sample was then authenticated by a Botanist at the Department of Pharmacognosy, KNUST, Kumasi, Ghana. It was air dried at room temperature of about 25° C and a sample was deposited at the Department of Pharmacognosy herbarium with a voucher number KNUST/HM1/2015/006. The dried stem bark was ground by an electric blender into powder with a net weight of 4.6 kg. Ethanol (95%) was used to extract the active ingredient by cold maceration. The hydro-ethanolic extract was concentrated and air dried with a hot air oven (Gallenk BS 300 Plus Series Electric Oven) at a temperature of 50° C for two to seventy hours to remove the alcohol from the extract. A semi-solid mass of 756.3 g was obtained and kept in the refrigerator at about 4 °C till it was used for dosing. The extract obtained will then be referred to as the alcoholic extract of K. senegalensis (KSE) in this study
Preliminary Phytochemical Analysis
Using the protocol by 7 the presence of some phytochemicals were tested on the raw plant material and the extract.
Body and Reproductive Organ Weight Measurement
The body weights of rats were measured weekly with KERN PCB 1000-1 precision balance. After 28 days, the rats were sacrificed by cervical dislocation, and their reproductive organs (testes and epididymis) weights were excised. Excess fat and interstitial tissues were trimmed off and organ weight was measured with a precision electronic balance (Acculab VIC-212 digital scale) obtained at the Pharmacology department. After weighing the organs, they were then fixed in 10% formalin for 24 h to prepare it for histology.
Epididymal Sperm Analysis
For this experiment, the method described by 8 was used. Four groups of adult male Wistar rats were used. There were five (5) rats in each group. Group IV served as the negative control group and received distilled water only while groups’ I-III received 100 mg/kg, 500 mg/kg, and 1000 mg/kg per os (po) of the ethanolic KSE at priori respectively for 28 days. On the last day of extract administration, the animals were weighed and euthanized by cervical dislocation. The wet weights of the left epididymis and testes were measured and recorded. The cauda epididymis was minced to release the sperm cells into the diluent (0.9% Sodium Chloride)
Sperm Morphology
For the morphological analysis of the samples, a modified method 8 was used. A semen-diluting fluid was prepared by mixing 5 gm of sodium bicarbonate with 1 ml of 10% formalin and 100 ml of distilled water. An aliquot of 3 ml of the semen-diluting fluid was added to the sample in a plain tube. An aliquot (0.5 µl) was pipetted on a clean slide and a thin smear was done and allowed to air dry and later fixed by heat. 1% of chloramine solution was added for several minutes to remove excess mucus. It was washed by blotting with filter paper and stained for five minutes with two parts of Zeil-Neelson, carbol fuchsin, and one part of concentrated alcoholic solution of eosin and one part of alcohol 95%. It was later washed with water and counterstained with Loefller's methylene blue for a few seconds and washed dry. The prepared slide was examined at a magnification of X40 using Leica DM 500 digital microscope.
Sperm Concentration (Count)
To assess sperm count, semen-diluting fluid was added to the sample as described above. Using a pasture pipette, an improved Neubauer counting chamber was filled and allowed to stand for about two minutes to allow the sperm cells to settle. Using Leica DM 500 digital microscope, sperm cells were counted using 40X objective, and the sperm cells were counted as in WBC count. 9
Sperm Viability
After sperm count was done on the sample, the aliquot was mixed with eosin and examined under the microscope with the Leica DM 500 digital microscope at 40X magnification. It is based on the assumption that cells that absorbed dead cells are permeable to eosin dye hence it will stain dark while viable cells do not pick up the dye.
Preparation of Serum for Competitive ELISA Serum (Hormonal Assay)
Following anesthesia with chloroform, blood samples were freshly collected by cardiac puncture and sacrificed immediately by cervical dislocation. A minimum of three milliliters (3 ml) of blood was collected into a serum separator tube. Using ACNA 90-1 Centrifuge, samples were centrifuged at 3500 RPM at a temperature of 2 °C for twenty minutes. The serum obtained was poured into Eppendorf tubes and stored at −80 °C till all samples were ready for analysis.
Serum Hormonal Assay
To run hormone analysis with the samples, they were taken out of the −80 °C and were allowed to thaw for about fifteen to twenty minutes (15-20 min). Samples were then arranged in order to make identification and recording of details easier. They were analyzed as stated in the ELISA kit manual.
Testosterone Assay
As stated in the Kit manual, WASH was prepared by adding one part of the wash buffer with thirty-nine parts of deionized water at a temperature between 15 °C-25 °C. Controls were prepared by pipetting 25 µl of calibrators A, B, C, and D into their respective wells. The samples were also pipetted at 25 µl to their wells as well. 200 µl of the conjugate was added and carefully mixed. It was incubated at 20 °C-25 °C for about one hour (1 h). It was washed three times with 400 µl of the wash solution and 200 µl of the substrate was pipetted into the various well and incubated at a temperature of 20 °C-25 °C for fifteen minutes. 100 µl of the stop buffer was pipetted and carefully mixed. As soon it was well mixed, it was then read with an automated chemistry analyzer at an absorbance of 450 nm and the results were obtained as such.
Luteinizing Hormone and Follicle-Stimulating Hormone Assay
Reagents and solutions for analyzing LH were done as stated in the manual provided for by the manufacturer. Working reagents were prepared by the mixing of one part of the WASH buffer to forty-nine parts of deionized water at a temperature between 15 °C-25 °C. 50 µl of calibrators, controls, and samples were distributed accordingly. 100 µl of the conjugate was pipetted and distributed as well. It was carefully mixed and covered with an adhesive strip to incubate for about one hour (1hr) at a temperature of 20 °C-25 °C. It was washed three times with 300 µl of the wash solution. 100 µl of the substrate was pipetted and distributed into their various well and incubated at a temperature of 20 °C-25 °C for about fifteen minutes and later terminated with a 50 µl of stop buffer which was carefully mixed. It was immediately put into the analyzer with an absorbance of 450 nm and analyzed as such.
Testicular Assay
After the rats were sacrificed by cervical dislocation, the left testes were excised for homogenization. Normal saline was used as a solvent for the homogenization process. The homogenate was centrifuged at 3500RPM at a temperature of 2 °C. The supernatants were pipetted into Eppendorf tubes and stored at −80 °C till later use.
Sample Preparation for Histology
The organs (right testis and epididymis) harvested after sacrifice were initially fixed in 10% formalin solution for 24 h. They were later embedded in paraffin and stained with hematoxylin and eosin (H&E). Slices were taken with a microtome blade horizontally. Using D.P.X mountant, the slide was fixed with a cover slid. It was examined microscopically by a Leica DM 500 digital microscope at 10X and 40X magnification. Photomicrographs were obtained at a magnification of X10 and X40. 10
Morphometric Analysis
Prepared testicular tissues were assessed in groups by two pathologists (NAT and BMD) who were blinded to the study groupings. Using Leica DM 500 digital microscope, slides were evaluated for testicular damage. To design the grading for scoring, the two pathologists first examined all the slides in all the groupings. The two pathologists agreed on slides artifacts, lesions and its parameters, and testes morphometrics (spermatozoa, spermatocyte, spermatogonia, and Leydig) before the start of the assessment. Scoring was based on the presence of severity of lesions. Two scoring systems were adopted (normal and ordinal). A binary nominal scoring (+/-) was used to show the presence or absence of lesion (degeneration) while 0–4 (0%–100%) was used to determine the severity (where 0 means normal 3 means severe form of degeneration)
Reagents and Instruments
In executing the project, reagents of analytical grade were used and they included: Zeil-Neelson, carbol fuchsin; Loefller's methylene blue, paraffin, stained with hematoxylin and eosin (H&E) all obtained from the Chemistry Department Laboratory of KNUST. ELISA for testosterone, Luteinizing Hormone and Follicle Stimulating Hormone (HUMAN, Max-Planck-Ring 21, 65205 Wiesbaden, Germany) and D.P.X Neutral Mounting Medium, Sigma-Aldrich, Germany.
The following instruments were used in the current research:
Automated-chemistry analyzer (LA Scientific Horizon 850 Analyzer, China) KERN PCB 1000-1 precision balance (KERN& SOHN Gmbh Balingen Germany) ASP 6025 Automated Vacuum Tissue Processor; Oven (Accumax, New Delhi, India) Leica RM2125 RTS Rotatory Microtome (Leica Biosystems, USA) Oven (Gallenk BS 300 Plus Series Electric Oven, Gallenkamp Weiss) Precision electronic balance (Acculab VIC212 digital scale, Sartorius Mechatronics, Germany) Leica DM 500 digital microscope (Leica Microsystems (Schweiz) AG Max Schmidheiny Strasse 201 CH-9435 Heerbrugg, Switzerland) ACNA 90-1 Centrifuge, Jiangsu Zhengji, China.
Statistical Analysis
Data were presented as mean and ± SEM. Significant differences between treatments and controls were analyzed by One-way ANOVA. It was further followed by a Dunette Post Hoc test using GraphPad Prism version 8.00 for Windows, GraphPad Software, La Jolla California USA, and IBM SPSS Statistics for Windows, Version 23.0. Armonk, NY: IBM Corp.
Results
Preliminary Phytochemical Analysis of KSE
Phytochemical screening showed the following results. Qualitative analysis of KSE showed the presence of saponins, alkaloids tannins and condensed tannins (Table 1).
Figure 1 below shows a comparison between the reproductive organ weights among the three dose groups and the results showed, that there was a weight gain in the epididymis among rats dosed with 1000mg/kg of KSE compared to distilled water.
From Figure 2 below, there was a weight gain in the epididymis among rats dosed with 1000mg/kg when compared to controls.
Effect of 28-day KSE Treatment on Sperm Parameters
From Table 2 below, there were no statistically significant differences between group means of sperm concentration and viability as determined by one-way ANOVA (p = 0.74 and 0.48 respectively).
Phytochemical Analysis of KSE.

Outcome of 28-day Treatment of KSE on Sperm Concentration and Viability.

The above table shows the mean and SEM figures of the non-reproductive organ weight. *P value < 0.05 was considered statistically significant. One-way ANOVA was computed to obtain the values.
From Figure 3 below, the highest mean of coiled tail occurred in the group treated with 1000mg/kg of KSE.
From Figure 4 below, testosterone increased in a dose dependent manner when compared to the control with the highest occurring 1000mg/kg.
The Figure 5 below shows ex-vivo testosterone levels dosed with KSE. From the figure below, testosterone increased in a dose-dependent manner when compared to the control with the highest occurring at 1000mg/kg.
Table 3 below shows majority (80%) of rats dosed with 1000 mg/kg of KSE had degeneration of their spermatogonia with minimal (20%) hyalinization. This observation was shown to be dose-dependent.
Scoring of the Testes for the Degeneration of Spermatogonia After 28-day KSE Treatment.

Sections of rats testes dosed with KSE (n = 5) * n = 4 Scoring less than 25% was considered normal, 25%-50% was considered as mild degeneration, 51%-75% was considered as moderate degeneration while above 75% was considered as severe degeneration.
The cross-section of the testis of KSE (1000 mg/kg, 500 mg/kg, and 100 mg/kg) showed mild to moderate inflammation with mild to moderate hyalinization, and reduced spermatogenesis. The inflammation accompanied by fibrosis observed was in a dose-dependent manner (Figure 6).
Discussions
The growing interest in alternate or herbal medicine calls for further studies into the efficacy and safety of medicinal plants. In as much as several studies are done with respect to the safety and efficacy of some of these plants,11,12 there is still the possible effect on non-target organs. K. senegalensis is one of such a kind that needs thorough investigation. The current study showed the presence of alkaloids, saponins, and tannins as reported by13,14 who demonstrated that K. senegalensis contains those phytochemicals.
Reproductive organ weight is used as an indicator for reproductive studies, hence variations may connote its healthy state or otherwise. 15 This assumes that the heavier the reproductive organ weight, the more optimal its function is. 16 Sperm production and maturation are also known to be associated with their respective testicular and epididymal weights. 17 The epididymis functions to give the enabling environment for the sperm cell to mature and gain the ability to fertilize. The relative weight gained in the epididymis may indicate a possible existing enabling environment for the matured sperm cell, which is provided by the blood-epididymis barrier. An efficient blood-epididymis barrier reduces the exchange of molecules between the blood and the lumen and also provides protection against immunological response for these sperm cells. 18
Scanlon in 2007 reported that an improvement of serum gonadal hormones is directly related to an improved reproductive function. In this study, KSE at all doses (1000 mg/kg 500 mg/kg, and 100 mg/kg) did not significantly reduce serum hormones when compared to the control (Figure 4). This could imply that KSE at these doses, the extract had not deteriorating effect. The rise in serum testosterone was again proportional to the rise in testicular testosterone but was not statistically significant. The results agree with19,20 that some plants have the intrinsic properties to increase serum hormones and increase sperm parameters like sperm count, motility, and morphology.

Reproductive organ weight of rats treated with KSE (100 mg, 500 mg and 1000 mg) for 28 days.
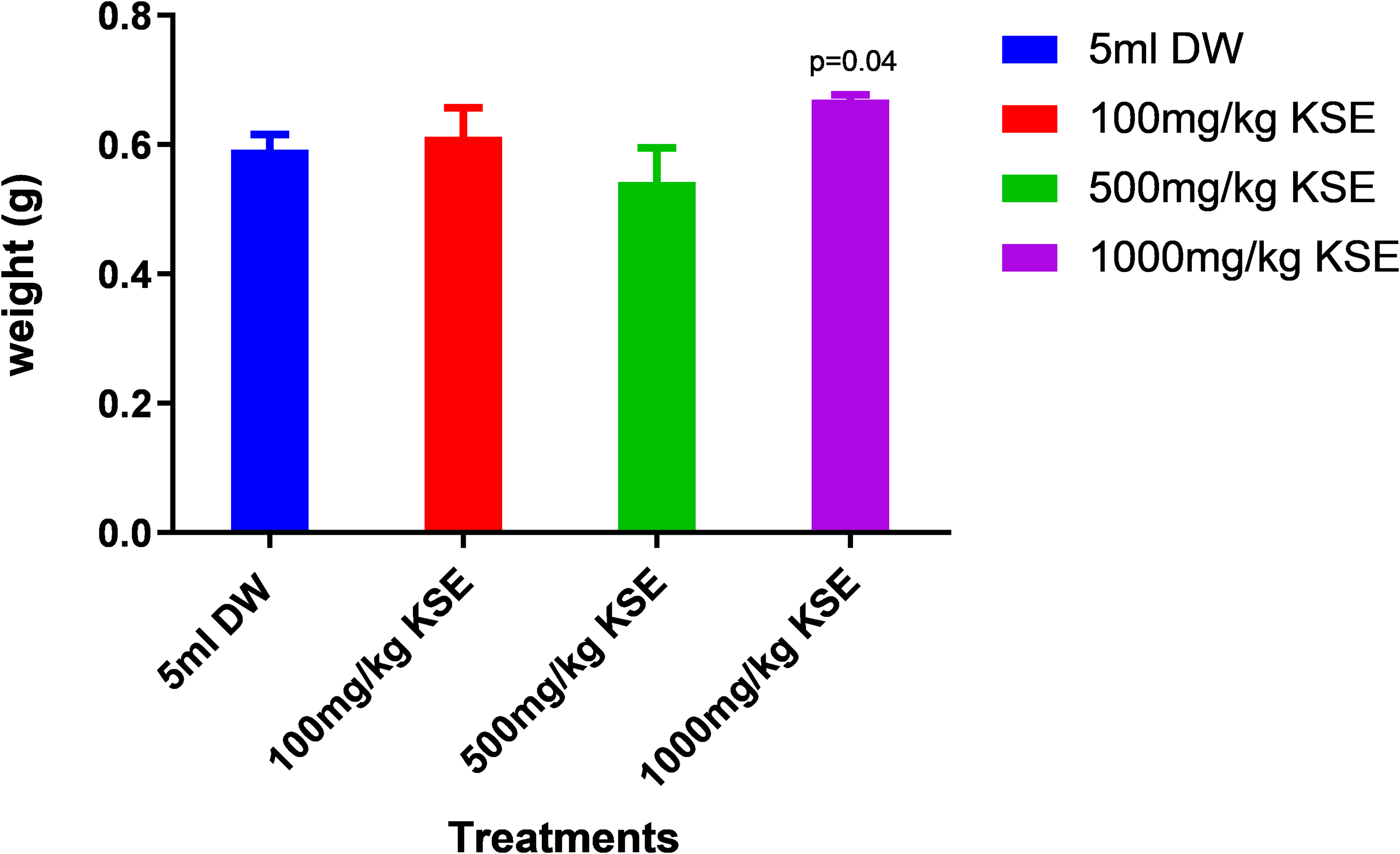
Effects of 100 mg/kg, 500 mg/kg and 1000 mg/kg KSE on epididymis wet weight in male rats.

Slides showing the morphology of rats treated with KSE at 1000 mg/kg, 500 mg/kg, 100 mg/kg, and 5 ml of DW.
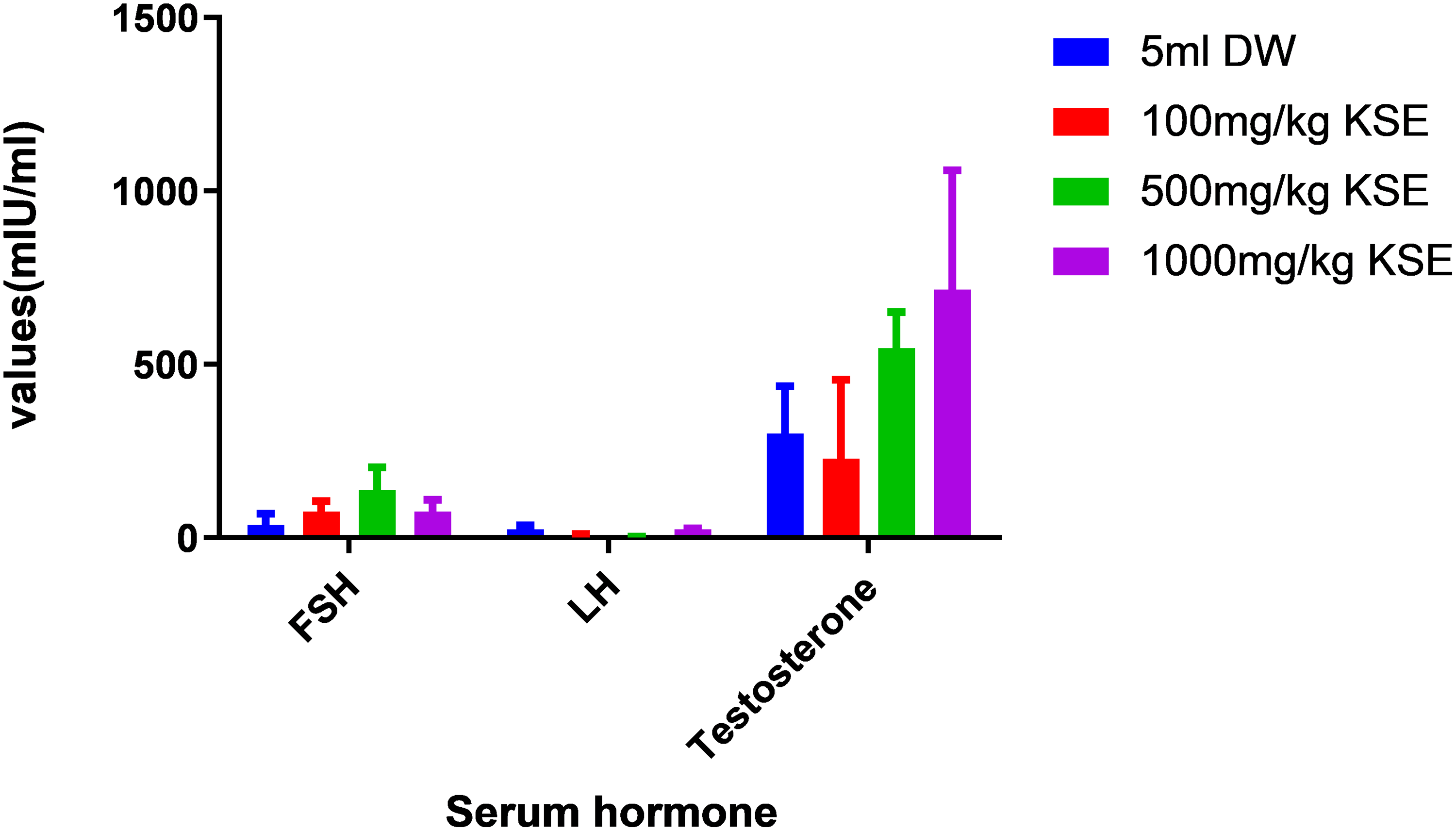
Effects of hydro-ethanolic KSE 100 mg/kg, 500 mg/kg and 1000 mg/kg, p.o. on serum hormones in male rats.

Effects of ethanolic KSE 100 mg/kg, 500 mg/kg and 1000 mg/kg, p.o. on Ex-vivo testosterone hormones in male rats.

Photomicrographs of H&E stained seminiferous tubule showing increased spermatogenesis distill water group.
Several xenobiotics disrupt the process of spermatogenesis by interfering with the stages. In this study, KSE appears to cause spermatogonia degeneration, fibrosis, and hyalinization of the seminiferous tubules in the testis at all dose levels compared to the control (Figures 7, 8 and 9). Sertoli cells and the Leydig cells were normal and not affected by KSE. The degeneration observed was in a dose-dependent manner. The highest (1000 mg/kg) received dose showed the severest form of degeneration as compared to the other dose group and the control which showed a mild form of degeneration (Figure 9). This result corroborates with 21 where stem bark extract of KSE could result in major organ damage.

Photomicrographs of H&E stained seminiferous tubule showing normal spermatogenesis in 100 mg/kg KSE treated group.
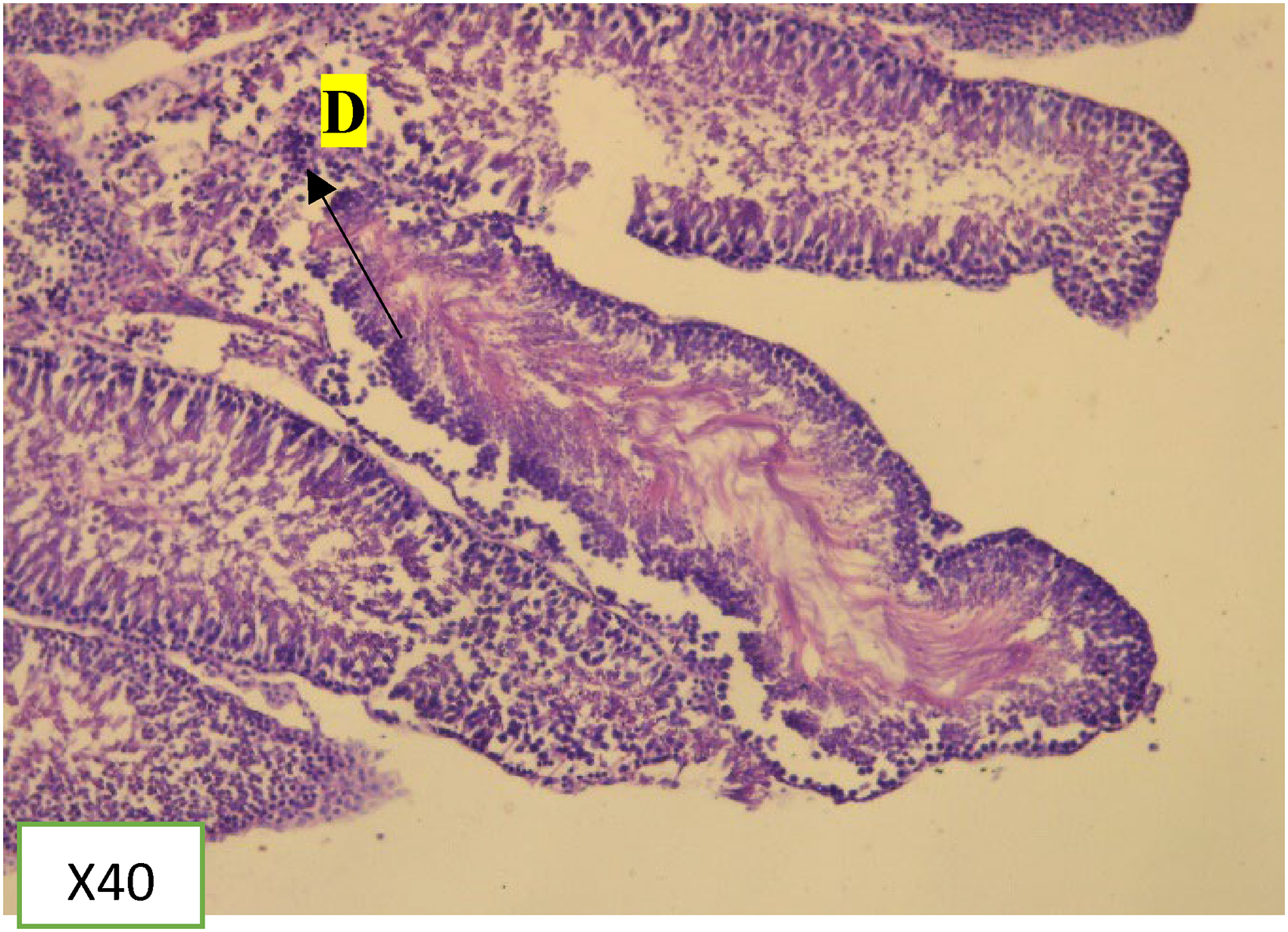
Photomicrographs of H&E stained seminiferous tubule showing reduced spermatogenesis KSE 500 mg/kg treated group.

Photomicrographs of H&E stained seminiferous tubule showing reduced spermatogenesis in KSE 1000 mg/kg treated group.
The distortion in the histoarchitecture of the testis is expected as reported by 21 that a dose of 1200 mg/kg, 1600 mg/kg, and 2000 mg/kg could cause massive distortions in the histoarchitecture of the testis in rats. Also, moderate degeneration which is occurring in the 1000 mg/kg dose group could be expected, because, some xenobiotics have been documented to increase the process of apoptosis in the reproductive organ. A. indica for instance is known for its antifertility effect and has been demonstrated in an in-vivo study that, the leaves has the potential to induce apoptosis in oocyte.22,23 In this study, the effect of KSE stem bark on the histoarchitecture of the testis could be due to an increase in ROS. Reactive Oxygen Species (ROS) is normally produced by sperm cells in the capacitation process and it is essential for sperm function. 24 The imbalance between the production of ROS and the scavenging abilities of antioxidants results in pathological conditions like degeneration and fibrosis. K. senegalensis leaves extract has been shown to increase total white blood cell, granulocytes, and lymphocyte counts. 25 As these cells are responsible for the inflammatory cytokines in damaged tissues, they may be responsible for the inflammation seen in the various dose groups.
Conclusion
The study suggests that K senegalensis can adversely affect the morphology of the testis in a dose-dependent manner. Users of the product should be interested in the dose they are consuming to reduce the unwanted effect such as testicular histoarchitecture distortions that could potentially lead to a decrease in fertility in the short term.
Limitations
The study did not look at the histoarchitecture of both the Sertoli and Leydig cell play a significant role in spermatogenesis and steroidogenesis respectively. Again, the study did not cover the effect of free radicals and their scavengers (antioxidants) which are equally important in evaluating sperm parameters.
Statement of Human and Animal Rights
All experimental procedures were carried out in accordance with the KNUST Animal Research Ethics Committed. The guideline outlined by the National Research Council for the Care and Use of Laboratory Animals was strictly adhered and efforts were taken to minimize the number of rats used but good enough to obtain data to make a valid conclusion. The study adhered to the ARRIVE guidelines (Animal Research: Reporting of in Vivo Experiments) in carrying out the work and reporting the findings.
Footnotes
Acknowledgment
We acknowledge the contribution of technical staff at the Pharmacology Department whose contribution greatly influenced the completion of this project.
Authors’ Contributions
All authors made a significant contribution to this study, whether that is in conception, data analysis or interpretation. All authors also took part in the drafting, revising, and gave approval for the publication of this manuscript.
Author Contributions
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The study received ethical approval from the Animal Research Ethics Committee of the Kwame Nkrumah University of Science and Technology (KNUST) with approval number KNUST/AREC/054.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Use of AI Tools Declaration
The authors declare they have not used Artificial Intelligence (AI) tools in the creation of this article.
