Objective: The genus Phrynium has medicinal value and is effective in relieving sore throat and mouth ulcers. However, the extraction and chemical analysis of essential oils from Phrynium tonkinense Gagnep, have not yet been reported. The present study aimed to extract, chemically characterize, and evaluate the antimicrobial properties of volatile oils from Phrynium tonkinense Gagnep (P. tonkinense). Methods: In the present study, essential oil was extracted from the leaves of P. tonkinense for the first time by the hydrodistillation method, and its chemical composition was determined by gas chromatography-flame ionization detection (GC-FID) and gas chromatography-mass spectrometry (GC-MS). The area normalization method was used to calculate the relative percentage content of each chemical constituent in the essential oil. Additionally, the antibacterial efficacy of the essential oil was tested against six pathogenic microorganisms by the agar diffusion method and the microdilution method. Result: The yield of essential oil obtained by hydrodistillation was 0.499%. Ninety volatile components were identified from the essential oil, with fatty acid compounds (47.36%) accounting for the largest proportion of these components. The main compounds were hexadecanoic acid (25.20%), (Z)-3-hexen-1-ol (10.31%), pentadecanoic acid (8.09%), and myristic acid (3.99%). The results of the in vitro antimicrobial assay showed that essential oils from P. tonkinense had a good inhibitory effect on the six selected pathogenic microorganisms and showed good bactericidal activity, with the minimum inhibitory concentration (MIC) value ranging from 31.3 to 250 μg/mL and the minimum bactericidal concentration (MBC) value ranging from 62.5 to 500 μg/mL. Notably, the essential oil exhibited the highest antibacterial activity against Staphylococcus aureus, with the MIC of 31.3 μg/mL and the MBC of 62.5 μg/mL. Conclusion: The findings of this study provided new insights into the medicinal functions of P. tonkinense and offered a scientific basis for the development and utilization of P. tonkinense as a natural antibacterial agent.
Introduction
Essential oil is generally a clarified, colorless to brownish oily liquid extracted from plants. It has a characteristic volatile aroma and is the most commonly used ingredient in natural flavor products.
1
The research indicated that most plant essential oils, as reported in previous studies, exhibited broad-spectrum bioactivities such as antibacterial, anti-inflammatory, antiviral, antioxidant, antitumor, and insecticidal properties, along with several other pharmacological effects.2–5 The bioactivities of plant essential oils are closely associated with their chemical composition.
6
For example, the essential oil extracted from the fruit of Amomum cannicarpum, was mainly composed of β-pinene and elemol, and it exhibited antibacterial activity against both Gram-positive and Gram-negative bacteria.
7
Hence, there has been a growing interest in the global study of essential oils to reveal their potential in medicinal and commercial applications.
The genus Phrynium, belonging to the family Marantaceae, comprises approximately 30 species in the world. These plants prefer shady, moist, and warm environments growing in tropical and subtropical rainforests.
8
Two species, Phrynium tonkinense Gagnep and Phrynium pubinerve Blume, mainly grow in China. Related studies have shown that plants of the genus Phrynium had biological effects,
9
such as relieving allergic inflammation,
10
controlling weeds,
11
inducing analgesic and antioxidant effects,12,13 repelling insects,
14
providing soothing effects,
15
and exhibiting antibacterial properties.
16
These plants can also be used to purify artificial ecological wetlands.17–19 Furthermore, as reported in the Chinese Pharmacopoeia, P. tonkinense had medicinal value and were effective in relieving sore throat and mouth ulcers.
20
It was also used as a material to wrap dumplings.21,22 However, to the best of our knowledge, there were still no reports in the literature of the extraction and chemical composition of essential oils from the P. tonkinense and their biological activities, which restricted the in-depth comprehensive medicinal application of P. tonkinense.
The present study determined the chemical composition and antimicrobial activity of essential oil extracted from P. tonkinense and may provide a theoretical basis for systematic research, rational development and utilization of P. tonkinense resources. Through further analysis and exploration, we hope to offer more scientific evidence for the application of essential oils, facilitating their use in the fields of pharmaceuticals and healthcare.
Materials and Methods
Plant Material and Microbial Strains
Plant material: P. tonkinense material was collected from Xishuangbanna State (21°41′N,101°25′E), Yunnan Province, China, in September 2022. The voucher specimens (No. 0179) were stored at the College of Tobacco Science and Engineering, Zhengzhou University of Light Industry, Zhengzhou, China.
Microbial strains: bacterial strains included Staphylococcus. aureus ATCC 25923, Bacillus cereus ATCC 10987, Escherichia coli ATCC 25922, and Pseudomonas aeruginosa ATCC 27853; fungal strains included Candida albicans ATCC 10231 and Aspergillus fumigatus ATCC 1022. All of above strains were purchased from the China Industrial Microbial Strain Preservation and Management Center.
Extraction of the Essential Oil
The collected materials of P. tonkinense were dried in a cool, shaded area at room temperature and then ground using a stainless grinder. After grinding, the mixture was sieved through a 40-mesh screen to obtain uniform and fine powders. According the method published in the 2020 edition of the Pharmacopoeia of the People's Republic of China,
23
and our laboratory methods in the previous studies with minor alteration, the essential oil of P. tonkinense was extracted from the powdered plant material through hydrodistillation using a Clavenger apparatus. Specifically, 25 g of the powder was accurately weighed and placed in a 1000 mL round-bottom flask, to which 400 mL of distilled water was added, maintaining a material-liquid ratio of 1:16. The upper end of the flask was connected to a volatile oil distillation apparatus and a reflux condenser to collect the extracted volatile oils. Then, the flask was heated using an electric heater, maintaining a gentle boil for 10 h until no further oil was collected in the receiver. After cooling, the volatile oils were collected, dried with anhydrous sodium sulfate, and stored in an amber glass bottle at 4 °C for further analysis.
24
Identification of the Chemical Components of the Essential Oil
The qualitative analysis of compounds in essential oils was conducted by GC-MS (8890A-5977B, Agilent Technologies, Santa Clara, CA, USA) equipped with a fused silica DB-5 ms capillary column (60 m × 250 μm × 0.25 μm). The following settings were used for the GC-MS instrument: injector temperature, 280 °C; carrier gas, high-purity helium; flow rate, 1.0 mL/min; injection mode, split mode; diversion ratio, 3:1; injection volume, 1 μL. The heating program was set as follows: (1) initial temperature was 50 °C and maintained for 3 min, (2) temperature was increased to 130 °C at the heating rate of 2 °C/min and maintained for 3 min, (3) temperature was further increased to 180 °C at the heating rate of 5 °C/min and maintained for 2 min, and (4) temperature was increased to 200 °C at the heating rate of 3 °C/min and maintained for 3 min, and finally increased to 280 °C at the heating rate of 1.5 °C/min and maintained for 5 min. MS was performed under the following conditions: SCAN mode, transmission line temperature: 280 °C, and ion source temperature: 230 °C, quadrupole temperature: 150 °C, ion source: electron bombardment ionization (EI), electron energy: 70 eV, solvent delay time: 10 min, and scanning range: 10∼800 amu.
The quantitative analysis of compounds in essential oils was performed using a 6890N device (Agilent Technologies) equipped with a flame ionization detector and electronic pressure control injector. The GC-FID analysis was conducted under the same experimental conditions using the same column as described for GC-MS. The relative percentages of components were calculated according to the peak areas by using the normalization method without correction factors. The analysis was performed in triplicate for the sample, and the final results were presented as average values. The components of essential oil were identified by matching retention times of available authentic standards, RIs, and mass spectra in the NIST 20 database.
Antimicrobial Activity Assay
The bacteria were inoculated into Nutrient Broth (NB) and incubated at 37 °C on a shaking incubator for 24 h. The fungi were inoculated onto Sabouraud Dextrose Agar (SDA) and incubated at 28 °C for 120 h.
Zone of Inhibition determination with the agar diffusion method: 0.1 mL of the tested strain suspension was evenly spread onto Nutrient Agar (NA) or SDA solid media, with a strain concentration of 106-107 CFU/mL. The essential oil was diluted with dimethyl sulfoxide (DMSO) to achieve a concentration of 1 mg/mL for in vitro bioassay. Next, 100 µL of the diluted essential oil was added to 6 mm wells created in agar plates coated with different test strains. The following antibiotics were used as positive controls: ampicillin for Gram-positive bacteria, gentamicin for Gram-negative bacteria, and amphotericin for the fungal and yeast strains. DMSO was used as the negative control. The antimicrobial activity was assessed by measuring the zone of inhibition diameter after incubation of the plates at 37 °C for 24 h under dark conditions. The zone of inhibition was measured in millimeters. The experiments were performed in triplicate.
MIC determination with the microdilution method: The MIC was calculated using the 96-well microdilution method. Two-fold serial dilutions of the essential oil were prepared in DMSO at concentrations ranging from 500 to 0.98 µg/mL and added to 96-well microtiter plates. This was followed by the addition of suspensions of the test strains to the plates containing varying concentrations of the essential oils. The plates were then incubated for 24 h at 37 °C and visually assessed for the presence of turbidity. The MIC value was determined by transferring 10 µL of the inoculum from the MIC plate to a nutrient agar plate, followed by incubation for 24 h at 37 °C. The minimum concentration of the essential oil that completely inhibited microbial growth was considered the MIC value. To calculate the MBC value, 10 µL of the inoculum from the MIC plate was transferred to a nutrient agar plate and incubated for 24 h at 37 °C. The concentration at which no visible bacterial growth was detected on the plate was considered the MBC value.
Statistical Analysis
The results of the antimicrobial activity tests were expressed as mean ± SD (n ≥ 3) and analyzed using R software version 4.3.1.
Results
Chemical Composition of the Essential Oil
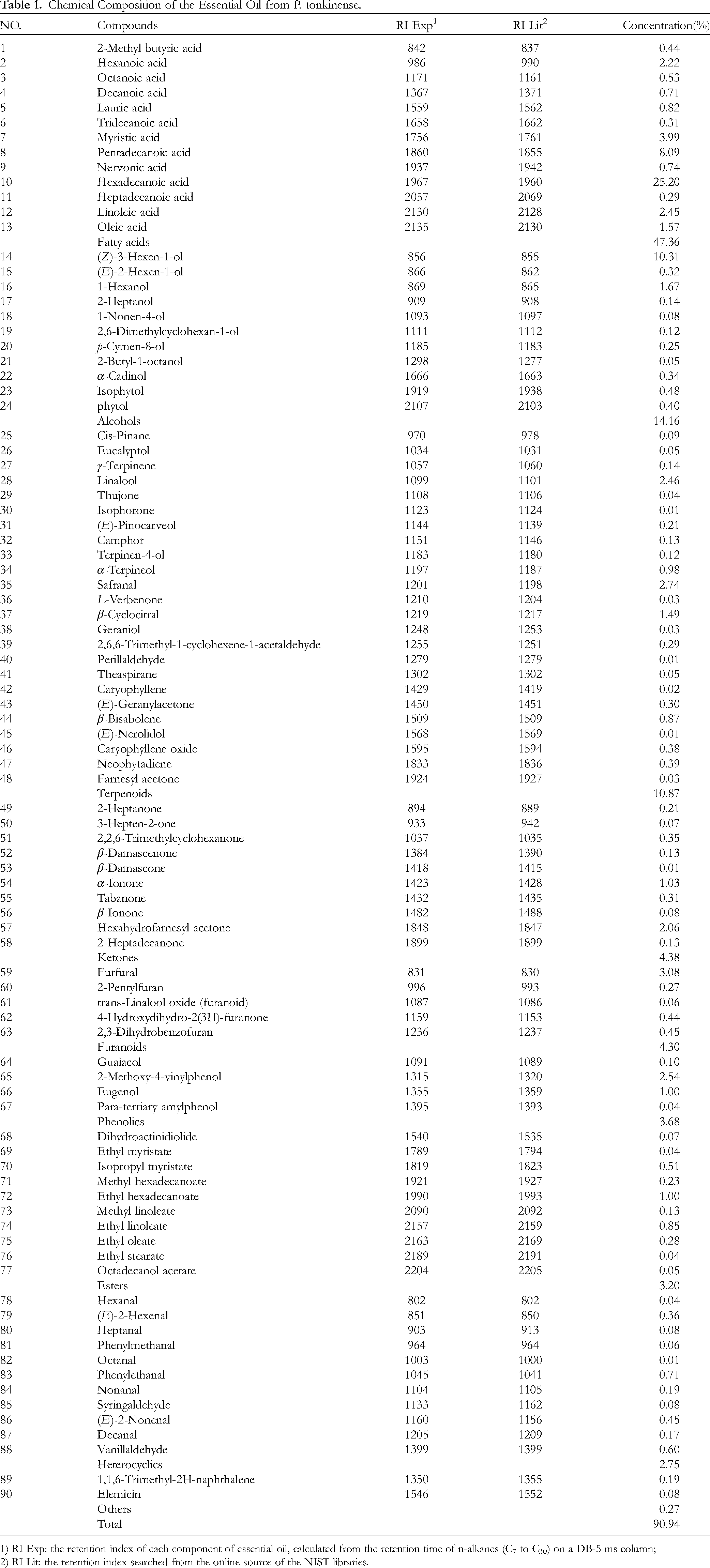
The yield of essential oil from P. tonkinense was 0.499%. (w/w relative to the weight of dry material). The composition of the essential oil was determined by GC-FID and GC-MS. 90 volatile compounds were identified, which accounted for 90.97% of the total oil. The volatile compounds were identified by matching their retention indices and retention times with those of the standards reported in the NIST20 database (Table 1). The essential oil is mainly composed of 13 fatty acids (47.36%), 11 alcohols (14.16%), 23 terpenoids (10.86%), 11 ketones (4.39%), 5 furanoids (4.30%), 4 phenolics (3.68%), 10 esters (3.2%), and 11 heterocyclics (2.75%). The major compounds included hexadecanoic acid (25.20%), (Z)-3-hexen-1-ol (10.31%), pentadecanoic acid (8.09%), and myristic acid (3.99%).
Antimicrobial Activity of the Essential Oil
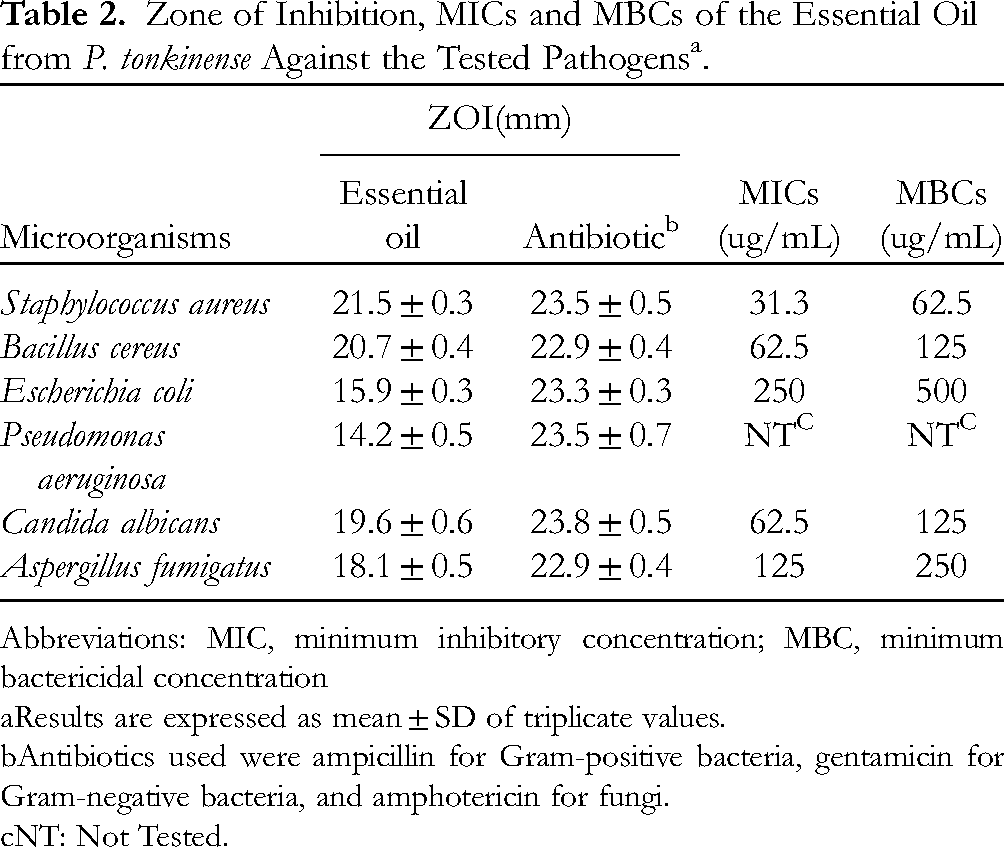
The agar diffusion assay and the microdilution method were used to determine the in vitro antimicrobial activity of the essential oil against six pathogenic microorganisms (Staphylococcus aureus, Bacillus cereus, Escherichia coli, Pseudomonas aeruginosa, Aspergillus fumigatus, and Candida albicans). Table 2 shows the diameter (mm) of the zone of inhibition and the minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of the essential oil against the tested species. The essential oil showed antimicrobial effects against all the six pathogenic species and formed a distinct zone of inhibition. The diameters of the zone of inhibition for S. aureus, B. cereus, C. albicans, and A. fumigatus were 21.5, 20.7, 19.6, and 18.1 mm, respectively. In the microdilution experiments, the essential oil had a good inhibitory effect on the six selected pathogenic microorganisms and showed good bactericidal activity, with the MIC value ranging from 31.3 to 250 μg/mL and the MBC value ranging from 62.5 to 500 μg/mL. It's worth noting that the essential oil exhibited the highest antimicrobial activity against S. aureus (MIC and MBC = 31.3 and 62.5 μg/mL, respectively), followed by that against B. cereus and C. albicans (MIC and MBC = 62.5 and 125 μg/mL, respectively). Moreover, the essential oils had a strong antibacterial effect on Gram-positive bacteria and fungi, while it had a weak antibacterial effect on Gram-negative bacteria.
Discussion
Ninety volatile compounds were identified in the essential oil. The major compounds included hexadecanoic acid (25.20%), (Z)-3-hexen-1-ol (10.31%), pentadecanoic acid (8.09%), and myristic acid (3.99%). Research shows that the major constituents for Phrynium pubinerve Blume were (Z)-3-hexen-1-ol (17.31%), (E)-2-hexenal (9.01%) and 1-hexanol (8.61%).
25
Higher levels of Phytol (11.40%) were detected in the ethanol extract of Phrynium capitatum.
12
These differences may be related to factors such as species diversity or extraction methods.
The strong antibacterial activity of the essential oil extracted from P. tonkinense is closely associated with its chemical composition. Research had found that medium-chain fatty acids (MCFAs), such as hexanoic acid, octanoic acid, decanoic acid, and lauric acid, exhibit potent antibacterial properties as they can penetrate bacterial semi-permeable membranes and disrupt internal cellular structures.
26
Additionally, hexadecanoic acid, tridecanoic acid, myristic acid, and linoleic acid had been reported to be effective against S. aureus.27–29 Among fatty acid esters, isopropyl myristate and ethyl myristate demonstrate antibacterial activity against P. aeruginosa and S. aureus, respectively.27,30
Terpenoids are the primary antimicrobial agents used against various microorganisms. They exerted antimicrobial effects through multiple mechanisms, including disruption of the cell membrane.
31
For example, nerolidol, linalool, and isophytol had shown antibacterial effects against Salmonella enterica, S. aureus, and Aspergillus niger.
32
Federico et al reported that compounds like eucalyptol, γ-terpinene, and terpinen-4-ol, individually, effectively inhibited the growth of C. albicans.
33
Other studies had reported that compounds like thujone and eucalyptol had antimicrobial properties against multiple bacterial strains.
34
Camphor had shown efficacy against S. aureus and Listeria monocytogenes.
35
Moreover, geraniol showed efficacy against E.coli, Helicobacter pylori, and S. aureus at higher concentrations.
36
Krist et al demonstrated that α-terpineol, linalool, and perillaldehyde exhibited antimicrobial activity against airborne microbes.
37
Thus, the combination of these biological compounds present in the essential oil of P. tonkinense may act collectively, causing antibacterial activity by synergistic effect, which is also the focus of our follow-up research work.
In the context of the increasing global challenge of antibiotic resistance, many common pathogens, such as Escherichia coli
38
and Staphylococcus aureus,
39
have demonstrated resistance to conventional antibiotics. For example, the emergence of methicillin-resistant Staphylococcus aureus (MRSA)
40
and extended-spectrum beta-lactamase-producing Escherichia coli strains
41
has become a significant challenge in clinical treatment. To explore the broad-spectrum antimicrobial properties of P. tonkinense essential oil and assess the potential for developing novel antimicrobial agents based on P. tonkinense,
42
this study selected clinically relevant pathogens that are responsible for various types of infections, including Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, and Candida albicans
43
for antimicrobial activity testing. The results revealed that compared to Gram-negative strains, essential oils of P. tonkinense exhibited better antibacterial effects on Gram-positive strains. This difference might be due to the double-layered cell membrane of Gram-negative bacteria.
44
According to previous studies,
45
there were significant differences in the structure and composition of the cell walls and cell membranes between Gram-positive and Gram-negative bacteria. In Gram-negative bacteria, the lipopolysaccharide-rich outer membrane constituted an effective barrier, which could prevent the diffusion of hydrophobic or many antibiotic molecules.
Limitations of the Study
Although essential oils are known to possess antimicrobial effects, the evaluation of their antimicrobial efficacy remains to be standardized, similar to that of antibiotics. Therefore, it is difficult to compare the antimicrobial activities of various essential oils.
46
The antibacterial activities of essential oils varies depending on their components and usually involves the synergistic action of major components, although some components seem to have independent effects. In our work, although the antibacterial efficacy of the essential oil was tested, the active compounds and underlying mechanisms have not been comprehensively and systematically investigated, and interrelationships of the various factors and their synergy has not been sufficiently quantified. These issues are also high priority for our subsequent research.
Conclusion
The present study reported for the first time on the chemical composition and antimicrobial activity of the essential oil from P. tonkinense. The main components of the essential oil were hexadecanoic acid, (Z)-3-hexen-1-ol, pentadecanoic acid, and myristic acid. Moreover, the essential oil showed promising antibacterial activities against S. aureus, B. cereus, C. albicans, and A. fumigatus. These results suggest that the essential oil of P. tonkinense may be served as a natural antimicrobial agent. Future studies should focus on elucidating the active ingredients and pharmacodynamic substance basis of antimicrobial activity, and the interactions between various compounds also need to be considered. Additionally, research should explore the development of P. tonkinense as a medicinal resource, based on its pharmacological effects, biological activities, and mechanisms of action.