Abstract
Objective
To investigate the impact of “Xinshuixiao” drug-containing serum on dDAVP-induced AQP2 expression in IMCD3 cells and to preliminarily explore its underlying mechanisms.
Methods
Rats were orally administered with “Xinshuixiao granules” containing medicinal compounds. The CCK-8 assay was employed to determine the appropriate drug serum concentration and its effect on IMCD3 cell viability. Additionally, we assessed the influence of “Xinshuixiao” drug-containing serum on IMCD3 cell activity following dDAVP intervention. Cells were divided into blank, model, low, medium, and high-dose “Xinshuixiao” drug serum groups, as well as a Tolvaptan group. After 24 h of drug treatment, ELISA was used to measure the levels of arginine vasopressin (AVP), cyclic adenosine monophosphate (cAMP), and protein kinase A (PKA) in the cell supernatant. Western blotting was employed to evaluate AQP2 protein expression, and real-time polymerase chain reaction (Real-time PCR) was used to assess AQP2 mRNA expression levels.
Results
In comparison to the blank group, the model group exhibited elevated expression of AVP, cAMP, PKA (P<0.05), as well as increased AQP2 mRNA and protein expression (P<0.05). When compared to the model group, all treatment groups displayed varying degrees of reductions in AVP, cAMP, and PKA expression (P<0.05), along with decreased AQP2 mRNA and protein expression, with statistically significant differences (P<0.05).
Conclusion
“Xinshuixiao” drug-containing serum exerts an inhibitory effect on the overexpression of AQP2 induced by dDAVP in IMCD3 cells, which may be associated with its suppression of the cAMP/PKA signaling pathway.
Introduction
Heart failure represents the ultimate stage of cardiovascular disease, and its prevalence has been steadily increasing in the context of both aging populations and rising cardiovascular disease incidence. Epidemiological data indicate that the prevalence of heart failure among adults in developed nations ranges from approximately 1% to 2%. 1 In China, an estimated 4 million individuals aged 35 to 74 years are currently living with heart failure. 2 Furthermore, with advancing age, the mortality rate associated with heart failure significantly escalates. Fluid retention stands as a pivotal pathophysiological alteration in chronic heart failure and is a primary mechanism underlying congestive heart failure. The judicious use of diuretics plays a paramount role in heart failure management, and diuretics should be prescribed to all heart failure patients with evidence of fluid retention. However, the emergence of diuretic resistance (DR) has impeded effective heart failure control and the amelioration of sodium and water retention. DR 3 is defined as a phenomenon wherein diuretics’ natriuretic and diuretic effects diminish or disappear following administration, or even when maximal diuretic doses are employed, normalizing blood volume remains elusive. DR occurs in approximately 20% to 35% of cases, and it significantly elevates mortality rates in patients with delayed diuretic responses.
Aquaporin 2 (AQP2) plays a pivotal role in regulating water balance within the kidneys and serves as a fundamental molecular component of fluid metabolism. Its primary regulation is orchestrated by arginine vasopressin (AVP). AVP binds to the vasopressin type 2 receptor (V2R) situated on the basolateral membrane of principal cells in the renal collecting ducts, thereby activating cyclic adenosine monophosphate (cAMP). This activation leads to the phosphorylation and/or upregulation of AQP2, subsequently enhancing water reabsorption. An aberrant increase in AQP2 levels represents a significant factor contributing to fluid retention.4,5 Consequently, AQP2 has emerged as a critical therapeutic target in the management of fluid imbalance, garnering considerable attention.
Current medical approaches in modern medicine to ameliorate Diuretic Resistance (DR) primarily involve escalating diuretic dosages, combination therapies with other drugs, or techniques such as ultrafiltration. However, these strategies have limitations, and a definitive treatment protocol remains elusive.3,6,7 Traditional Chinese Medicine (TCM) has emerged in recent years as a unique and advantageous avenue for addressing D.Renowned TCM expert, Professor Yu Kaicheng, has proposed a TCM pathogenesis model for late-stage heart failure, characterized by “renal essence deficiency” and “cardiac insufficiency.” He has applied the empirically formulated “Xinshuixiao Granules” to treat large number of late-stage heart failure patients. ‘Xinshuixiao Granules’ is mainly composed of Astragali Radix., Drabaemerosa hebecarpa, Cornus officinalis Sieb. et Zucc., Ophiopogonis, Dilophylaxis, Rhizoma Ligustici Chuanxiong, Radix et Rhizoma Ginseng, and Radix et Rhizoma Dioscoreae. Radix Astragali and its main active constituents (AS-IV and CAG) improve liver regeneration by upregulating the expression of AQP9 in hepatocytes to increase gluconeogenesis and reduce oxidative stress. 8 Cornel iridoid glycoside (CIG), the main component of Cornus officinalis Sieb. et Zucc. could attenuate brain edema of the cerebral ischemia/reperfusion rats by modulating the polarized Aqp4 through the interaction of AQP4-M23 with morroniside and loganin. 9 2,3,5,6-Tetramethylpyrazine (TMP) as an active ingredient extracted from a traditional Chinese herbal medicine Ligusticum chuanxiong Hort. inhibits the overexpression of AQP4 and Cx43 in astrocytes and activates the FGF2/PI3K/AKT pathway to promote neurovascular repair following ischemic strokeinhibits the overexpression of AQP4 and Cx43 in astrocytes and activates the FGF2/PI3K/AKT pathway to promote neurovascular repair following ischemic stroke. 10 To further elucidate the mechanisms underlying the efficacy of “Xinshuixiao Granules” in improving DR among heart failure patients, this study, with reference to the method proposed by Hiroko Iwama et al,11–13 we collected drug-containing serum from rats and cultured mouse renal inner medullary collecting duct 3 epithelial cells through and in vitro. It aims to investigate the influence of “Xinshuixiao” drug-containing serum at varying concentrations on the expression of Aquaporin 2 (AQP2), cAMP, and protein kinase A (PKA) in IMCD3 cells induced by desmopressin (dDAVP). This research seeks to unravel the mechanisms through which “Xinshuixiao” drug-containing serum impacts AQP2 expression and improves water metabolism.
Materials and Methods
Animal and Cells
A total of 30 specific-pathogen-free (SPF) male Sprague-Dawley (SD) rats, aged 8 weeks, with body weights ranging from 220 to 250 grams, were procured from Changchun Yisi Experimental Animal Technology Co., Ltd The experimental animal permit number is SYXK (Ji) 2018–0014. Laboratory facility usage was granted under certificate number SCXK (Ji) 2020–0002. This study received ethical approval from the Changchun University of Traditional Chinese Medicine Animal Ethics Committee. IMCD3 cells derived from murine kidneys were obtained from Shanghai Fuheng Biotechnology Co., Ld
Pharmaceutical Sources
The “Xinshuixiao Granules,” composed of Astragali Radix, Lepidium apetalum Willd, Cornus officinalis Sieb. et Zucc., Ophiopogon japonicus (L.f)Ker-Gawl., Pheretima, Ligusticum chuanxiong Hort., Codonopsis pilosula (Franch.)Nannf.and Dioscorea opposita Thunb., were procured from the Third Affiliated Clinical Hospital of Changchun University of Traditional Chinese Medicine. dDAVP and Tolvaptan were both acquired from MCE (MedChemExpress) Company.
Reagents
IMCD3 complete culture medium and serum-free cell freezing solution (Shanghai Fuheng);Trypsin from Gibco;Dimethyl sulfoxide (Sigma);Cell counting kit-8 from MCE (MedChemExpress);cAMP, AVP, and PKA assay kits (Jiangsu Jingmei Bio);Trizol and reverse transcription kit (Tiangen);PCR primers (Shanghai Shenggong);Protein Marker, PVDF membrane, electrophoresis buffer, TBS buffer, and phosphate-buffered solution (Solarbio);AQP2 primary antibody, β-Actin, goat anti-rabbit IgG, goat anti-mouse IgG (Abcam, UK);BCA protein concentration assay kit (Beyotime).
Instruments
CO2 incubator and enzyme-linked immunosorbent assay (ELISA) reader (Thermo Fisher Scientific, USA);High-speed refrigerated centrifuge (Eppendorf, Germany);Clean bench (Suzhou Purification Equipment Co., Ltd);Forced air drying oven (Shanghai Boxun Medical Biological Instrument Corporation);Protein electrophoresis system, transfer apparatus, and real-time fluorescence quantitative PCR instrument (Bio-Rad, USA);Inverted phase contrast microscope (Olympus, Japan);Chemiluminescence gel imaging system (ProteinSimple, USA).
Preparation of Drug-Containing serum
Thirty specific-pathogen-free (SPF) male Sprague-Dawley (SD) rats were randomly divided into two groups: the control group and the “ Xinshuixiao granules ” group, with each group consisting of 15 rats. After 7 days of acclimatization feeding, the “ Xinshuixiao granules ” group was orally gavaged with a clinically equivalent dose of 15.75 g kg−1·d−1, while the control group received an equal volume of physiological saline, administered twice daily for 7 consecutive days. Following the approach of Shasha Shi et al 14 Briefly, an hour after the final gavage, the rats were anesthetized by sodium pentobarbital, and blood samples were collected from the abdominal aorta. After standing at room temperature for 4 h, the serum was separated by centrifugation at 3000 rpm for 15 min. The supernatant was collected, heat-inactivated at 56°C for 30 min, aseptically filtered through a 0.22 μm microporous membrane in a clean bench, and then stored at −80°C for later use (Figure S1).
IMCD3 Cell Culture
Mouse inner medullary collecting duct (IMCD3) cells were seeded in T25 cm 2 culture flasks with 5 mL of complete culture medium. The flasks were incubated at 37°C in a 5% CO2 cell culture incubator for 24 h. Experiments were conducted using cells in the logarithmic growth phase.
CCK-8 Cell Viability Assay
Cells in the logarithmic growth phase were collected after trypsin digestion, counted, and adjusted to a density of 2×104 cells/mL. They were seeded in 96-well microplates with 5 replicate wells for each group. After cell attachment, different concentrations of drug-containing serum (0%, 5%, 10%, 15%, 20%, 25%, 30%) were added for 24 h. Following incubation, 10 μL of CCK-8 solution was added to each well, and after 2 h, the culture was terminated. Absorbance (OD value) at 450 nm was measured using a microplate reader. Different concentrations of dDAVP (10−5 to 10−12 mol/L, 10-fold dilution gradient) were added to each well, and cell viability was determined as described above.
Preparation of Experimental Drugs
Dissolve 10 mg of dDAVP in ultrapure water, and volume to 10 ml to make a 1 mg/ml dDAVP reservoir. This stock solution was further divided into 1.5 mL Eppendorf tubes and stored at −20°C. A 2 mM stock solution of tolvaptan was prepared by dissolving 5 mg of tolvaptan in 5.6 mL of dimethyl sulfoxide (DMSO). This stock solution was also aliquoted into 1.5 mL Eppendorf tubes and stored at −20°C. Prior to use, the drug stock solutions were diluted with complete culture medium as needed.
IMCD3 Cell Grouping and Interventions
IMCD3 cells were treated with complete culture medium containing 10−9 mol/L dDAVP for 24 h to establish the model. IMCD3 cells were seeded in six-well plates at a density of 1×105 cells per well and divided into the following groups: Blank control group (C), dDAVP model group (M), dDAVP + different concentrations of drug-containing serum groups (10%, 15%, 20%), and dDAVP + tolvaptan for subsequent research. The blank control group received no treatment and was cultured with normal complete culture medium. The remaining groups were induced with dDAVP, and after 24 h, the model group received normal culture medium, while the drug-containing serum and tolvaptan groups continued to be cultured with different concentrations of drug-containing serum and 10−6 mol/L tolvaptan for 24, 48, or 72 h.
ELISA Detection of AVP, cAMP, and PKA Expression in IMCD3 Cells
Cells were collected after trypsin digestion, centrifuged at 1000 rpm for 5 min, and the supernatant was aspirated and stored at −80°C. Relevant indicators were measured following the instructions of the assay kits.
Western Blot Analysis of AQP2 Protein Expression in IMCD3 Cells
After the completion of cell interventions, the supernatant was removed from each treatment group, and 1 mL per well of pre-chilled PBS was added, followed by two washes. RIPA cell lysis buffer was added, and the cells were repeatedly pipetted and mixed. Protein extraction was performed on ice for 20 min, followed by cell scraping and centrifugation at 12,000 rpm for 20 min at 4°C. The supernatant was collected for BCA protein quantification. A 10% SDS-PAGE separation gel and a 5% stacking gel were prepared. A total of 40 μg of total protein was loaded, and electrophoresis was performed under constant voltage (80 V for stacking gel, approximately 30 min; 110 V for separation gel, approximately 90 min). Wet transfer was conducted onto a polyvinylidene fluoride (PVDF) membrane for 1.5 h. After blocking with 5% BSA-TBST for 1 h at room temperature, primary antibodies (AQP2, 1:2000, β-actin, 1:5000) were added and incubated overnight at 4 °C with gentle shaking. Following three washes with TBST for 10 min each, secondary antibodies (dilution 1:10,000) were added and incubated for 1 h at room temperature, followed by three washes with TBST for 10 min each. ECL chemiluminescence was used for band visualization, and Image J software was used for band grayscale analysis.
RT-PCR Detection of AQP2 mRNA Expression in IMCD3 Cells
Total RNA was extracted using the Trizol method, and RNA concentration, purity, and integrity were assessed. Reverse transcription of cDNA was performed using total mRNA as a template, following the manufacturer's instructions. Primers were designed using Primer Premier 6.0 and synthesized by Shanghai Sangon Biotech. Primer sequences are shown in Table 1.
Real-time PCR reactions were carried out, and each sample was detected in triplicate. The relative expression of the target gene was calculated according to the 2−ΔΔCt formula.
Statistical Methods
Using GraphPad Prism 9.0 software, the data were analyzed using a one-way ANOVA followed by a post hoc Tukey test, with statistical significance set at P < 0.05.
Results
Effect of “Xinshuixiao” Drug-Containing Serum on IMCD3 Cell Proliferation
As illustrated in Figure 1, utilizing CCK8 analysis, it was observed that, in comparison to the control group, there were no statistically significant differences in the cell viability of IMCD3 cells when exposed to “Xinshuixiao” drug-containing serum at concentrations of 5%, 10%, 15%, and 20% (P > 0.05). However, as the concentration of “Xinshuixiao” drug-containing serum increased to 20% and 25%, there was a pronounced inhibition of cell activity, resulting in a noteworthy decrease in cell viability compared to the control group (P < 0.05). Consequently, this study elected to employ concentrations of 5%, 10%, and 15% “Xinshuixiao” drug-containing serum as low, medium, and high doses for subsequent experiments.
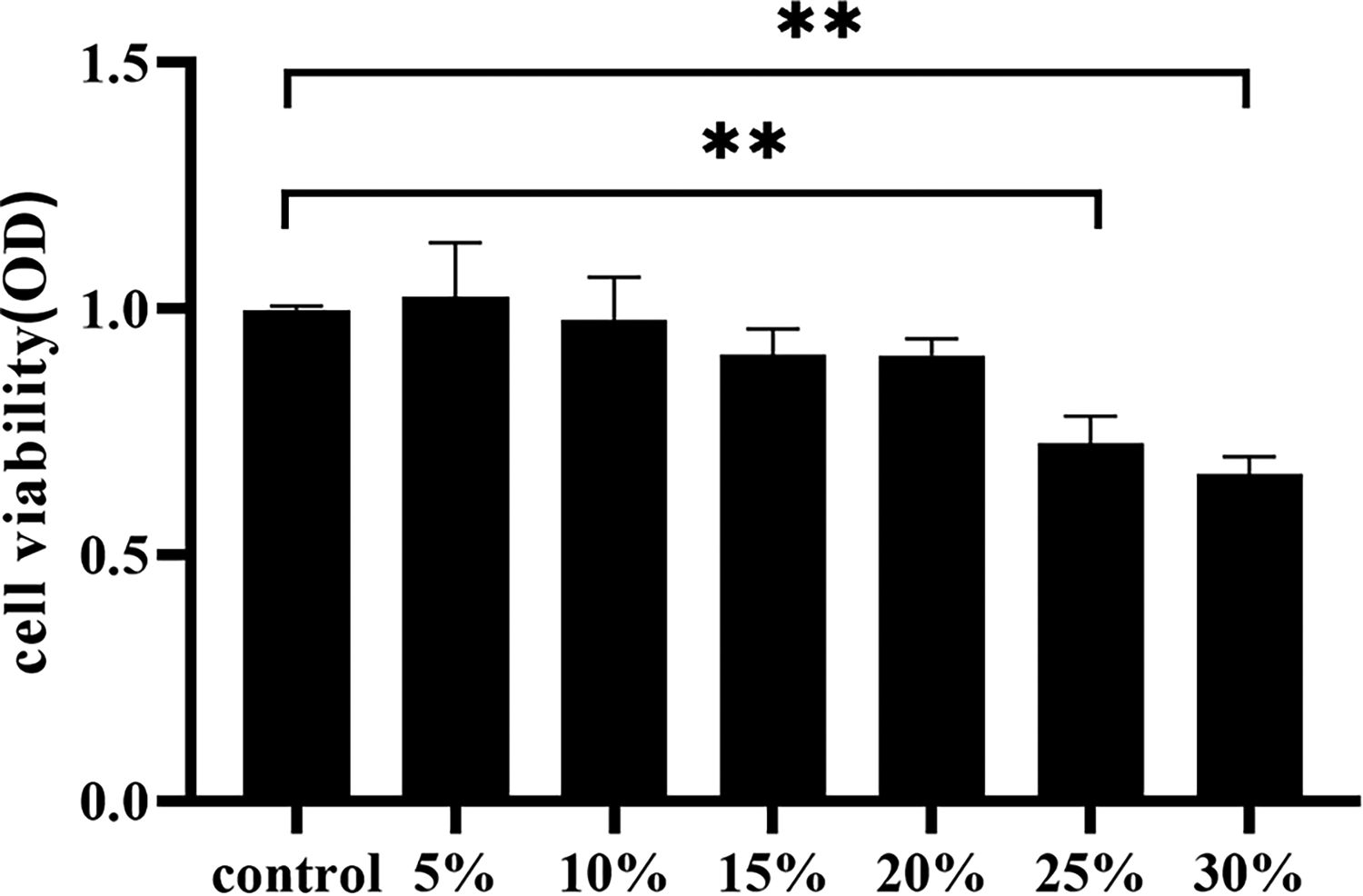
Comparative analysis of IMCD3 cell viability with varying concentrations of “Xinshuixiao” herbal serum. Penta-holes were used for each test. Data are expressed as mean ± SD. An asterisk indicates significant difference (P< 0.05).
As illustrated in Figure 2, CCK8 analysis revealed that different concentrations of dDAVP (ranging from 10−5 to 10−12 mol/L with a tenfold gradient) had no statistically significant impact on IMCD3 cell activity after a 24-h intervention (P > 0.05). Consequently, all concentrations of dDAVP within this range were considered suitable for subsequent experiments. For experimental convenience, 10−9 mol/L was ultimately chosen as the optimal intervention concentration for dDAVP.

Impact of different concentrations of dDAVP on cellular activity. Penta-holes were used for each test. Data are expressed as mean ± SD. An asterisk indicates significant difference (P < 0.05).
As depicted in Figure 3, when compared to the control group, the 10−9 mol/L concentration of dDAVP did not exhibit statistically significant differences in IMCD3 cell viability (P > 0.05). Likewise, when compared to the control group, the 5%, 10%, and 15% “Xinshuixiao” drug-containing serum did not show statistically significant differences in cell viability after dDAVP intervention (P > 0.05).

Influence of “Xinshuixiao” herbal serum on IMCD3 cell viability following dDAVP intervention. Penta-holes were used for each test. Data are expressed as mean ± SD. An asterisk indicates significant difference (P < 0.05).
As demonstrated in Figures 4, 5, and 6, in comparison to the control group, the model group exhibited a significant increase in AVP, cAMP, and PKA levels, with statistical significance (P < 0.05). Conversely, when compared to the model group, the “Xinshuixiao” low, medium, high-dose groups, and the tolvaptan group showed a marked reduction in AVP, cAMP, and PKA levels (P < 0.05), with statistically significant differences.

Effect of “Xinshuixiao” herbal serum on AVP levels. Triplicate wells were used for each test. Data are expressed as mean ± SD. An asterisk indicates significant difference (P < 0.05).

Modulation of cAMP levels by “Xinshuixiao” herbal serum. Triplicate wells were used for each test. Data are expressed as mean ± SD. An asterisk indicates significant difference (P < 0.05).
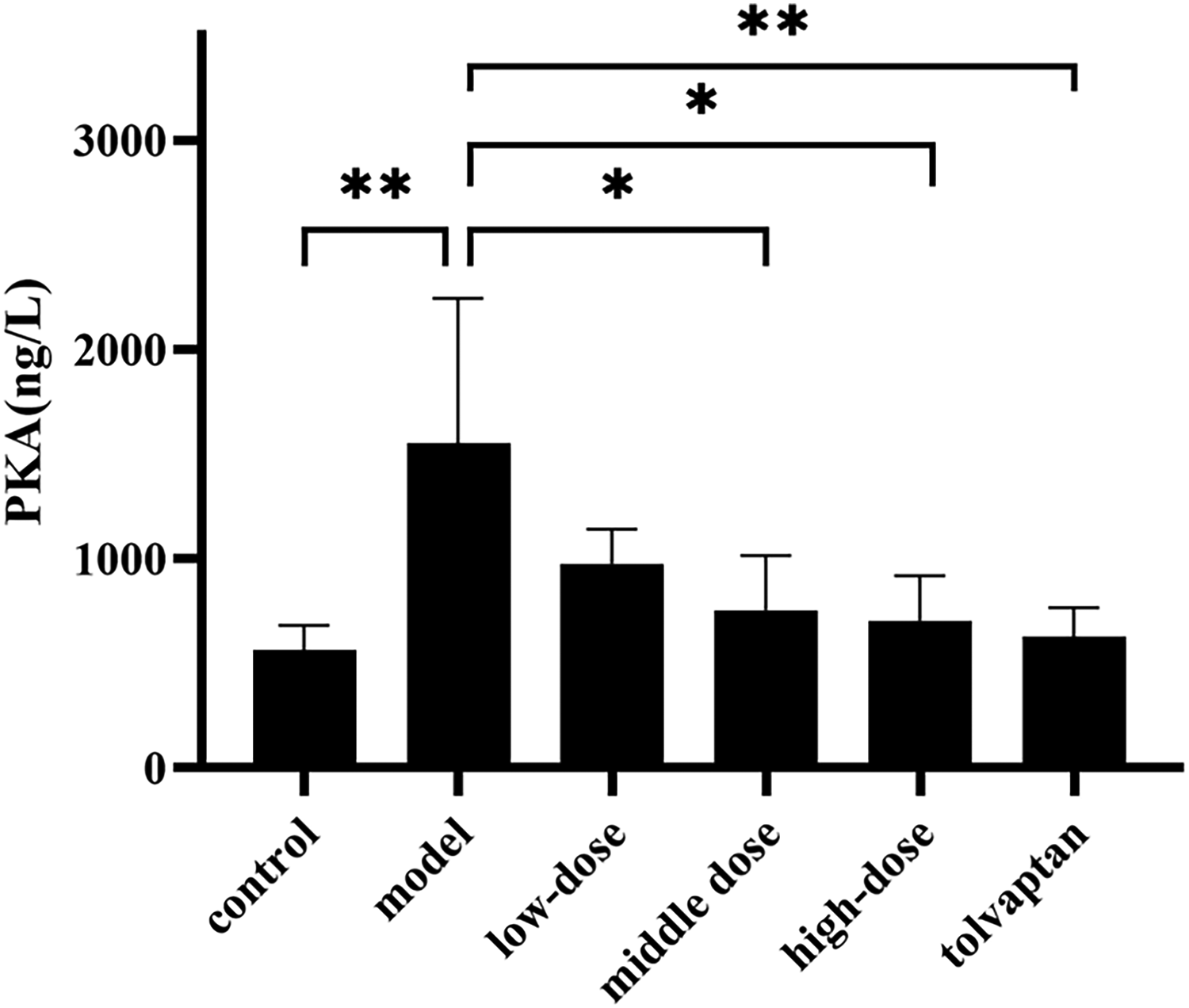
Impact of “Xinshuixiao” herbal serum on PKA activity. Triplicate wells were used for each test. Data are expressed as mean ± SD. An asterisk indicates significant difference (P < 0.05).
As illustrated in Figure 7, when compared to the control group, the model group exhibited an increase in AQP2 protein expression (P < 0.05). Conversely, when compared to the model group, all treatment groups demonstrated a decrease in AQP2 protein expression, with statistically significant differences (P < 0.05).

Alteration of AQP2 protein expression by “Xinshuixiao” herbal serum. Repeat the test three times. Data are expressed as mean ± SD. An asterisk indicates significant difference (P < 0.05).
As presented in Figure 8, when compared to the control group, the model group displayed an elevation in AQP2 mRNA expression (P < 0.05). Conversely, when compared to the model group, all treatment groups showed a reduction in AQP2 mRNA expression (P < 0.05).
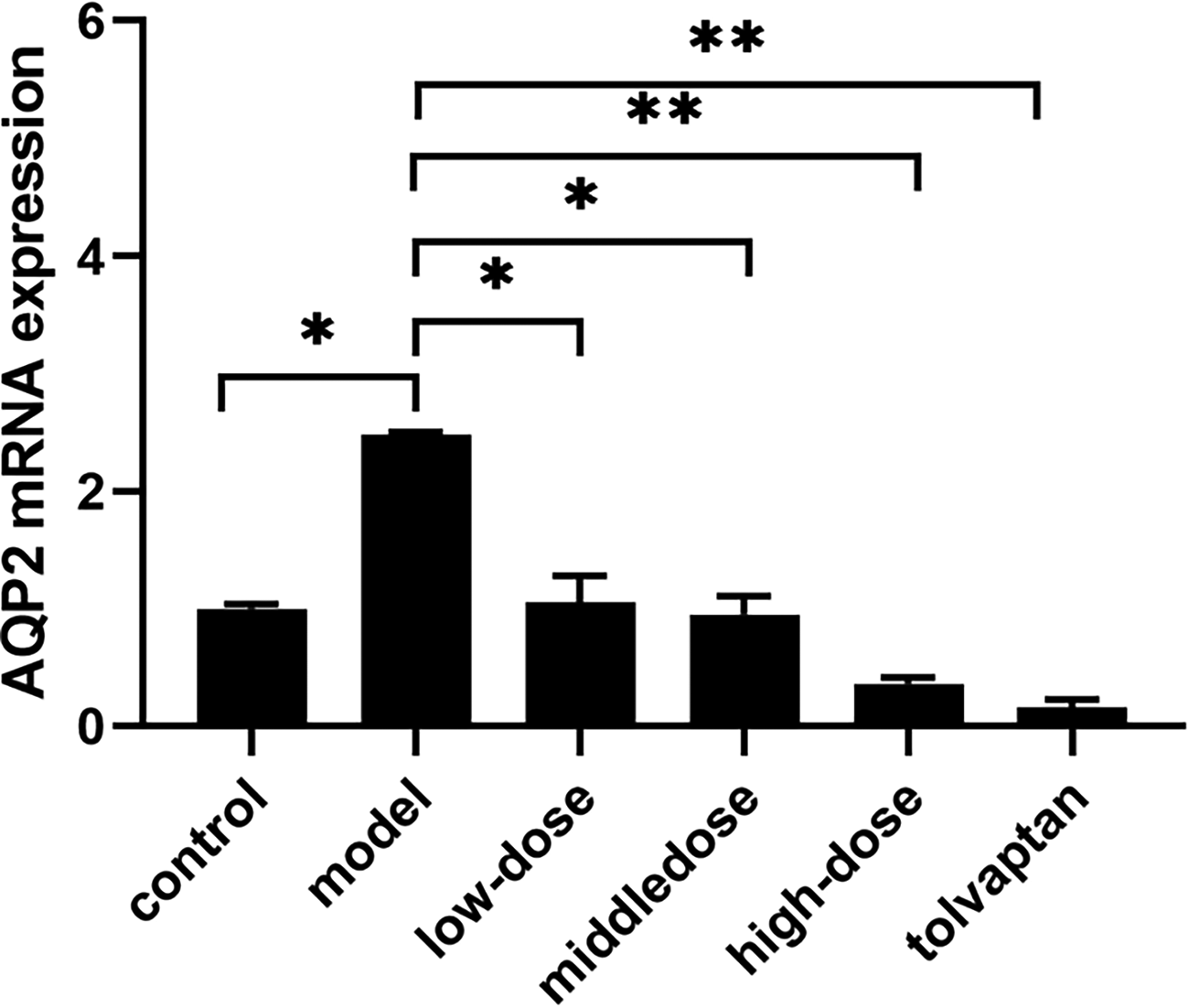
Modulation of AQP2 mRNA expression by “Xinshuixiao” herbal serum. mRNA expression was evaluated by quantitative RT-PCR; values were normalized using the 2−ΔΔCT method and were reported as mean expression ± SD. Triplicate wells were used for each test. An asterisk indicates significant difference (P < 0.05).
Heart failure (HF) is a clinical syndrome characterized by impaired cardiac function, reduced ejection fraction, and insufficient perfusion of various organ systems. Its primary symptoms include breathlessness, edema, and fatigue. Diuretics, as a cornerstone of heart failure treatment, alleviate water and sodium retention, improving clinical symptoms. However, with disease progression, Diuretic Resistance (DR) becomes a frequent concern. Modern medicine attributes the mechanism of DR to multiple factors, including changes in diuretic pharmacodynamics, alterations in renal tubular function, decreased renal blood flow, and conditions like hyponatremia and hypoalbuminemia.15,16
TCM lacks a specific concept of D.Based on symptoms such as shortness of breath, a heavy sensation in the body, inability to lie down, palpitations, restlessness, and abdominal fullness, which often accompany heart failure and DR, these can be attributed to various TCM conditions like “Heart Qi Deficiency,” “Heart Distention,” “Heart Cough,” “Asthma Patterns,” and “Edema.” Most TCM scholars believe that this condition is primarily characterized by “virtual” and “real” elements. The “virtual” aspect mainly involves Qi deficiency and Yang deficiency, while the “real” elements pertain to water retention, blood stasis, and phlegm accumulation, which may coexist. Professor Yu Kai-Cheng, on the other hand, emphasizes the close relationship between the heart and kidneys. As the kidneys govern water, their Yang plays a crucial role in fluid metabolism. He proposed the academic concept of “nourishing the kidneys to strengthen the heart” and developed the “Xinshuixiao granules” based on this principle. This formulation consists of seven Chinese herbal ingredients, including Astragali Radix., Cornus officinalis Sieb. et Zucc., Dioscoreae Rhizoma, Lepidium apetalum Willd, Ophiopogon japonicus (L.f)Ker-Gawl., Ligusticum chuanxiong Hort., and Dioscoreae Rhizoma. It is tailored in clinical practice according to syndrome differentiation. Astragali Radix., as the monarch herb in this formula, supplements Qi, promotes Qi circulation, and replenishes insufficient central Qi. It circulates bodily fluids throughout the body. It is reminiscent of Zhang Bing-Cheng's “Ben Cao Bian Du,” which states that “Astragalus supplementation excels at reaching the surface, boosting the defense, warming the muscles, and enriching the hair follicles. It invigorates Yang Qi and facilitates its smooth circulation.” Astragali Radix. also aids in diuresis and edema reduction. HuangQi injection, the top-selling among TCM injections, is made of AR aqueous extract. It has an indication for the treatment of viral myocarditis and cardiac insufficiency in the clinical practice. It can also improve myocardial contractility, boost circulation and preserve myocardial cells from failure. 17 Astragaloside IV, one of the major compounds in the aqueous extract of Astragali Radix., is a cyclic artemisinane-type triterpene glycoside chemical. Several cellular and animal model studies have shown that AS has potent protective effects in cardiovascular, lung, kidney and brain. 18 Astragalus polysaccharides can regulate the miR-16/NF-κB signaling pathway, thereby affecting the levels of MDR1 and P-gp170, and reducing the inflammation in the kidney tissues in the adriamycin nephropathy rats. 19 Cornus officinalis Sieb. et Zucc. and Dioscoreae Rhizoma serve as ministers, with Cornus officinalis Sieb. et Zucc. being sour and astringent in taste and slightly warm in nature. It possesses strong astringent properties, reinforcing Kidney Yang, nourishing Qi and Blood, and promoting diuresis and cardiac strengthening.Corni Fructus inhibits ECM deposition and TGF-β1/Smad signaling by inhibiting the activity of NF-κB through inhibition of oxidative stress in the UUO. We found that CF impacted renal fibrosis and inflammation in a rat model of UUO. 20 As active constituents of CF, 21 Loganin and morroniside are being actively studied for their renoprotective effects in diabetes.22–24 loganin protects the heart from I/R injury by inhibiting I/R-induced oxidative stress and cellular apoptosis via activation of JAK2/STAT3 signaling. 25 Total triterpene acids, active ingredients from Cornus officinalis Sieb. et Zucc., attenuate diabetic cardiomyopathy by normalizing ET pathway and expression of FKBP12.6 and SERCA2a in streptozotocin-rats. 26 Dioscoreae Rhizoma has a sweet taste, and it is mildly warm and balanced. It nourishes Yin, eliminates cold and heat pathogenic factors, tonifies the spleen, and supplements Qi and Yang. It enhances muscle growth, strengthens Yin, soothes the soul, and replenishes deficient heart Qi, opening the heart orifices, and reinforcing Kidney Qi. Dioscoreae Rhizoma polysaccharides reduce cerebral infarct size, up-regulate antioxidant enzyme activities, down-regulate inflammatory factors, and also affect the activation of the Nrf2/HO-1 signaling pathway, possibly by modulating the expression of CaMMKβ, which has a pharmacodynamic effect on IR injury. 27 Total saponin of Dioscoreae hypoglaucae rhizoma ameliorates glomerular and tubular pathologic changes in diabetic rats Nephroprotective effect in experimental diabetic nephropathy 28 Lepidium apetalum Willd and Ophiopogon japonicus (L.f)Ker-Gawl. serve as assistants, with Lepidium apetalum Willd facilitating lung Qi descent, resolving phlegm, calming wheezing, and promoting diuresis and edema reduction.2-phenylacetamide Separated from the seed of Lepidium apetalum Willd. inhibited renal fibrosis via MAPK pathway mediated RAAS and oxidative stress in SHR Rats. 29 Cis-desulfoglucotropaeolin (cis-DG) and trans-desulfoglucotropaeolin (trans-DG) were isolated from Lepidium apetalum Willd, which could protect NRK52e cells from hypertonicity-induced adhesion and inflammation by inhibiting the MAPK signaling pathway and RAAS. 30 Ophiopogon japonicus (L.f)Ker-Gawl. nourishes Lung and Kidney Yin, clears heart and liver heat, and harmonizes Yin and Yang. The polysaccharide (OJP1), extracted from the root of Ophiopogon japonicus (L.f)Ker-Gawl., may produce significant cardioprotective effects against ISO-induced damage by potentiating endogenous antioxidants. 31 Ligusticum chuanxiong Hort. And Dioscoreae Rhizoma are envoys in the formula, promoting blood circulation, Qi flow, clearing heat, and stopping wheezing. Ligustrazine (2,3,5,6-tetramethylpyrazine) is a major active ingredient of the Szechwan lovage rhizome and is extensively used in treatment of ischemic cerebrovascular disease. 32 Liguzinediol, 2, 5-dimethyl-3, 6-dimethyl-pyrazine, is a compound synthesized after the structural modification of ligustrazine (one active ingredient of Szechwan Lovage Rhizome), which could alleviate HF caused by MI in rats, and the protective effect was associated with the regulation of the TGF-β1/Smads pathway. 33 Water extract of earthworms mitigates kidney injury triggered by oxidative stress via activating intrarenal Sirt1/Nrf2 cascade and ameliorating mitochondrial damage. 34 The entire formula, through the dual reinforcement of Yin and Yang, stabilizes the foundation and fosters the origin, eliminating Qi stagnation, blood stasis, and water retention – common manifestations of this condition. Moreover, modern pharmacological research suggests that kidney-tonifying Chinese herbs possess heart-strengthening, diuretic, immune-enhancing, and blood pressure-regulating properties. The polysaccharides in Astragali Radix. and saponins in Dioscoreae Rhizoma have been found to protect myocardial cells and correct heart failure.35,36 While TCM doesn't directly recognize Diuretic Resistance, it identifies a range of related syndromes associated with heart failure. The “Xinshuixiao granules”, formulated with the principles of nourishing the kidneys and strengthening the heart, demonstrate promise in managing these conditions. Further research and clinical exploration are warranted to better understand and harness the therapeutic potential of this TCM.
Diuretic Resistance (DR) in the context of heart failure (HF) is the result of various factors working in concert. Its fundamental pathology lies in the ineffective relief of fluid retention. Notably, elevated levels of angiotensin II and AVP are among the principal causes of sodium and water retention, leading to hyponatremia. The renal collecting duct (CD) plays a pivotal role in the final concentration of urine. Under the influence of AVP, the permeability of the renal collecting duct to water rapidly increases, thereby enhancing the reabsorption of water. Consequently, AVP is widely recognized as a central element in the retention of sodium and water in chronic heart failure, ultimately regulating the expression of aquaporin water channel proteins to modulate fluid metabolism.37–39 Aquaporin-2 (AQP2) is a crucial molecular component for regulating water reabsorption in the body, primarily distributed in the kidneys. It is predominantly located in the apical membrane of principal cells and vesicles near the luminal side of the collecting duct. AQP2 is the key protein responsible for regulating water permeability in the renal collecting duct and is considered essential for maintaining water balance in the human body. Research has indicated an upregulation of the renal AQP2 gene expression in patients with chronic heart failure, which is positively correlated with the degree of heart dysfunction. Moreover, both AQP2 gene mRNA and protein expression are significantly enhanced in congestive heart failure animals. Studies involving high-dose diuretic treatment in refractory heart failure patients have observed a significant decrease in AQP2 levels over time. The more prolonged the treatment, the more pronounced the decline in AQP2, indicating that the reduction of AQP2 can serve as a reference for assessing therapeutic progress. Additionally, after AQP2 reduction, renal sodium excretion capacity significantly improves, and left ventricular ejection fraction significantly increases. This observation might be attributed to the continuous elevation of AVP levels, which induce the activation, transcription, and translation of the AQP2 gene, resulting in increased AQP2 protein expression and, subsequently, enhanced renal water transport capabilities.40–42 Experimental studies have found that TCM and its active constituents play a significant role in improving the mechanisms related to fluid retention, closely linked with AQP2. For example, Wuling Powder 43 can inhibit AQP2 expression in renal tissue, reduce cAMP levels, and consequently diminish water reabsorption. Zhenwu Decoction 44 increases AQP2 expression, suggesting its potential to alleviate renal edema induced by kidney damage through the regulation of AQP2 and miR-92b. Qilin Strong Heart Capsules 45 can reduce plasma AVP and AngII levels, downregulate V2R and AT1R protein expression, and further decrease the phosphorylation of AQP2 and AQP2 protein expression. Similarly, the results of this experiment demonstrate that “Xinshuixiao” herbal serum at different doses exerts an inhibitory effect on the expression of AQP2 protein and mRNA induced by dDAVP in IMCD3 cells. Diuretic Resistance in heart failure is a multifaceted condition, with AVP playing a central role. AQP2, as a key molecule in regulating water reabsorption, is intricately related to the mechanisms underlying fluid retention. TCM, exemplified by “ Xinshuixiao granules”, has demonstrated its potential to influence AQP2-related pathways, providing a promising avenue for managing this condition. Further exploration is warranted to unlock the full therapeutic potential of these interventions.
In the context of heart failure, a diminished sensitivity of atrial stretch receptors leads to the inability to inhibit AVP release, resulting in elevated plasma AVP levels. The classical regulatory pathway of AVP involves its binding to the vasopressin V2 receptor (V2R) located on the basolateral membrane of principal cells in the renal collecting duct. This interaction activates cAMP production, thereby increasing intracellular cAMP levels and modulating aquaporin-2 (AQP2) expression to enhance water reabsorption. Under normal circumstances, activated V2R can stimulate both type III and type VI adenylyl cyclases (AC), which convert cellular ATP into cAMP. Subsequently, cAMP further activates protein kinase A (PKA), ultimately leading to the phosphorylation of AQP2.46,47 We chose dDAVP to mimic the action of natural antidiuretic hormone (AVP). dVAVP activates the cAMP-PKA signaling pathway by binding to the V2 receptor in the renal collecting ducts, leading to the phosphorylation and transport of AQP2. Phosphorylated AQP2 is transported to the cell membrane, allowing AQP2 to be embedded in the parietal membrane of the collecting duct host cell, increasing the number of water channels and water reabsorption capacity, and mimicking urine storage. The present results showed that cardioverse-containing serum was able to reduce the expression levels of AVP, cAMP, and PKA, and decrease the total protein expression of AQP2, thereby reducing the formation of water channels, inhibiting the water reabsorption capacity, and decreasing the volume of urine, thus alleviating urinary retention. In essence, during heart failure, the dysregulated AVP system contributes to fluid imbalance. The conventional AVP signaling pathway, which normally stimulates water reabsorption, appears to be modulated by “ Xinshuixiao granules”. These findings suggest a potential therapeutic avenue for restoring fluid balance in heart failure patients.
In this study, we investigated in vitro only the effect of “Xin Shuixiao” herbal serum on cAMP/PKA signaling pathway. The “Xinshuixiao” herbal serum may exert its inhibitory effect on the excessive expression of AQP2 through the suppression of the cAMP/PKA signaling pathway. However, as a traditional Chinese herbal compound with multifaceted and multi-pathway actions, the clinical applications and the mechanistic impact of “ Xinshuixiao granules ” on other relevant signaling pathways require further exploration. “ Xinshuixiao granules ” presents a promising avenue of investigation in clinical practice, shedding light on its potential applications and its broader influence on interconnected signaling pathways. Further research will be instrumental in unraveling the full spectrum of “ Xinshuixiao granules ” therapeutic capabilities.
Conclusions
“Xinshuixiao” drug-containing serum exerts an inhibitory effect on the overexpression of AQP2 induced by dDAVP in IMCD3 cells, which may be associated with its suppression of the cAMP/PKA signaling pathway. However, ‘Xinshuixiao granules’, as a TCM compound, is characterized by multiple effects and pathways, so it is necessary to continue to explore the value of ‘Xinshuixiao granules’ in clinical practice and its influence on other related signaling pathways.
Oligonucleotide primers for the amplification and detection of rat.

Supplemental Material
sj-docx-1-npx-10.1177_1934578X241289213 - Supplemental material for Investigating “Xinshuixiao” Serum's Impact on dDAVP-Induced AQP2 Expression and Mechanisms in IMCD3 Cells
Supplemental material, sj-docx-1-npx-10.1177_1934578X241289213 for Investigating “Xinshuixiao” Serum's Impact on dDAVP-Induced AQP2 Expression and Mechanisms in IMCD3 Cells by Qian Wei, Yang Li, Yanxin Wang, Cheng Luan and TianYang Zhang in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X241289213 - Supplemental material for Investigating “Xinshuixiao” Serum's Impact on dDAVP-Induced AQP2 Expression and Mechanisms in IMCD3 Cells
Supplemental material, sj-docx-2-npx-10.1177_1934578X241289213 for Investigating “Xinshuixiao” Serum's Impact on dDAVP-Induced AQP2 Expression and Mechanisms in IMCD3 Cells by Qian Wei, Yang Li, Yanxin Wang, Cheng Luan and TianYang Zhang in Natural Product Communications
Footnotes
Author Contributions
Yang Li: Conceptualization, Methodology. Qian Wei: Writing-Original Draft, Investigation. All authors contributed to the article and approved the submitted version.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplemental Material, further inquiries can be directed to the corresponding authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Ethics Committee of the Affiliated Hospital of Changchun University of Traditional Chinese Medicine.
Funding
This study was supported by Department of Science and Technology of Jilin Province Natural Science Foundation of Jilin Province (No. YDZJ202201ZYTS231); Administration of Traditional Chinese Medicine of Jilin Province (No. 2021065); Health Commission of Jilin Province (No. 2021JC077); Scientific Research Program of Jilin Provincial Department of Education (No.JJKH20231194KJ); Research Project of Undergraduate Teaching Reform in Jilin University (No. 2021XZC087); Research Projects of Higher Education in Jilin Province (No. JGJX2021D53).
Informed Consent
This article does not involve any studies with human subjects.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Statement of Human and Animal Rights
This study does not contain any studies on human subjects. The standard Guidelines of the Ethical Committee for animals were followed.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
