Abstract
Nephrolithiasis is a prevalent condition characterized by the formation of urinary tract stones, with a high incidence rate worldwide. The recurrent nature of this disease often directs the public to seek alternative therapies, including herbal medicine, due to a common perception of its safety. However, emerging evidence emphasizes the potential for herb–drug interactions, which may pose serious risks to patient health, particularly among elderly individuals and those with chronic diseases. In light of the increasing use of herbal remedies worldwide, this review aims to investigate the potential interactions between conventional drugs and commonly used antiurolithiatic medicinal plants, based on findings from a recent ethnobotanical survey. Seven frequently used medicinal plants in Iraq were identified and reviewed, utilizing data published between 1900 and 2025, with a particular focus on pharmacokinetic and pharmacodynamic interaction mechanisms. This review intends to help healthcare professionals identify and manage potential herb–drug interactions, thereby enhancing patient safety and informed clinical decision-making in nephrolithiasis management.
Introduction
Nephrolithiasis, also known as kidney stone disease, is a common condition worldwide characterized by the formation of crystalline stones of varying composition within the urinary tract. 1 It is associated with a high incidence and recurrence rate in various regions of the globe, 2 as well as in the Africo-Asian region known as the stone-forming belt. 3 Iraq, as geographically positioned in this area, demonstrates a significant proportion of kidney stone disease among all patients with urologic conditions.4,5 The recurrent nature of kidney stone disease directs patients to explore alternative herbal therapies arising from their belief in the safety of these products. 6 However, accumulating evidence suggests that the concurrent use of herbal remedies and conventional medications may result in clinically significant herb–drug interactions, particularly in elderly patients and those exposed to polypharmacy, where the use of multiple drugs increases the risk of adverse interactions.7,8 In Iraq, the use of medicinal plants for nephrolithiasis management remains common. 9 A recent Scopus-indexed cross-sectional survey conducted in 2024 among Iraqi herbalists identified seven widely used medicinal plants that are frequently administered alongside conventional pharmacotherapy. 10 Despite their extensive use, systematic evaluations of potential herb–drug interactions involving these plants remain limited.
Therefore, this study aims to address this gap by examining available pharmacological, phytochemical, and clinical evidence related to potential drug–herbal interactions, to support healthcare professionals in improving patient safety and preventing adverse outcomes in nephrolithiasis management.
Review Methodology
This mini-review was designed to examine the herbal-drug interactions of commonly used medicinal herbs in managing kidney stones in Iraq, using relevant scientific data extracted from various sources, including PubMed, Web of Science, HerbMed, NatMed Pro (formerly Natural Medicines), UpToDate Lexidrug, Medline, and Embase. Different articles and studies were reviewed for the last 20 years and extended later than 2000 in some cases to gather relevant data due to the minimal studies available in this area; the search involved using the scientific names of the selected herbs in addition to keywords such as “drug-herbal interaction,” “adverse effects,” “safety,” “toxicity,” “pharmacokinetic, pharmacodynamic interactions”. The inclusion criteria were all studies and reviews, including in vivo and in vitro studies, as well as case reports. only English-based articles were included. Studies were selected based on their most recent publication and screened for their titles, abstracts, and full texts to obtain the necessary data. The tables demonstrating interactions were arranged and designed according to the NatMed Pro database stop light rating system, which is explained in Figures 1 and Table 1. This shows the occurrence/severity rating and explanations, in addition to the level of evidence used, respectively.

Stop light rating system adapted from the NatMed Pro database.
Level of Evidence and Definitions Adapted from the NatMed Pro Database.

Evidence Synthesis
This study gathered data on the general information of the seven selected herbs, focusing on their potential drug-herbal interactions through a thorough review of a large number of articles across various resources. Results are represented in detail in Tables 1 to 7.
Adiantum capillus-veneris
The first plant in this review is Adiantum capillus-veneris (A. capillus-veneris) of the Adiantiaceae, commonly named Maidenhair fern. This herb is traditionally used in the management of respiratory disorders such as cold, asthma, cough, and pneumonia,11,12 Several studies examined its pharmacological effects as an antioxidant, anti-inflammatory, antidiabetic, antibacterial anti cancer, antidiarrheal, and antispasmodic13–16; in addition to its role in hypothyroidism, and activity against testosterone-induced alopecia.17,18 It acts as a lithiasis agent in the treatment of kidney stones, as demonstrated by several studies, and inhibits crystal aggregation, particularly of calcium crystals.19–21 The phytochemical constituents responsible for the medicinal use of this plant appear to be more than 130 chemicals related to different chemical classes, namely triterpenoids, phyto steroids, flavonoids (quercetin, kaempferol, rutin and gallic acid), phenyl propanoids, and other phenolic compounds, such as coumarins, carotenoids, in addition to fatty acids, mucilage, and minerals.11,17
Regarding the plant's safety, limited studies have been conducted on the adverse effects and toxicity. In the evaluation of the acute toxicity in animals, mice administered A. capillus-veneris extract at doses up to 2 g/kg body weight exhibited no signs of mortality or apparent behavioral abnormalities, even at this elevated dose 17 ; however, this plant is contraindicated in pregnancy due to its anti- implantation property 22 ; and additional toxicological studies are still needed to certain their safety in humans.17,22
Drug-herbal interactions for this plant have not been investigated; despite extensive searching of all databases, the author reported theoretical interactions based on the plant's pharmacological activities derived from some studies, which are presented in “Table 2”; with monitoring tips presented in Figure 2.

Drug-herbal interaction of Adiantum capillus-vene and monitoring tips (created using BioRender).
The Theoretical Drug-Herbal Interactions of Adiantum capillus-veneris.

Ammi visnaga
Ammi visnaga L. is a short biennial plant native to the Mediterranean area, commonly known as khella. 24 The oral decoction of khella has been utilized in both modern and traditional medicine for the treatment of various diseases and conditions, including cardiovascular disorders, respiratory conditions, kidney stones, and abdominal cramps, as well as antibacterial, antiviral, and antifungal activities.25–27 Several studies have investigated the phytochemicals responsible for the pharmacological activity of the plant, including γ-pyrones such as visnadin, visnagin, and khellin, which exhibit vasodilating and bronchodilating effects, as well as coumarins, flavonoids, and essential oils, which demonstrate antibacterial and antioxidant properties.25,28,29 The antilithiogenic activity of Ammi visnaga is primarily attributed to visnagin and khellin, which act through their vasodilating effect and interference with citrate metabolism, thereby inhibiting calcium oxalate crystallization.27,30,31
The oral dose of khella extract is standardized to 12% khellin. An average dose of Ammi visnaga is stated to be 20 mg of the khellin component per day. 27 Regarding plant safety, Studies are conflicting. Some studies have reported hepatotoxicity at high, prolonged doses, as well as an increase in liver enzymes.27,32,33 Another study showed no effect on the liver in a 28-day trial on rats 34 ; thus, it is essential to avoid using this herb in patients with liver function conditions and to monitor liver enzymes during use. Reports also stated its contraindication for use in pregnancy due to the uterine stimulant activity of khellin.27,35 It was reported that the median lethal dose (LD50) was 3.6 g/kg for intraperitoneal administration and 10.1 g/kg for oral administration in rats. 36
Drug-herbal interactions for khella have not been extensively investigated; some of the listed interactions are based on studies proving its pharmacological activity, while other interactions are based on the actual references as presented in “Table 3”; and monitoring tips in Figure 3.
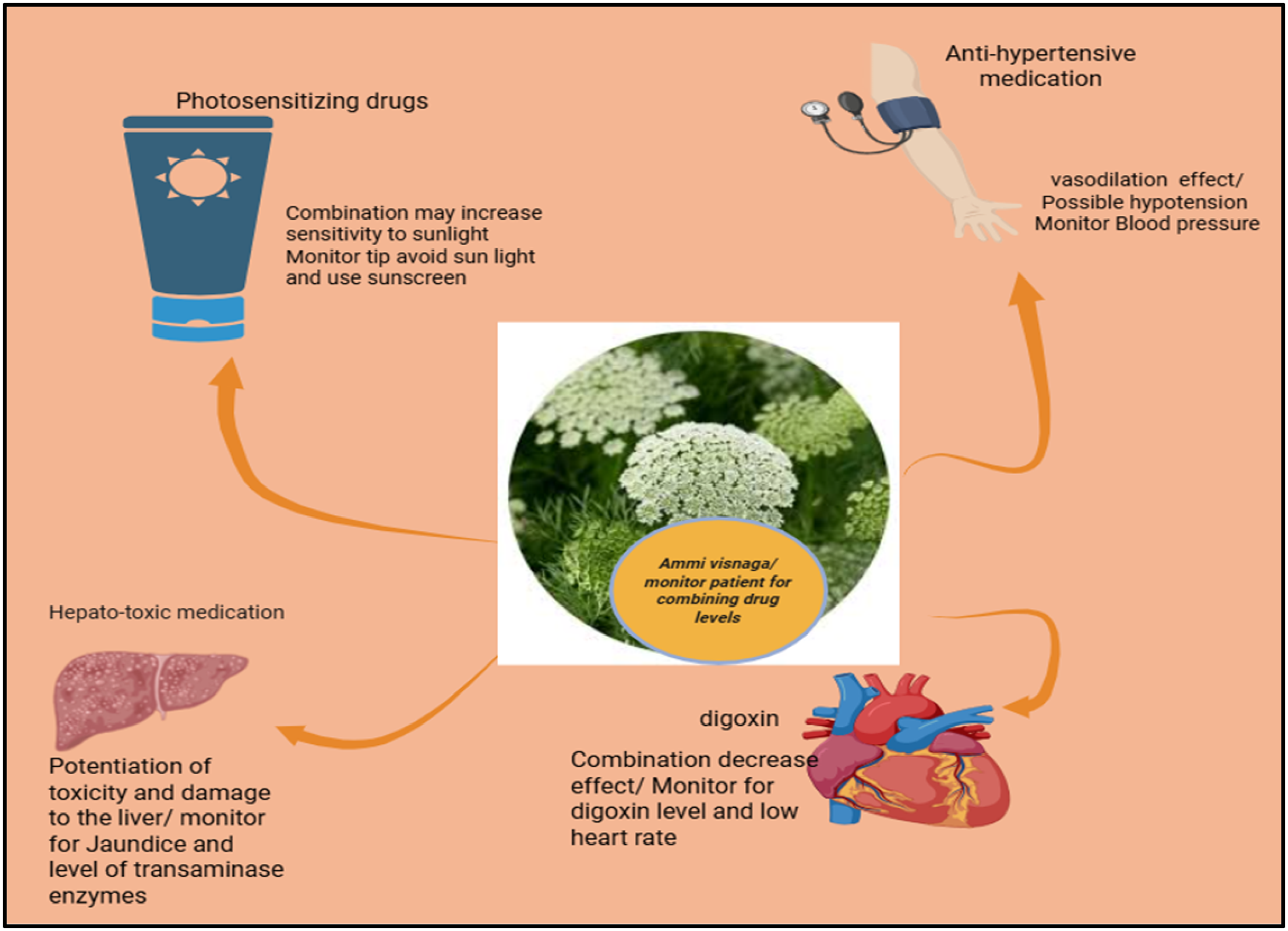
Drug/ herbal interaction of Ammi visnaga monitoring tips (created using BioRender).
The Drug-Herbal Interactions of Ammi visnaga.

Petroselinum sativum
Petroselinum sativum Hoffm. (synonyms of Petroselinum crispum), commonly known as parsley and belonging to the family Apiaceae, is a fragrant herb with a rich cultural history of use in traditional medicinal practices. 38 Parsley has its origins in the Mediterranean area but is now grown worldwide. 39 Traditionally, parsley has been used to manage various ailments, including diabetes, hyperlipidemia, hyperuricemia, renal disease, cardiovascular disease, and hypertension. 40 Several studies have reported the pharmacological activity of this herb, including antimicrobial, estrogenic activity, antioxidant, antiplatelet, antilithiatic, and diuretic properties. 41 The phytochemical constituents of parsley include flavonoids, furanocoumarins, carotenoids, essential oils (phenylpropane and terpene compounds as major constituents), phytoestrogens, and minerals. 40 The role of Petroselinum sativum as an antilithic agent is thought to be through reducing urinary calcium excretion and alkalinizing the urine, thereby decreasing the formation of stones.42,43
Regarding the safety of this herb, clinical data are lacking to provide dosing recommendations. In general, Petroselinum sativum is considered safe for use as a food, based on the U.S. Food and Drug Administration (FDA), and is likely safe when used in an appropriate amount for the short term with no toxicity reported.44–47 A study performed in rats suggested a dose of 200–600 mg/kg body weight for the treatment of kidney stones. 42 However, High doses of parsley extract, as 1 g/kg body weight, may lead to substantial increases in liver enzymes, indicating hepatotoxicity. In addition, the increase in blood urea nitrogen and creatinine levels suggests kidney toxicity. 48 Data also confirmed the unsafe use in pregnancy and possible congenital malformation. 49
Several drug-herbal interactions have been reported; detailed information is presented in “Table 4 and Figure 4”.

Drug/herbal interaction of Petroselinum sativum and risk management (created using BioRender).
The Drug-Herbal Interactions of Petroselinum sativum.

Senegalia Senegal (Acacia senegal)
Senegalia Senegal, also known as acacia gum or Arabic gum (AG), is an edible constituent widely used as an excipient in both the food and industrial sectors. AG is a gummy exudate derived from the air-dried sap exuded from the branches and trunk of Senegalia Senegal of the family Leguminosae. 59 Plant species of this genus are naturally found in tropical and subtropical regions worldwide, including Africa, Australia, Central America, the Middle East, and South Asia. 60 A literature review revealed the traditional use of this plant in managing gastrointestinal tract conditions, promoting oral hygiene, and addressing disorders such as obesity and diabetes. Reports also suggest its role in hyperlipidemia, gingival inflammation, and antibacterial activities, as well as its therapeutic role in managing chronic kidney conditions.59–62 Its role in urolithiasis is not directly related to its effect on kidney stones, but it is prebiotic, anti-inflammatory, and antioxidant, which plays an essential role in preventing kidney damage.61,63 GA reduced plasma urea and creatinine levels, improved creatinine clearance, and significantly altered electrolyte excretion, resulting in improved overall water and electrolyte balance in healthy mice. 61 GA is a complex combination of polysaccharides and glycoproteins, which accounts for its water solubility and low viscosity. The main components are the arabinogalactan protein complex. This compound consists of several minerals, such as magnesium, calcium, and potassium salts of arabic acid. 62
Regarding safety, AG is widely identified as safe for human utilization and has been classified as “Generally Recognized As Safe” (GRAS) by the FDA in the United States. It is also considered safe for use as a food additive by the European Food Safety Authority.61,64,65 According to references, it is considered safe to be taken orally in a dose of up to 30 grams of powdered gum daily for 3 months66,67; however, reliable information on its use during pregnancy and lactation is not available. 65 Research is limited for typical dosing information.
The drug herbal interactions of AG are related to the suspending osmotic property in drug preparations. AG may enhance the bioavailability of water-insoluble drugs, such as naproxen, but reduce the absorption of polar drugs, like amoxicillin. 68 To avoid changes in absorption, it is advisable to take gum arabic 30–60 min after oral medications. These interactions are represented in “Table 5 and Figure 5.”

The drug-herbal interactions of Arabic Gum and monitoring tips ( created using BioRender).
The Drug-Herbal Interactions of Arabic Gum (AG).

Tribulus terrestris
Tribulus terrestris (TT), commonly known as “Al-Gutub” in Iraq, manuscriptto the family Zygophyllaceae. TT is a small annual plant that grows in Mediterranean, subtropical, and desert climate regions worldwide, including India, China, the southern United States, Mexico, Spain, and Bulgaria.69,70 Traditionally used as an aphrodisiac, in addition to other folk uses, including the management of kidney disorders, antioxidant, anti-inflammatory, respiratory, and skin conditions71,72; it has also shown effectiveness as an antibacterial, antidiabetic, and cardioprotective agent.71,73–75 Its role as an antilithiatic was documented by the fact that TT extract suppresses the formation and growth of calcium oxalate crystals, as well as having a protective effect at the cellular level.69,76 A wide range of bioactive and chemically diverse phytoconstituents has been associated with the various uses of TT. These include steroidal saponins, flavonoids, tannins, terpenoids, glycosides, amide derivatives, amino acids, protein, polyphenol carboxylic acids, and alkaloids.69,72
For the safety of TT extract, studies have shown that oral doses of 750–1500 mg per day are apparently safe for up to 90 days of use, as well as the powdered TT fruits in an oral dose of 6 g per day for 2 months; however, there is no documentation for its safety beyond three months of treatments.77–80 Studies have found that chronic use of large doses can adversely affect liver and kidney function. 81 While there are no available studies on the use during lactation, its use in pregnancy is not recommended due to its effect on fetal development. 82
Literature reveals that TT has a hormonal effect and is therefore used as an aphrodisiac, although drug interactions with hormones have not been investigated. Studies suggest its careful use due to its potential to disrupt the endocrine system.83–85 Other studied interactions are illustrated in “Table 6 and Figure 6”.

The drug-herbal interactions of Tribulus terrestris and monitoring tips (created using BioRender).
The Drug-Herbal Interactions of Tribulus terrestris.

Trigonella foenum-graecum
Trigonella foenum-graecum, commonly known as “halba” or “fenugreek,” is an annual plant of the family Leguminosae. It is grown in the Mediterranean region, Western Asia, and Southern Europe, and is a well-known spice in human food, as well as one of the oldest botanical herbs used in ancient history.95,96 Traditionally, fenugreek has been used to treat different conditions, including digestive disorders, skin inflammation, as a demulcent, a lactation stimulant, and a laxative.95,97 Several pharmacological properties have been documented for fenugreek, including antioxidant, anti-inflammatory, antihyperlipidemic, antidiabetic, antibacterial, and anticancer effects.98,99 These medicinal properties are attributed to the bioactive compounds, including alkaloids (trigonelline, choline), steroidal sapogenins (yamogenin, diosgenin, smilagenin, sarsasapogenin), saponins, flavonoids (quercetin, rutin, vitexin), fiber, lipids, and amino acids. 100 Its role in kidney stone prevention is attributed to the ability of the seed extract to reduce and prevent the growth of calcium oxalate crystals, a mechanism thought to be mediated through diuretic and antioxidant activity, thereby lowering the risk of stone formation.100,101
In regard to the safety of this herb extract, references suggested its safety when used in an appropriate amount, which is represented by fenugreek seed powder in an oral dose of 5–10 grams daily for up to 3 years, and fenugreek seed extract in a dose of 1 gram daily for up to 3 months is considered safe.102–106 A study based on an animal model suggested that oral doses for humans should not exceed 350 mg/kg (21 g per 60 kg) to prevent accidental overdose. 107
The use of fenugreek during pregnancy in an amount greater than that found in food has potential oxytocic and uterine stimulant activity, and cases have been reported for congenital malformations.107,108 The use during lactation is possibly safe if used for a short term, although most available clinical data lack testing on lactating women or nursing infants.109,110 Some studies suggest that a dose of 1725 mg, administered three times daily for three weeks, may be considered safe. 111
The drug herbal interaction of fenugreek is represented in “Table 7 and Figure 7.”
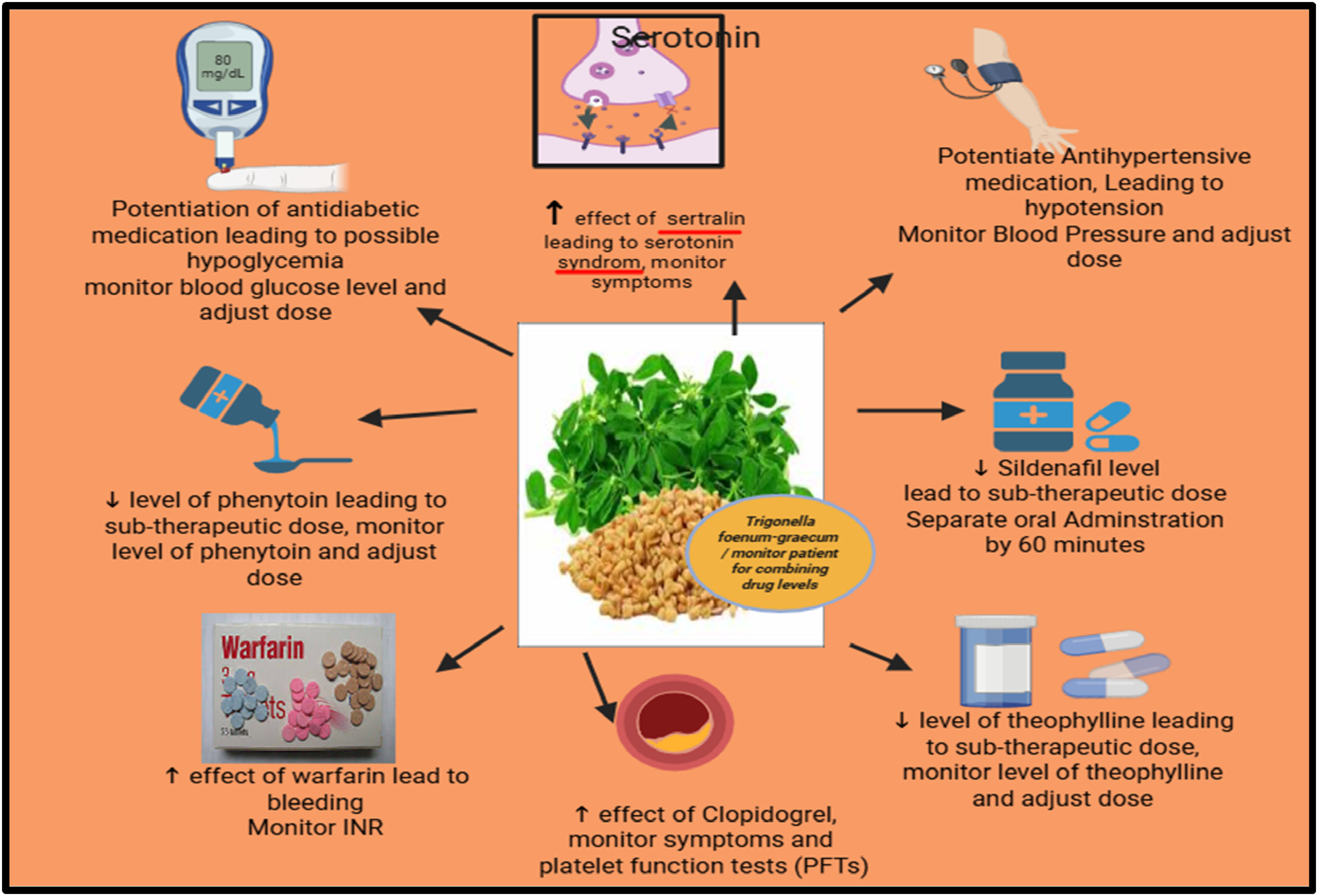
The drug-herbal interactions of Trigonella foenum-graecum and monitoring tips (created using BioRender).
The Drug-Herbal Interactions of Trigonella foenum-graecum.

Zea mays (Corn Silk)
Corn silk (CS), the elongated stigma of the female flower of Zea mays (F. Poaceae), is considered the third most widely grown crop worldwide and a primary cereal in various countries and regions, including Southern and Eastern Africa, Central America, and Mexico. 124 It is a widely available agricultural byproduct often considered a waste product and traditionally used in the management of diabetes, obesity, prostate disorders, as a diuretic, and kidney disorders. 125 Recent research has documented the pharmacological activities of CS extract, including antioxidant, anti-inflammatory, anticancer, hypotensive, anti-diabetic, hypolipidemic, antibacterial, antifatigue, antidepressant, and anticoagulant properties.125–128 The bioactive components of CS are reported to include polyphenolic compounds, flavonoids, tannins, alkaloids, volatile oils, steroids (sitosterol and stigmasterol), saponins, proteins, various minerals, and vitamins.128–130 Regarding the antiurolithiatic activity, a recent study reported that the mechanism involved both diuretic activity and alkalinization of urinary pH, thereby preventing the formation of calcium oxalate crystals. Moreover, CS antibacterial and antioxidant activity aid in kidney protection. 131
Concerning the safe consumption of this herb, references suggest that corn silk and its extract, when used in amounts found in food, have a Generally Recognized as Safe (GRAS) status in the US. 44 Recent studies showed that an oral dose of CS extract at 500 mg/kg body weight is generally safe, but doses equal to or higher than 2000 mg/kg for longer durations is considered toxic and should be avoided; thus, the study recommended a safe dose of less than 1000 mg/kg body weight for the therapeutic purposes.128,132 The use in pregnancy in larger amounts is considered unsafe due to the uterine stimulant effects of CS 133 ; however, there is no available data on its use during lactation.
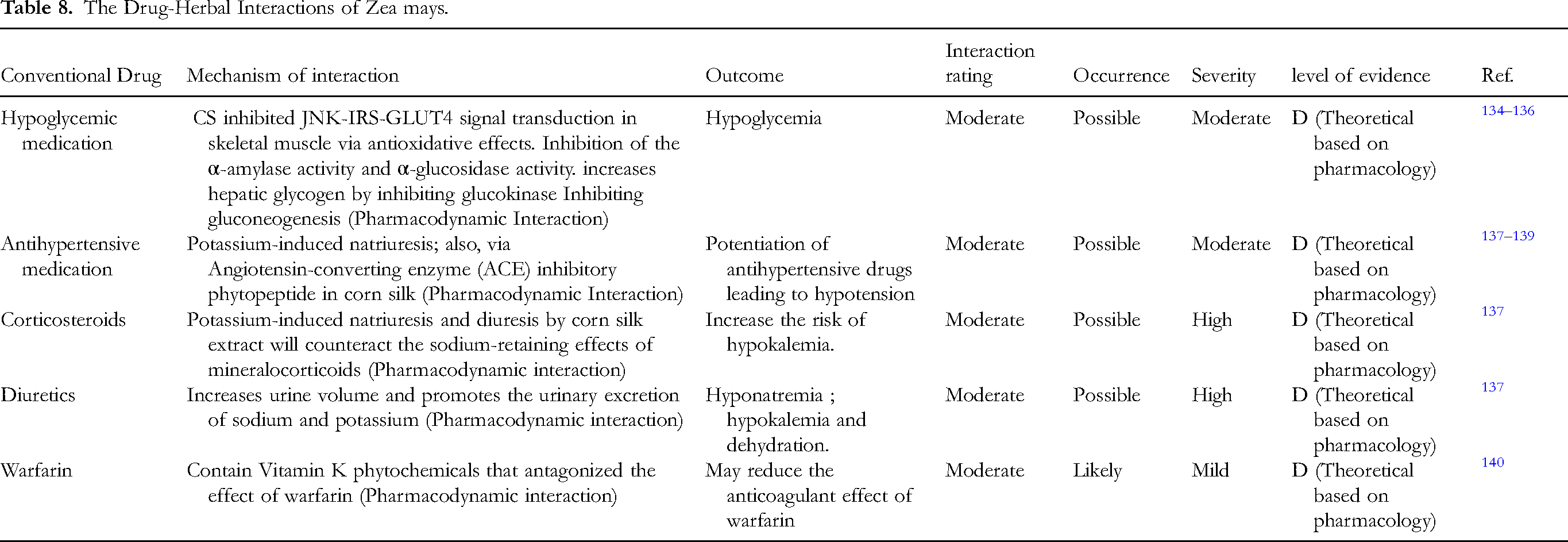
The drug-herbal interaction reported in the literature is demonstrated in “Table 8 and Figure 8.”
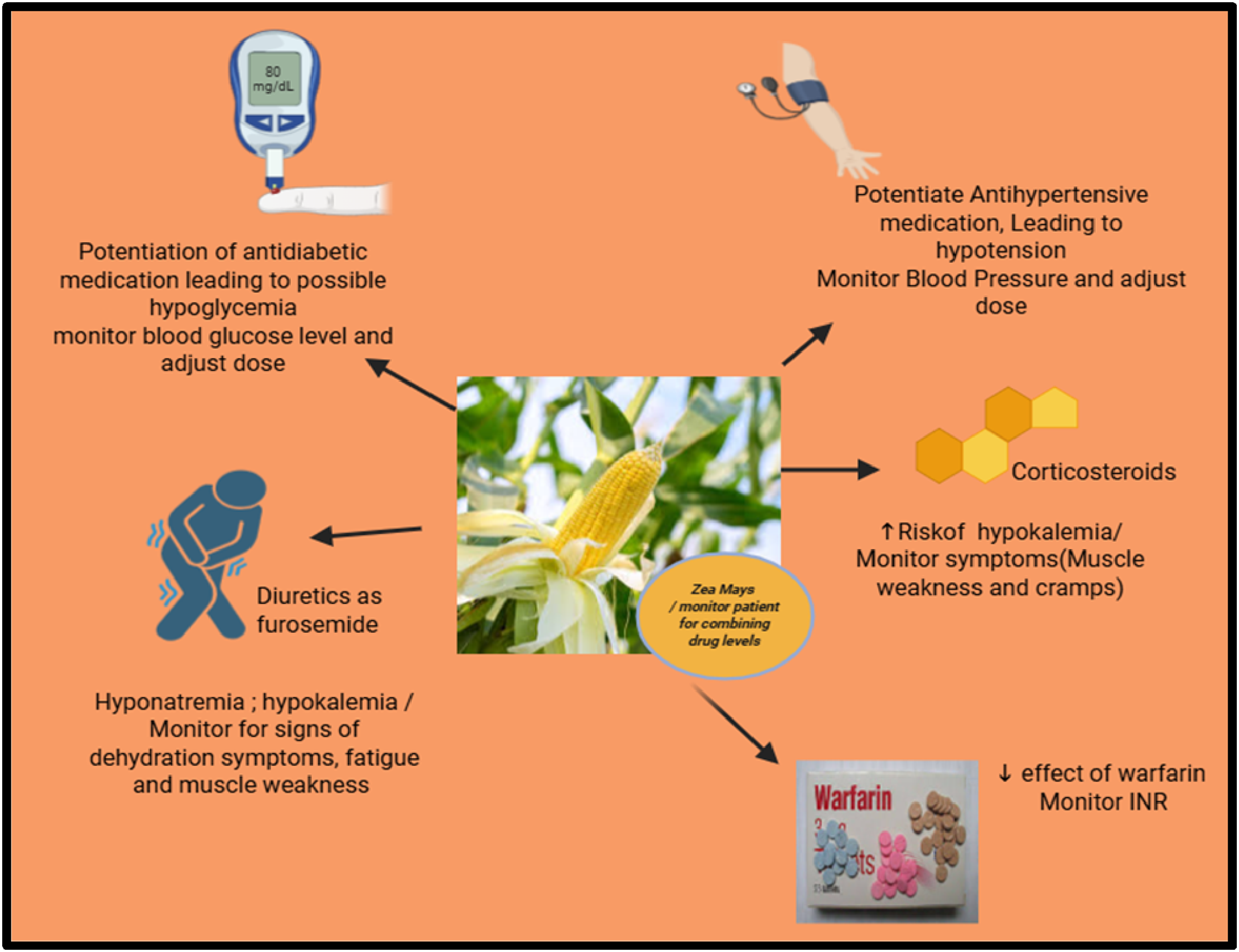
The drug-herbal interactions of Zea mays and monitoring tips (created using BioRender).
The Drug-Herbal Interactions of Zea mays.
Clinical Implications of Herb–Drug Interactions
The prevalence of botanical use of herbs has increased significantly worldwide, with a public usage rate as high as 80%, according to the literature. 141 People culturally believe in the safety of these natural products, and this belief has extended from early civilizations to the present day.142–144 Patients often do not disclose their use of supplements and herbal products to their healthcare providers due to this belief and a lack of awareness of potential interactions that could occur with their current disease state or conventional medications.145,146 This practice exposes patients to unwanted side effects and reactions that put them at risk and hinder the management of their condition by primary care personnel. Several research studies have addressed this risk and discussed herbal drug interactions in general147,148; however, to the author's knowledge, there have been limited studies that specifically examined herbal interactions in the management of kidney stones. Thus, this review aimed to discuss the potential drug-herbal interactions for commonly used herbs in the management of urolithiasis in Iraq, as this condition is one of the major urological conditions in this region. 10 This review is crucial for clinicians and pharmacists to understand the potential interactions between patients’ medications and herbal products, thereby preventing adverse effects on patient health and providing guidance on monitoring and managing these interactions. Seven medicinal herbs were selected for this review based on a previous survey on the most commonly used herbs in the management of kidney stones. 10 These herbs were selected based on the highest use value (UV) reported in the mentioned study. An extensive review of various data sources was then conducted, examining related studies in this context. Some of the interactions were based on the theoretical explanation of the pharmacological properties of the studied herbs, such as A. capillus-veneris and Ammi visnaga, as there were limited studies regarding the safety of consuming these products with other medications, as compared to the rest of the studied herbs, where more articles were documenting their interactions. The results showed that the most frequently interacting medications were related to cardiovascular disorders (anticoagulants and antihypertensive medications) and Diabetes (antidiabetic medications), which are the two most common disorders affecting patients worldwide, 149 with some drugs having low therapeutic indices (as warfarin and digoxin). 150 These interactions would affect patients of different age levels, particularly the elderly, due to their age-associated physiological alterations in organ systems, which in turn significantly modify drug pharmacokinetics and pharmacodynamics, thus imposing an even greater risk of harmful effects. 151
The literature survey also explained the mechanism of drug-herb interactions as cited in the Tables (2-8), but the exact mechanisms behind many reported interactions between drugs and herbal medicines remain largely unclear. 152 This may be due to the fact that herbal products consist of different phytochemicals that exert varying effects, making it challenging to evaluate these interactions based on the percentage of these chemicals in the studied herb.150,153 The chemical metabolite profiles are also influenced by plant species, chemotype, cultivation conditions, harvesting practices, extraction, and preparation methods. 152 The difficulty extends to the fact that most herbs are not used as a single entity but rather as mixtures of herbs in a single herbal product, thus predicting the exact influence on concurrent medication use is difficult. Some of the listed interactions in this review were related to the pharmacokinetic interactions, such as the modulation of drug absorption (as Senegalia Senegal, Tribulus terrestris, and Trigonella foenum-graecum) or metabolism through inhibiting CYP450 (Petroselinum sativum, Tribulus terrestris, Trigonella foenum-graecum) or alteration in drug excretion (Tribulus terrestris, Trigonella foenum-graecum, and Zea mays); other interactions are pharmacodynamic, which is represented by the synergistic effect of the herbal products, thus potentiate the effect of the concurrent drug.
Conclusion
This review highlights potential drug-herbal interactions associated with the use of commonly used medical herbs in the management of kidney stones. It also emphasizes raising awareness among healthcare practitioners as well as the public regarding the possible potential risks of using herbs concurrently with conventional therapy and highlights the importance of reporting any interactions or adverse reactions that may result from this use.
Footnotes
Acknowledgment
The author hereby acknowledges Assistant Professor Dr Salam Ahjel for his help in providing some of the references needed for this review as well as the Library staff In Al-Zahrawi University/ College of Pharmacy
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
