Abstract
Introduction
Atherosclerosis (AS) is a primary cause of cardiovascular morbidity and mortality worldwide. Accounting for approximately 20% of total global deaths, cardiovascular diseases pose a significant threat to public health.1,2 Despite the global increase in living standards, the incidence of AS has rapidly increased in low- and middle-income countries. With extended lifespans, this trend is expected to increase. AS is the leading cause of mortality worldwide. According to data from 2019, cardiovascular diseases, notably coronary and cerebral AS, accounted for approximately 18 million deaths globally, representing over 30% of the total fatalities. 3 In China, cardiovascular diseases are the primary cause of mortality in both urban and rural populations. As of 2020, the mortality rate attributed to cardiovascular diseases was 48.00% and 45.86% in rural and urban areas, respectively. The escalating prevalence of cardiovascular disease has substantially increased the economic burden on both residents and society, highlighting its significance as a major public health concern. 4 Consequently, there is an urgent need to develop safe and effective strategies for the prevention and treatment of AS.
Current approaches for AS treatment predominantly involve surgery and drug therapies. Surgical interventions such as percutaneous transluminal coronary artery angioplasty (PTCA), percutaneous intracoronary stenting (PCI), coronary artery bypass grafting, and transcatheter atherosclerotic plaque exfoliation (TAE), are commonly employed.5–7 Despite their ability to swiftly alleviate stenosis induced by AS and restore blood flow in obstructed vessels, these surgical procedures have notable limitations, including the occurrence of vessel restenosis postoperatively. Consequently, AS cannot be effectively treated with single surgical interventions.
Currently, several lipid-regulating and antithrombotic drugs are clinically used for AS treatment, including statins, probucol, fibrates, aspirin, clopidogrel, and certain anticoagulants. 8 Among these, statins are particularly notable and currently the preferred choice for clinical AS management. 9 Clinical research has demonstrated the effectiveness of statins in ameliorating high blood lipid and cholesterol levels in AS patients, thereby stabilising atherosclerotic plaques and preventing their progression. Nevertheless, lipid-lowering medications have limitations such as incomplete AS eradication and potential long-term toxicity. 9 Consequently, monotherapy with lipid-lowering drugs alone may not result in complete remission of AS. Therefore, there is an urgent need to explore alternative medications with enhanced safety profiles, efficacy, and low toxicity for the treatment of AS.
In this study, we primarily employed a high-cholesterol diet (HCD)-induced AS model in zebrafish to assess the anti-atherosclerotic properties of DOKM.
Results
To analyse potential targets of DOKM active ingredients for the treatment of AS, 145 active ingredients were screened and numbered. The correspondence between the numbers and chemical names is provided in Attachment Table 1. A total of 680 targets were identified using target prediction. After de-weighting the individual database genes, 1583 disease targets were identified (Figure 1A). Subsequently, 680 active ingredient action targets and 1583 disease targets were screened, and the predicted targets were mapped to the disease targets to draw a Venn Diagram (Figure 1B). This process yielded 202 intersecting genes that represent potential therapeutic targets of DOKM for AS.

Binding Energy of Compound to Core Target.

Note: The table presents the binding energies of the five compounds with 10 core target molecules through molecular docking. The binding energy (measured in kcal/mol) indicates the strength of the interaction between the compound and target molecule, with lower values representing tighter binding. A binding energy of −5 kcal/mol was considered the threshold for a stable interaction.
Abbreviations: EGFR, epidermal growth factor receptor; JAK2, Janus Kinase-2; PDGFRA, PDGFRα, Platelet-Derived Growth Factor Receptor-α; SRC, Proto-oncogene tyrosine-protein kinase Src.
Network Diagram of “Active Ingredient-Target-Disease”
The “active ingredient-target-disease” network graph comprises 349 nodes and 1788 edges. Analysis by degree using the “analyze Network” function of Cytoscape 3.9.1 software revealed the top five compounds to be gigantol, chrysotobibenzyl, naringenin chalcone, hesperetin, and tangeretin, respectively. The network diagram of “active ingredient-target-disease” is depicted in Figure 2.

Network diagram of “active ingredient-target-disease”.
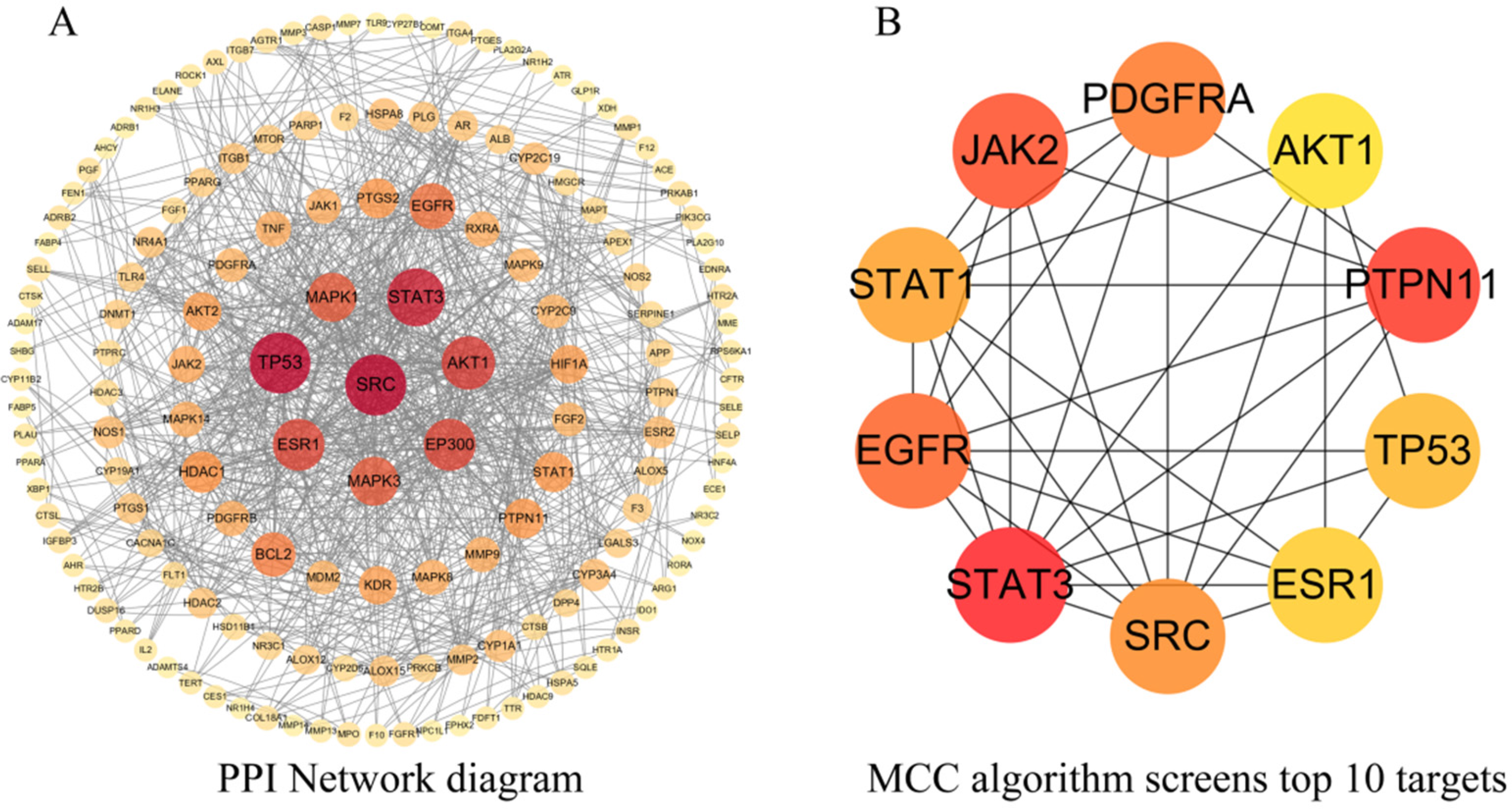
Protein-Protein Interaction (PPI) Network and Core Targets
The 202 overlapping targets obtained from the Venn analysis were uploaded to the STRING database, which was utilised to construct a PPI network with non-overlapping targets hidden. A PPI network model comprising 154 target protein nodes was generated. The resulting Tabular Separated Values (TSV) format file was saved and imported into Cytoscape 3.9.1 software for visual analysis (Figure 3A). Subsequently, the maximal clique centrality (MCC) algorithm was employed for the calculation, and the top 10 targets were identified as core targets (Figure 3B). The colours of the core targets, arranged in descending order, transitioned from dark to light shades. These core targets included STAT3, PTPN11, JAK2, epidermal growth factor receptor (EGFR), platelet-derived growth factor receptor alpha (PDGFRA), proto-oncogene tyrosine-protein kinase Src (SRC), STAT1, TP53, ESR1, and AKT1, indicating a significant association with DOKM in AS treatment.
GO Function and KEGG Pathway Enrichment Analysis
Using the Metascape database, potential therapeutic targets for AS in DOKM were subjected to GO function and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses, and the results were visualised. GO functional enrichment analysis yielded 2090 GO biofunctional entries. Among these, the biological process (BP)-related entries were the most numerous, comprising 1822. These entries were primarily involved in responses to hormones, cellular responses to nitrogen compounds, cellular responses to organonitrogen compounds, peptides, cellular responses to hormone stimuli, cellular responses to peptide stimuli, peptide hormones, cellular responses to peptide hormone stimuli, insulin, and cellular responses to insulin stimuli. Entries related to cellular components (CC) totalled 85, encompassing membrane rafts, membrane microdomains, caveolae, plasma membrane rafts, glutamatergic synapses, postsynapses, late endosomes, receptor complexes, external sides of plasma membranes, and sides of membranes. There were 183 entries associated with molecular functions (MF), including nuclear receptor activity, ligand-activated transcription factor activity, transcription factor binding, DNA-binding transcription factor binding, RNA polymerase II-specific DNA-binding transcription factor binding, transcription coregulator binding, transcription coactivator binding, nuclear receptor binding, DNA-binding transcription activator activity, and RNA polymerase II-specific DNA-binding transcription activator activity. Based on the

Go function enrichment analysis graph. DOKM,
KEGG pathway enrichment analysis revealed 200 AS signalling pathways associated with AS treatment in DOKM. These pathways primarily included: Lipid and AS, PI3K-Akt signalling pathway, AGE-RAGE signalling pathway in diabetic complications, mitogen-activated protein kinase (MAPK) signalling pathway, EGFR tyrosine kinase inhibitor resistance, endocrine resistance, efferocytosis, hepatitis B, thyroid hormone signalling pathway, and insulin resistance. The KEGG pathway data were filtered based on high
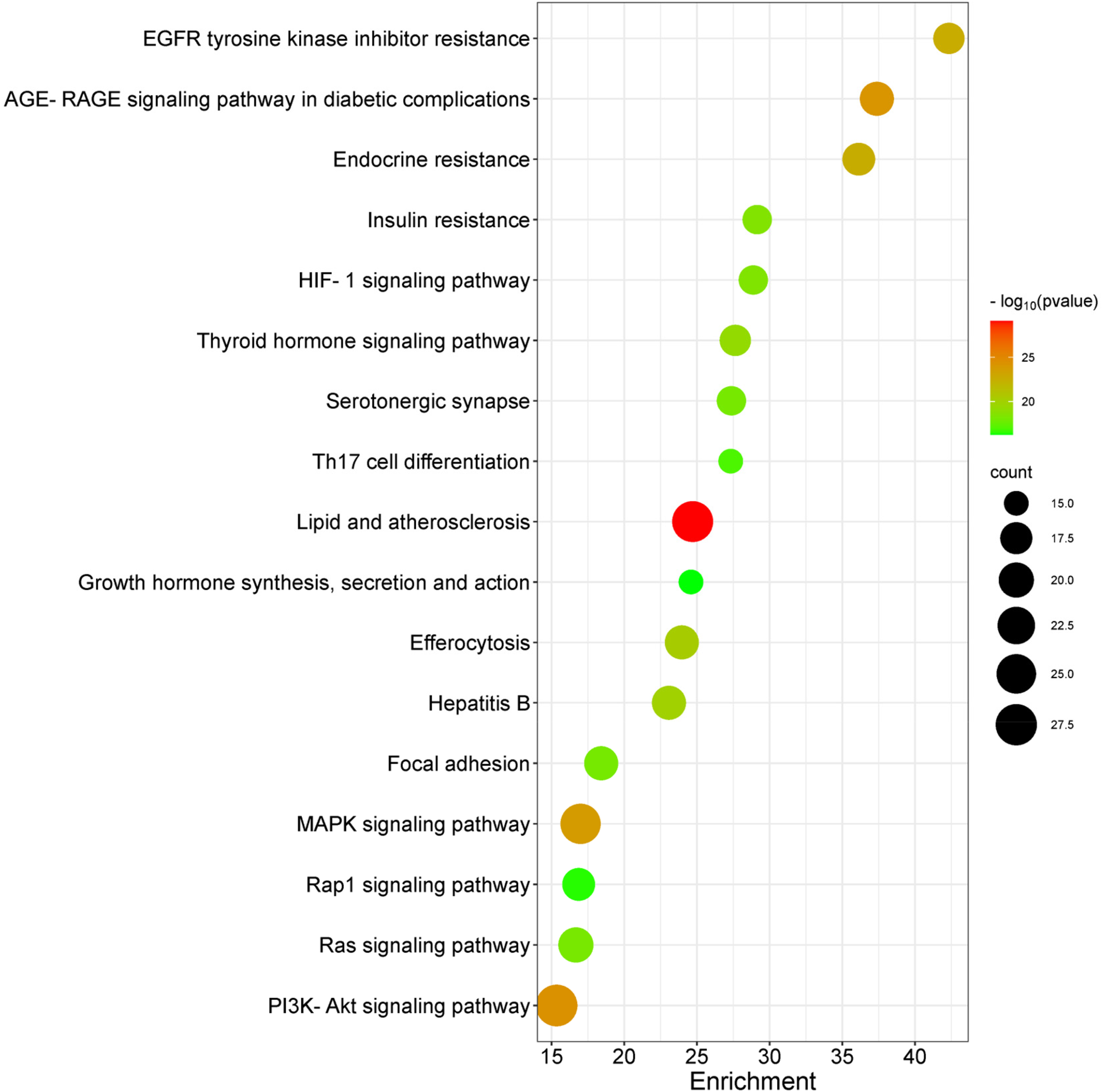
KEGG pathway enrichment analysis.
Molecular Docking Results
The five compounds with the highest degree, namely gigantol, chrysobibenzyl, naringenin chalcone, hesperetin, and tangeretin, were identified as the top 10 PPI analysis targets using the MCC algorithm. Molecular docking validation was performed and the binding energies of the main active ingredients to the key core targets were obtained, as shown in Table 1. Lower binding energy values indicate more stable conformations, with binding energies of all active ingredients to key core targets below −5.0 kcal/mol, suggesting good binding affinity and stable conformations. 22 Figure 6 visually presents the molecular docking of compounds with core targets.
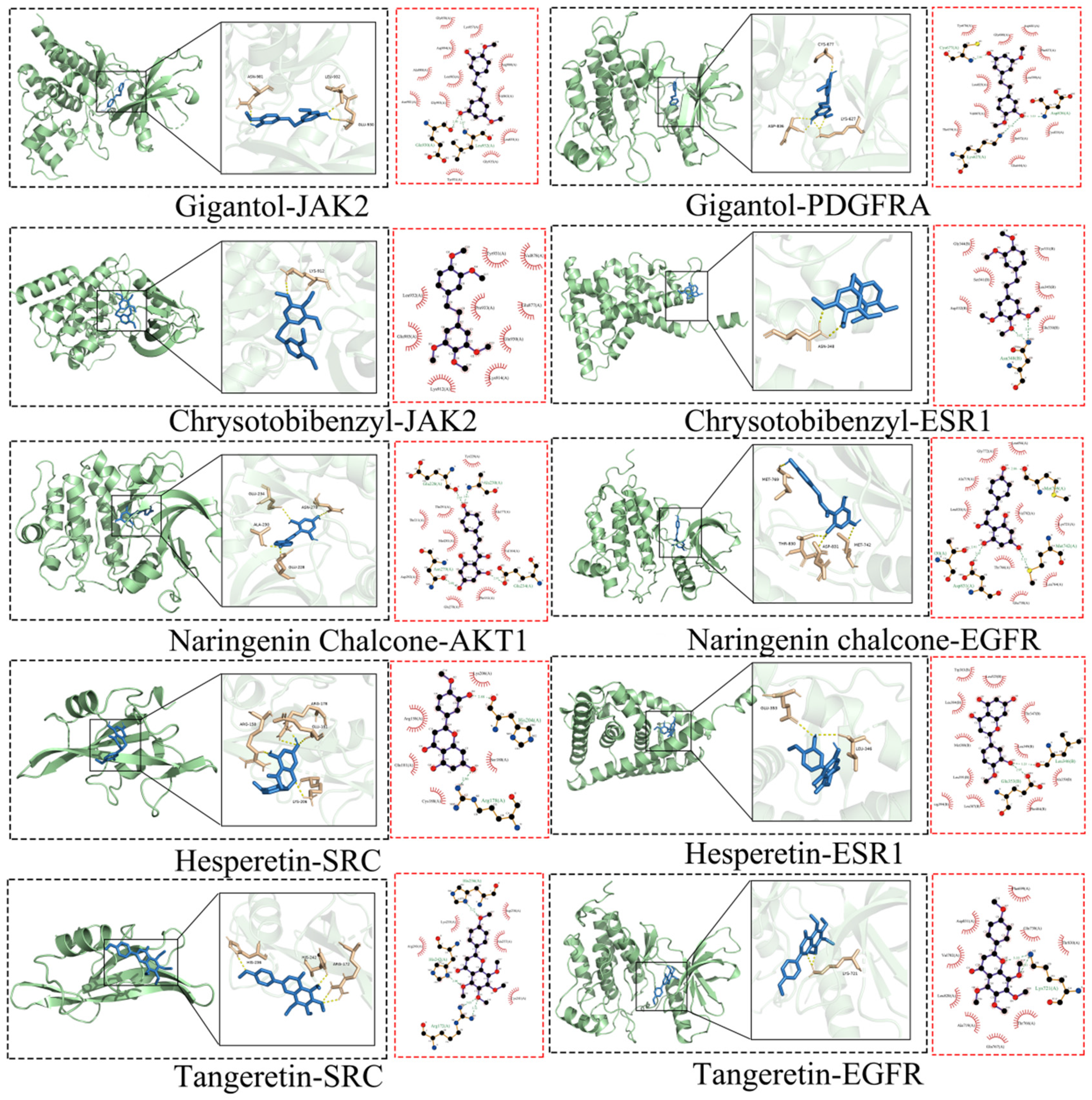
Molecular docking. Note: The black box contains the 3D molecular docking image, whereas the red box contains the 2D molecular docking image. The images include amino acid residues of the interacting protein molecules.
DOKM Mitigates Cholesterol Accumulation in the Blood Vessels of AS Zebrafish
As shown in Figure 7, there was no significant cholesterol accumulation in the blood vessels of the zebrafish in the control group. Compared to the control group, a large amount of cholesterol accumulation was observed in the AS model group. Compared to the AS model group, both 1 mg/L and 10 mg/L DOKM treatments were effective in reducing cholesterol accumulation in blood vessels.
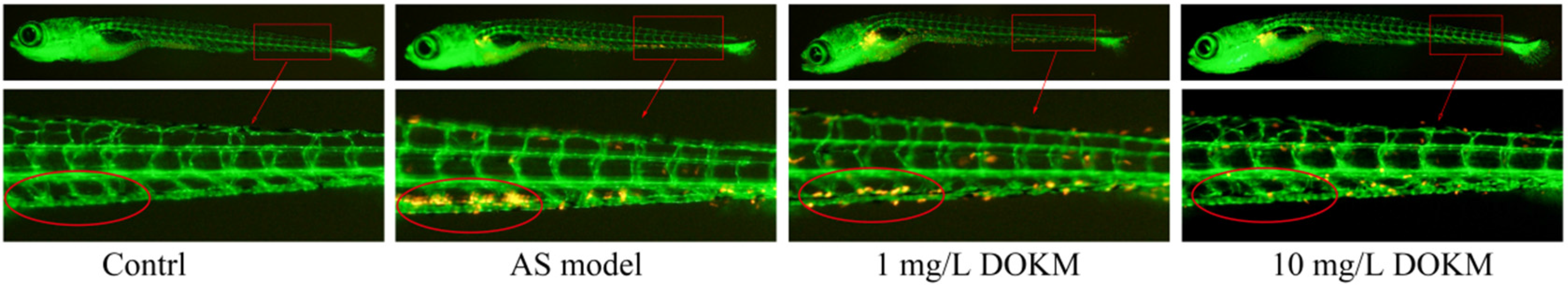
DOKM alleviates cholesterol accumulation in AS zebrafish vessels (
DOKM Attenuates Macrophage Infiltration in the Blood Vessels of AS Zebrafish
As shown in Figure 8, there was no obvious macrophage infiltration into the blood vessels of the zebrafish in the control group. Compared to the control group, a large number of macrophages appeared in the blood vessels of zebrafish in the AS model group. Compared to the AS model group, both 1 mg/L and 10 mg/L DOKM treatments were effective in reducing the number of macrophages in the blood vessels.

DOKM alleviates macrophage infiltration in AS zebrafish vessels (
DOKM Attenuates Neutrophil Accumulation in the Blood Vessels of AS Zebrafish
The results are shown in Figure 9, which indicates that there was no obvious accumulation of neutrophils in the blood vessels of zebrafish in the control group. Compared to the control group, a large number of neutrophils appeared in the blood vessels of zebrafish in the AS model group. Both 1 and 10 mg/L DOKM treatments were effective in reducing the number of neutrophils in blood vessels when compared to the AS model group.

DOKM alleviates neutrophil accumulation in AS zebrafish vessels (
DOKM Attenuates Plaque Formation in the Blood Vessels of AS Zebrafish
Plaque formation in the blood vessels of zebrafish in each group was detected by haematoxylin and eosin (HE) staining and Verhoeff's Van Gieson (EVG) staining. The results are shown in Figure 10. Plaques were not formed in the blood vessels of zebrafish in the control group and no blockage occurred in the blood vessels. Compared to the control group, plaques were formed in the blood vessels of zebrafish in the AS model group, and severe vascular blockage occurred. Compared to the AS model group, both 1 mg/L and 10 mg/L DOKM treatments were effective in reducing plaque formation in blood vessels.

DOKM alleviates plaque formation in AS zebrafish vessels (
DOKM Attenuates Lipid Levels in AS Zebrafish
The results are shown in Figure 11, compared with the control group, the lipid levels of the zebrafish in the AS model group were significantly higher than those in the control group. Compared to the AS model group, both 1 mg/L and 10 mg/L DOKM treatments were effective in reducing lipid levels. The results showed that the total cholesterol (TC), TG, and low-density lipoprotein (LDL)-C levels of zebrafish in the AS model group were significantly elevated compared to those in the control group, whereas both 1 mg/L and 10 mg/L DOKM treatments were effective in reducing lipid TC, TG, and LDL-C levels (Table 2). These results suggested that DOKM attenuates AS by improving lipid metabolism.

DOKM alleviates lipid levels in AS zebrafish (
Effect of DOKM on Lipid of AS Zebrafish (n = 6).

Abbreviation: DOKM,
Effect of DOKM on pcsk9, srebf2, and hmgcr Genes in AS Zebrafish
To further investigate the possible mechanism of action of DOKM, its effect on lipid metabolism-related genes (mainly zebrafish
Effect of DOKM on mRNA Expression of pcsk9, srebf2, and hmgcr in AS Zebrafish (n = 3).

Abbreviation: DOKM,
Discussion
Hypercholesterolaemia is a significant risk factor for AS, and one of the most common methods for creating animal models of AS involves the use of a HCD.23,24 In hypercholesterolaemia, excess cholesterol in the bloodstream directly damages the vessel intima, leading to the deposition of fatty streaks on the vessel wall, an early sign of AS. 25 As cholesterol accumulation in the intima worsens and endothelial damage occurs, the infiltration of neutrophils and macrophages is triggered, inducing vascular inflammation and gradually leading to plaque formation, ultimately resulting in blood vessel stenosis. 25 In the present study, we used an HCD to induce AS in a zebrafish model. We observed that HCD led to cholesterol accumulation, neutrophil aggregation, macrophage infiltration, and plaque formation in zebrafish blood vessels, causing vascular stenosis. This finding suggests that constructing an AS model using an HCD is feasible. Treatment with the aqueous extract of DOKM significantly mitigated HCD-induced cholesterol accumulation, neutrophil aggregation, macrophage infiltration, and plaque formation in the zebrafish vasculature. These results indicated that DOKM effectively attenuated HCD-induced AS.
Lipid metabolism disorders are common risk factors for AS, particularly low-density lipoprotein cholesterol (LDL-C), which is an independent risk factor.26,27 LDL-C enters the vascular endothelial cells and undergoes oxidative modification, resulting in oxidised LDL (OX-LDL), which can directly damage the vascular endothelial cells and contribute to AS. OX-LDL is further phagocytosed to form foam cells, initiating early lipid streaks in AS, which may progress to atherosclerotic plaques under the influence of inflammatory mediators and fibroblast growth factors. 28 In this study, we observed significantly elevated lipid levels and LDL-C content in zebrafish fed an HCD, along with increased expression of genes involved in lipid metabolism, such as PCSK9, SREBF2, and HMGCR.29–31 These findings indicated a lipid metabolism disorder in the zebrafish AS model, resembling the phenotype of AS disease, further supporting zebrafish as a suitable animal model for AS model construction. Treatment with DOKM significantly reversed the lipid metabolism disorder phenotype in atherosclerotic zebrafish, as evidenced by reduced lipid levels, decreased LDL-C content, and significantly reduced expression of the PCSK9, SREBF2, and HMGCR genes. These results suggest that DOKM effectively improves lipid metabolism disorders and that its therapeutic effect on AS may be related to its effect in improving lipid metabolism disorders.
Gigantol exerts anti-inflammatory effects by inhibiting the release of inflammatory mediators and reducing the inflammatory response, thereby reducing inflammation-induced tissue damage and disease progression. 32 Previous studies demonstrated that gigantol partially alleviates CCl4-induced liver inflammation by inhibiting the c-Jun N-terminal Kinase (JNK)/cPLA2/12-LOX pathway. 33 Moreover, gigantol targets cancer stem cells (CSCs) in tumours and reduces tumour integrity by inhibiting the PI3 K/AKT/mTOR and Janus Kinase (JAK)/STAT pathways. 34 Fu et al 35 proposed that inhibiting the activation of the JAK/STAT pathway in vivo may alleviate atherosclerotic inflammation, thus presenting a novel target for AS treatment. Platelet-derived growth factor D (PDGFD,) a recognised molecular characteristic of AS, mediates the risk of AS by promoting inflammatory responses. 36 Molecular docking results suggested that gigantol may treat AS by targeting JAK and PDGFD. The results demonstrated that gigantol tightly binds to the JAK2 and PDGFRα targets. Gigantol forms one hydrogen bond each with glutamic acid (GLU)-930 and leucine (LEU)-932 on the JAK2 target. Additionally, it interacts with PDGFRα by forming one hydrogen bond each with lysine (LYS)-627, cysteine (CYS)-677, and aspartic acid (ASP)-836. These interactions suggest that gigantol's binding to these specific residues is a potential mechanism for its therapeutic effects on AS.
Chrysotobibenzyl inhibits the migration of lung cancer cells and sensitises them to cisplatin-mediated apoptosis. 37 A meta-analysis revealed a correlation between polymorphisms in ESR1 and the severity of coronary artery disease (CAD). 38 Molecular docking results indicated that chrysotobibenzyl has strong binding energy with both JAK2 and ESR1 targets, suggesting its potential as a compound for AS treatment. Specifically, Chrysotobibenzyl interacts with the JAK2 target by forming a hydrogen bond with LYS-912. Additionally, chrysotobibenzyl exhibits strong binding affinity to the ESR1 target by forming two hydrogen bonds with Asparagine (ASN)-348. These interactions highlight chrysotobibenzyl's significant binding capabilities with these targets.
Naringenin chalcone inhibits cholesteryl ester transfer protein (CETP), leading to an increase in high-density lipoprotein (HDL) cholesterol levels against AS. 39 CETP is a key lipid transfer protein involved in the exchange of cholesteryl esters between HDL and LDLs. Increased CETP activity is typically associated with reduced levels of HDL-C, which are known to have anti-atherosclerotic properties. Furthermore, excessive CETP activity may promote inflammatory responses and oxidative stress, both of which are critical factors in the development of AS. 40 Uncontrolled EGFR expression promotes tumourigenesis by inhibiting apoptosis, angiogenesis, and tumour-associated inflammation. 41 Molecular docking studies suggested that naringenin chalcone may be involved in AS treatment by targeting EGFR and AKT1. Specifically, naringenin chalcone forms four hydrogen bonds with the AKT1 target at GLU-228, Alanine (ALA)-230, GLU-234, and Asparagine (ASN)-279, indicating strong interactions. Additionally, Naringenin-Chalcone forms four hydrogen bonds with the EGFR target at Methionine (MET)-742, MET-769, Threonine (THR)-830, and ASP-831. These interactions suggest that Naringenin-Chalcone may have potential therapeutic value.
Hesperetin is a natural flavonoid with antioxidant and anti-inflammatory properties. 42 Studies have shown that hesperetin attenuates cardiovascular oxidative and pro-inflammatory effects induced by diesel exhaust particles in Wistar rats. 43 Moreover, Hesperetin exhibits antioxidative stress and anti-inflammatory activities by blocking sphingosylphosphorylcholine (SPC)-induced tyrosine kinases of the SPC/Src family, thereby inhibiting SPC-induced contraction of porcine coronary artery smooth muscle. 44 SRC kinase (Src) plays a key role in macrophage activation and is implicated in immune responses and inflammatory diseases, such as AS. 45 Molecular docking results suggested that hesperetin targeted SRC and ESR1, indicating its therapeutic potential. Specifically, hesperetin forms two hydrogen bonds with Arginine (ARG)-178 and HIS-204 on the SRC target. Additionally, it interacts with the ESR1 target by forming two hydrogen bonds with GLU-353 and LEU-346. These interactions indicate that Hesperetin engages effectively with these targets.
Tangeretin has antioxidant, anti-inflammatory, antitumour, hepatoprotective, and neuroprotective effects. 46 Studies have shown its therapeutic effect on osteoarthritis (OA) mediated through Nrf2/NF-κB and MAPK/NF-κB pathways. 47 Molecular docking studies indicated strong binding energy between tangeretin and SRC and EGFR targets, suggesting its potential as a new therapeutic agent. Specifically, Tangeretin forms two hydrogen bonds each with HIS-236 and HIS-242 on the SRC target, and it also forms two hydrogen bonds with Arginine (ARG)-172. Additionally, Tangeretin interacts with the EGFR target by forming two hydrogen bonds with Lysine (LYS)-721. These interactions highlight Tangeretin's strong binding affinity with these targets.
Network pharmacology has identified targets, such as JAK2, PDGFRA, ESR1, AKT1, EGFR, and SRC, which play roles in AS through various signalling pathways. JAK2 functions via the JAK/STAT pathway by phosphorylating STAT3 to regulate gene expression, which is crucial for inflammatory responses and vascular smooth muscle cell (VSMC) proliferation. Additionally, EGFR activation enhances inflammatory responses via the JAK2-STAT3 pathway.48,49 PDGFRA promotes VSMC proliferation and migration leading to arterial wall thickening and plaque formation. PDGFRA and EGFR signalling pathways jointly facilitate VSMC proliferation and AS progression. 50 ESR1, an oestrogen receptor, reduces inflammatory responses and oxidative stress, thereby inhibiting the onset and progression of AS. Cross-regulation of the ESR1 and AKT1 signalling pathways enhances its anti-inflammatory effects. 51 AKT1 operates via the PI3 K/AKT pathway and promotes cell survival, proliferation, and metabolic activity. AKT1 modulates endothelial cell function, suppresses inflammatory responses, and along with SRC, regulates cell migration and proliferation.49,50 EGFR and SRC activate the rat sarcoma virus protein (Ras)-rapidly accelerated fibrosarcoma kinase (RAF)-mitogen-activated protein kinase kinase (MEK)-extracellular signal-regulated kinase (ERK) and PI3K-AKT pathways, promoting cell proliferation, migration, and survival, which exacerbate the formation and instability of atherosclerotic plaques. Moreover, EGFR interacts with JAK2 and SRC pathways to regulate inflammation and cell proliferation.51,52
SREBF2 is a transcription factor that regulates cholesterol synthesis and uptake by activating downstream genes, such as HMGCR and low-density lipoprotein receptor (LDLR), thereby promoting cholesterol production and absorption. In AS, the overactivation of SREBF2 leads to increased cholesterol synthesis, which contributes to cholesterol deposition in the arterial walls and plaque formation. 53 HMGCR is the rate-limiting enzyme in cholesterol synthesis that catalyses the conversion of HMG-CoA to mevalonate. High HMGCR expression results in excessive cholesterol synthesis and accumulation and promotes plaque formation and progression. Inhibition of HMGCR activity is an effective method for reducing cholesterol levels. 54 PCSK9 regulates cholesterol metabolism by promoting LDLR degradation, thereby reducing the number of LDLRs on the cell surface. This decreases the ability of the liver to clear LDL cholesterol from the blood, leading to elevated blood cholesterol levels. The inhibition of PCSK9 can increase LDLR levels, enhance cholesterol clearance, and reduce the risk of AS. 55 Experimental results show that DOKM significantly reduced the expression of SREBF2, HMGCR, and PCSK9. However, the specific active constituents responsible for this regulation have yet to be identified. Further research is needed to determine whether the reduction in gene expression is due to the direct interactions of DOKM compounds with these targets or through indirect regulatory mechanisms. This will be an important area for future research to fully understand the therapeutic potential of DOKM for the treatment of AS.
Conclusions
In conclusion, this study delineated gigantol, chrysobibenzyl, naringenin chalcone, hesperetin, and tangeretin as active constituents within DOKM, along with core therapeutic targets comprising STAT3, PTPN11, JAK2, EGFR, PDGFRA, SRC, STAT1, TP53, ESR1, and AKT1 for AS management, as elucidated through comprehensive network pharmacological analyses. Experimental validation in zebrafish underscored the anti-atherosclerotic efficacy of DOKM, which was potentially mediated by its ameliorative effects on lipid metabolism. These insights provide a foundational rationale for the therapeutic exploration of DOKM for AS management and prophylaxis. Nonetheless, this study has some limitations, including the elusive mechanistic underpinnings of DOKM's anti-atherosclerotic actions and the inherent challenges associated with detecting protein-level alterations in zebrafish models because of the unavailability of species-specific antibodies.
Materials and Methods
Construction and Screening of the Active Components of DOKM
To identify the active components of DOKM extract, a multi-step approach was employed. Initially, a comprehensive review of literature and public databases such as PubMed, the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP) (https://old.tcmsp-e.com/tcmsp.php), and the Encyclopedia of Traditional Chinese Medicine (ETCM) (http://www.tcmip.cn/ETCM/index.php/Home/Index/index.htm) was conducted to collect information on the compounds contained in DOKM. Subsequently, the collected compounds were verified for canonical SMILES numbers through databases such as PubChem (https://pubmed.ncbi.nlm.nih.gov/) and ChemSrc (https://www.chemsrc.com/). These canonical SMILES numbers were then entered into the Swiss Target Prediction database (http://www.swisstargetprediction.ch/) to predict the target of action of each potential active ingredient.
Analysis of the Targets of DOKM in the Treatment of AS
To identify the targets of DOKM in the treatment of AS, relevant target information was retrieved from multiple databases including GeneCards (http://www.genecards.org/), DrugBank (http://go.drugbank.com), OMIM (http://www.omim.org/), and the Therapeutic Target Database (https://db.idrblab.net/ttd/) using the keyword “atherosclerosis”. The gene search results from each database were then integrated and de-duplicated to obtain AS-related genes. These AS-related genes were further intersected with the targets of DOKM active ingredients to obtain the targets of action of DOKM for the treatment of AS. This integration and analysis were conducted using the Metascape platform for Wayne diagram analysis (https://metascape.org/gp/index.html).
Construction of the “Active Ingredient-Target-Disease” Network
To construct the “active ingredient-target-disease” network, two essential files were created: the network file containing the network structure and the attribute type file specifying the type of each node. These files were then imported into Cytoscape 3.9.1 software. In the network diagram, the active ingredients, targets, and diseases are represented by nodes, whereas the links between them are represented by edges, forming an effective network diagram. Subsequently, a software network analyser tool was used to calculate the degree of each node. Based on this analysis, core active ingredients with therapeutic effects were identified and screened from the network.
Construction of PPI Network and Screening of Core Targets
DOKM therapeutic AS targets were imported into the STRING database (http://string-db.org/). We selected the “Multiple proteins” option and set the species to “
Molecular Docking
The structural formulas of the active ingredients were downloaded from the TCMSP database and converted to the appropriate format using the AutoDockTools software for use as ligands. Protein receptors were downloaded from the Protein Data Bank (PDB) database, and water molecules and small-molecule ligands were removed using the PyMOL software. Molecular docking was performed using the Vina software to dock the active compounds gigantol, chrysobibenzyl, naringenin chalcone, hesperetin, and tangeretin with their respective core targets identified among the top 10 by the MCC algorithm. The results were visualised and analysed using PyMOL software.
Reagents
Juvenile zebrafish basic feed was purchased from Shanghai Haisheng Biological Experimental Equipment Co. DOKM was purchased from the Anhui Dendrobium Shengji Biotechnology Co. An orange fluorescent-labelled cholesterol standard was purchased from Sigma-Aldrich. A cholesterol standard was purchased from Beijing Solebao Biotechnology Co. The remaining reagents were of analytical grade.
Instrumentation
Z-C-D6 centralised zebrafish culture units were purchased from Shanghai Haisheng Biological Experimental Equipment Co. An SZX7 Olympus stereoscope was purchased from Olympus Corporation, an HH-4 digital thermostatic water bath (Guohua Electric Co., Ltd), a LEGEND Micro 21R centrifuge (Thermo Scientific, USA), and a BS210S electronic balance (Sartorius) were also obtained.
Animals
Wild-type AB strain zebrafish, transgenic Tg(
Zebrafish AS Modelling
In this study, a zebrafish AS model was established by feeding zebrafish with a HCD [13]; 5 d post-fertilisation (dpf), zebrafish larvae were fed a normal basal diet for 3 d, followed by an HCD containing 8% cholesterol for 45 d.
Preparation of DOKM Water Extract
The stems of dried DOKM were crushed using a grinder. A total of 50 g was added to 500 mL distilled water for reflux extraction, and an extraction was taken again at 1 h. The aqueous extract was combined and filtered through a 200-mesh filter cloth. The aqueous extract was concentrated to 100 mL by using a rotary evaporator. The concentrate was freeze-dried into a powder and weighed. Finally, 4.68 g of powder was obtained with an extraction rate of 9.36%.
Experimental Grouping and Treatment
In this study, we divided zebrafish into the following four groups: control, AS model, and 1 and 10 mg/L DOKM. Each group was treated as follows: control group, 5 dpf zebrafish juveniles were selected and fed a normal basal diet for 3 d and then continued to feed on a normal basal diet for 45 d. AS model group: Zebrafish larvae at 5 dpf were fed a normal basal diet for 3 d and then an HCD containing 8% cholesterol for 45 d. One mg/L DOKM group: 5 dpf zebrafish larvae were fed a normal basal diet for 3 d and then fed an HCD enriched with 8% cholesterol for 45 d and treated with 1 mg/L DOKM. Ten mg/L DOKM group: 5 dpf zebrafish larvae were fed a normal basal diet for 3 d and then fed HCD enriched with 8% cholesterol for 45 d and treated with 10 mg/L DOKM.
Detection of Cholesterol Accumulation
The zebrafish AS model was established by selecting 5 dpf Tg(
Detection of Macrophage Infiltration
The zebrafish AS model was established by selecting 5 dpf Tg(
Detection of Macrophage Infiltration
Zebrafish with 5 dpf of Tg(
Detection of Lipid Levels
Wild-type AB strain zebrafish were used to construct the AS model, and the experimental groups and treatments were consistent with those in Materials and Methods “
Wild-type AB strain zebrafish were used to construct the AS model, and the experimental groups and treatments were consistent with those in Materials and Methods “
Detection of Plaques
Wild-type AB strain zebrafish were used to construct the AS model, and the experimental grouping and treatment were the same as in Materials and Methods “
Detection of pcsk9, srebf2, and hmgcr Gene Expression in Zebrafish
Wild-type AB strain zebrafish were used to construct the AS model, and the experimental groups and treatments were consistent with those in Materials and Methods “
Primer Sequence.

Reverse transcription reaction of RNA: RT reaction solution (5 × PrimeScript RT Buffer 2 μL, total RNA, RNase Free dH2O supplemented to 10 μL) was prepared according to the instructions, and placed in a reverse transcription apparatus to reverse into cDNA.
Using the above reverse transcription cDNA as a template, RT-PCR was used to detect the expression of
Data Analyses
Statistical treatment was performed by applying SPSS 10.0 software, origin 8.0 to process the images, data were expressed as the
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241282497 - Supplemental material for Dendrobium Officinale Kimura & Migo Attenuates High Cholesterol Diet-Induced Atherosclerosis
Supplemental material, sj-docx-1-npx-10.1177_1934578X241282497 for
Supplemental Material
sj-jpg-2-npx-10.1177_1934578X241282497 - Supplemental material for Dendrobium Officinale Kimura & Migo Attenuates High Cholesterol Diet-Induced Atherosclerosis
Supplemental material, sj-jpg-2-npx-10.1177_1934578X241282497 for
Footnotes
Acknowledgments
This work was supported by the Shihezi University.
Data Availability
All data used to support the findings of this study are included within the article.
Authors’ Contributions
Yang Zhang and Peng Zhao contributed equally to the work. Yang Zhang and Peng Zhao conceived and designed the experiments and wrote the paper. Yang Zhang, Peng Zhao, Qing Hao, Aiyun Guo, Song Han, Huanhuan Ren, Jichun Han, Nannan Jiang, and Qiusheng Zheng provided technical support. Nannan Jiang and Qiusheng Zheng supervised the whole project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Development Fund of Shanghai Pudong New Area, Young Medical Talents Training Program of Pudong Health Bureau of Shanghai, (grant number PW2022B-18, PWRq2021-26).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
