Abstract
Background
Staphylococcus aureus (S. aureus) is a leading cause of soft tissue and skin infections. 1 MRSA can cause a wide range of infections, from minor skin disorders like cellulitis and boils to more serious diseases including pneumonia, bacteremia, and septic arthritis. 1 Life-threatening complications of invasive MRSA infections might include necrotizing fasciitis and endocarditis. 1 Due to their role in hospital-acquired diseases and their ability to resist antibiotics, these bacteria pose serious challenges in healthcare settings. Because of its adaptability, MRSA serves as a reminder of the urgent need for innovative, creative approaches to tackle anti-microbial resistance (AMR) and to develop more efficient therapies for infections brought on by these microbes.2,3
In light of the urgent need for innovative approaches to combat AMR and develop more efficient therapies against infections caused by pathogens like MRSA, exploring natural plant extracts presents a promising avenue. Natural plants, long revered in traditional medicine, offer a rich array of secondary metabolites known for their anti-microbial properties. 4 Capitalizing on traditional knowledge, scientists are increasingly screening these natural products to decipher their mechanisms of action and potential therapeutic applications.5,6 Moreover, the cost-effectiveness of extracting anti-microbial agents from natural plants and their reported fewer side effects than synthetic agents underscores their potential as accessible and affordable treatments, particularly in regions where infectious diseases are endemic.7,8 Thus, investigating plant-derived anti-microbial compounds aligns with the pressing need for alternative, safe, and effective therapies against resistant pathogens like MRSA, emphasizing the synergy between traditional wisdom and modern scientific inquiry in addressing global health challenges.2,3
Prosopis juliflora (P. juliflora), native to America, Africa, and Asia, has long been utilized in traditional medicine to treat a variety of medical conditions, including asthma, diabetes, diarrhea, fever, liver infections, malaria, and rheumatism. 9 Recent studies have revealed that P. juliflora has a high antibacterial potential. 10 Methanolic and methanol extracts from leaves and seed pods, respectively, have been shown to be effective against a variety of bacteria, including Gram-negative and Gram-positive strains. 11 Notably, juliflorine, julifloricine, and a benzene-insoluble alkaloidal fraction from P. juliflora have shown therapeutic potential in the treatment of S. aureus skin infections,12‐14 and a methanol extract inhibits the growth of Plasmodium falciparum. 15 These findings identify P. juliflora as an interesting candidate for further investigation in the development of new antibacterial drugs.
Acacia ehrenbergiana (A. ehrenbergiana), scientific name Vachellia flava (Forssk.) Kyal. & Boatwr (Plants of the World Online. Royal Botanic Gardens, Kew), another plant distributed in North and East Africa and the Arabian Peninsula, known for its therapeutic characteristics, has been extensively studied for its antibacterial efficacy in vitro assays. 16 Previous research has shown that extracts from this plant exhibit concentration-dependent antibacterial activity, with substantial potency against both Gram-negative and Gram-positive bacterial strains,16,17 including Bacillus and Staphylococcus aureus species. 16 Interestingly, geographical differences in antibacterial efficacy against different bacterial strains have emerged when comparing in vitro investigations conducted in Sudan, Egypt, Saudi Arabia, and Qatar. 16 Nevertheless, these findings collectively highlight the potential of A. ehrenbergiana (Hayne) as a source of antibacterial agents with regional variations in effectiveness against different bacterial strains.
Traditional in vitro methods of antibacterials’ screening, focused on the direct effects on microorganisms, face critical limitations with compounds exhibiting novel mechanisms of action, challenges associated with the culture of specific microorganisms, potential toxicity, and resistance barriers. As a consequence, there is a need for more robust in vivo multicellular models in anti-microbial drug discovery. Caenorhabditis elegans (C. elegans), a small, transparent nematode commonly used in different research areas thanks to its short life cycle, genetic tractability, and physiological similarities to higher organisms, provides a strong platform for anti-microbial drug development in a variety of experimental settings. 18 Survival assays offer a quick and effective platform for the discovery of novel anti-microbials and for the assessment of their effectiveness while additionally providing insights into the organisms’ response to pathogenic stresses. 19
In vitro studies have outlined the ethnopharmacological and antibacterial profiles of A. ehrenbergiana (Hayne) and P. juliflora.16,20 However, data from in vivo models is not available. To fill this knowledge gap, we investigated, for the first time, the anti-infective properties of these plants on methicillin-susceptible and -resistant S. aureus (MSSA and MRSA, respectively), using C. elegans as a live infection model. We report here that both A. ehrenbergiana (Hayne) and P. juliflora extracts increase the lifespan of S. aureus-infected worms, and we provide insights into potentially involved mechanistic aspects.
Results
A. ehrenbergiana (Hayne) and P. juliflora Extracts Enhance the Survival of Methicillin-Susceptible- and Methicillin-Resistant S. aureus-Infected Worms
In initial studies, killing assays were performed to determine mean time to death (TDmean) following worms’ exposure to MSSA 25923, MRSA 195, and MRSA 3820 strains (Figure 1a). Worms’ lifespan was similarly significantly reduced in the presence of all strains tested. Consistent with previous studies,21,22 a TDmean of 72 ± 1.3 h was observed for worms fed with MSSA 25923, MRSA 195, and MRSA 3820, with <20% of them surviving on day 7. In contrast, worms exposed to control E. coli OP50 remained alive throughout the assay.

Survival of bacteria-infected C. elegans in the presence or absence of A. ehrenbergiana (Hayne) and P. juliflora ethanol extracts. (a) Methicillin-susceptible and methicillin-resistant S. aureus kill C. elegans on solid agar medium. Kaplan–Meier survival curves refer to wild-type (N2) C. elegans continuously feeding on lawns of MSSA 25923, MRSA 195 or MRSA 3820 grown on LB agar medium. Worms feeding on control E. coli OP50 survived for > 7days. In contrast, worms grown on pathogenic strains MSSA 25923, MRSA 195 or MRSA 3820 strains showed significant lifespan reductions. Mean time to death (TDmean) for C. elegans-infected with MSSA 25923, MRSA 195 or MRSA 3820 was 72 ± 1.3 h. Data are presented as mean ± SEM of six replicates (35 nematodes/replicate) from a representative experiment out of three independently performed assays. (b-g) Survival of C. elegans worms following exposure to MSSA 25923, MRSA 195 or MRSA 3820 on solid agar medium 72 h post-infection. (b-d and e-g) Mean percentage of survival of infected worms in the presence or absence of different concentrations of A. ehrenbergiana (Hayne) (AEH) or P. juliflora (PJ) ethanol extracts. While only 20% of worms infected with MSSA 25923, MRSA 195 and MRSA 3820 survived in control conditions, the indicated A. ehrenbergiana (Hayne) and P. juliflora extracts concentrations significantly increased worms’ survival. Curcumin (100 µg/ml) was used as a positive control. Data, presented as mean ± SEM of three replicates (30 nematodes/replicate), refer to one representative experiment out of three independently performed assays. *P < 0.05, **P < 0.001 & ***P < 0.0001.
Effects of A. ehrenbergiana (Hayne) and P. juliflora extracts on the survival of C. elegans-infected with MSSA 25923, MRSA 195, and MRSA 3820 were then analyzed (Figure 1b-g, Supplementary Figure 1). Lifespans of MSSA 25923-, MRSA195-, and MRSA 3820-infected C. elegans were significantly increased after 72 h infection in the presence of both extracts under investigation. In particular, A. ehrenbergiana (Hayne) at 300 µg/ml and 500 µg/ml concentrations increased worms’ survival in the presence of all strains by 65–70% (Figure 1b-d), whereas a 100 µg/ml concentration was only effective against MSSA 25923 infection.
Instead, P. juliflora extract improved worms’ survival upon infection with all three strains by 50–70% at 100, 300 and 500 µg/ml concentrations (Figure 1e-g). Notably, the highest effectiveness of P. juliflora was observed at 300 µg/ml with 78 ± 5.6% and 76.3 ± 3.8% survival for worms infected with MRSA 195 and MRSA 3820 strains, respectively.
Overall, medium and high concentrations of A. ehrenbergiana (Hayne) and P. juliflora extracts showed in vivo antibacterial effects comparable to those observed in the presence of a 100 µg/ml concentration of curcumin, a known anti-infective compound.
A. ehrenbergiana (Hayne) and P. juliflora Extracts Fail to Inhibit Methicillin-Susceptible and Methicillin-Resistant S. aureus Proliferation
To obtain mechanistic insights into the effects of A. ehrenbergiana (Hayne) and P. juliflora extracts on the lifespan of MSSA- and MRSA-infected worms, we explored their direct antibacterial potential.
Minimum Inhibitory Concentration (MIC) assays performed with a range of concentrations, spanning between 25 and 1000 µg/ml for both A. ehrenbergiana (Hayne) and P. juliflora, showed no inhibition of MSSA 25923, MRSA 195 and MRSA 3820 growth. In contrast, gentamycin (30 µg/ml) inhibited MSSA 25923 proliferation, while vancomycin (50 µg/ml) inhibited bacterial proliferation of all three strains tested (data not shown).
Effect of A. ehrenbergiana (Hayne) and P. juliflora Extracts on Caenorhabditis elegans Pharyngeal Pumping Rate and Intestinal Colonization by Methicillin- Susceptible and Methicillin-Resistant S. aureus
S. aureus and MRSA colonization of worms’ gut has been shown to correlate with C. elegans mortality.21,23 Although S. aureus does not persist in the host's intestine, prolonged contact leads to host death. Similar studies have shown that S. aureus, E. faecalis, and P. aeruginosa bacterial load can reach up 103–105 CFU/worm.22‐25
A CFU assay was conducted to assess the effects of the plant extracts on S. aureus levels in the worm intestine. In vivo CFU counts were measured at 12 and 24 h, with >70% of the untreated, infected worms remaining alive.
After 24 h exposure to MSSA 25923, MRSA 195, and MRSA 3820 in the absence of plant extracts, all bacterial strains effectively colonized worms’ intestines, with 4.3 × 104, 4.8 × 104, and 4.9 × 104 CFU per worm, respectively (Figure 2a and b). These findings align with published data.21‐23
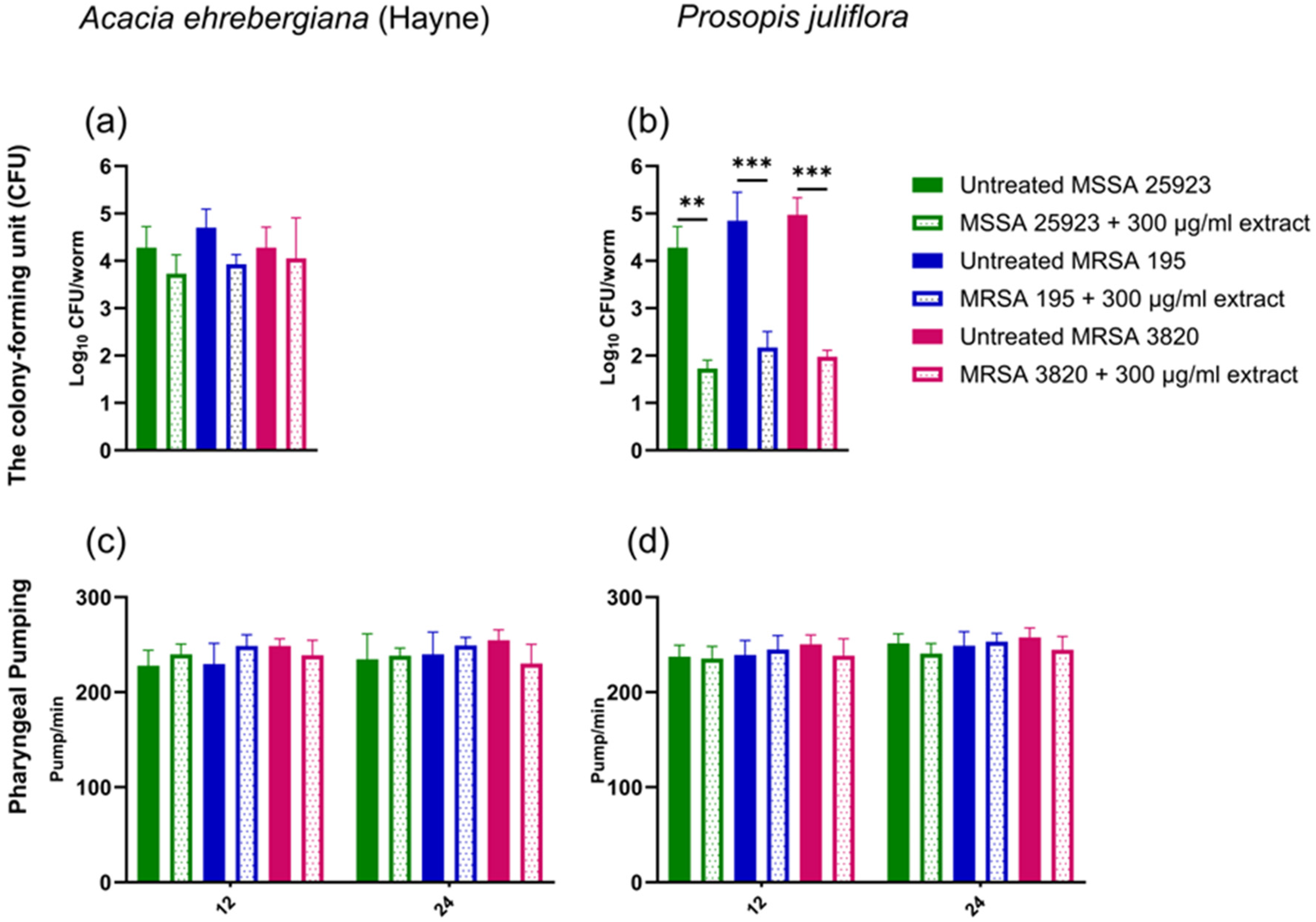
Effects of A. ehrenbergiana (Hayne) and P. juliflora ethanol extracts on the numbers of MSSA and MRSA strains in infected C. elegans. Bacterial CFU from intestines of MSSA 25923-, MRSA 195- and MRSA 3820-infected worms were enumerated 24 h post-infection in the presence or absence of plant extracts at 300 μg/mL concentration. (a and b) Bacterial CFU in the presence of A. ehrenbergiana (Hayne) and P. juliflora. (c and d) The feeding behavior was measured by counting pharyngeal pumping rates in the presence of A. ehrenbergiana (Hayne) and P. juliflora at 12 h and 24 h post-infection. Data are presented as mean ± SEM of four replicates (4 nematodes/replicate) from one representative experiment out of four independently performed assays. *p < 0.05, **p < 0.001 & ***p < 0.0001.
However, in the presence of 300 μg/mL concentration of P. juliflora, a significant reduction in the number of bacteria collected from intestines of C. elegans (1.8 × 102, 2.2 × 102, and 1.9 × 102 CFU/worm) fed with MSSA 25923, MRSA 195, and MRSA 3820, respectively (Figure 2b), was detectable. In contrast, no reduction in the number of bacteria was observed in the presence of A. ehrenbergiana (Hayne) extract (Figure 2a).
To investigate whether bacterial load in the C. elegans could impact feeding behavior, we evaluated pharyngeal pumping 26 in the presence of A. ehrenbergiana (Hayne) and P. juliflora extracts at 12- and 24-h interval post-infection. However, no reduction in pharyngeal pumping was observed in the presence of either plant extract (Figure 2 c and d).
Overall, this data suggests that the reduction in bacterial content in the intestine of the C. elegans observed upon P. juliflora treatment is not associated with defective feeding behavior.
Discussion
S. aureus, a Gram-positive bacterium commonly associated with community- and hospital-acquired infections, has acquired resistance to penicillin and semi-synthetic methicillin.27,28 Due to their ability to produce potent toxins and enzymes, along with the formation of biofilms, these bacteria are formidable pathogens. MRSA has become prevalent in various environments, and the emergence of new strains poses new challenges as they progressively develop heightened resistance to the limited number of available antibiotics.28‐30 The identification of novel antibacterial products is urgently necessary. To address these issues, we leveraged the C. elegans survival assay, hinging on the colonization and subsequent proliferation of pathogenic bacteria in the worm gut, to assess the ability of A. ehrenbergiana (Hayne) and P. juliflora ethanol extracts to inhibit the lethal impact of staphylococcal infection on the host.22,24,25,31,32
In this study, we have demonstrated that A. ehrenbergiana (Hayne) and P. juliflora significantly extended the lifespan of MSSA 25923- and MRSA 195- and MRSA 3820-infected worms similarly to curcumin, a potent anti-infective compound, active against many bacterial strains including S. aureus and, in particular, MRSA.32‐35 Importantly, neither extract was able to directly kill MSSA or MRSA bacteria. Therefore, it is reasonable to hypothesize that A. ehrenbergiana (Hayne) and P. juliflora extracts may limit pathogens’ virulence in vivo without compromising their viability,36,37 or, possibly, modulate host immune responses.38,39 These characteristics appear to be favorable in the quest for new therapeutic agents against pathogenic bacteria, since they do not contribute to increased selective pressure on bacteria, leading to the development of antibiotic resistance.36,37,40 Furthermore, these findings further underline the superiority of the C. elegans model in comparison with traditional in vitro assays, which usually overlook compounds that do not directly impact on bacterial replication.
Interestingly, P. juliflora showed potent anti-infective properties against MSSA and MRSA, possibly due to its rich phytochemical content. This plant, endemic to dry and semi-arid parts of Africa, the Americas, and Asia, has been the focus of many research studies.9‐15 Plants within the Prosopis L. genus, part of the Fabaceae family and Mimosoideae subfamily, are particularly popular in traditional medicine for their potential applications in anticancer, antidiabetic, anti-inflammatory, and antibacterial treatments. In our experiments, P. juliflora extract not only enabled the infected C. elegans to survive infection by methicillin-susceptible and -resistant S. aureus strains, without direct bactericidal effects in vitro, but also prevented intestinal colonization without affecting feeding rate. These observations suggest that this plant extract might enhance the host immune system's ability to eliminate bacteria or disrupt bacterial virulence factors favoring pathogen accumulation in the intestine. On the other hand, A. ehrenbergiana (Hayne) prolonged the lifespan of MSSA- and MRSA-infected C. elegans without reducing S. aureus accumulation in the gut or negatively impacting pharyngeal pumping. Thus, its favorable effects were still detectable in the presence of a substantial bacterial load in the gut.
However, our data suggest that while neither P. juliflora nor A. ehrenbergiana (Hayne) are endowed with direct bactericidal potential, partially different, indirect, antimicrobial mechanisms might be operational. Indeed, despite similar capacities to support worm survival and feeding behavior, the two extracts differentially impact intestinal bacterial colonization. Different components of the C. elegans immune system might play critical roles in the eliciting these effects.
In utilizing C. elegans as an in vivo model to screen for novel plant-based anti-infective compounds against S. aureus, we acknowledge several limitations inherent to this model system. Notably, C. elegans possesses only innate immunity, lacking the adaptive immune responses found in mammalian hosts. 41 This limitation restricts the scope of immune mechanisms that can be explored and may not fully recapitulate the complexity of host-pathogen interactions seen in mammals. 42 Furthermore, while our study demonstrated the ability of A. ehrenbergiana (Hayne) and P. juliflora extracts to extend the lifespan of S. aureus-infected worms, it is essential to note that neither extract exhibited direct bactericidal activity. This suggests that their anti-infective effects may be mediated through indirect mechanisms, such as modulation of host immune responses or limitation of pathogen virulence.
Additionally, the differential impact of the two extracts on intestinal bacterial colonization underscores the complexity of their anti-microbial mechanisms. It highlights the need for further research to elucidate the active compounds and associated anti-infective mechanisms.
Despite these limitations, our study represents the first demonstration of the anti-infective properties of A. ehrenbergiana (Hayne) and P. juliflora against MSSA and MRSA strains using the C. elegans model, emphasizing the model's relevance in pre-clinical infection biology studies and paving the way for future mechanistic investigations.
Conclusion
In this study, we have demonstrated for the first time the anti-infective properties of A. ehrenbergiana (Hayne) and P. juliflora ethanol extracts in a live-infection intact host model, C. elegans. Although these plant extracts did not directly kill the bacteria, they markedly enhanced the survival of MRSA-infected worms. Nevertheless, the mechanism by which these plants enhance immunity in C. elegans should be examined.
Materials and Methods
Plant Extract Preparation
A. ehrenbergiana's (Hayne) stem and P. juliflora's leaves were collected from the Kordofan region of western Sudan (Voucher number: MAPRI-A.E.168/19). Samples were prepared and identified at the Medicinal and Aromatic Plants and Traditional Medicine Research Institute in affiliation with the National Centre for Research in Khartoum, Sudan. Following the methodology outlined by Harborne, 43 with slight adjustments. Samples were dried under shade and then packed and transported under controlled conditions, ensuring no contamination. The dried plant was then powdered into a fine powder using a mechanical grinder (500 g). The powder was mixed and immersed in 2500 ml of 80% ethanol (analytical grade, 95%) and continuously agitated for 72 h at room temperature. To ensure the absence of ethanol in the final product, the extract underwent an additional drying process using a vacuum oven (38 °C) till it was fully dry. The resulting supernatants were filtered through a 0.45 µm pore size filter paper. This process was repeated twice, and dried extracts were stored in dark vials at −20 °C.
Bacterial and Nematode Strains Collection and Preparation
MRSA ATCC BF195 and BC 3820, hereafter referred to as MRSA 195 and MRSA 3820, were obtained from the Department of Microbiology, College of Medicine at Alfaisal University, KSA. MSSA ATCC 25923 was used as a reference strain. The MSSA and MRSA strains were grown with aeration in Trypticase Soy (TS) media for 16 h at 37 °C while the Escherichia coli OP50 strain was grown in Luria-Bertani (LB) broth supplemented with streptomycin (100 µg/ml). Wild-type C. elegans N2 strains were maintained at 20 °C on a nematode growth medium (NGM) and fed on E. coli OP50 as a standard laboratory food source 44 (Table 1).
C. elegans and Bacterial Strains Used in This Study.

*Caenorhabditis Genetics Center (CGC).
Caenorhabditis elegans Killing Assay
The killing assay was adapted from a previous study. 21 An overnight culture of MSSA 25923, MRSA 195, and MRSA 3820 strains grown in TS broth (50 µl) was spread on 35 mm TS agar plates and incubated at 37 °C for 4 h. Plates were then removed and left to cool at room temperature for 1 h before use. A total of 35 young adult worms (L4 + 1 day) were transferred to assay plates and incubated at 25 °C. E. coli OP50 was used as a negative control. Three replicas were independently obtained. Numbers of dead and alive worms were counted every 24 h following exposure to the pathogen. Worms were transferred to new plates every 48 h due to the presence of new progeny. A worm was considered dead if it failed to respond to plate tapping or gentle touch by the platinum wire.
Anti-Infective Screening of A. ehrenbergiana (Hayne) and P. juliflora
Anti-infective assays were performed on agar media, as described previously.25,31 A 2 ml TS agar was added to 35 mm assay plates. While TS agar was still in liquid form, different concentrations of A. ehrenbergiana (Hayne) and P. juliflora (50–700 µg/ml) were added and thoroughly mixed. As positive control, we used curcumin at a 100 µg/ml concentration. Plates were left to set at room temperature for 24 h before seeding with 50 µl of different MSSA and MRSA strains and subsequent incubation at 37 °C for 4 h. Consequently, plates were removed and left to cool at room temperature before use. As negative controls, extracts were replaced with 1% ethanol, and MSSA 25923, MRSA 195 or MRSA 3820 were substituted with E. coli OP50. Age-synchronized adult worms (35 worms) were transferred to assay plates and incubated at 25 °C. Their survival was recorded manually every 24 h for seven days. Worms were transferred to new plates every 48 h due to the presence of new progeny.
Determination of MIC of A. ehrenbergiana (Hayne) and P. juliflora
Bactericidal and bacteriostatic effects of A. ehrenbergiana (Hayne) and P. juliflora were evaluated using broth microdilution MIC assay. 45 A range of concentrations of A. ehrenbergiana (Hayne) and P. juliflora was prepared in a 96-well plate. The concentrations of MRSA and MSSA were adjusted to 108 CFU/mL (MacFarland standard) and then inoculated into the wells at a final concentration of 106 CFU/mL.
Evaluation of Pharyngeal Pumping Rate in Caenorhabditis elegans
Worms were prepared and exposed to different bacterial strains, as described in the killing assay protocol, in the presence or absence of A. ehrenbergiana (Hayne) and P. juliflora extracts. The pharyngeal pumping rate of 10 worms was counted for 1 min at 12- and 24-h intervals. A single pump was scored when a backward movement of the terminal bulb grinder was completed. Four independent repeats were conducted.
Colony-Forming Unit (CFU) Assays of Live Methicillin-Susceptible and Methicillin-Resistant S. aureus Recovered from Caenorhabditis Elegans’ gut
After exposing C. elegans to MSSA 25923, MRSA 195 or MRSA 3820 for 24 h in the presence or absence of A. ehrenbergiana (Hayne) and P. juliflora as described,32,46 10–12 live worms were randomly selected and briefly anesthetized in 25 mM sodium azide. Subsequently, they were washed in a 200 μl antibiotic cocktail containing 25 mM Levamisole and 10 μg/ml gentamicin and incubated for 45–60 min to eliminate bacteria from their cuticles. After three washes with 200 μl of 25 mM Levamisole, worms were counted, mechanically disrupted in 50 μl of 1% Triton X, and subjected to serial dilutions. Ten microliters of each dilution were spotted on TS agar with 5 μg/ml nalidixic acid, and bacterial colonies were counted after overnight incubation at 37 °C. Bacterial CFU per worm were calculated using the formula from Ooi et al. 24
Statistical Analysis
Data from the killing assays were analyzed using the Kaplan-Meier Cumulative Survival Plot for time (nonparametric survival analysis). The Log-rank (Mantel-Cox) significance test was used for comparisons between survival curves. Results from the anti-infective assays were transformed into bar charts, and the statistical significance of differences between groups was evaluated using one-way analysis of variance (ANOVA). When appropriate, Tukey's post hoc multiple comparisons test was applied for ranking. Differences with p < 0.05 were considered statistically significant. Data are presented as mean ± standard error of the mean (SEM) and are representative of three or more independent experiments. All analyses were performed using GraphPad Prism version 10.2.3.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241279065 - Supplemental material
Supplemental material, sj-docx-1-npx-10.1177_1934578X241279065
Footnotes
Acknowledgements
We thank Giulio C. Spagnoli, National Research Council, Institute of Translational Pharmacology, Rome, Italy, for valuable improvement and discussions of the manuscript draft. We thank Caenorhabditis Genetics Center (CGC) for providing the C. elegans and E. coli OP50 strains.
Author Contributions
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Alfaisal University Internal Research Grant (IRG), grant number 2331, to Samah H. O. Zarroug. Professor Khaled Al-Kattan, the Dean of the College of Medicine at Alfaisal University, funded the APC.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
