Abstract
Introduction
The investigation of novel biological sources continues to be an important strategy in the discovery of new and biologically active chemical diversity. 1 Fungal endophytes represent a relatively new and largely unexplored source of biodiversity2-4 that are currently providing a remarkable diversity of structurally unique bioactive natural products.5-8 In our continuing search for biologically active natural products from endophytic fungi of Canadian medicinal plants,9-12 we have investigated the EtOAc extract of an Aureobasidium pullulans endophyte (JAJ1-073) isolated from the needles of white cedar (Thuja occidentalis).
The JAJ1-073 extract selectively inhibited the growth of Mycobacterium tuberculosis in our bioassay screening and subsequent examination by NMR spectroscopy indicated that it was unique within our collection of fungal extracts and contained
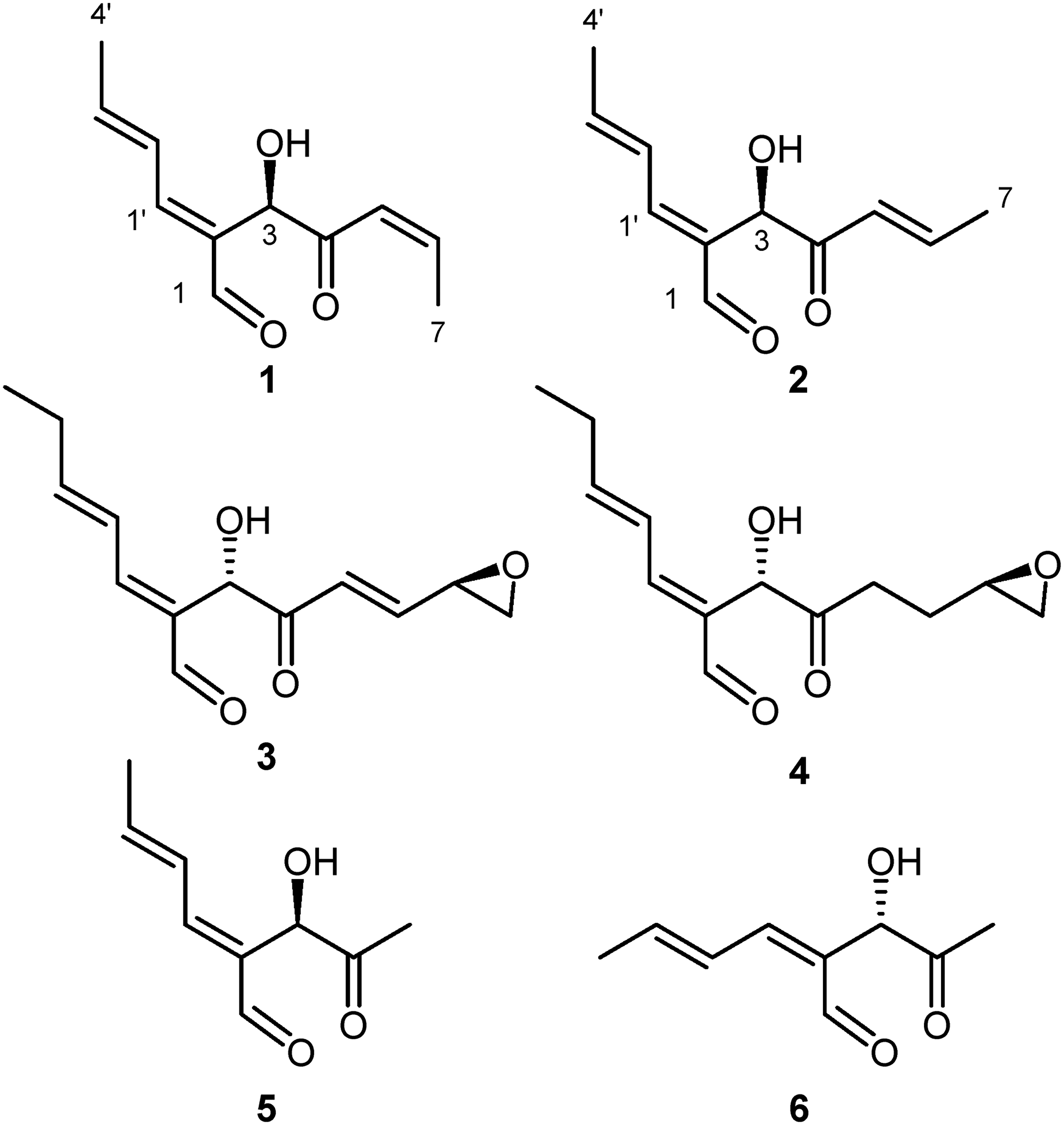
Results and Discussion
Fungal isolate JAJ1-073 was obtained from needles of Thuja occidentalis collected in Saint John, NB, Canada through an indirect isolation protocol on malt extract agar. The isolate was identified as Aureobasidium pullulans by morphological examination of colonies on various solid media (supporting information, Figure S1) and ITS DNA sequencing. An extract of the isolate obtained from a 2-week liquid fermentation of JAJ1-073 in potato dextrose broth was subjected to reversed-phase flash chromatography and HPLC to afford
1H NMR (400 MHz) and 13C NMR (100 MHz) data for

The 1H, 13C, and HSQC NMR data of compound

Key COSY and HMBC correlations for
The NMR data obtained for compound
Repeated attempts to prepare α-methoxy-α-trifluoromethylphenylacetoyl esters or a 2,4-dinitrophenylhydrazone derivative to assign the absolute stereochemistries of
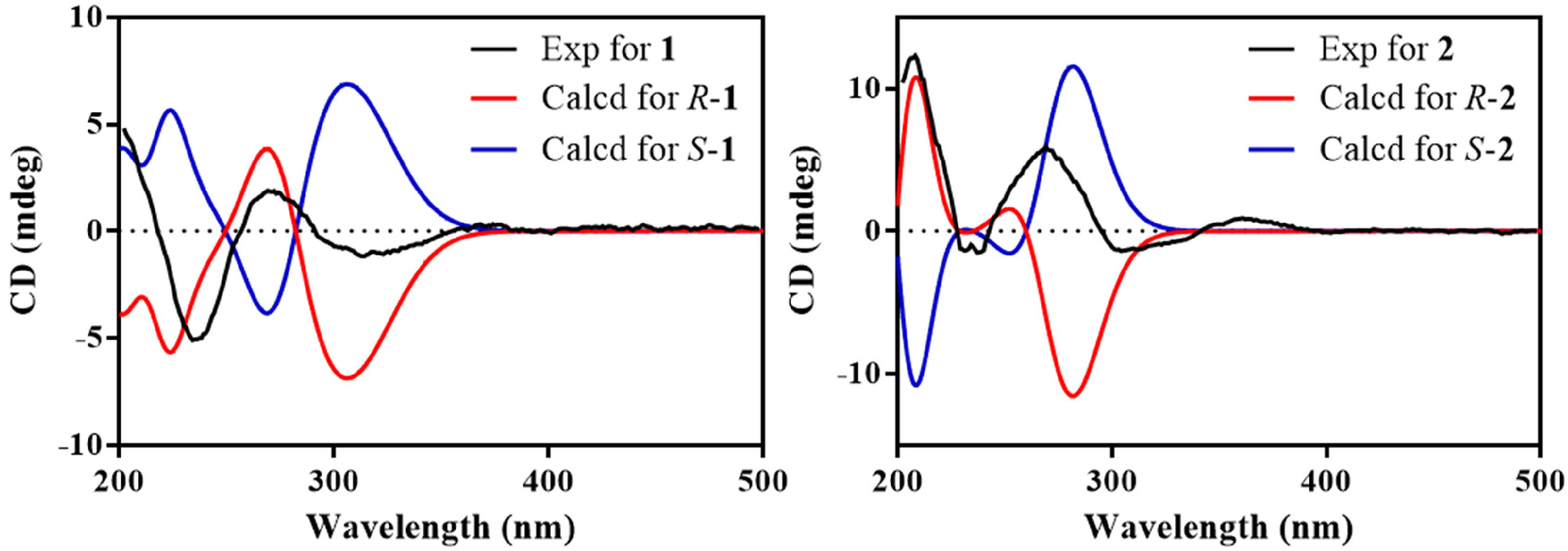
Experimental and calculated ECD spectra of
Experimental and calculated ORD values for R-

We therefore used ORD data to confirm the assignment of configurations (Table 2) and the calculated ORD data for the R-series enantiomers provided large, negative values at all four wavelengths that decrease in magnitude with increasing wavelength (Table 2). These data provide a good match with the trends seen in the experimental values of
Surprisingly few natural products that contain the highly functionalized structural motif present in the aureobasidols have been isolated, with all of the related polyketides being of fungal origin: mollipilin A (
Conclusion
Two new highly functionalized C11-polyketides, aureobasidol A (
Experimental
General Experimental Procedures
Optical rotations were recorded in MeOH on a Bioevopeak POL-537 polarimeter at 633, 589, 546, and 436 nm. UV spectra were measured on a Shimadzu UV-1280 spectrophotometer. Electron circular dichroism was measured on a JASCO-810 spectropolarimeter at room temperature, using a 1-mm quartz cuvette. NMR spectra were recorded on an Agilent 400-MR DD2 instrument in deuterated solvents and were calibrated to residual protonated solvent resonances. HR-MS data were recorded on a Thermo LTQ Exactive instrument with an electrospray ionization (ESI) source. Solvents for extraction and isolation were purchased from Fisher Scientific (Ottawa, ON, Canada) and deuterated solvents for NMR spectroscopy were purchased from Sigma-Aldrich (Oakville, ON, Canada). Flash chromatography was performed using a Biotage Flash + chromatography system fitted with C18 (reversed-phase) SiliaSep cartridges (40-63 µm, 60 Å, 25 g; SiliCycle, QC, Canada). Semi-preparative reversed-phase HPLC was performed on a Phenomenex Luna C18 column (250 × 10 mm, 10 µm, 100 Å) using an Agilent 1100 HPLC system comprising a G1311A binary pump and a G1315C diode array detector.
Endophyte Isolation
JAJ1-073 was isolated from the needles of Thuja occidentalis collected from Millidgeville, Saint John, NB, Canada (45°18′22″ N, 66°5′4″ W) in Fall 2015 and a voucher specimen has been deposited at the New Brunswick Museum (voucher number: NBM VP 40880). Needle surfaces were sterilized by sequential immersion in 70% ethanol (EtOH) for 5 s and rinsed with sterile distilled water (H2O) for 10 s and then immersed in 6% aqueous sodium hypochlorite (NaOCl) for 10 s and rinsed with sterile distilled H2O and blotted dry on autoclaved paper towel. Sterile tissue was immediately aseptically cut into pieces (1 cm) that were placed onto 2.0% malt extract agar and incubated at room temperature under ambient light.
Endophyte Identification
Isolate JAJ1-073 was identified as Aureobasidium pullulans through the examination colony morphology when grown on cornmeal, Czapek-Dox, malt extract, and potato dextrose agar (Supplementary Figure S1). The taxonomic classification was confirmed by comparison of the internal transcribed spacer (ITS) and 5.8S rRNA gene deoxyribonucleic acid (DNA) regions with corresponding sequences available in the GenBank database (National Center for Biotechnology Information, US National Library of Medicine, Bethesda, MD, USA). The genomic DNA of JAJ1-073 was isolated using a DNEasy plant mini kit (Qiagen, Toronto, Ontario) as directed by the manufacturer; the ITS gene was amplified by polymerase chain reaction using the ITS1 and ITS4 universal fungal primers (Invitrogen, Burlington, Ontario) as previously described, 18 and the amplified ITS DNA was sequenced by Genome Québec (Montreal, Québec). The JAJ1-073 DNA sequence was checked for ambiguity before being compared with existing GenBank sequence data using the Basic Local Alignment Search Tool. The ITS gene sequence of JAJ1-073 was found to have >99% homology with numerous conspecific A. pullulans isolates and has been deposited in GenBank (accession number: MT738220).
Fermentation and Isolation
JAJ1-073 was fermented in 1.2% potato dextrose broth at room temperature with shaking (150 rpm) for 2 weeks (5 L; 50 × 100 mL batches in 250 mL Erlenmeyer flasks stoppered with foam baffles). Fermentation cultures were sonicated for 30 s, the fungal material was removed by filtration, and the spent broth was extracted with ethyl acetate (EtOAc, 3 × 1.6 L). The organic fractions were combined and concentrated in vacuo (650 mg). The PDB extract was subjected to C18 flash chromatography (stepwise gradient from 100% H2O to 100% acetonitrile [CH3CN] in 10% increments) to give 11 fractions. Fraction 3 (4:1 H2O/CH3CN; 146 mg) was subjected to further isocratic reversed-phase HPLC (7:3 H2O/CH3CN) to give
(R,2E,5Z)-2-((E)-But-2-en-1-ylidene)-3-hydroxy-4-oxohept-5-enal (1)
-160.8 (c 4.00, MeOH); UV (MeOH) λmax (log ε) 227 (2.84), 275 (2.68) nm; CD (c 3.4 mM, MeOH) λmax (Δε) 200 (0.48), 217 (0), 234 (-0.46), 255 (0), 274 (0.17), 290 (0), 313 (-0.11), 353 (0) nm; 1H and 13C NMR data in CDCl3, see Table 1 and Supplementary Figures S2–S7; HR-ESI-MS m/z 217.0834 [M+Na]+ (calcd for C11H14O3Na+, 217.0835).
(R,2E,5E)-2-((E)-But-2-en-1-ylidene)-3-hydroxy-4-oxohept-5-enal (2)
-41.6 (c 1.00, MeOH); UV (MeOH) λmax (log ε) 217 (2.87), 272 (2.64) nm; CD (c 20 mM, MeOH) λmax (Δε) 207 (0.19), 227 (0), 239 (-0.03), 245 (0), 270 (0.09), 295 (0), 306 (-0.02), 341 (0), 362 (0.1) nm; 1H and 13C NMR data in CDCl3, see Table 1 and Supplementary Figures S9–S14; HR-ESI-MS m/z 217.0835 [M+Na]+ (calcd for C11H14O3Na+, 217.0835).
ECD and ORD Computational Methods
An exhaustive molecular mechanics conformer search of
Electronic transition and rotational strength calculations were performed by TDDFT for 10 transitions/excited states at the CAM-B3LYP-D3BJ/def2-TZVPD[CPCM]//B3LYP-D3BJ/def2-TZVPD[CPCM] level (ORCA 5.01) and optical rotatory dispersion calculations were carried out by DFT for wavelengths unaffected by electronic absorptions
21
(ie >420 nm) at the CAM-B3LYP/def2-TZVPD[CPCM]//B3LYP-D3BJ/def2-TZVPD[CPCM] (Gaussian16
22
) on the conformers that made up 99.9% of the Boltzmann distribution at the B3LYP-D3BJ/def2-TZVPD level (18 for
Antimycobacterial Activity
Antimycobacterial activity was determined against Mycobacteria tuberculosis strain H37Ra (ATCC 25177) using a microplate resazurin assay as previously reported. 24
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231210086 - Supplemental material for Aureobasidols A and B: New C11-Polyketides From an Endophytic Aureobasidium pullulans Isolate
Supplemental material, sj-docx-1-npx-10.1177_1934578X231210086 for Aureobasidols A and B: New C11-Polyketides From an Endophytic Aureobasidium pullulans Isolate by Nicholas J. Morehouse, John A. Johnson and Christopher A. Gray in Natural Product Communications
Footnotes
Acknowledgements
The authors are grateful to Hilary Jenkins (McMaster University), Larry Calhoun (UNB), and Josh Kelly (UPEI) for acquisition of 700 MHz NMR, 400 MHz NMR, and high resolution ESIMS data, respectively.
Authors’ Contributions
N.J.M. and J.A.J. isolated and identified JAJ1-073 and confirmed its taxonomy. N.J.M isolated and elucidated the structures of compounds
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this research was provided by the Natural Sciences and Engineering Research Council of Canada (Discovery Grant 2019-04114 to CAG and CREATE Grant 510963 supporting the CREATE Training Program in BioActives) and the New Brunswick Innovation Foundation (Research Assistantship Initiative grants 2018-033 and 2019-024 to CAG).
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
