Abstract
Background
Traditional Chinese Medicine Astragalus was commonly used to assist chemo-drug treatment in clinical cancer patients. However, whether Astragalus injection (AGI) reverses drug resistance remains unclear. Therefore, a meta-analysis is necessary to assess the effect of AGI on resistance reversement in cancer patients.
Method
The studies presenting tumor response rates in cancer patients receiving chemo-drugs treatment in combination with AGI were systematically searched from six common scientific databases until February 2024. The relative risks (RRs) indicating the tumor response rate, 1-year survival rate, and quality of life improvement in clinical patients among two groups were calculated in metan package. The pooled RRs with 95% confidence intervals (CIs) were used to explore the effect of AGI on enhancing drug sensitivity in terms of tumor response, 1-year survival rate and quality of life improvement in cancer patients.
Result
Fifty-one studies were included for meta-analysis following a thorough screening process that adhered to the inclusion and exclusion criteria. A total of 4613 patients were enrolled in all included trials. Results obtained indicated that AGI significantly enhanced the sensitivity of cancers to chemo-drugs with pooled RRs (95% CI) of 1.25 (1.18, 1.32), prolonged the 1-year survival rate of patients with pooled RRs (95% CI) of 1.34 (1.14, 1.57), improved the quality of life with pooled RRs (95% CI) of 1.61 (1.34, 1.93). Similarly, Astragalus's main component, astragaloside polysaccharides (APS), also enhances drug sensitivity in clinical cancer patients.
Conclusion
Our findings demonstrated that AGI had the ability to resensitize cancers to chemo-drugs and improve the quality of life of cancer patients. Therefore, AGI could potentially be used as a drug resistance reversal for resistant cancer treatment.
Introduction
Cancer is still the leading cause of mortality in human beings globally. 1 In 2019, the World Health Organization (WHO) estimated that cancer is the first or second leading cause of death before the age of 70 in 112 out of 183 countries. 2 Cancer incidence and mortality are rapidly increasing worldwide due to population aging, growth, and changes in cancer risk factors associated with socioeconomic development. 3 With decades of research progress, the number of treatment approaches for cancer has increased, including radiotherapy, chemotherapy, target therapy, immunotherapy, etc, wherein cytotoxic chemotherapy remains a mainstay of cancer treatment. 4 However, resistance to chemotherapy drugs is a common occurrence in clinical practice. 5
Drug resistance of cancers results in chemotherapy failure and cancer relapse, finally leading to the death of cancer patients.6–8 Therefore, screening the resistance reversal agent is critical to eliminate the resistant cancer cells and finally cure the resistant cancers. The preferred approach to screen resistance reversal agents is to initially identify resistance genes and consequently screen small molecules that target resistance genes.9,10 A previous study identified a resistance signature and screened JmjC KDMs inhibitors to target taxane-platin-resistant NSCLCs by suppressing resistant genes. 11 However, this study validate the effect of resistance reversal inhibitors in cell lines instead of primary lung cancer specimens, 11 which resulted in the screened resistance reversal agent being inefficiently applied in clinical cancer patients.
Another approach is to screen the resistance reversal agent from a natural herb that is well used in clinics. 12 Presently, natural compounds from traditional Chinese medicine have garnered much attention as resistance reversal agents for decades.13,14 Over 30% of Food and Drug Administration (FDA) approved tumor resistance reversal agents were small molecule compounds from natural products and derivatives. 15 However, resistance reversal agents need an extremely high concentration to effectively eliminate resistant cells, consequently causing toxicity to the normal cells.16,17 Conversely, a natural compound that renders the resistant cancer cells sensitive to chemo-drugs rather than killing the resistant cancer cells is more promising in clinical treatment for resistant cancer patients.
Astragalus injection (AGI) is a lipid-soluble drug extracted from Astragalus, widely used for advanced cancer treatment.18–20 Wan et al found that AGI could enhance the chemotherapy efficacy of clinical malignant cancer patients. 21 Zou et al also revealed that AGI could increase the effective rate and quality of life in NSCLC patients receiving chemotherapy. 22 However, there are also other studies suggesting that there was no significant improvement effect of AGI when it was used in combination with chemo-drugs.23,24 The conflict of the existing evidence showed that the enhancement of AGI on the effectiveness of chemotherapy remains uncertain and needs to be conclusively studied. Additionally, Pharmacological studies have shown that Astragalus comprises multiple active substances, including astragaloside polysaccharides (APS), which have been proven to improve the effectiveness of chemotherapy drugs.25,26 Similarly, some studies still showed that APS did not significantly improve the chemo-drug effect. 27 Taken together, Whether AGI or APS injection could significantly and effectively enhance the chemo-drug sensitivity to cancer cells needs to be explored systematically by meta-analysis to make a clear conclusion. Therefore, a systematic meta-analysis of the included eligible studies was performed in our study to evaluate the effect of Astragalus or its main component, APS, on the resensitization of cancer cells to chemo-drugs and clarify the exact therapeutic value of AGI or APS injection in clinically resistant cancers.
Materials and Methods
Search Strategy
Potentially relevant research was thoroughly explored using combined medical subject heading (MeSH) terms in six common databases, including PubMed, EMBASE, Cochrane Library, ISI Web of Science, VIP and CNKI until February 2024 without a lower limit on publication date. MeSH terms included Astragalus (or AS) combined with words correlating to chemotherapy (chemo-drug or chemo*), and terms referring to cancer (cancer* or adenocarcinoma* or carcinoma* or tumor*) were used to search for eligible studies. The entry terms for the same MeSH term were initially combined using the logical word “OR,” subsequently, different combinations for the MeSH term were re-combined using the logical word “AND”. The detailed search strategy referred to other publications.28,29
Selection Criteria for Included Studies
Only studies that satisfied the following conditions were included: (1) the study involved human cancer patients; (2) all patients underwent basic treatment; (3) all patients were randomly classified into two groups; one group received only basic treatment while the other group received basic treatment combined with AGI-related therapeutic agents; (4) presenting tumor response events in both groups. Studies that met the following criteria were excluded as described previously 29 : (1) duplicative analysis; (2) abstracts, case reports, reviews, news reports, or protocols; (3) subjects were cell lines or xenografted animals using patient-derived cancers; (4) AGI combined with other Chinese medicines.
Study Selection Process
All searched studies were imported into Endnote and independently reviewed by three researchers (Y.T.J, Y.X.F, and L.Y.L). Duplicated studies were automatically screened and removed using Endnote. Subsequently, articles of the following types - reports/reviews, conferences, books/letters, patents, notes, news, and surveys - were excluded. Afterward, through reviewing the abstract and keywords in the remaining studies, those that did not meet the screening criteria were removed. Finally, unsatisfied studies were removed after reviewing the full text. Detailed screening process as described in the previous study.29–31 During the screening process, any dissenting opinions were repeatedly discussed by the three researchers until reaching a consensus.
Data Extraction
Studies satisfying both inclusion and exclusion criteria were independently reviewed in detail by two investigators (Y.T.J. and L.Y.L.) according to previous study. 32 Then, critical information was extracted for further analysis. Disagreements were proposed and discussed until all issues reached a consensus. The basal information, including first author's name, year of publication, Chinese medicine type, first-line therapeutic approach, cancer type, sample size, and intervention, was obtained from all studies. The outcomes representing the effect of AGI on tumor patients, such as tumor response (TR) event, survival rate event (SR), and Karnofsky Performance Score (KPS) increase event, were carefully extracted from studies. For any study lacking the above variables, researchers will strive to contact the authors or locate related studies to obtain the necessary variables.
Outcome Measures
The events of tumor response, 1-year survival, and KPS increase from the control groups (chemotherapy alone) and the combination group (AGI or APS with chemotherapy) were extracted by two of the reviewers independently. Clinically, the therapeutic effects of tumors are divided into complete remission, partial remission, stable, and progression. Treatment efficacy includes complete remission and partial remission. Following treatment, the patient was followed up for one year. The outcome of survival could be used to evaluate the effectiveness of the therapy. The KPS increase is utilized to assess the quality-of-life improvement, with an increase of ≥10 points being considered a quality-of-life improvement. The events for each outcome from two groups were input to calculate the RRs in the metan package, indicating the risk of an event in the combination group versus the control group.
Quality Assessment for Included Studies
The risk of bias in the randomized controlled trials (RCT) from the included studies was assessed using Rob2 tools, which is an update to the original risk of bias tool that was launched in 2008.33,34 The RoB2 consists of five domains covering all types of bias that are currently understood to affect the results of RCT. The five domains consist of the randomization process bias, bias caused by deviations from intended interventions, bias caused by the absence of outcome data, bias in measuring the outcome, and bias in selecting the reported result, as described in other study. 35 Every domain was rated as low, some concern, or high risk of bias according to the principles of protocol. The ultimate choice will be made by the third author (Wenhao Xiu).
The GRADE approach was used to assess the confidence of the effects estimates for each outcome, taking into account the limitations of the studies (risk of bias), inconsistency, imprecision, indirectness, and publication bias, as previously described. 35 Two independent researchers assessed the certainty of the effect estimates for every outcome, categorizing them as high, moderate, low, or very low. Dissensions were discussed until reaching a consensus. The GRADE summary of findings table was generated using GRADEprofiler. Our study was structured under the guidance of PRIMSA. 36
Statistical Analysis
The type of extracted data was count and pooled together using the random (M-H) effects model. Pooled RRs with 95% CIs, evaluating the effect of AGI or APS on the resensitization of cancer patients to chemo-drugs, were represented using a forest plot. A pooled RR > 1 indicates AGI enhanced the effect of chemo-drug in cancer treatment. Cochran's Q test and I2 statistic were used to evaluate the heterogeneity from the pooling model, where I2 ≤ 50% indicates no or moderate heterogeneity and I2 > 50% indicates strong heterogeneity, along with phet <0.05 presenting significance. 29 If heterogeneity occurred, the subgroup analysis and regression analysis were conducted to analyze the heterogeneity source. To assess the stability of the pooling model for pooled RRs, a sensitivity analysis was conducted by sequentially removing one study at a time. A funnel analysis was carried out using Begg's test to estimate publication bias, where a significance with a p-value less than 0.05. The statistical analysis was done using STATA software version 17.0 (STATA Corporation, College Station, TX, USA). All p values were shown in the tables and figures.
Results
Literature Search Results
After an initial search, 1840 original articles and 4 articles from the references in the publications about the study of AGI were extracted in total. Afterwards, 270 duplicative records were removed from the total articles. Next, 530 records were removed, including cases, conferences, books, patents, notes, news and surveys. Furthermore, 906 studies were excluded after reading the abstract and keywords due to those unrelated to cancer or the studies unrelated to clinical cancer patients. Finally, another 87 publications were excluded after carefully reading the full text of the remaining publications due to the reasons as shown in Figure 1. Hence, a meta-analysis was conducted on 51 studies. Figure 1 shows a detailed screening process.

The selection process of papers included for meta-analysis.
Characteristics of the Included Studies
The main characteristics of the included studies are shown in Table 1. A total of 4613 patients from 51 selected studies were included in the analysis. All studies adopted the chemo-drugs, with the different Chemotherapy regimens for specific cancer types. Forty-four studies used AGI, while seven studies applied APS injection. Meanwhile, twenty of the 51 studies had a sample size of over 100 patients. Tumor response events in two groups were recorded in all studies. Ten of the 51 articles reported a 1-year survival rate (Table 1). Additionally, 27 studies reported patients with non-small cell lung cancer.
Main Characteristics of Included 51 Studies.

The author name and publication years was recorded as study ID; Chinese medicine type (CM), first-line therapeutic approaches, tumor type, outcomes and sample size of the studies were extracted. Meanwhile, the intervention method was also extracted. NSCLC, non-small cell lung cancer; EC, endometrial cancer; OC, ovarian cancer; EC, esophagus cancer; GC, gastric carcinoma; CC, colon cancer; BC, breast cancer; ALL, acute lymphocyte leukemia; mixed, numerous different cancers; GT, gastrointestinal tumors; AGI, Astragalus injection; TP: taxol and cisplatin; EP: Platinum, Etoposide, and Paclitaxel; VMP: Changchun Flower Alkaline Amide, Mitomycin, and Cisplatin; PUL: cisplatin, tegafur with uracil, and leucovorin; FOLFOX4: L-OHP, LV, 5-FU; VDLP: vincristine, daunorubicin, L-asparaginase, prednisone; GP: gemcitabine and cisplatin; AP: pemetrexed, cisplatin; FOLFOX: oxaliplatin, calcium folinate, fluorouracil.TR, tumor response; SR, survival rate.
Methodological Bias of the Included Studies
Based on the Cochrane Handbook for the Systematic Evaluation of Randomized Controlled Trials (RCTs), methodological quality assessment forms were developed. Two independent reviewers evaluated the methodological quality of all studies. Discrepancies were resolved by discussion. The bias risk across all the included RCTs was evaluated (Figure 2). Four trials did not describe the method of randomization and were considered as having bias, and twenty-four trials were rated with some concern in the randomization process. According to the deviations from intended interventions, two trials were considered as having bias, while fifteen trials had some concerns. Eight trials had some concerns about the measurement of the outcome. Summary information for the methodological quality is as shown in Figure 2.

Methodological bias risk of all studies. Summary figure for bias risk.
GRADE Evidence Quality
The GRADE evidence quality assessment, which is exhaustively reported in Table 2, revealed that none of the studies showed methodological limitations that may weaken confidence in their effect size estimations. Taken together, the recommendation level for tumor response and 1-year survival rate was certainly strong, indicating the high-quality evidence presented.
GRADE Summary of Three Outcomes in Included Studies.

The quality of the evidence for three outcomes including tumor response, 1-year survival rate and quality of life improvement were graded with high or moderate grade.
AGI and APS Enhanced Tumor Response to Chemo-Drugs
The tumor response events in both the control group and combination group from included 51 studies were extracted, and input into metan package to obtain the RR. Consequently, the pooled RRs for enhancement of tumor response rate were calculated to assess the effect of AGI, or APS combined therapeutics on tumor response enhancement using a random-effect model. The pooled RR (95% CIs) for tumor response rate was 1.25 (1.18, 1.32) (Figure 3), showing AGI or APS enhanced the sensitivity of cancer to chemo-drugs. These findings indicated that AGI or APS possessed the ability to reverse the resistance of cancers to chemo-drugs.

The pooled studies evaluating risk ratios (RRs) for tumor response. Forest plots of pooled RRs for 51 studies presenting tumor response in various cancer patients treated with chemotherapy. CI, confidence interval; HR, Hazard ratio; phet, p value for heterogeneity; I2, I2 statistic value.
Astragalus membranaceus is a traditional Chinese herb commonly used in cancer treatment. Numerous studies have proved that AGI has a chemoresistance reversal effect; however, whether its effective component, such as APS, also has a similar effect remains unclear. Consequently, the pooled RRs for tumor response in patients treated with AGI or APS plus chemo-drugs were analyzed respectively. The result showed that pooled RR for AGI was 1.24 (1.17, 1.32) while pooled RR for APS was 1.27 (1.08, 1.5), which demonstrated that both AGI and APS could enhance the chemo-sensitivity of cancer patients (Figure 4A and 4B).
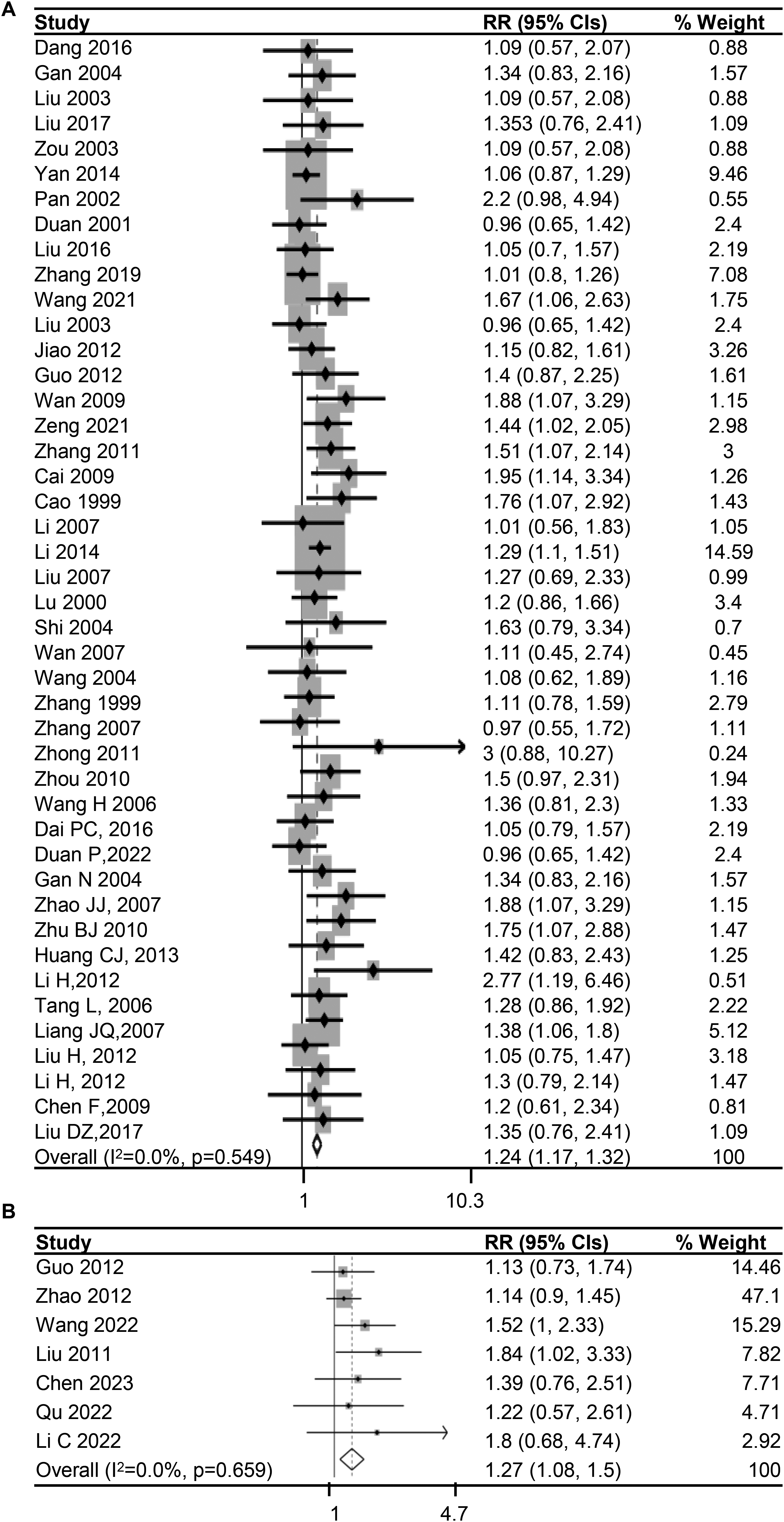
The pooled RRs evaluating tumor response in patients treated with AGI and APS. (A) Forest plots of pooled RRs for 44 studies presenting tumor response rate in cancer patients treated with AGI plus chemo-drugs. (B) Forest plots of pooled RRs for 7 studies presenting tumor response rate in cancer patients treated with APS plus chemo-drugs. CI, confidence interval; HR, Hazard ratio; phet, p value for heterogeneity; I2, I2 statistic value.
AGI and APS Enhanced 1-Year Survival Rate
Moreover, the effect of chemo-therapeutics on cancer patients could also be assessed by survival rate; hence, 1-year survival events from 8 studies were calculated as pooled RRs of 1.34 (1.14, 1.57) (Figure 5A), implying that combination treatment significantly prolonged survival compared to chemo-drugs alone. Therefore, these results revealed that AGI might increase the sensitivity of cancer patients to chemo-drugs.

The pooled RRs evaluating the effect of AGI on the improvement of 1-year survival rate and quality of life. (A) Forest plots of pooled RRs for 1-year survival rate of cancer patients with combined treatment versus chemotherapy alone from 10 studies. (B) Forest plots of pooled RRs for improved quality of life in patients with combined treatment versus chemotherapy alone from 15 studies. CI, confidence interval; HR, Hazard ratio; OR, odds ratio; phet, p value for heterogeneity; I2, I2 statistic value.
AGI Improved Quality of Life in Cancer Patients
KPS was used to evaluate the behavioral ability of cancer patients, with higher scores indicating better health. An increase of KPS over 10 points is considered to improve quality of life. Among the combination-treated groups and chemo-drug treated groups, nine studies presented events of cancer patients with increased KPS (Table 3). Since QOL improvement of cancer patients reflected treatment effects to some extent, we calculated and pooled the RRs of KPS increase from combination-treated groups versus chemo-drug treated groups to assess the AGI effect on QOL improvement. The pooled RRs (95% CIs) were 1.61 (1.34, 1.93) for KPS increase (Figure 5B). The pooled RRs and lower 95% CI values were greater than 1, thus demonstrating that AGI had indeed enhanced the effect of chemo-drug on the improvement of quality of life in cancer patients.
the Statistical Data from the Included 51 Studies.

The events and non-events for three outcomes including tumor response, 1-year survival and QOL improvement was extracted and listed in this table.
The Subgroup Analysis for Tumor Response
To investigate whether AGI enhanced the sensitivity of chemo-drugs in various cancer types, subgroup analysis for tumor response in different cancer types was conducted. The pooled RRs for tumor response rate in lung cancer was 1.27 (1.16, 1.38), which indicated that AGI significantly increased the chemo-sensitivity of NSCLC patients. However, the pooled RR for tumor response rate in gastric cancers was 1.18 (0.99, 1.42) (Table 4), with the lower 95% CI below 1, suggesting there is no significant enhancement of AGI for chemo-sensitivity in gastric cancers. Additionally, the pooled RRs for tumor response rate in other cancer patients revealed AGI significantly enhanced chemo-sensitivity. Taken together, AGI increased chemo-sensitivity in cancer patients except for gastric cancer.
Analysis of Pooled RRs for Tumor Response Estimates in Different Subgroups.

The cancer type was divided into lung cancer (LC), gastric cancer (GC), acute lymphocyte leukemia, pan cancer (meta) and Other uncommon tumors (other). Sample size was divided into the group with less than 100 patients and group with over 100 patients. According to Chinese medicine type, patients were divided into AGI treated patients and APS treated patients. No., number; RR, risk ratio; CI, confidence interval; AGI, Astragalus injection; APS, Astragalus polysaccharide.
Because the sample size of cancer patients enrolled in clinical studies might affect the overall effect of AGI, subgroup analysis for sample size was also conducted. The result showed that the pooled RRs of sample size < 100 was 1.27(1.16, 1.39) and of sample size >100 was 1.23(1.14, 1.34) (Table 4), which indicated that the effect of AGI on enhancing chemo-sensitivity was not varied by sample size.
Heterogeneity
The I2 value for pooled RRs from 51 studies was 0.0%, with phet equal to 0.656, which demonstrated that no heterogeneity existed among the 51 studies (Figure 3). The I2 value of the pooled RRs for survival rate was 32.6% and phet > 0.05 (Figure 5A), while the I2 value of the pooled RRs for KPS increase was 24.7% with phet > 0.05 (Figure 5B). When the studies were pooled in different subgroups, the analysis of overall heterogeneity showed no heterogeneity (Table 4).
Sensitivity Analysis
The leave-one-out approach was used in the sensitivity analysis to investigate the effect of each study on the pooled RRs, and to determine the stability of the pooled model. The pooled RRs were recalculated after removing one study from the pool at a time to assess the impact of eliminating the study on the pooled outcomes. The pooled model remained steady even after every study was eliminated sequentially, suggesting that none of these studies had significant effects on the pooled results (Figure 6).

Sensitivity analysis for the pooled RRs presenting tumor response rate. X-axis, the ranges of RRs; Y-axis, the study ID.
Publication Bias
Begg's funnel plot was utilized to evaluate the publication bias of all included studies, and it was found that the plot was asymmetric (Figure 7). Meanwhile, the p-value is less than 0.01 in Begg's test (Table 5). It was concluded a significant publication bias existed in the meta-analysis. After removing five studies (Zhong,2011; Cai,2009; Pan,2002; Zhao,2007; Liu,2012), the p-value is over 0.05 in Begg's test, suggesting no evidence to support the significant publication bias.

Begg's funnel plot analysis. X axis, the ranges of selogRRs; Y axis, the logRR.
Begg's Test for Funnel Plot of the Tumor Response Estimates of 51 Included Studies.

Pr, probability; Z, Z-score; Std. Dev., Standard Deviation.
Discussion
Chemo-drugs were the first-line therapeutic approach for advanced cancers. However, chemo-treatment often leads to drug resistance, and about 70% of cancer patients develop recurrence or drug resistance, as demonstrated by multiple studies.82,83 Although novel targets identified from resistant cancer cells could be used as therapeutic targets, resistance issues still occurred. Therefore, resensitization of resistant cancer cells to chemotherapy might be a promising approach for resistant cancers. AS, a traditional Chinese medicine herb, was frequently administrated to assist the treatment of chemo-drugs in advanced cancer patients. However, it was still unclear whether AGI could increase sensitivity to chemo-drugs and reduce the toxicity of chemo-drugs in clinical cancer patients. Therefore, our study innovatively explored the function of AGI in enhancing the therapeutic effect of chemo-drugs and improving the life quality of clinical cancer patients systematically.
Thus far, various studies have revealed that a Chinese herbal compound containing Astragalus significantly enhanced the chemosensitivity of cancer patients. However, there is still no systematically conclusive data revealing the function of a single AGI in improving chemosensitivity in various cancer patients.84–87 Additionally, one systematic analysis showed that AGI significantly enhanced chemosensitivity in advanced NSCLC patients. However, it did not include other types of cancer patients. 88 In our study, studies unveiling the effect of AGI injection or its main ingredient in combination with chemo-drugs in various cancer patients were extracted from the database for analysis. The pooled RRs for tumor response rate in all cancer patients were significantly over 1, especially the RRs for TR were over 1 in NSCLC, which was in line with the previous study. 89 Strangely, the pooled RRs of gastric cancer were over 1, but there was no obvious significance, which implied that it could not be concluded that AGI could strengthen the sensitivity of gastric cancer patients to chemo-drugs. The possible reason is that the number of included studies about gastric cancer was not insufficient.
Another issue is that there was no obvious heterogeneity among the included studies, yet publication bias existed. The RR for tumor response rate was significantly higher in five studies, compared to other studies, likely due to the varied dosage of AGI resulting in a greater difference in tumor response outcome under combination treatment for cancer. On the other hand, some studies pooled various cancers together to explore the combination effect, which also possibly resulted in a more effective enhancement of chemo-sensitivity in cancer patients. The removal of these five studies significantly eliminated publication bias, further proving that it originated from those studies.
Although our findings proved that AGI or its main ingredient enhanced the sensitivity of cancer patients to chemo-drugs with higher tumor response and 1-year survival rates, there are still limitations to this study. It's impossible to eliminate all potential biases, so there may be some degree of bias despite rigorous methodology. Firstly, the number of included studies was insufficient for robust conclusions. Moreover, some studies have applied APS treatment, resulting in a varied combination effects from AGI. Additionally, the therapeutic dosage of AGI or APS in the regime also caused variations in combination effects for cancer patients.
Conclusion
This systematic study demonstrated that AGI could significantly enhanced the tumor response to chemotherapy in vavious cancers, indicating the combination of AGI and chemo-drug had a better effect to eliminate cancer cells than chemodrug alone. More importantly, AGI significantly improved the 1-year survival rate and quality of life of cancer patients who received chemotherapy. Taken together, these findings indicate that AGI can effectively resensitize cancer patients to chemo-drugs in resistant cancer patients. Therefore, AGI could potentially serve as resistance reversal agents for the treatment of resistant cancer patients.
Footnotes
List of Abbreviations
Acknowledgments
We gratefully thank Qiming Zhou from Tsinghua University for his help in operating stata17.
Authors’ Contribution
Y.T.J. and Y.X.F. were responsible for the initial idea, data collection, analysis, and manuscript review. X.W.H. aided in data gathering, analysis and manuscript review. Q.C.T. and Y.Y.X. obtained data and revised the manuscript. Y.J.Z., Z.L.M., and W.X.L. contributed to manuscript revisions. The study idea was conceived by L.Y.L. and X.W., who also collected and analyzed the data, interpreted the findings, and drafted and revised the manuscript. All authors have carefully read and approved the final version of the manuscript.
Availability of Data and Materials
The datasets analyzed in this study are available from the corresponding author upon reasonable request.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by the Natural Science Foundation of China (NSFC) grants (No. 82204673; No. 82305335), the budget research project of Shanghai University of Traditional Chinese Medicine (No. 2021LK018), and the Science and Technology Development Fund of Shanghai University of Traditional Chinese Medicine (No. 23KFL036).
Registration of the Study
The study have been registered with INPLASY (No.INPLASY202470009).
