Abstract
The drug resistance of
Foodborne pathogens are a global concern and seriously threaten human health. The Centers for Disease Control and Prevention (CDC) estimated that 31 major pathogens in the United States caused 9.4 million foodborne diseases, 55 961 hospitalizations, and 1351 deaths each year
1
; 1.8 million people died from diarrhoeal diseases in 2005 alone all over the world.
2
Radix
Based on our previous studies,
28
we found that different AD extracts could stifle biofilm formation of
Materials and Methods
Bacterial Strains and Culture Conditions
Extraction of AD
AD root was dried, powdered, sieved, and then soaked in 75% ethanol solution for 3 hours. The mixture was heated under reflux for 3 times, each time for 3 hours. The liquids were then combined, the insoluble material removed by filtration, and the filtrate evaporated to dryness in a rotary evaporator under reduced pressure. The dry extract was dissolved in pure water and extracted 3 times with ethyl acetate. The solvent extracts were combined and concentrated to dryness under reduced pressure to give the dry extract, which was dissolved in water, filtered, and sterilized by passage through a 0.22-µm sterile filter membrane, prepared into certain concentrations of an aqueous solution, and then stored at low temperature for the next experiment.
Observation of Biofilm Formation
After biofilm formation had been observed, approximately 20 mL of a 106 CFU/mL suspension was added to a clean glass slide that had been autoclaved and sterilized under ultraviolet (UV) light for 24 hours. After incubation for 1, 3, 5, and 7 days at 37 °C, crystal violet stain was applied, and the slides were observed under an optical microscope.
Effects of Extracts on Biofilm Formation
The overnight culture suspension was diluted to 106 CFU/mL, and 100 µL was inoculated into a 96-well plate. Different extract concentrations were added to obtain final concentrations of 1/8, 1/4, 1/2, 1.5, and 2 minimum inhibitory concentrations (MICs). The mixture was subjected to static culture at 37 °C for 24 hours. The supernatant was removed, and then crystal violet staining was performed. Finally, absorbance was measured at 570 nm on a microplate reader, and the experiment was repeated 3 times to obtain an average value.
Destruction of Mature Biofilms
Given that the concentration of coumarins in the extract is low, accurately determining their effect on mature biofilms is difficult. Therefore, standard coumarins were used in this experiment (identification reports are shown in the Supplemental Material).
The mature biofilm cultured for 3 days was washed 3 times with phosphate-buffered saline (PBS). Approximately 100 µL of medium and 100 µL of coumarin of different concentrations were added to the mature biofilm to obtain final concentrations of 0.1, 0.2, 0.4, 0.6, 0.8, and 1.0 mg/mL, with anhydrous ethanol serving as the control group. The mixtures were statically cultured for 24 hours, and 20 µL of TTC was added to each well and cultured for 4 hours (20 r/min) in the dark. The color change of each well was observed. This experiment was repeated 3 times, and the results were averaged. Approximately 10 µL of the bacterial suspensions with different drug concentrations were aspirated from the above 96-well plates in LB solid medium, uniformly coated, and cultured at 37 °C for 24 hours to determine the minimum bactericidal concentration (MBC) of coumarins on the mature biofilm.
C-A Combination Treatment
The antibiotics selected for
The mature biofilm cultured for 3 days was washed 3 times with PBS. To each well were added antibiotics (final concentrations of 3, 4, 5, 6, 7, and 8 µg/mL) and coumarin (final concentrations of 0, 10, 20, 30, 40, and 50 µg/mL). After incubation at 37 °C for 24 hours, the supernatant was removed, and crystal violet staining was performed. Finally, absorbance was measured at 570 nm, and the average value was obtained from 3 measurements.
Statistical Analysis
Three independent biological replicates were performed for all experiments. Statistical analysis was performed using a 2-tailed paired Student’s
Results
Determination of Biofilm Formation
With prolonged culture time, the amount of biofilm produced by the strain gradually increased (Figure 1). When the strains were cultured to the fifth day, the amount of biofilm tended to be constant, indicating that the mature biofilm had been formed. The results of optical microscopy are shown in Figure 2. With prolonged time, the structure of the biofilms gradually changed from loose to dense. When their culture reached the third day, mature biofilms began to form.

Biofilm formation measurement with semiquantitative adherence assay, *

Observation of biofilm formation with optical microscope (100×).
Inhibition of Bacterial Biofilm by Extracts
The inhibitory effect of the extract on the biofilms was gradually enhanced after various concentrations of the extract were applied to the strain for 24 hours (Table 1).
Effects of

Abbreviations: MBC, minimum bactericidal concentration; MIC, minimum inhibitory concentration.
The Effect of Extracts on Biofilm Formation
Figure 3 showed that the biofilm yield of the strain gradually decreased with increasing coumarin concentration. When the drug concentration was either greater than or equal to the MIC, the strain biofilm formation could be completely inhibited.
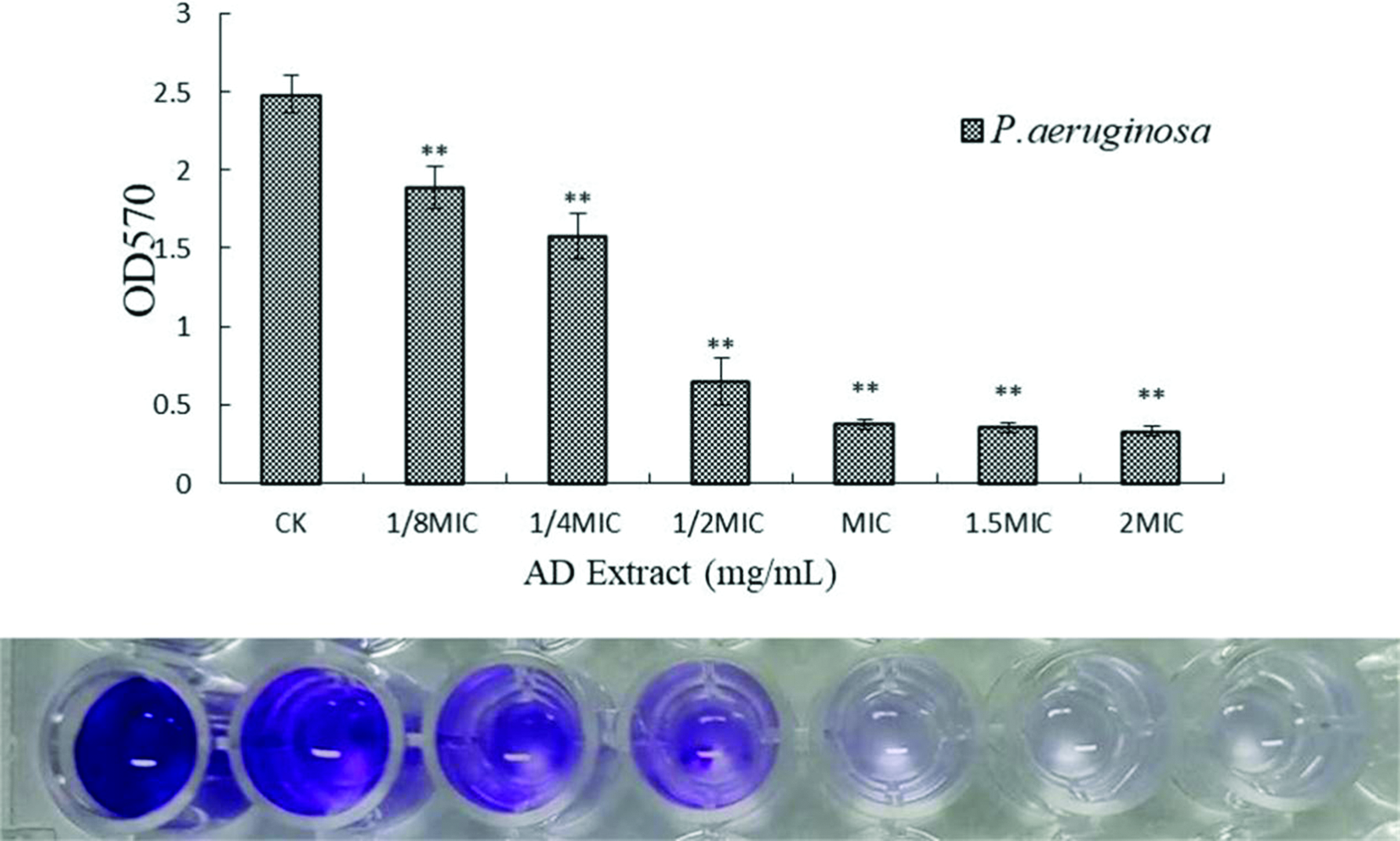
Effect of
Determination of MIC and MBC of the Extracts on Bacterial Mature Biofilm
The results recorded in Table 2 show that the extract minimally affected mature biofilms due to its low coumarin concentration. When used alone, coumarin reacted positively on the mature biofilms but could not completely destroy them.
Effects of Coumarin at MIC and MBC on Mature Biofilms.

Abbreviations: MBC, minimum bactericidal concentration; MIC, minimum inhibitory concentration.
C-A Combination Treatment of P. aeruginosa Mature Biofilm
Simple coumarin and antibiotics
Figure 4 showed that coumarin alone, in all groups, inhibited the formation of

Effect of simple coumarin combined with ampicillin and ceftazidime on biofilms.
Imperatorin and antibiotics
Figure 5 showed that the inhibitory effect on

Effects of imperatorin combined with ampicillin and ceftazidime on biofilms.
Isoimperatorin and antibiotics
As showed in Figure 6, when isoimperatorin was combined with either amipicillin or ceftazidime, the inhibitory effect on

Effects of isoimperatorin combined with ampicillin and ceftazidime on biofilms.
Discussion
Antibiotics resistance has become a threat to peoples’ health, and, therefore, some novel treatments should be explored to treat it, including some traditional medicines and herbs.
1,33
-35
AD has been used as a medical food for thousands of years in southwest Asia and shows valuable bioactivities.
35
-38
In this study, AD extracts showed promising anti-QS activity on a
Supplemental Material
Supplementary Material 1 - Supplemental material for An In Vitro Coumarin-Antibiotic Combination Treatment of Pseudomonas aeruginosa Biofilms
Supplemental material, Supplementary Material 1, for An In Vitro Coumarin-Antibiotic Combination Treatment of
Footnotes
Acknowledgments
The authors are grateful for financial support from the State Administration of Traditional Chinese Medicine of China, Science and Technology Department of Sichuan Province and China Scholarship Council.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Fourth National Census of Traditional Chinese Medicine Resources in China (No. 2018pc02), “13th Five-Year” Breeding Tackling Project of Sichuan Province (No. 2016NYZ0036), and China Scholarship Council (No. 201906910095 ).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
