Abstract
Introduction
Candida albicans is a commensal of the gastrointestinal tract, vagina, oral cavity, vascular system and the skin of humans that reportedly contributes to the maintenance of normal physiological balance (homeostasis).1,2 However, as revealed by Talapko et al, 2 disturbances in normal homeostasis such as a weakened immune system, prolonged antibiotics use, hormonal changes, or alterations in the local micro-environment result in its transition into an opportunistic pathogen. According to Bhatt et al, 1 its excessive proliferation as an opportunistic pathogen in the gastrointestinal tract of at-risk individuals has been associated with mucocutaneous infections (candidiasis and oral thrush), systemic infections (candidemia) as well as gastrointestinal and respiratory tract infections among adults and those living with HIV, especially in health-care settings.3,4
During infection with candida, hydrolytic enzymes such as lipases, phospholipases B enzymes, and secreted aspartyl proteinases (SAP) are reported to be elaborated by Candida species and they influence its virulence.5,6,7 Several studies have further revealed that Candidapepsin-2 also known as SAP-2 is among the sap proteins usually expressed during infections such as systemic candidiasis, vaginal candidiasis, cutaneous and other superficial candida infections thus, induces immunological responses.6,8,9,10,11,12,13,14 Specifically, these proteins aid host colonization, cell membrane rupture, the spread of infections and host immune evasion.6,8,9,14
Among these proteins, SAP-2 stands out. It is a protein composed of at least 60 amino acids made from a pre-proenzyme5,6 and it is linked to increased virulence. An earlier study showed an upregulation of the expression of the SAP-2 gene due to constant exposure to drugs against yeast infections. 13 SAP-2 reportedly degrades α2-macroglobulin and cystatin A leading to the breakdown of Factor H involved in the control of the complement system and other host immune receptors such as FHR-1, CR4 and CR3 used for pathogen recognition, interfering with pathogen recognition and complement complexes. 11 Furthermore, it is regarded as a potent drug target due to its role in cleaving host hydrophobic protein building blocks at phenylalanine sites which are involved in protein folding, stability, and interaction with other molecules.14,15,16,17,18,19,20
Several antifungal agents such as pyrimidine analogues, morpholines, echinocandins, polyenes and azoles among others have extensively been utilized in the management of C. albicans-related infections.16,21,22,23,24 Among the routinely used azoles such as fluconazole, itraconazole, voriconazole and posaconazole, the fluconazole is the most commonly used.17,22,23 Fluconazole inhibits the synthesis of ergosterol, a vital component essential for fungal membrane integrity and function by interacting with lanosterol 14-α-demethylase, a cytochrome P450 enzyme involved in ergosterol synthesis.25,26 By blocking this enzyme, fluconazole disrupts ergosterol production, leading to the accumulation of abnormal sterols and destabilization of the fungal cell membrane, increasing membrane permeability, leakage of intracellular contents and fungal growth inhibition. 26 However, unpleasant side effects and the development of resistance to these drugs including azoles have consistently been reported.18,19,20,23,24
Beside the undesirable side effects and toxicities associated with conventional drugs, fungal pathogens have been reportedly neglected in the past,21,27,28 resulting in the emergence of difficult-to-treat drug-resistant candida-related infections. Furthermore, as revealed by World Health Organization (WHO), this organism has recently been classified as a critical pathogen for which new, effective and safer therapeutics are urgently needed.29,30,31,32 One such novel therapeutic options is the use of alternative treatment such as ethnomedicinal plants including coconut plants and their products.33,34
Coconut oil is one of the major edible products derived from coconut fruit due to its high medium-chain fatty acids content such as lauric acid, monoglycerides, capric acid and caprylic acid, among others.1,35 Studies have revealed that coconut oil possesses anti-inflammatory, antioxidant, antimicrobial, anticancer, and anti-diabetic properties.1,32,35,36 Several studies based on the in-vitro assessment of coconut oil against C. albicans exist.35,37,38 To the best of our knowledge, these studies did not predict the pharmacokinetics nor perform molecular docking of its bioactive compounds against a potential drug target. As a critical pathogen, there is a need for the utilization of a rational drug approach in the search for anti-candida drugs. One of such approaches is the use of molecular docking; an established in-silico approach to rational drug design as it allows for the discovery of lead compounds.36,37,38,39,40,41,42 Given these desirable properties of coconut oil, the study was designed to evaluate its antifungal property against drug-sensitive and drug-resistant C. albicans species singly and in combination with fluconazole as well as the in-silico interaction of the lead compounds against candidapepsin-2.
Results and Discussion
Coconut oil is currently an emerging product that is gradually gaining prominence among consumers in most countries of the world due to its excellent and valuable potential including its medicinal properties against a myriad of infections and diseases such as fungal infections.1,39,43 In this study, the two species of C. albicans: drug-sensitive species and drug-resistant species exhibited considerable susceptibilities against the test extract (100% neat) compared to the observed susceptibility to the conventional antifungal agent (fluconazole 128 mg/ml) (Figure 1). This observation is consistent with those of earlier reports.11,44,45 Consistently, the drug-sensitive C. albicans species exhibited significant sensitivity (59–76 mm) to the extract compared to the drug-resistant species (47–55 mm). On a whole, fungal susceptibility to the extract was higher (47–76 mm) compared to that exhibited towards fluconazole (12–42 mm). Even at lower concentrations of both the extract and the azole, fungal susceptibility to the extract was observed to be higher compared to that exhibited against the control drug (azole). This is in line with the report of Akula et al, 45 where they observed inhibition even at low concentrations of the extract. The antimicrobial properties of coconut oil extract observed in this study may be because coconut oil reportedly inhibits microbial growth by preventing the binding of pathogens to the host cell membrane, induces the breaking of the lipid membrane and impede pathogen maturation.46,47

Antifungal activity of cold-pressed coconut extract against drug-sensitive and drug-resistant Candida albicans species determined by well-diffusion method.
(Table 1).
Antimicrobial Susceptibility Profile of Test Isolates Exposed to Varying Concentrations of the Extract and Fluconazole in Triplicates (a-c).

Key: FLU = Fluconazole; S = Sensitive; R = Resistant; I = Intermediate.
Similarly, the cold-pressed coconut oil recorded minimum inhibitory and cidal concentrations (MICs and MFCs) ranging from 6.25–25% and 12.5–50.00%, respectively for drug-sensitive and drug-resistant species of C. albicans compared to MICs and MFCs of (25–50.00%) and (50–100%), respectively exhibited by azole against the two species of C. albicans. The cold-pressed coconut oil had the lowest MIC (1.57%) against the drug-sensitive species compared to the drug-resistant species where its MIC was observed to be 6.25% (see Table 2). In addition, the MFCs of the extract against the test organisms ranged from 3.13% to 12.5% for drug-sensitive and drug-resistant C. albicans, respectively. When compared with the conventional drug (fluconazole), MICs of 6.25% and 12.5% as well as MFCs of 12.5% and 25.0% were recorded for the drug-sensitive and drug-resistant test isolates, respectively. This observation is similar to that of Ogbolu et al 35 and Akula et al. 45 The antimicrobial potential of coconut oil observed in this study may be due to the presence of some unique bioactive compounds in the extract. 48 Akula et al, 45 observed that caprylic acid and lauric acid had anti-candidal potential against C. albicans. Gunsalus et al, 49 observed that medium-chain fatty acids of coconut oil actively inhibited the gut colonization of C. albicans due to a decrease in fatty acid utilization and an increase in glucose utilization which is usually in limited supply in murine guts thereby, decreasing the expression of several genes. Murzyn et al, 50 reported that capric acid, a component of coconut oil decreased C. albicans’ HWP1 transcription thereby reducing the cell wall protein encoding potential of the hyphae responsible for biofilm formation and adhesion as well as other virulence factors of the pathogen. In addition, Takahashi et al, 51 reported that some components of coconut oil (caprylic and capric acids) inhibit C. albicans yeast and mycelial growth even at very low concentrations and serve as a potent candidate for Candida albicans treatment options.
Minimum Inhibitory and Cidal Concentrations of Coconut oil Extract and Fluconazole Against Test Isolates.

Key: MIC = Minimum inhibitory concentration; Minimum fungicidal concentrations (MFC); DR = Drug-resistant; DS = Drug-sensitive.
In addition, Shino et al, 44 reported that the anti-fungal potential of coconut oil was similar to that of azole. However, this is however, contrary to the report of Ghavam, 52 who noted that the inhibitory and lethal activity of essential oil from Heracleum persicum Desf. ex Fisch against was a degree weaker against Candida albicans compared to nystatin. Besides its antifungal potential, this nature's wonder has been reported to possess other properties including antiviral, antibacterial, and antioxidant properties among others.47,53 Verallo-Rowell et al 54 reported the efficacy of coconut oil in removing colonized Staphylococcus aureus in 26 patients aged 18 to 40 years with atopic dermatitis (AD). Fischer et al, 55 observed the efficacy of coconut oil extract on Gram-positive and negative bacteria. Ghavam, 56 reported that the inhibitory potential of essential oil against microorganisms such as Escherichia coli, Klebsiella pneumoniae, Candida albicans and Shigella dysenteriae (∼ 8.50 mm) was comparable to 62.5 μg/mL of rifampin. Nafar et al 57 observed that coconut oil prevented beta toxicity of cortical neurons via the signaling of cell survival pathways. Furthermore, Nitbani and Jumina, 58 observed that some components of coconut oil such as monoglycerides act by interacting with ergosterol through van der Waals forces resulting in cell apoptosis. Thus, the susceptibility pattern of the test isolates observed is in line with the growth kinetics of the test isolates evaluated over time.
Microbial growth kinetics at different concentrations of an antimicrobial agent over time reveal the relative sensitivity of the microorganisms to the tested compound and further confirm the end-point drug susceptibilities of organisms. In this study, evaluation of the growth kinetics of the test isolates exposed to varying concentrations of the extract over time revealed an increasing concentration-dependent effect on C. albicans’ growth dynamics. The result showed that the fungal isolates (DR and DS) when exposed to increasing concentrations of the coconut oil extract over a 72hr period exerted a concentration-dependent effect on C. albicans growth dynamics (Figures 2 and 3). The observed growth dynamics correlates the end-point drug susceptibility profiles earlier recorded in the study. Drug-sensitive C. albicans (Sa−c) at 0.0% concentration of the extract showed an increase in its cell number (optical density) from 0.90 to 12.78 following incubation for 72 h. Similarly, cells exposed to low concentrations (0.19 to 0.78%) of coconut oil extract were observed to increase in optical density (OD) ranging from 0.81 to 8.54; however, at increasing concentrations of the extract up to 100%, the test organism was observed to decrease in OD to 0.001 after 72 h of incubation. Furthermore, the drug-resistant C. albicans (Ra-c) also grew at 0.00% concentration of the extract. At low concentrations, the organism had an extended lag phase, but at increasing concentrations of the extract, low optical density (0.06) was observed (Table 3). This indicates that microbial cells at low concentrations of the extract were increasing exponentially but at higher concentrations, the optical densities of the test isolates gradually decreased with increasing extract concentration. This observation correlates report from an unrelated study of emerging multidrug-resistant bacteria. 59 Tjin et al 60 revealed that coconut oil inhibited the growth of Candida albicans on day two of their study. However, the drug-sensitive C. albicans species recorded a shorter lag phase and exhibited higher sensitivity (OD = 0.001) to increasing concentrations of the extract compared to the drug-resistant species which showed a rather extended lag phase with the lowest OD of 0.06. The extended lag phase observed with drug-resistant test isolate may be due to the absence of lethal injury, stiffened microbial cell wall due to selective pressure or inherent static properties of the antimicrobial agent. 59
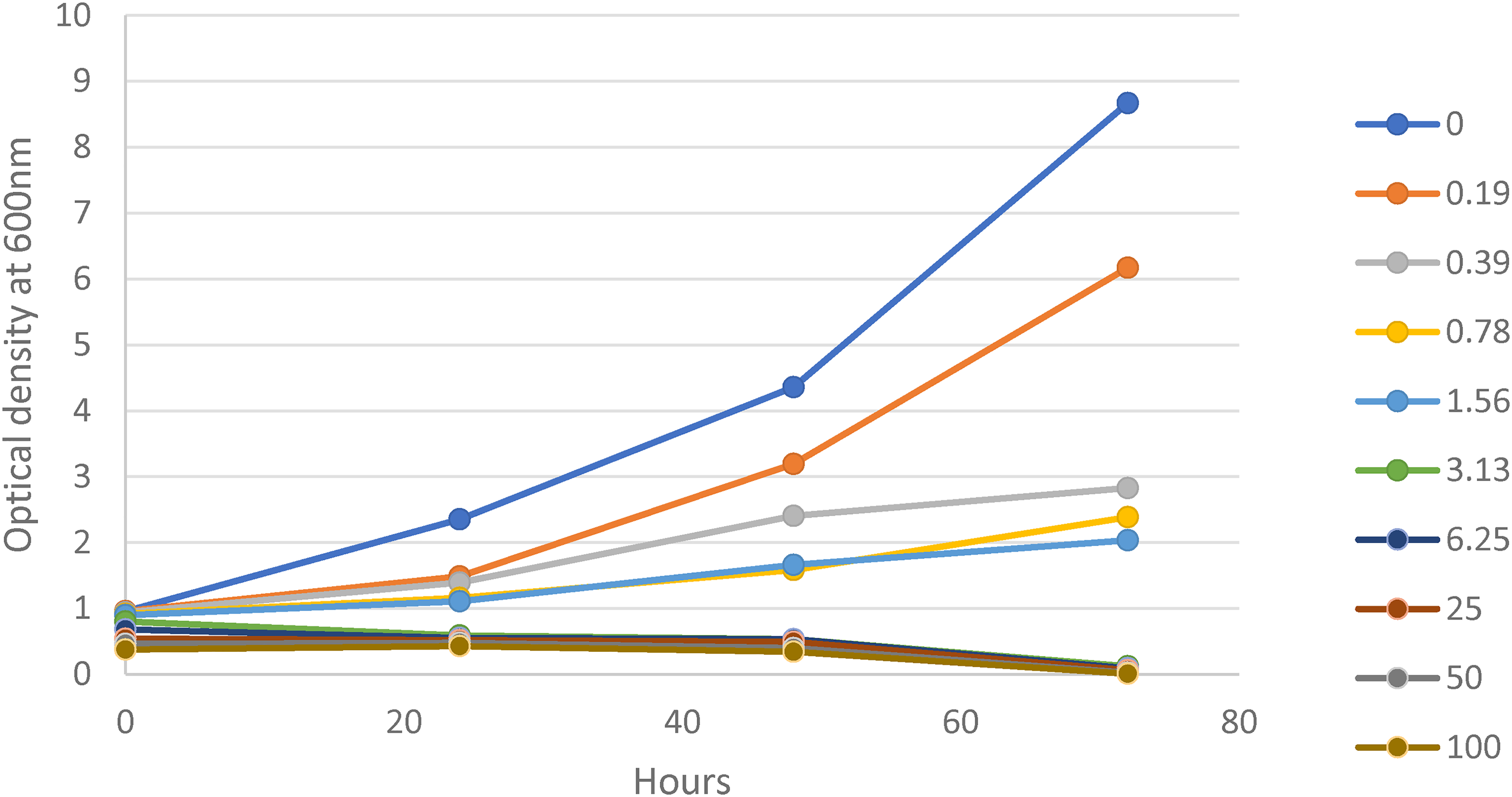
Growth kinetics of DS C. albicans at different concentrations (0-100%) of extract determined after various time intervals (0 to 72 h) of exposure. Key: DS = drug-sensitive Candida albicans.

Growth kinetics of DR C. albicans at different concentrations (0-100%) of extract determined after various time intervals (0 to 72 h) of exposure. Key: DR = drug-resistant Candida albicans.
Antifungal Effect of Combinations Consisting of Extract and Fluconazole Against Test Isolates Assessed with Broth Dilution Method.

Key: DS = Drug sensitive; DR = Drug resistant; FLU = Fluconazole; FIC = Fractional inhibitory concentration.
As revealed by LaFleur et al, 61 azole is one of the potent antifungals that act by impeding α-14 lanosterol demethylase in the ergosterol biosynthesis pathway. However, due to their possession of efflux pumps and high propensity to form a biofilm, azole-related resistance has been reported.62,63 Studies have consistently revealed the potential of existing antimicrobial agents with natural products is gradually becoming a potent strategy for extending the lifespan of existing antimicrobial agents while reducing the emergence and impact of drug resistance.64,65 As further noted by Sadeer & Mahomoodall, 64 utilizing potentiators such as essential oils with antimicrobial agents significantly inhibit efflux pumps and results in a four-fold increase in antimicrobial potencies. The enhanced antifungal properties of an antimicrobial agent combined with coconut oil may be due to the oil's ability to alter cell permeability by forming micelles that perforate the cell's membrane, solubilize lipids/phospholipids in the pathogen's envelope as well as its propensity to interfere with signal transduction in cell replication.66,67 This facilitates the uptake of the antimicrobial agent and subsequent achievement of the target by the antimicrobial agent. In addition, there is a possibility that both coconut oil and azole have similar sites of action in the fungus.68 In this study, the potential of using coconut oil as an adjunct to fluconazole therapy for a synergistic relationship against drug-resistant C. albicans was investigated.
When coconut oil extract was combined with fluconazole, the coconut oil-FLU mixture was found to enhance the antifungal activity of both C. albicans species. Based on FIC index, the antifungal potential of fluconazole was greatly enhanced (synergy) by the combination with coconut oil extract against both drug-sensitive and drug-resistant C. albicans species. The combination in varying proportions showed value-added synergistic potential, reduced the MIC value with little or no antagonistic effect thus, indicating that the drug-resistant fungus may be treatable with the clinically recommended dosage. Thus, combinations containing equal and/or higher proportions of the extract such as 0.5:0.5 and 0.5:0.25 extract: fluconazole exerted greater synergistic (inhibitory potentials) on the test isolates compared to those with low extract proportions (0.25:0.5) where the synergies were indifferent (intermediate) as shown in Table 3. Thus, the coconut oil plus FLU mixture reduced fluconazole resistance in the test organism by lowering the MIC value which further confirmed a positive association between coconut oil and azole. This observation is consistent with similar reports.
Ogbolu et al 35 in their study reported variable susceptibilities to the various species of Candida utilised in their study to fluconazole and coconut oil utilised singly. However, their test C. albicans showed the highest susceptibility (100%) to coconut oil and a MIC of 1:4 compared to that of fluconazole that was 1:4. In another study, the combination of coconut oil and fluconazole as well as oregano oil with fluconazole showed altered properties and antifungal properties as observed in our study. 69 Antifungal property against C. albicans that were intermediate between those of probiotics and clotrimazole have also been reported with zones of inhibition comparable to our findings. 70 Similarly, Bae and Rhee, 71 revealed that the caprylic acid of coconut oil when in synergy with other essential-oil-derived bioactive compounds disrupts more than 83% of the fungal cell wall membrane and inhibits efflux pumps thus, enhancing the penetration of antifungals into the cytoplasm and the subsequent destruction of the pathogen. The in-vitro results obtained in this study complement the quantitative and in-silico components of the work.
The bioactive compounds recovered from cold-pressed coconut oil extract ranged from amino acids, fatty acids, organic acids, alkanes, alcohols, and related hydrocarbons among others in varying concentrations ranging from 0.723 to 53.212 (Table 4) and of different peaks (Figure 4) and chemical structures (Figure 5). The bioactive compounds obtained were 2-trifluoroacetoxydodecane, aziridine, 1, -(1-butenyl)-, (Z)-, octanoic acid oct-3-en-2-yl ester, 1,3- butadiene-1- carboxylic acid and pentadecane among others. This observation is consistent with previous reports where coconut oil was reported to contain medium-chain saturated fatty acids, antioxidants and nutritional components.1,46,72 These bioactive compounds were then used to perform molecular docking against candidapepsin-2.

Chromatogram showing peaks of bioactive compounds present in cold pressed coconut oil.

Chemical structures of bioactive compounds revealed by GC-MS.
GC-MS Profiles of Bioactive Compounds Present in Cold-Pressed Coconut oil.

Key: Conc. = Concentration; S/N = Serial number; IUPAC
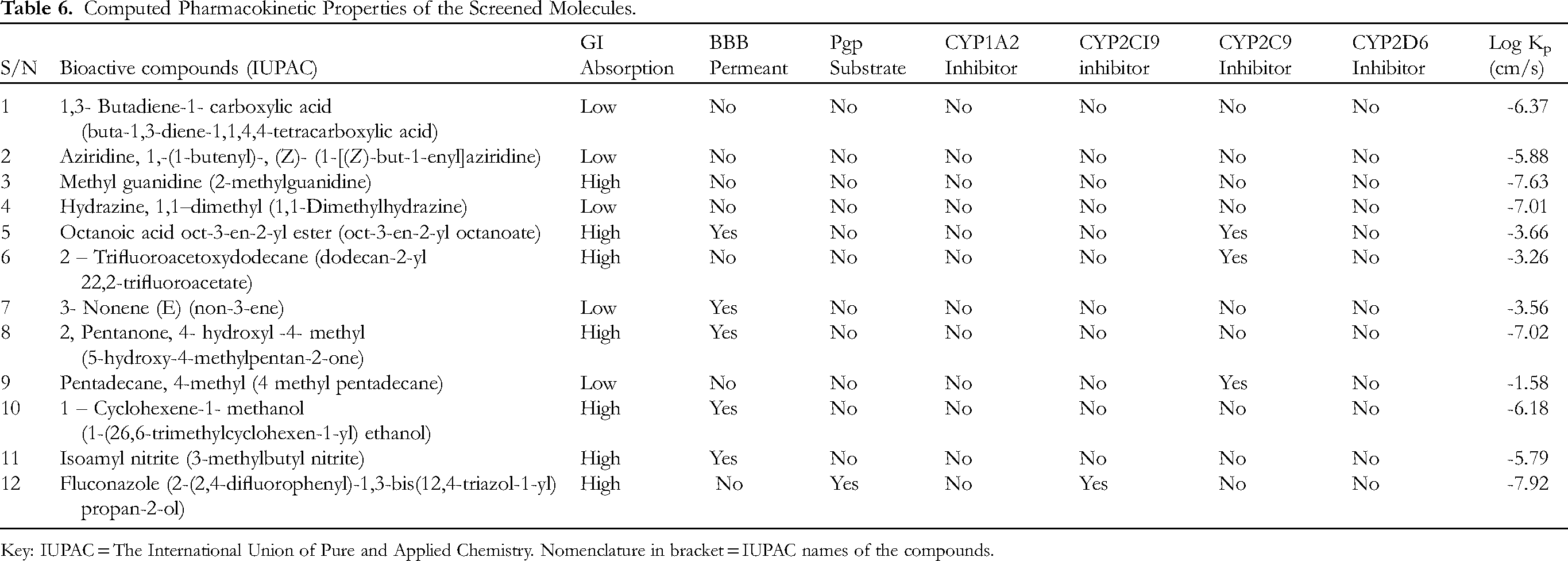
Molecular docking is one of the techniques employed in the preliminary discovery and development of novel therapeutic agents including phytomedicines. 73 Studies revealed that Lipinski's rule of five is used as a measure of the durability of a molecule to be considered an oral drug73,74,75 and is a crucial tool in molecular docking during the early stages of drug discovery. It reportedly serves as a valuable filter, eliminating compounds that are less likely to succeed as drugs due to unfavourable pharmacokinetic properties thus, in combination with molecular docking enhances the drug discovery process by ensuring a more comprehensive approach, improving the chances of identifying successful drug candidates with optimal oral bioavailability and effective binding to the target protein.76,77 The screened bioactive compounds showed little or no adverse effects or toxicity. The physicochemical properties of bioactive, their pharmacokinetics and drugability are presented in Tables 5–7, respectively. The bioactive compounds recovered from the coconut oil extract obeyed Lipinski's rule of five: (molecular weight ≤500; LogP ≤5.6; Number of hydrogen bond donors ≤5; Number of hydrogen bond acceptors ≤10.6 and the topological surface area (TPSA) < 40A) (Tables 5 and 7). Among the compounds that met Lipinski's rule of five, two further returned one violation each for the Veber and Egan parameters. Drugability assessment of the bioactive compounds showed that 81.8% of the compounds obeyed Lipinski's rule of five while 63.6% and 72.7% followed Veber and Egan parameters, respectively with bioavailability score ranging from 0.55 to 0.56 (Table 7). Overall, the bioactive compounds showed favourable oral bioavailability, low toxicity as well as good absorption rates. In addition to these rules, the bioactive compounds showed favourable absorption properties (non-Pgp substrates, GIT permeant and LogKP) as well as favourable distribution potential (BBB permeant).75,76,77 As shown in the hard-boiled structure, all the bioactive compounds as opposed to fluconazole were not substrates to p-glycoproteins, further enhancing their GI absorption. 77 Similarly, approximately 45.5% of the compounds showed permeability through the brain-barrier (BBB permeant) (Figure 6). Furthermore, the bioactive compounds exhibited skin permeability (LogKP) potential ranging from −1.58 to-7.63 cm/s (Table 6). In addition, all the bioactive compounds were not substrate to CYP1A2, CYP2CI9, and CYP2D6 thus, implying that these bioactive compounds effluated despite the specificity of the P450 cytochrome enzymes for substrates.78,79,80,81

Boiled egg graph showing the absorption potential of the various bioactive compounds and positive control done using SWISSADME tool.
Physicochemical Characteristics of the Screened Molecules Related to Lipinski's Rule.

Key: H = Hydrogen; MW = Molecular weight; TPSA = topological polar surface area; IUPAC
Computed Pharmacokinetic Properties of the Screened Molecules.
Key: IUPAC
Computed Drugability Characteristics of the Screened Molecules.

Key: IUPAC
Key: BBB = Represents molecules predicted to passively permeate through the blood brain barrier; HIA = Represents molecules predicted to be passively absorbed by the gastrointestinal tract; PGP+ = Represents (blue dot) molecules predicted to be effluated from the central nervous by the P-glycoprotein; PGP– = Represents molecules (red dots) predicted not to be effluated from the central nervous by the P-glycoprotein. The unlabeled compound represents molecule 6 (the nomenclatures of the various molecules are presented in the various tables).
Candidapepsin-2 is one of the acidic hydrolases considered the organism's key virulence factors implicated in all candida-related infections. In this study, the protein (Candidapepsin-2) was evaluated in-silico for its affinity against a set of ligands recovered from coconut oil extract and revealed varying interactions largely influenced by unique amino acid residues and various bonds such as hydrogen bonds, alkyl groups and attractive charges (Figure 7 and Table 8). The result of binding affinity for the candidiasis inhibitory efficiency of the ligands showed Aziridine, 1,-(1-butenyl)-, (Z)- (1-[(Z)-but-1-enyl]aziridine) to have binding affinity values of −2.0 kcal/mol, Methyl guanidine (2-methylguanidine) and 2, Pentanone, 4- hydroxyl −4- methyl (5-hydroxy-4-methylpentan-2-one) to have a binding affinity of −3.0Kcal/mol, respectively. Hydrazine, 1,1–dimethyl (1,1-Dimethylhydrazine) had a bonding affinity of −2.2Kcal/mol and Isoamyl nitrite (3-methylbutyl nitrite) had a bonding affinity of −2.8Kcal/mol (Table 8). From the results obtained, the lead compounds showed better inhibition potency with the receptor protein than the standard drug Fluconazole (2-(2,4-difluorophenyl)-1,3-bis(12,4-triazol-1-yl) propan-2-ol. The amino acid residues THR A215, ILE: A216, TYR: A218; VAL: A:284 LEU: A244, SER: A272, VAL: A275, ARG: A305 and ASN: A302 had favourable affinity with the Sap-2 protein with bond lengths ranging from 2.37 to 5.46 (Figure 7). The most commonly occurring amino acids in this study were THR and ILE. This is consistent with the report of Biswal et al, 82 who observed that Threonine-222 was the predominant amino acid with good bonding affinity against a similar protein.

Molecular docking interactions of the various ligands against candidapepsin-2 done using Biovia (discovery tool) with default settings. Key: I = Aziridine, 1, -(1-butenyl)-, (Z)- (1-[(Z)-but-1-enyl] aziridine), II = Methyl guanidine (2-methylguanidine), III = Hydrazine, 1,1–dimethyl (1,1-Dimethylhydrazine), IV = 2, Pentanone, 4- hydroxyl −4- methyl (5-hydroxy-4-methylpentan-2-one) and V = Isoamyl nitrite (3-methylbutyl nitrite) all docked against Candidapepsin-2.
Docking Affinity Results with Amino Acid Residue of the Ligands with the Receptor Proteins.

Among the bonds formed in the interaction of lead compounds with the test protein, hydrogen bonds are reportedly the most significant in computational biology.83,84,85 This may be due to the fact that hydrogen bonds contribute to the specificity of ligand binding to the target protein, structural stability of the ligand-protein complex, and the overall binding affinity between the ligand and the protein.84,85 In addition, this bond type has been observed to be essential for molecular recognition, enabling the ligand to fit into specific binding pockets on the protein surface. 83
Hydrogen bonds were observed in the protein interaction with the ligands: methyl guanidine (2-methylguanidine) (amino acid residue THR: A215 with bond lengths of 2.88 and 2.91, respectively), hydrazine, 1,1–dimethyl (1,1-Dimethylhydrazine) (amino acid residues SER: A272 and VAL: A275 with bond lengths of 2.54 and 2.76, respectively), 2, Pentanone, 4- hydroxyl −4- methyl (5-hydroxy-4-methylpentan-2-one) (amino acid such as ARG: A305 and ASN: A302 with bond lengths of 2.37 and 2.50, respectively).
The non-formation of hydrogen bonds in the interaction between Aziridine and Sap-2 protein observed in this study may be due to the fact that this compound exhibits limited hydrogen bond donor and acceptor capabilities due to having only one hydrogen atom directly bonded to nitrogen; making it a weaker hydrogen bond donor compared to molecules with multiple hydrogen atoms bonded to nitrogen or oxygen.86,87 Additionally, while the nitrogen in aziridine could act as a hydrogen bond acceptor, it is less effective than oxygen or fluorine due to its lower electronegativity thus, aziridine have limited potential for extensive hydrogen bonding interactions hence, impacting its binding affinity and specificity with target proteins in molecular docking studies. 87 The standard drug (fluconazole also known as (2-(2,4-difluorophenyl)-1,3-bis(12,4-triazol-1-yl) propan-2-ol)) utilized in this study did not interact with the study protein (Sap-2). This may be due to structural mismatch as well as binding site characteristics. This drug reportedly targets lanosterol 14-alpha-demethylase involved in ergosterol synthesis. 26 Thus, the interaction between fluconazole and candidapepsin-2 may be indirect, as fluconazole's primary mode of action is related to membrane integrity rather than direct interference with specific virulence factors like proteases. The binding of the ligands to the Sap-2 protein indicates that these ligands could be utilized in the development of drugs targeting SAP enzymes in the management of drug-resistant candida infections. This observation is consistent with similar reports.8,16,80
Conclusion
Uniquely, the study has made some contribution to knowledge. First, the results indicates that cold-pressed coconut oil inhibited the growth of both drug-sensitive and resistant isolates in-vitro with susceptibility comparable to fluconazole. Second, the combinations of coconut oil extract with fluconazole showed synergism that could further improve the activity of fluconazole in managing drug-resistant C. albicans infection. Third, although, all the bioactive compounds examined in this study showed favourable pharmacokinetics, only five (5) interacted with the test protein during docking. Furthermore, the standard drug (Fluconazole) also failed to interact with the test protein. In addition, the most frequent amino acid residues in the molecular docking were THR and ILE with docking scores ranging from −2.3 to −3.0 indicating strong interactions between the bioactive compounds and the Sap-2. Unlike aziridine, other lead compounds formed hydrogen bonds with the test protein depicting their suitability as potential anti-candida agents. Furthermore, in-vitro and in-vivo studies utilizing purified bioactive compounds, targeting various virulence factors of many test isolates are highly needed to validate the potential of these lead compounds as drug candidates in the management of candida-related infections and diseases.
Limitations
The present study has several limitations. Firstly, the in-vitro assay was carried out using crude extracts even though the various bioactive compounds were identified using GC-MS. Secondly, the isolates were identified using cultural and biochemical properties. Thirdly, the molecular simulation was not done further to evaluate the potentiality of the lead bioactive compounds. In addition, efficacy of coconut oil was done using only two isolates.
Materials and Methods
Preparation of Cold-Pressed Coconut oil
This was carried out following the chilling and thawing method previously described but with some slight modifications. 35 Briefly, ready-to-eat, mature and fresh coconut (Cocos nucifera) were obtained from local markets in the Calabar metropolis and transported within an hour to the biological science department for identification and certification. The fresh coconut meat was shredded into tiny pieces using a clean blender (Dongguan Sanzhong Machinery Co., Ltd China), mixed with lukewarm water (37–40.5 °C) and filtered using a sterile sieve to produce the coconut milk. The coconut milk was then transferred to a sterile container and allowed to stand undisturbed at room temperature for 12–18 h during which the oil naturally formed a distinct layer on top separating from the thick coconut milk that settled at the bottom. The mixture was then transferred to a suitable container and chilled at 10 °C for 12–15 h after which the chilled container was removed and kept to thaw at room temperature. The liquefied coconut oil was subsequently strained using a fine-mesh filter to remove any remaining impurities. The pure and clear oil extract was then transferred into air-tight Bijou bottles and stored at 4 °C until needed. 89
Sterility Test of the Extracted Coconut oil
This was carried out as described previously to ascertain the microbial sterility of the cold-pressed oil extract. 35 Briefly, the extract was cultured onto separate plates containing freshly prepared MacConkey, Sabouraud dextrose and chocolate agar using both spread and pour plating techniques, and the plates were inverted and incubated at 37 °C for 24–48 h. The MacConkey and Sabouraud dextrose agar plates were incubated aerobically while the Chocolate agar plates were incubated in a candle extinction jar.
In-Vitro Assay
The in-vitro assay was done using various methods that included antimicrobial assay via the doubling dilution technique, growth kinetics and antagonistic/synergistic effect of fluconazole and the cold-pressed coconut oil against C. albicans.42,88
Preparation of the C. albicans Isolates
The C. albicans (drug-sensitive and drug-resistant) species (n = 10) used in this study were obtained from the stock collection unit of the Department of Microbiology, Faculty of Biological Sciences, University of Calabar, Calabar. The test isolates were isolated and characterized in our previous study. 43 Subsequently, they were revived via sub-culturing onto freshly prepared SDA agar plates and incubated for 24–48 h at room temperature. Following incubation, the revived test isolates were preserved in freshly prepared SDA slants in Bijou bottles at 4 °C for further use.
Susceptibility Profiling of the Test Isolates to the Standard Drug
The susceptibility profile of the test isolates against the standard drug, fluconazole was evaluated via the agar well diffusion technique with some modifications. The stocked isolates were revived via inoculation onto separate 10 ml test-tubes (Pyrex,Corning Incorporated) containing 5 ml of freshly prepared Sabouraud dextrose broth (SDB) and incubated with regular agitation at 35 °C for 24 h. The turbidities of the test isolates were adjusted visually to 0.5 McFarland standards (1–5 × 106cell/mL). Afterwards, 0.2 ml aliquots of the test isolates were inoculated onto plates containing freshly prepared Muller Hinton agar (MHA) (supplemented with 0.2% glucose and 0.5 g/mL methylene blue dye), respectively and spread uniformly over the agar surface and allowed to stand for 10–15 min, four wells, each 4 mm in diameter, were cut out of the agar, and 20 µL of FLU was placed into each well and then incubated at 37 °C for 24 h following methods previously described.90,91 Zone diameter (≤12 mm) was used as interpretative breakpoints for resistance, dose-dependent susceptibility (13–18 mm) or intermediate and susceptible (≥19 mm).90,92
Susceptibility of the Test Isolates to Coconut oil Extract
A doubling dilution was carried out following methods previously described by Kamga et al, 93 with some modifications. Briefly, 5mls of cold pressed coconut oil was dispensed into a 10 ml Pyrex test tube containing 5 ml lecithin and this constituted the stock. Afterwards, 2mls of water was each dispensed into five tubes and appropriately labeled: 1:2, 1:4, 1:8, 1:16 and 1:32, respectively. Furthermore, exactly 2 ml of the stock solution was aseptically transferred into the first test tube (1:2) and agitated vigorously after which 2 ml was transferred from the first tube to the second tube (1:4), mixed well and then 2 ml transferred from 1:4 to 1:8. This was repeated for the fourth test tube (1:16) and 1:32), respectively. Following thorough agitation, 2 ml was withdrawn from the fifth test tube (1:32) and discarded. Similarly, 100 to 0.19% dilutions of the fluconazole drug were also carried out (used as the positive control with water as the diluent).
The anti-candida activity of coconut oil extract was evaluated against two C. albicans species using the well diffusion assay previously described.91,92 Briefly, 2–5 colonies of the test isolates were introduced, respectively into tubes containing 2 ml of 0.85% sterile saline and the turbidity adjusted to 1.0 McFerland Standards. Then, 0.2 ml aliquots of the drug-sensitive and the drug-resistant test isolates were dispensed in triplicates onto sterile plates containing MHA (supplemented with 0.2% glucose and 0.5 g/mL methylene blue dye) and uniformly spread on the surface of the plates to ensure the test organism made adequate contact with the medium. Subsequently, the inoculated plates were allowed to stand for 10–15 min to allow the isolates to absorb on the agar. Using a sterile agar borer, wells were cut on the agar 4 mm in diameter. Afterwards, 100 µL of the various concentrations of the coconut oil extract (100 to 0.19%) and fluconazole drug were added to wells, respectively. The plates were incubated at 37 °C for 24 h after which the diameters of the zone of inhibition were measured.
Minimum Inhibitory and Cidal Concentrations of Coconut oil Extract Versus Fluconazole
This was carried out using the doubling dilution technique described previously,35,93 with some modifications. Exactly, 5 ml of coconut oil was diluted into 5 ml of 1% alcohol to form the Neat (100%). Using sterile distilled water as a diluent for both coconut oil and fluconazole (400 mg), the 1% ethanol-100μl neat solution of coconut mixture and fluconazole stock were both diluted down to 1:512 (0.19%).
Each dilution for the cold-pressed coconut oil and the control (fluconazole) was added to the prepared inocula 1–5×106Cells/ml/ (equivalent to 0.5 McFarland Standards), incubated for 24 h and observed for turbidity. For each dilution, a control (inocula without extract or fluconazole) was also set up. The minimum inhibitory concentrations (MICs) of the extract and fluconazole (FLU) were recorded as the lowest concentration where no turbidity was observed in the tubes after 24 h of incubation. The minimum fungicidal concentrations of the test organisms were determined via inoculating 10 µl from each of the tubes onto plates containing freshly prepared SDA and incubating for 24 h. Dilutions that showed no growth after 24 h incubation was taken as fungicidal values.
Growth Kinetics of the Test Isolates in Coconut oil Extract and Drug
This was carried out as described previously94,95 with some modifications. Briefly, 5 ml of coconut oil was introduced into a 10 ml Pyrex test tube containing 5 ml of lecithin to make the Neat. Similarly, 400 mg of fluconazole was introduced into another tube containing 10 ml of sterile distilled water (diluent) to make the stock. The stock solutions were then respectively utilized to carry out doubling dilutions (ie, 1:2, 1:4, 1:8, 1:16, 1:32, 1:64, 1:128, 1:512, 1:1024 and 1:2048). Subsequently, 50 µL of each concentration of the coconut oil extract and fluconazole (400 mg) in triplicates were respectively introduced into a set of 20 tubes containing 50 µL Muller Hinton broth (MHB) medium. Afterwards, 0.2 ml aliquots of the test organisms (1–5×106 cells/ml) that have been previously incubated on SDA broth for 24 h and adjusted to 0.5 McFarland standards were mixed, respectively with the drug-MHB dilutions in the tubes and incubated at 37 °C for 0–72 h (a−c). Three tubes containing drug alone, medium + organism and medium alone were utilized as controls. Growths were monitored at intervals of 0, 24, 48 and 72 h using a spectrophotometer at a wavelength of 600 nm as 20 µL was withdrawn at each interval from each tube and used for the assessment of growth.
Synergistic Effect of Coconut oil Extract with Fluconazole
The antifungal effect of the combination (coconut oil with fluconazole) was evaluated following methods previously described95,96,97,98,99 with some modifications. Briefly, doubling dilutions of antifungal agent (fluconazole) to at least double the MICs of the test isolates (ie, 12.5% for drug-sensitive and 25.0% for drug-resistant test isolates, respectively) were prepared before testing and mixed in a set of 20 tubes (Pyrex) containing 50 μl of varying concentrations (0.5 to 0.25 g/ml) of the coconut oil extract and 50 μl of the MHB medium (coconut oil extract-MHB medium). Afterwards, 50 μl of the fungal isolates that has been adjusted to 0.5 McFarland standards (1–5 × 106 cells/ml) were introduced into the coconut oil extract + MHB + Fluconazole medium and the tubes were incubated at 35 °C for 24 h. Additionally, 40 test tubes with each containing 50μl of test organisms and either varying concentrations of coconut oil extract (100% to 0.19%) or varying concentrations of fluconazole (100% to 0.19%) were also incubated. Following incubation, the MIC was regarded as the concentration of the extract + fluconazole with the least number of cells and/or turbidity. The following formulas: fractional inhibitory concentration (FIC index): FIC coconut oil extract = (MIC coconut oil extract in combination)/ (MIC coconut oil extract alone); FIC fluconazole = (MIC fluconazole in combination)/ (MIC Fluconazole alone), and the FIC index = FIC Coconut oil + FIC fluconazole were used to validate the Minimum inhibitory concentration (MIC). The FIC indices were employed to characterize antifungal interactions as follows: Synergy (when the combination of compounds results in an FIC value of < 0.5, it decreases the MIC (increases the inhibitory activity) of one or both compounds when compared with the compounds alone), indifference or additivity (when the combined compounds give an FIC value of 0.5-4, in which case there is no increase in inhibitory activity and/or a slight increase in inhibitory activity from the combined effect of both compounds) and antagonism (when the combined compounds results in an FIC value > 4, it increases the MIC or lowers the activity of the compounds.
In-Silico Evaluation of the Bioactive Compounds Against Candidapesin-2
GC-MS Analysis of the Cold-Pressed Coconut oil
Cold pressed coconut oil was reacted with trimethylsilyl (TMS) reagent (derivatization). Afterwards, an Agilent 6890N gas chromatography equipped with an auto-sampler connected to an Agilent Mass Spectrophotometric Detector was used for the GC-MS. 99 One microlitre of the sample was injected in the pulsed spitless mode onto a 30 m×0.25 mm ID DB 5MS coated fused silica column with a film thickness of 0.15 micrometre. Carrier gas used was helium and the column pressure kept at 20 psi to give a constant of 1 ml/min. Other operating conditions were pre-set. The initial column temperature was maintained at 55 °C for 24 s, then increased to 200 °C at a rate of 25 °C/mins, thereafter 280 °C at a rate of 8 °C/mins and then taken a final temperature of 300 °C at a rate of 25 °C/mins that was held 2 min. Components with lower retention time eluted first before those with higher retention time. The interpretation of GC-MS was conducted using the database of National Institute Standard and technique (NIST).
Retrieval of the Structure of Candidapepsin-2 Protein and Ligands and Active Site Prediction
The 3D structure of the Candidapepsin-2 of Candida albicans (Figure 8) was obtained from the Research Collaboratory for Structural Bioinformatics (RCSC) protein database. The properties of the retrieved protein (source, name of the protein, PDB ID, Uniprot name, Uniprot Accession ID, Uniprot taxonomic ID and organism) were sc-PDB, Candidapepsin-2, 1eag, CARP2_CANAL, P28871, 5476, and Candida albicans, respectively. Others were the protein resolution and the default binding sites of the protein which were 2.100 and 41.4702, 24.8575 and, 13.1694, respectively for binding centres X, Y and Z.

Structure of candidapepsin-2 from C. albicans.
Assessment of drug-likeness of the bioactive compounds
This was carried out following methods previously described.82,100,101 Briefly, canonical strings of the various compounds returned by GC-MS were produced and used to perform the adsorption, distribution, metabolism, excretion and toxicity (ADMET) parameters via the SWISSADME and MCULE toxicity checker tools. Using Lipinski's rule of five (molecular weight (MW) ≤ 500, iLogP/ A log P (the octanol/water partition coefficient) ≤ 5, the number of hydrogen bond acceptors (HBAs) ≤ 10.6, the topological polar surface area (TPSA) < 40 Å2 and the number of hydrogen bond donors (HBDs) ≤ 5), the drug-likeness of the bioactive compounds were evaluated. In addition to the physicochemical properties (Lipinski's rule of five), we also examined the lipophilicity and pharmacokinetics of the bioactive compounds and that of fluconazole (See supplementary data for more details).
Molecular Docking of the Ligands and the Protein
To confirm the binding affinity of the investigated compounds, we conducted molecular docking studies with Candidapepsin-2 protein using Autodock Vina software. Molecular docking is a robust computational technique utilized to assess the strength of interaction between proteins and ligands. 102 In an effort to discover potential drugs via molecular docking within the active site of the Candida albicans pathogen, which assesses their inhibitory effectiveness, we retrieved the C. albicans protein 4ybf from the PDB database (www.RSCPDB.org). The preparation of these proteins was performed using Discovery Studio, which involved correcting any deformities in the amino acid sequence, eliminating water molecules, and adding polar hydrogen atoms and charges to render the protein molecules suitable for the docking process. The XYZ coordinates for the protein's binding sites were determined as follows: 14.267500, 0.863750, and 28.575750. Figure 8 illustrates the structure of the receptor protein. For the ligands used in the docking, structural optimization was carried out using Gaussian09 W software at the B3LYP/6–311++G(d,p) level. The resulting binding affinities, orientations, binding scores of the various ligands against the Candidapepsin-2 as well as the amino acid residues involved in the bonding were noted for each ligand.
Statistical Analysis
All the data generated was managed using Microsoft Excel 2016, version 2016. Descriptive statistics such as mean, percentages, and ratios were utilised in the analysis of the data. The replicate readings for the sensitivity results were first converted into mean values that were classified further as S = sensitive, R = resistant, and I = intermediate. The concentrations of the extracts were expressed as ratios and percentages. The optical densities of the various extract concentrations were also plotted against time (hours).
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241275798 - Supplemental material for In-vitro Evaluation of the Antifungal Property of Cold-Pressed Coconut oil Against Drug-Resistant Candida albicans and in-Silico Bioactivity Against Candidapepsin-2
Supplemental material, sj-docx-1-npx-10.1177_1934578X241275798 for In-vitro Evaluation of the Antifungal Property of Cold-Pressed Coconut oil Against Drug-Resistant Candida albicans and in-Silico Bioactivity Against Candidapepsin-2 by Elizabeth Nkagafel Mbim, Uwem Okon Edet, Francisca O. Nwaokorie, Henshaw U. Okoroiwu, Oju R. Ibor, Ini Ubi Bassey, Antai E. Ekpo, Bassey Okon Edet, Glory P. Bebia, Curtis Tega, Clement I. Mboto, and Ani E. Nkang in Natural Product Communications
Footnotes
Acknowledgements
Not applicable.
List of Abbreviations
Authors’ Contributions
The conceptualization and design of the study was done by ENM and UOE The microbiological analyses (sensitivity testing, culturing and characterization) were jointly managed by ENM, UOE, CT, BOE, IR, AEE, IUB and FON. Softwares, molecular docking, ADMET analysis was done by UOE, ENM and HUO. All the authors took part in the writing of the manuscript and editing. Correspondence (submission and revisions) was handled by ENM, UOE and HUO. All authors approved the final manuscript draft.
Availability of Data and Materials
All data generated in this study are presented in the study and the supplementary data.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Consent to Participate
Ethical clearance for this study was obtained from the research and linkages committee of Arthur Jarvis University (No: AJU/RC/9/21). All the protocols compiled with the institutional guideline for the conduct of research.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
