Abstract
Background
Virgin Coconut Oil (VCO) is rich in medium-chain fatty acids and bioactive constituents such as vitamin E and polyphenols, rendering it an appealing natural ingredient for medicinal and cosmeceutical skin applications. Its use on the skin warrants cytotoxicity assessment to ensure safety and efficacy. This study evaluated VCO's antioxidant properties and its cytotoxicity in human keratinocytes (HaCaT).
Methods
Fatty acid composition was analyzed using gas chromatography with a flame ionization detector (GC-FID). The antioxidant potential of serial doses of VCO (45%, 22.5%, 11.3%, 5.6% and 2.8%) was assessed by 2,2-Diphenyl-1- picrylhydrazyl (DPPH) and ferric-reducing/antioxidant power (FRAP) assays. Cytotoxicity was determined using the 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay on human keratinocytes.
Results
GC-FID analysis revealed lauric acid as the predominant fatty acid, accounting for 46.4% of the total composition. VCO exhibited dose-dependent antioxidant activity in the DPPH assay, with statistically significant differences across concentrations (F (4, 320) = 143.2, P < .0001) and compared to positive control Trolox (F(1, 320) = 13,201, P < .0001). The FRAP assay showed a statistically significant difference in ascorbic acid equivalent antioxidant capacity (AEAC) among various concentrations (F(4, 35) = 17.65, P = .02). A dose-dependent decrease in HaCaT cell viability was observed with IC50 identified at 17.8% v/v. VCO was mildly cytotoxic to HaCaT at 11.3% v/v (74.36 + 5.41% cell viability). One-way ANOVA showed significant differences across concentration groups (F(5, 150) = 69.68, P < .0001). Tukey's post-hoc indicated significant differences from the negative control (P < .0001) and between the pairwise groups of 11.3% and 22.5% v/v (P = .0001).
Conclusion
VCO possesses antioxidant properties, but future formulation and application for skin may consider using an optimal concentration below 11.3% v/v.
Introduction
Skin is the largest organ of the human body that plays a vital role as a physical barrier against external factors such as microorganisms and environmental stressors. External stressors, such as sun radiation, pollution, and even certain lifestyle factors like smoking or an unhealthy diet, can trigger the formation of reactive oxygen species (ROS) in the skin. 1 These ROS are highly reactive molecules that, in excess, can damage skin cells and their components, including DNA, proteins, and lipids. 2 To counteract the oxidative damage and maintain homeostasis, endogenous antioxidants of the human skin, such as glutathione and superoxide dismutase, help neutralise ROS. 2 However, constant exposure to external stressors over time can overwhelm this defence system, leading to the accumulation of ROS and the disturbance of the delicate balance, a condition known as oxidative stress. 3 Increased risk of skin disorders such as psoriasis, acne, atopic dermatitis and other problems such as delayed wound healing, inflammation, premature ageing, and skin cancer are associated with oxidative stress.4–6 To mitigate oxidative stress, exogenous antioxidants like vitamin C, vitamin E, or polyphenols, often sourced from natural plant extracts, play a crucial role in reducing the harmful effects of ROS and promoting skin health. 7 Dietary and topical application of fatty acids and sterols has also been shown to benefit ageing skin. 8 These vitamins, phyto-antioxidants and oils are abundantly available in various botanical sources. Research findings supported that the regular intake of vitamins, fatty acids, and polyphenols sourced from plants can be beneficial in averting premature ageing, skin inflammations, dryness, and skin cancer.
Virgin coconut oil (VCO) is a multifunctional food oil renowned for various health benefits. Unlike regular coconut oil, VCO is extracted from mature coconut (Cocos nucifera) kernels using a cold-pressing method without excessive heat or chemical treatments, thus preserving the oil's natural properties and nutritional benefits. 9 Evidence of health benefits with VCO intake includes improved cardiovascular health, citing its main constituents, medium-chain triglycerides (MCTs), as the key attributes. 10 Phytochemical studies demonstrated that VCO is a source of various bioactives, especially lauric acid, caprylic acid and capric acid of the medium-chain triglycerides and the long-chain saturated fatty acids such as myristic acid, palmitic acid, and stearic acid. 11 In addition to these, VCO also contains a spectrum of other bioactive compounds, including linoleic acid or omega-6 fatty acids, phenolic compounds, vitamin E, polyphenols, squalene and phytosterols, among others. 12 Due to their unique composition of fatty acids and other bioactive compounds, there is enduring interest in using VCO for medicinal and cosmeceutical purposes to improve skin health. Its applications encompass remedies for dry skin, infections, wound healing, and anti-ageing.13,14 These benefits are closely linked to VCO's demonstrated anti-inflammatory, antimicrobial, antioxidant, and hydrating properties.15,16
The topical application of naturally-derived vitamins and antioxidants to the skin can produce undesirable effects, including contact dermatitis, skin and eye irritation, allergic reactions, and photosensitivity. 17 Toxicological examination of plant-derived compounds becomes imperative due to their potential to induce adverse effects such as cytotoxicity, cell death, and inflammation. The human keratinocyte (HaCaT) cell line is commonly used for this purpose. VCO features skin-protective characteristics, rendering it a popular ingredient in skincare and moisturising products. The widespread utilisation of VCO in the domain of skincare necessitates an in vitro evaluation of its cytotoxic potential to ensure safety and efficacy for cutaneous application. A previous study indicates that VCO is non-irritant when applied to rabbit eyes, supporting its use as an ocular rewetting agent. 18 However, the existing literature on the cytotoxic potential of VCO in human keratinocytes is limited. As such, this study examined VCO's antioxidant properties and further ascertained its cytotoxicity when applied to human keratinocytes. The investigation employed the DPPH and FRAP assays to ascertain VCO's antioxidant properties, while cytotoxicity was assessed using the MTT assay. The findings from this study offer valuable insights into the potential utilisation of VCO in skin care formulations, along with its safety and efficacy.
Materials and Method
Plant Material
The VCO used in this research was cold-pressed and procured from Yahanana Manufacturing at Bagan Datuk Best Farm, located on Jalan Simpang Empat, 36400 Bagan Datuk, Perak, Malaysia. The VCO has received international standard certification from UKM UNIPEQ (ULUKM/1850/20).
GC-FID Analysis for Fatty Acids in VCO
The analysis of fatty acid in the VCO was conducted by Agilent 6890N system gas chromatography equipped with a flame ionization detector (GC-FID). A DB-23 column from Agilent Technologies was used for the separation. 19 The GC parameters included an oven temperature program starting at 50 °C, with ramping rates leading to final temperatures of 175 °C and 230 °C, and a total run time of 29.75 min. The front inlet was maintained at 250 °C, and the FID detector operated at 280 °C. The protocol for sample preparation followed methods described previously. 20 About 100 mg of oil was weighed and dissolved in 5 mL of hexane, followed by the addition of 250 µL of sodium methoxide (0.5M in methanol). The mixture was vortexed for 1 min, pausing every 10 s, then 5 mL of saturated NaCl was added, vortexed for 15 s, and left to settle for 10 min. The hexane layer was then separated and transferred to a vial containing sodium sulphate, and after a 15-min wait, the analysis was initiated. A Supelco 37 Component FAME Mix (Sigma-Aldrich) was used as the standard and peaks identified were compared with the standard.
DPPH Radical Scavenging Assay
The antioxidant activity of the VCO was assessed using the 2,2-Diphenyl-1-picrylhydrazyl (DPPH) assay, denoted by the colour change of the dark purple DPPH into a yellow hue.21,22 Serial dilution of VCO was prepared (45%, 22.5%, 11.3%, 5.6% and 2.8%) by mixing the oil sample with ethanol (EtOH) solvent. Then, 100 µL of each oil concentration was added into six wells of a 96-well microplate, while the designated blank wells contained only 200 µL of ethanol. 100 µL of DPPH solution was subsequently added to each well except the blanks, and the plate was kept at room temperature in the dark for 45 min, after which their absorbance was measured at 517 nm using a microplate reader (Thermo Scientific USA). A positive control using Trolox prepared in the same serial dilution range was tested alongside the VCO samples. The calculation for the percentage of radical inhibition used the following formula:
FRAP Assay
The Ferric Reducing Antioxidant Power (FRAP) assay was performed to measure the antioxidant capacity of the VCO by reduction of ferric ions (Fe 3+) to ferrous ions (Fe 2+). 23 Six wells of a 96-well microplate were each loaded with 50 μL of serially diluted VCO (45%, 22.5%, 11.3%, 5.6%, and 2.8% v/v), followed by adding the FRAP reagent. The serial dilution of VCO was prepared by combining the oil sample with ethanol (EtOH) solvent. Subsequently, the microplate was incubated at 37 °C for 5 min. Following incubation, the absorbance was measured at 595 nm using a microplate reader (Thermo Scientific USA). The absorbance was directly proportional to the antioxidant's reducing power in the VCO samples. Serial dilution of ascorbic acid (50, 25, 12.5, 6.25, 3.125 µg/mL) was prepared as a positive control and tested alongside VCO samples. A standard curve was then generated using known concentrations of the positive control to determine the antioxidant capacity of the VCO. The VCO's FRAP values were converted to Ascorbic Acid Equivalent Antioxidant Capacity (AEAC) to determine the ascorbic acid required to achieve a comparable antioxidant effect.
Cell Culture
The human keratinocytes (HaCaT) were obtained from Elabscience (USA). The preparation of the culture medium and HaCaT cell revival and subculturing procedures were carried out according to the previously published protocol. 24 A frozen vial containing the stock cells was retrieved from a −80 °C freezer and promptly thawed in a 37 °C water bath until it was partially liquefied. Subsequently, the cryo vial was left at room temperature until completely liquefied. Following the thawing process, the cells were transferred to a 15 mL centrifuge tube containing 3 mL of enriched Dulbecco's modified Eagle medium (DMEM) (Sigma-Aldrich Germany) supplemented with 15% Fetal Bovine Serum (FBS) (Capricorn Germany) and 1% Penicillin-Streptomycin (Pen-Strep) (Sartorius Germany). The tube was centrifuged for 3 min at 1600 rpm to form a cell pellet, and the supernatant was discarded. The cell pellet was resuspended in 1 mL of DMEM, and the resulting cell suspension was transferred into a T-25 flask containing 4 mL of DMEM. The cells were examined under an inverted microscope to ensure uniform cell distribution and subsequently incubated for 24 h at 37 °C in an environment with 5% CO2. Following the incubation period, cell contamination and confluency were observed.
When the HaCaT cells reached 80% confluence, subculturing was initiated. Initially, the old media was discarded from the T-25 flask, and the cells underwent two washes with 5 mL of phosphate buffer solution (PBS) (Mp Biomedicals USA). Subsequently, the cells were trypsinised by adding 3 mL of trypsin-EDTA solution into the T-25 flask, followed by a 10-min incubation at 37 °C in 5% CO2. After the incubation period, cell detachment was observed using an inverted microscope, and the trypsin-EDTA solution was neutralised by adding 7 mL of enriched DMEM. The contents of the T-25 flask were then transferred to a 15 mL centrifuge tube and centrifuged for 3 min at 1600 rpm. The supernatant was discarded from the tube, and 1 mL of DMEM was added to resuspend the cell pellet. Cell counting was carried out to determine the appropriate volume of the cell suspension to be added to a new T-75 flask containing 13 mL of fresh DMEM.
Approximately 10 μL of the cell suspension was aliquot for cell counting, and this was mixed with 290 μL of Trypan blue, giving a 30-fold dilution. Subsequently, 10 μL of the cell and Trypan blue suspension was transferred into the hemocytometer, and cell counting was performed using an inverted microscope at 10x magnification. Viable cells were counted from the gridlines in the four corner squares of the hemocytometer and then averaged. The total cell count or cell concentration (expressed as the number of cells per mL) was calculated by multiplying the average cell count by the dilution factor and 104. By employing the dilution equation, M1V1 = M2V2, the volume of the cell suspension seeded into a new T-75 flask was determined. The cell suspension introduced into a new T-75 flask containing 13 mL DMEM was adjusted to a concentration of 1 × 106 cells/mL. The T-75 flask subculture was subsequently incubated at 37 °C with 5% CO2 and monitored daily for any signs of contamination and confluency.
MTT Assay
The 3-(4,5-dimethylthiazol-2-yl)-2,5 diphenyl tetrazolium bromide (MTT) assay was employed according to the previously established protocol to assess the cytotoxic effect of VCO on HaCaT cells. 25 Firstly, HaCaT cells were seeded into a 96-well microplate with 200 μL aliquots of cell suspensions transferred into six replicate wells, each achieving a concentration of 1 × 105 cells/mL. Then, the microplate was incubated at 37 °C with 5% CO2 overnight to allow the attachment of cells to the wells.
After a 24-h incubation period, the DMEM culture medium in each well was aspirated and washed with PBS. Serial dilution of treatment using the VCO (45%, 22.5%, 11.3%, 5.6% and 2.8%) was carried out aseptically in DMEM containing 15% FBS and added to the designated treatment wells. The range of VCO concentrations used in the serial dilution was established by preparing a working solution that combines VCO with solvents to account for VCO's hydrophobic properties. This formulation includes 10% DMSO and 0.4% Tween 80, resulting in a final mixture constituting approximately 90% v/v VCO (827.2 mg/mL). The working solution was subsequently subjected to 2-fold serial dilution in the cell culture medium, resulting in VCO concentrations spanning from 45% to 2.8% v/v (413.6 to 26 mg/mL). Table 1 presents the corresponding DMSO and Tween 80 concentrations for each VCO concentration tested following the serial dilutions.
The Concentrations of DMSO and Tween 80 in the VCO's Tested Range of Concentration After Serial Dilution.

Serial dilution of the positive control menadione was prepared in the same concentration range. Negative control wells consisted of cells in the DMEM medium without treatment, while blank wells contained only the culture medium. Along with the VCO treatment on HaCaT, the influence of DMSO and Tween 80 solvents on HaCaT cells was also evaluated across the range of concentrations used in serial dilutions of VCO to distinguish the potential effects of the VCO treatment from solvents. The microplate was then subjected to an additional 24-h incubation.
The next day, 20 μL of a 5 mg/mL MTT solution was introduced into each well, and the plate was incubated further for 4 h. Following this incubation, 190 μL of supernatant was removed from each well, and 200 μL of DMSO was added. The microplate was incubated for 15 min and then gently agitated on an orbital shaker for 5 min to completely dissolve the purple formazan crystal precipitate. A microplate reader (Thermo Scientific USA) read the absorbance at 570 nm. The MTT assay was performed six times, and the calculation of the percentage of viable cells used the following equation: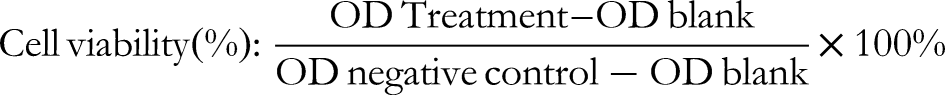
The dose-response curve determined VCO and menadione's inhibitory concentration (IC50), which kills 50% of HaCaT cells.
Statistical Analysis
The results are presented as the mean ± standard error of the mean (SEM). The normality distribution of the data was assessed using the Shapiro-Wilk test in GraphPad Prism 10. A two-way analysis of variance (ANOVA) was employed to assess the antioxidant activities of VCO in the DPPH assay, examining different VCO concentrations and comparing them to the positive control. For the FRAP assay, a one-way analysis of variance (ANOVA) was conducted to determine variations in ascorbic acid equivalent antioxidant capacity (AEAC) among the various concentrations. Additionally, a one-way ANOVA was performed to compare cell viability across different concentrations of VCO, with statistical significance set at P < .05.
Results
GC-FID Analysis of Fatty Acid Profiles in VCO
The GC-FID analysis of the VCO (Figure 1) revealed the presence of several fatty acids, primarily medium-chain fatty acids (MCFA). The identified fatty acids with their retention time and percentage composition were listed in Table 2. The major peaks identified include lauric acid (C12:0), which accounts for 46.44% of the total area, followed by myristic acid (C14:0) at 18.70%, and palmitic acid (C16:0) at 9.24%. Other significant components include caprylic acid (C8:0) at 7.89%, capric acid (C10:0) at 5.96%, and linolelaidic acid (C18:2, trans) at 6.23%.
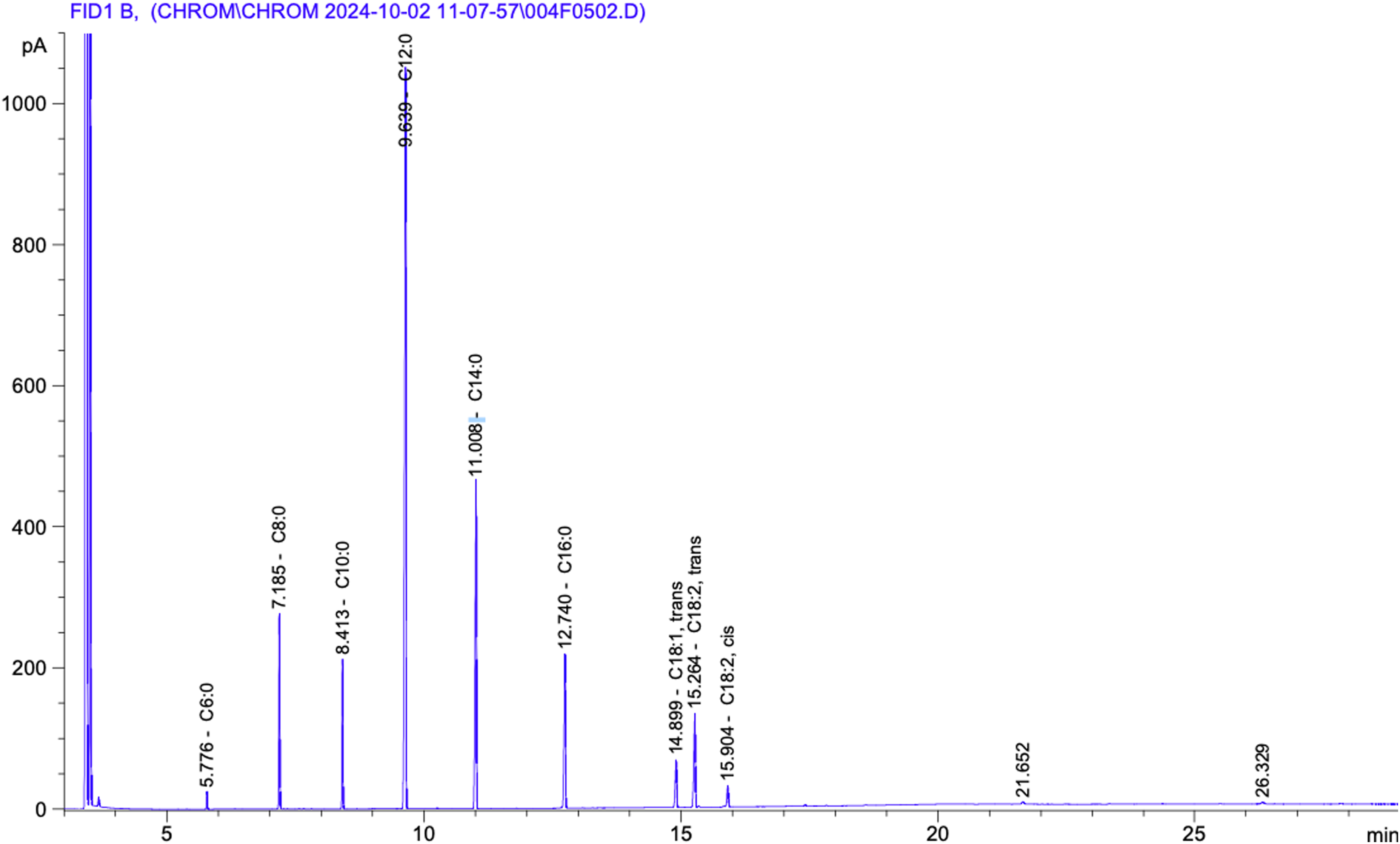
GC-FID chromatogram of VCO.
Fatty Acid Composition of VCO and Percentage Area.

Antioxidant Properties
DPPH Inhibition Activity
Figure 2 shows the VCO's antioxidant activity tested at different concentrations by DPPH assay. The percentage of DPPH inhibition increased with increasing concentration of VCO in a dose-dependent manner. An IC50 value, which denotes the concentration at which VCO effectively inhibits half of the DPPH radicals, was unattainable within the range of concentrations tested. Analysis with the two-way ANOVA revealed a statistically significant difference in the DPPH inhibition across different VCO concentrations tested (F (4, 320) = 143.2, P < .0001) and when VCO was compared to the positive control Trolox (F (1, 320) = 13,201, P < .0001).
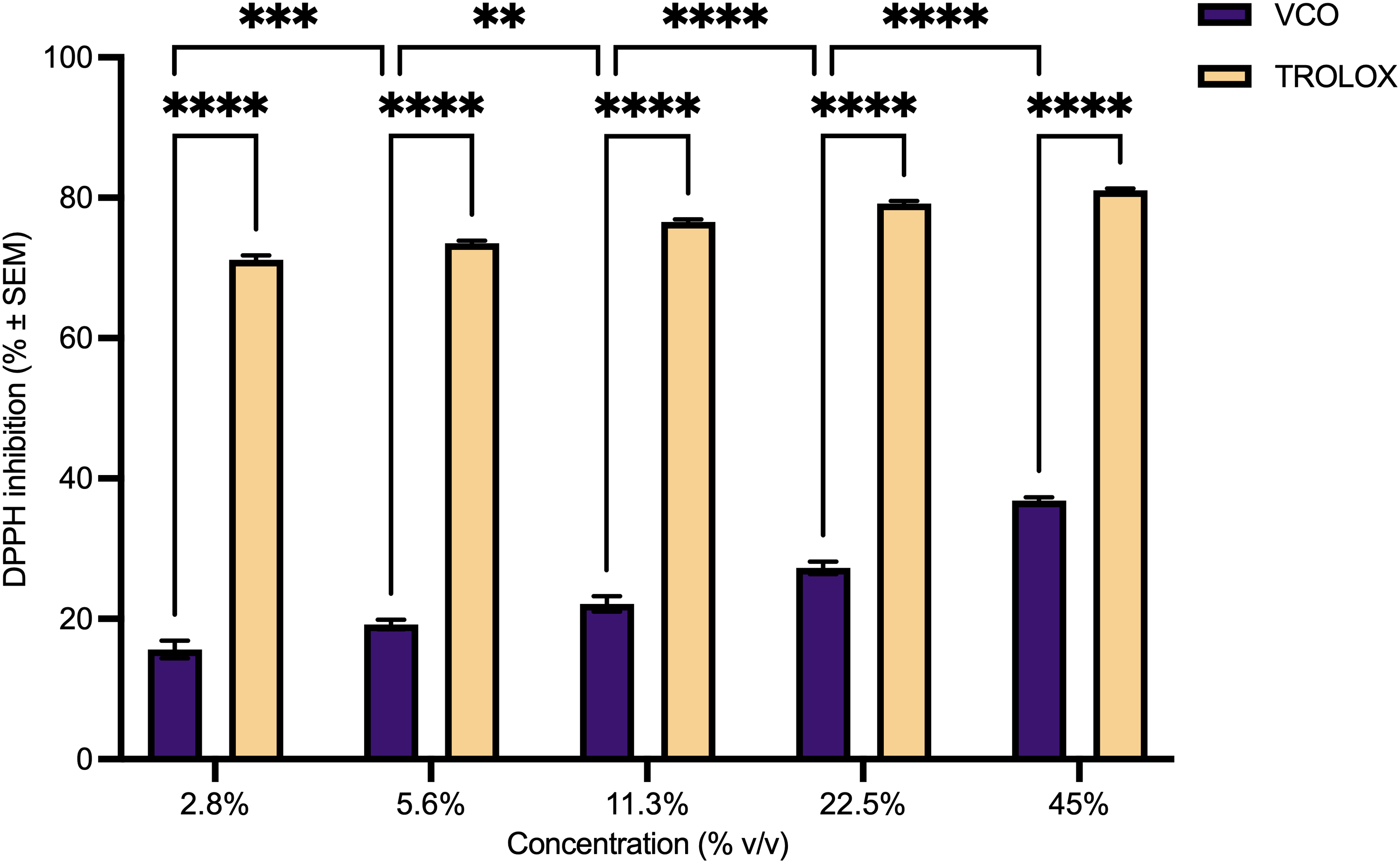
DPPH inhibition of various concentrations of VCO and control trolox. Values are expressed as mean ± SEM (n = 6). Statistical analysis by two-way ANOVA with Sidak's post-test. Significant differences denote by ** P ≤ .01, *** P ≤ .001, and **** P < .0001.
Antioxidant Capacity
As shown in Figure 3, the FRAP assay results indicated that VCO exhibits antioxidant capacity. There was a statistically significant difference in ascorbic acid equivalent antioxidant capacity (AEAC) across different concentrations. The highest VCO concentration of 413 600 μg/mL showed an AEAC of 4.4 ± 1.3 AEAC, which was significantly different from lower concentrations, including 206 800 μg/mL (15.9 ± 1.8 AEAC), 103 400 μg/mL (14.8 ± 0.8 AEAC), 51 700 μg/mL (13.8 ± 0.3 AEAC), and 26 000 μg/mL (13.2 ± 0.5 AEAC) of VCO as determined using a one-way ANOVA.
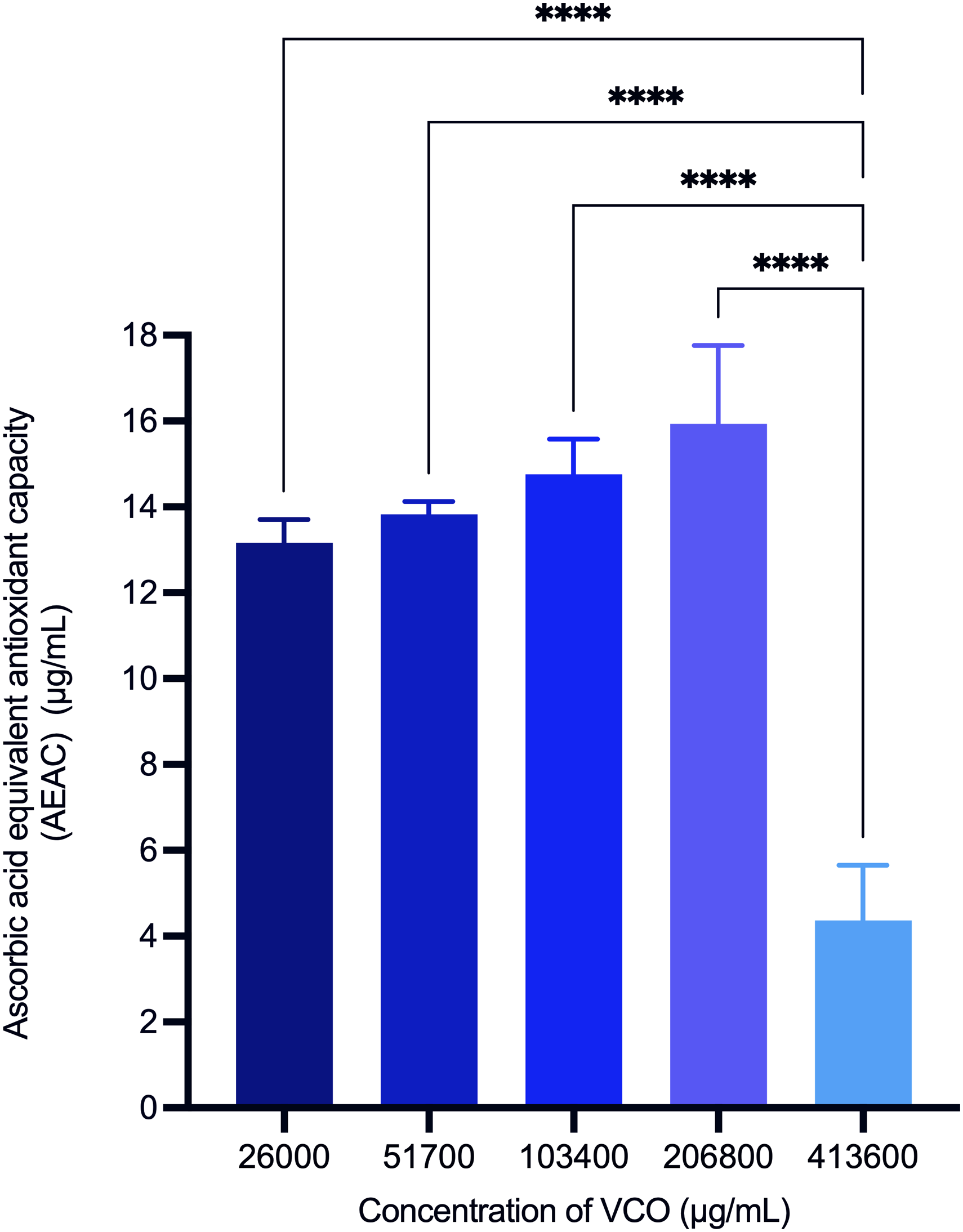
Total antioxidant capacity equivalent to ascorbic acid (AEAC) of virgin coconut oil measured with FRAP assay. Values are expressed as mean ± SEM (n = 6). Statistical analysis by one-way ANOVA with Tukey's post-test. Significant differences denote by **** P < .0001.
Cytotoxic Effects of VCO on HaCaT
The cytotoxic effects of positive control menadione and VCO on HaCaT cells within a similar range of concentrations are depicted in Figure 4. The results showed that HaCaT cell viability was higher in the VCO treatment group compared to the menadione control. At a concentration of 5.6% v/v, menadione is very toxic to HaCaT cells, with an IC50 at 4.6% v/v. Reduced viability of HaCaT cells was concentration-dependent when treated with VCO, with the IC50 identified at 17.8% v/v. HaCaT cells were most viable at 2.8% (102.5 ± 5.54%), followed by 5.6% (94.31 ± 5.04%) and 11.3% (74.36 ± 5.41%) v/v, after which declines toward higher concentration tested of 22.5% (34.94 + 2.31%) and 45% v/v (19.9 ± 2.82%). One-way ANOVA indicated a statistically significant difference in the HaCaT cell viability among the different concentration groups (F(5, 150) = 69.68, P < .0001). Post-hoc analysis using Tukey showed that cell viability in all groups was significantly different from the negative control (P < .0001). Pairwise comparisons between adjacent treatment groups showed a significant difference between 11.3% and 22.5% v/v (P = .0001) but not between other pairs.
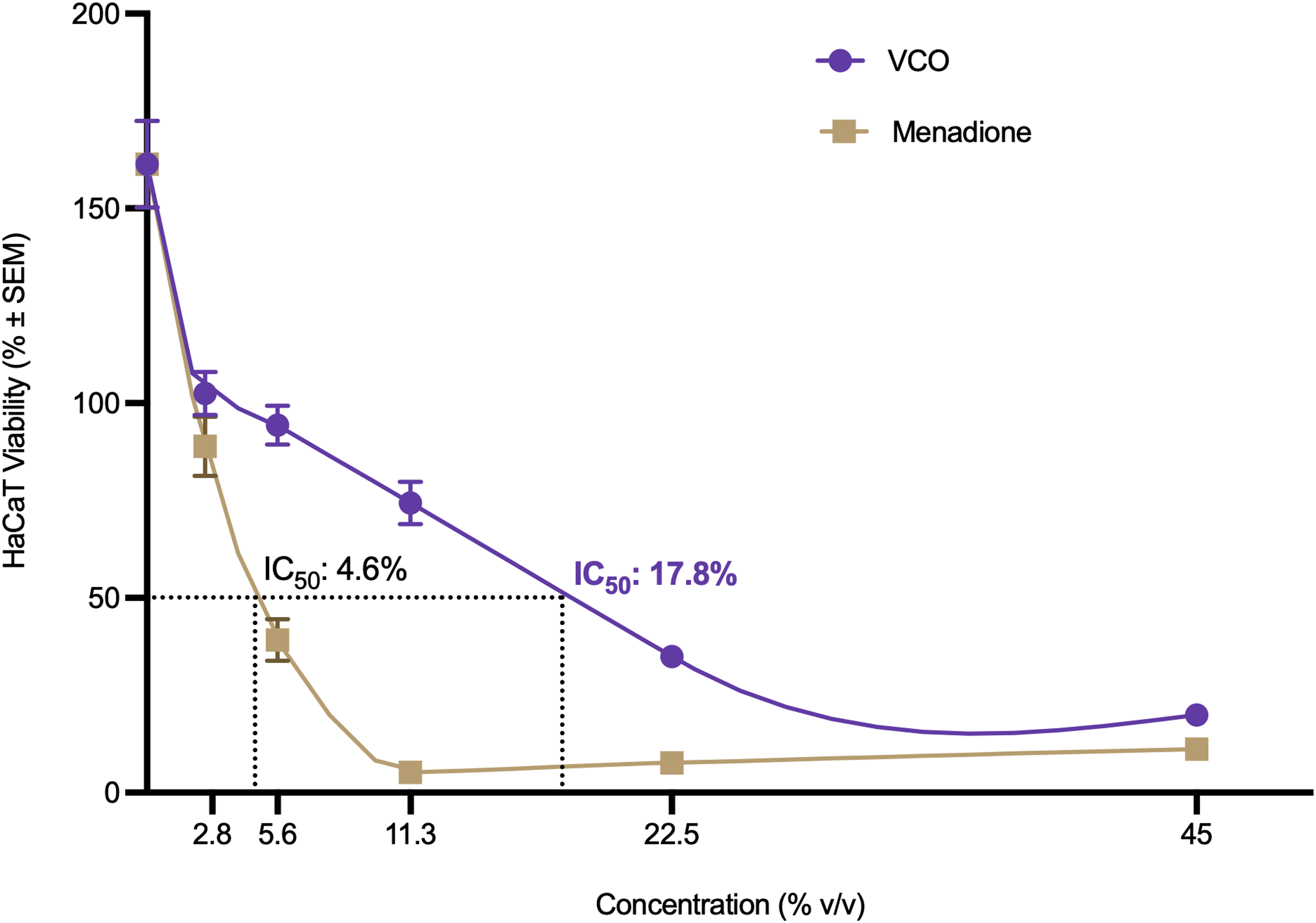
Cytotoxic effect of various VCO and menadione concentrations on HaCaT cells. Values are shown as mean ± SEM (n = 6).
Discussion
In recent years, there has been a rising interest in research focusing on uncovering plant-derived compounds with the potential to protect the skin from the adverse effects of oxidative stress. This interest stems from the increasing consumer demand for cosmetic products and dermatological treatments crafted from natural ingredients. VCO's research and commercial appeal emanates from its active ingredients that benefit the skin. The GC-FID analysis in the study confirmed that the overall fatty acid composition is consistent with that expected for VCO, where medium-chain fatty acids (MCFA) dominate the profile. This composition aligns with the fatty acids profile of commercially available VCO previously studied in the Malaysian market and comparable to Asian and Pacific Coconut Community (APCC) standard range for VCO.26,27 MCFA is saturated fatty acids with carbon chains ranging from 6 to 12, notably represented by caproic acid (C6:0), caprylic acid (C8:0), capric acid (C10:0) and lauric acid (C12:0). 28 These fatty acids contribute to strengthening the skin's protective barrier, providing energy through oxidation for normal skin functions, and inhibiting skin cancer. 28
The antioxidant properties of VCO have been previously evaluated by various methods.29–36 In the study, the concentration-dependent antioxidant activity of VCO showed the highest inhibition of the DPPH radical (36.8 ± 2.84%) at the highest tested concentration of 45% v/v. Compared to the standard Trolox, VCO has lower antioxidant activity. The FRAP assay indicated that VCO exhibited antioxidant capacity across various concentrations. The highest concentration tested showed a lower AEAC value than the lower concentration range. The findings align with earlier reports of a concentration-dependent activity against DPPH radicals.29,36 More recently, a relatively lower antioxidant activity was indicated by a 56.2% DPPH radicals inhibition using pure VCO. 29 They further classified VCO as a weak antioxidant based on an IC50 value of 313.46 mg/mL for VCO's phenolic extract. In the DPPH assay, the IC50 value represents the concentration of antioxidants required to decrease the initial DPPH concentration by 50%. A lower concentration of VCO indicates higher antioxidant activity. 37
However, a recent study observed that a mere 5% concentration of VCO resulted in an inhibition of 48.07, which substantially exceeded the antioxidant activity levels reported in this study. 30 An earlier study observed a range of lower IC50 values (7.49 to 104.52 mg/mL) for the DPPH radical-scavenging activity of VCO's phenolic compounds, with variability attributed to the specific processing methods employed. 33 Notably, the wet process, such as chilling and centrifugation methods or fermentation, exhibited superior antioxidant activity to the dry process in producing VCO. 33 Consistent findings were also reported previously, revealing that VCO obtained through the dry method exhibited the least amount of antioxidant activity among various extraction methods. 31 In this study, the VCO obtained was cold-pressed, a method often classified as a ‘dry’ approach to coconut oil extraction, and thus could partly contribute to the variations observed in the antioxidant outcomes.
Another possible factor that correlates with the antioxidant activity of plant oil is its total phenolic content, which also changes with processing methods. 34 Higher total phenolic content in plant oils often corresponds to enhanced antioxidant activity, possibly due to the preservation of phenolic compounds in wet processing versus dry processing methods. 38 For instance, the hot VCO extraction method has been found to yield higher total phenolic content compared to fermentation and mechanical (dry) extraction methods. 39 While the present study did not measure the total phenolic content of the cold-pressed VCO, a recent study using VCO extracted through cold-press corroborates these earlier findings. 32 A relatively lower total phenolic content in the cold-pressed VCO from various coconut varieties was reported, along with higher IC50 values for the DPPH radical-scavenging activity that fell within the range of 857.19 ± 14.99 mg/mL to 1282.5 ± 18.3 mg/mL. 32
The study employed the MTT assay to evaluate the cytotoxic effects of VCO extract on HaCaT cells. Treatment with VCO showed that the HaCaT cell viability was concentration-dependent. The classification of substance cytotoxicity based on percentage of cell viability under ISO 10993-5 indicated that VCO is non-cytotoxic to HaCaT at concentrations of 5.6% and lower, mildly toxic at 11.3%, and highly toxic at concentrations beyond 22.5%. The cytotoxicity of VCO in this investigation was lower than that reported in a previous study, which demonstrated an IC50 of 787.15 µg/mL for VCO's cytotoxicity to HaCaT cells. 16 The impact of solvents on cell viability is well-documented, with variations depending on the specific cell type and concentration used.40,41 A prior study reported a cytotoxic effect of DMSO in HaCaT cells when used at a concentration of 3.1% v/v. 42 Consequently, incorporating DMSO as a solvent in a similar concentration for the highest VCO concentration tested in the study contributed to the observed cell death in HaCaT cells.
The therapeutic potential of Virgin Coconut Oil (VCO) is influenced by its bioactive profile, which can vary based on extraction and processing methods, coconut varieties, and geographical origins. The cold-pressed VCO used in this study may have unique bioactive components contributing to its observed therapeutic effects. However, this variability in bioactive constituents presents a limitation, as the specific components responsible for these effects have yet to be fully characterized. GC-FID analysis provided valuable insights into the fatty acid composition may not encompass all relevant bioactive compounds. Therefore, further characterization of these bioactive constituents is essential to enhance understanding of VCO's therapeutic potential and elucidate its effects on the skin, ultimately optimizing the formulation and improving efficacy. Additionally, further studies are warranted to explore the mechanisms underlying these effects.
Conclusion
The analysis of virgin coconut oil (VCO) in this study demonstrated its rich composition of medium-chain fatty acids (MCFAs), with lauric acid being the predominant component. The antioxidant properties of VCO were substantiated through both the DPPH assay and the FRAP assay, revealing significant dose-dependent inhibition of DPPH radicals and notable antioxidant capacity. The treatment with VCO resulted in reduced cytotoxicity at lower concentrations, with only a mild cytotoxic effect on HaCaT cells induced at 11.3% v/v concentration. In light of the cytotoxicity findings, lower concentrations of VCO are suggested as optimal for prospective skincare formulations and applications.
Footnotes
Acknowledgements
The authors thank the staffs at Faculty of Health Sciences Universiti Kebangsaan Malaysia for supporting and assisting this study.
Data Availability Statement
The data used and analyzed during the current study are all provided in the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Dana Inovasi UKM (INOVASI-2021-012).

