Abstract
Introduction
Viral diseases, primarily caused by viruses, pose significant threats to both humans and animals. Although Before viral diseases were well known before discovery of viruses, the history of virology is relatively recent and will continue to evolve. 1 Life-threatening viruses cause several viral infections including influenza A, respiratory viruses, human immunodeficiency viruses, SARS CoV-2, coronavirus and hepatitis viruses, YVF (yellow virus fever), malaria, DENV-2 (dengue-virus type 2), and coxsackie virus. 2 The consequences of viral infection can be extremely diverse, ranging from asymptomatic infections severe diseases and death. Viral diseases pose a permanent challenge not only to humans and animals but also to plants, protozoa, fungi and bacteria. Viruses can disrupt an organism's normal physiological processes through several mechanisms including homeostasis disruption, immunosuppression, oncogenesis, direct cell-induce death, immunopathology and cellular transformation. These mechanisms make viral infections a major concern for human health. Viruses have developed resistance to drugs and spread rapidly, leading to an unexpectedly high death rate worldwide. Each year, infectious diseases cause the deaths of more than 20% of the global population. Consequently, viral infections have become a major challenge for healthcare professionals globally due to uncontrolled rate of mortality and morbidity. 3
Over the past few years, several synthetic antiviral agents (such as interferon, ribavirin, etc) have been explored to treat different viral infections. Currently, investigations are being carried out to provide protection against viral load using existing antiviral drugs. Unfortunately, uncountable, various adverse effects have been associated with these synthetic antiviral drugs including ineffectiveness against resistant viral strains, discrepancies in efficacy between in vitro and patient treatment and high cost, making them unaffordable for people in developing countries. Given the current situation, there is a pressing need for research to find effective and affordable antiviral drugs.3,4
Herbal medicine can serve as an effective alternative to conventional antiviral drugs. Medicinal plants, along with their biologically active metabolites, are becoming increasingly popular due to their anti-inflammatory, antibacterial, antiviral, anticancer, and antioxidant properties. 5 From crude extracts of terpenes, flavonoids, alkaloids, proteins, and various glycosides several phytochemicals were isolated, identified, and purified. Compounds including rutin, which is present in medicinal plants, exhibit antiviral activities. 6 The protection of aquatic animals (such as fish and shrimp) from viral infections by using medicinal plants has been extensively reported. 7 Plant-derived compounds display diverse modes of action, allowing them to inhibit various vrial mechanisms including blocking viral replication, preventing intracellular interactions of virus particles, reducing virus infectivity and inhibiting enzymes (RNA polymerase and DNA polymerase). Compared to classic synthesized medicines, medicinal plants offer several advantages. They consist of eco-friendly bioactive compounds in abundance, have lower negative influences on the environment and serve as immunostimulants to fight pathogens. Due to these beneficial properties, medicinal plants are considered as potential alternatives to conventional synthetic drugs. 7
Conventional solvents including n-hexane and alcohols are used for to recover different plant-based compounds such as phenolic and flavonoids from various parts of plant matrices. 8 These bioactive compounds can be employed to develop different modern medicines, thus their separation and isolation using different solvents is important. Solvents used for extraction purposes should be highly selective, safe, can easily be separated form recovered compounds, higher volatility and economical. 9 These conventional solvents have several disadvantages. They are required in bigger volume, leading to enhance the cost of analysis. These solvents are often toxic and leave residual solvents in the recovered extracts. Furthermore, their volatile nature poses a considerable risk of air pollution, contributing to global warming. 10
Thus, green solvents, including Natural Deep Eutectic Solvents (NADES) are required minimize the disadvantages of traditional solvents. NADES have attracted significant attention from scientists due to their excellent potential as extracting media. The composition of NADES mainly includes natural metabolites found in all living organisms and cells such as urea, sugars (glucose, sucrose), choline chloride, and organic acids. NADES possess many magnificent advantages like low cost, biodegradability, sustainability, tuneable properties (viscosity, pH and density) and also provide stability to the bioactive compounds. 11
The primary aim of this project was to assess the analytical characterization, antioxidant, anti-microbial and anti-viral properties of selected medicinal plants mainly including S. alexandrina, P. harmala, and C. colocynthis. This study employed NADES and water for extracting bioactive components from selected plats, focusing on their antiviral properties. Additionally, this research includes a comparative analysis and characterization of extracts obtained using NADES and water. To evaluate antiviral potential, NADES and water extracts of selected medicinal plants were applied to virally infected plants to evaluate their anti-viral potential.
Material and Methods
Chemicals and Reagents
Various chemicals and reagents including Folin-Ciocalteu reagent (99.9% purity, Sigma Aldrich), Quercetin (98% purity, AppliChem), DPPH (99% purity, Alpha Chemical), Na2CO3 (99.9% purity, Daejum), methanol (99% purity, Merck), gallic acid (98% purity, AppliChem), AlCl3 (99% purity, Merck), citric acid, glucose, NaNO3 ((97% purity, RDH)) and distilled water (Made in the lab) were obtained from store of Government College Women University of Faisalabad.
Experimental Procedure
How to Prepare NADES
To prepare NADES (300 mL), 100 g of each glucose and citric acid were dissolved in distilled water by stirring on hot plate at 60 °C till the solution became clear.
Preparation of Plant Extracts
S. alexandrina was collected from the local market of Faisalabad, while, P. harmala, and C. colocynthis were obtained from surrounding village of Hasilpur, Punjab, Pakistan. The collected sample were identified by a taxonomist at Department of Botany, Government College Women University Faisalabad Pakistan. These plants samples were washes, shade-dried and ground to 80 mesh. Each sample (20 g) was blend with in distilled water (100 mL) using blender for 5–10 min, followed by pouring the sample into a conical flask (500 mL) which was processed for maceration for 3 days. After the required processing time, samples were filtered Whatman No. 1 filter paper and excessive solvent in the resultant filtrate was evaporated using rotavapor. After the evaporation of solvent, crude concentrated extract was collected and its extraction yield was calculated. The recovered extracts were stored at −4 °C in glass vials. The same experiment was performed to get NADES extract using NADES as solvent. All the abbreviations of sample extracts and their NADES were mentioned in Table 1.
Abbreviations of Samples Extracts and Their NADES.

Total Phenolic Content (TPC) and Total Flavonoid Content (TFC)
TPC was determined using a modified version of Folin-Ciocalteu reagent method as outlined by Ahmad et al. 12 The dried residue of the sample was reconstituted in water (1 mg/mL). An aliquot (1.0 mL) of this solution was then combined with Folin-Ciocalteu reagent (0.5 mL) and deionized water (7.5 mL), followed by the addition of sodium carbonate (1.5 mL; 20% (w/v)) after 10 min of incubation. The resulting mixture was then allowed to heat in a water bath (40 °C) for 20 min and subsequently cooled in an ice bath, followed by recording of absorbance at 755 nm using a spectrophotometer (Hitachi U-2001, model 121-0032). The standards (5 to 100 ppm) of gallic acid were processed under similar experimental conditions. TPC was estimated as gallic acid equivalents (GAE) per gram of dry residue using the calibration curve obtained from gallic acid standards. Each sample was run in triplicate to ensure accuracy.
TFC was quantified using a method described by Ahmad et al. 9 The reconstitution of the dried residue was done in water at aa concentration of 100 mg/mL. An aliquot (1.0 mL) of this solution was transferred to volumetric flask (10 mL), followed by the addition of distilled water (5.0 mL) and NaNO2 (5%; 0.3 mL). After five mins of incubation, AlCl3 (10%; 06 mL) and NaOH (1 M; 2 mL) were mixed into final solution whose volume was made up with distilled water and its absorbance was observed at 510 nm. A series of standards (5 to 100 ppm) of catechin were also run under the similar experimental conditions. TFC was estimated as catechin equivalents (CE) of per dry matter with the help of calibration curve obtained from catechin standards. Each sample was process in triplicate to ensure accuracy.
Determination of Antioxidant Activity
The antioxidant activity of tested extracts and NADES was determined using 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical assay following a method as described by Tepe et al
13
Aliquots (50 µL) of the extracts at varying concentrations (0.10 to 5.0 mg/mL) were mixed methanolic solution of DPPH (0.004%; 5 mL). The resulting solutions were incubated for 30 min at room temperature, the absorbance was measured at 517 nm: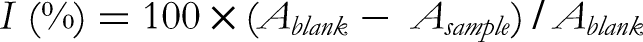
Characterization of Plant Extracts (HPLC)
The phenolic and flavonoid components in water and NADES extracts of three medicinal plants have been separated, characterized, identified and quantified. HPLC analysis was performed by following the protocol. 14 A Shimadzu HPLC system (Shimadzu Japan) equipped with a Shim-Pack CLC-ODS (C18 column: 25 cm×4.6 mm, 5 µm) was employed for identification of bioactive components. Two mobile phases: phase A (water: acetic acid, 94:6, pH 2.27) and phase B (absolute acetonitrile) were utilized which were delivered at pressures ranging from 500 to 5000 psi. An LC- 10 AT pump maintained at flow rate of 1 mL/min at room temperature. Detection of bioactive components was performed using UV-VIS spectrophotometer (SPD – 10AV) at 280 nm.
FTIR
This technique has extensively been utilized to characterize different materials. Through this method, the active compounds present in the sample were identified. The tested plant extracts and NADES were processed for FTIR analysis. It was carried out on a drop of water and NADES extracts by using an FTIR spectroscope. Each sample was scanned in a range from (400 to 4000 cm−1). 15
Anti-Microbial Activity
Anti-microbial potential of water and NADES extracts of investigated plants was assessed following the agar well diffusion method.
16
For this purpose, plates were prepared by nutrient agar media and then inoculated by bacterial strains culture by streaking method. In this research, we used S. aureus and E. coli strains. To make wells bottom of micro-tip after sterilization is used. 100
Antiviral Capacity
Nicotiana benthamiana plants were cultivated in order to perform antiviral capacity of tested extracts and NADES and the Cotton Leaf Curl Multan virus was propagated on these plants. Each plant extract was the individually applied to the infected plants to assess their ability to inhibit viral growth. The extracts were diluted with solvent or DMSO prior to application and subsequent analysis.
Statistical Analysis
The results were presented as mean value ± standard deviation (SD) with each experiment was conducted in triplicate. Statistical analysis was performed using analysis of variance (ANOVA) to differentiate the experimental results at the 95% confidence level. This analysis was carried out using STATISTICA 5.5 software (Stat Soft Inc, Tulsa, Oklahoma, USA).
Results and Discussion
Determination of Total Phenolic and Flavonoid Content
TPC was calculated using Folin-Ciocalteu method (Figure 1), where, results were estimated as mg GAE/g of dry extract weight with the help of calibration curve (

TPC (a) and TFC (b) of water and NADES extracts of selected plants.
TFC was assessed using aluminum chloride method, where quercetin was employed as standard to exhibit TFC in mg QE/g with the help of calibration curve (
Determination of Antioxidant Activity
Various plant species with high antioxidant potential have long been used to treat various.
19
In this study, antioxidant capacity of selected medicinal plants was assessed using DPPH free radical scavenging assay. Extracts from selected plants, recovered with water and NADES, were evaluated under UV light, where, samples demonstrated varying levels of UV absorption, with SW, SN, KW, HN, KN and HW showing absorbance value of 1.052, 1.325, 1.653, 0.816, 1.028 and 1.189 cm−1, respectively. The inhibition (%) of DPPH free radicals was as follow: SW (47.6%), SN (34%), KW (17.7%), HN (59.4), KN (48.8%) and HW (40.8%). Water and NADES extract of samples showed % inhibition in the order: HN > KN > SW > HW > SN > KW. NADES extract of P. harmala absorbed minimum UV radiation thus shows maximum % inhibition and as a result higher anti-oxidant activity. On the other hand, water extract of C. colocynthis absorbs maximum UV radiations and it shows less % inhibition thus having lower antioxidant activity. NADES extract of C. colocynthis displayed higher antioxidant capacity compared to the water extract extract of S. alexandrina, which, in turn, demonstrated greater antioxidant potential than the water extract of P. harmala. However, the water extract of P. harmala showed higher antioxidant ability than NADES extract of S. alexandrina. Figure 2 represented the comparison of the antioxidant activities of different extracts (water and NADES) of selected plants with ascorbic acid (3.276 µl/mL) with the help of calibration curve of ascorbic acid (
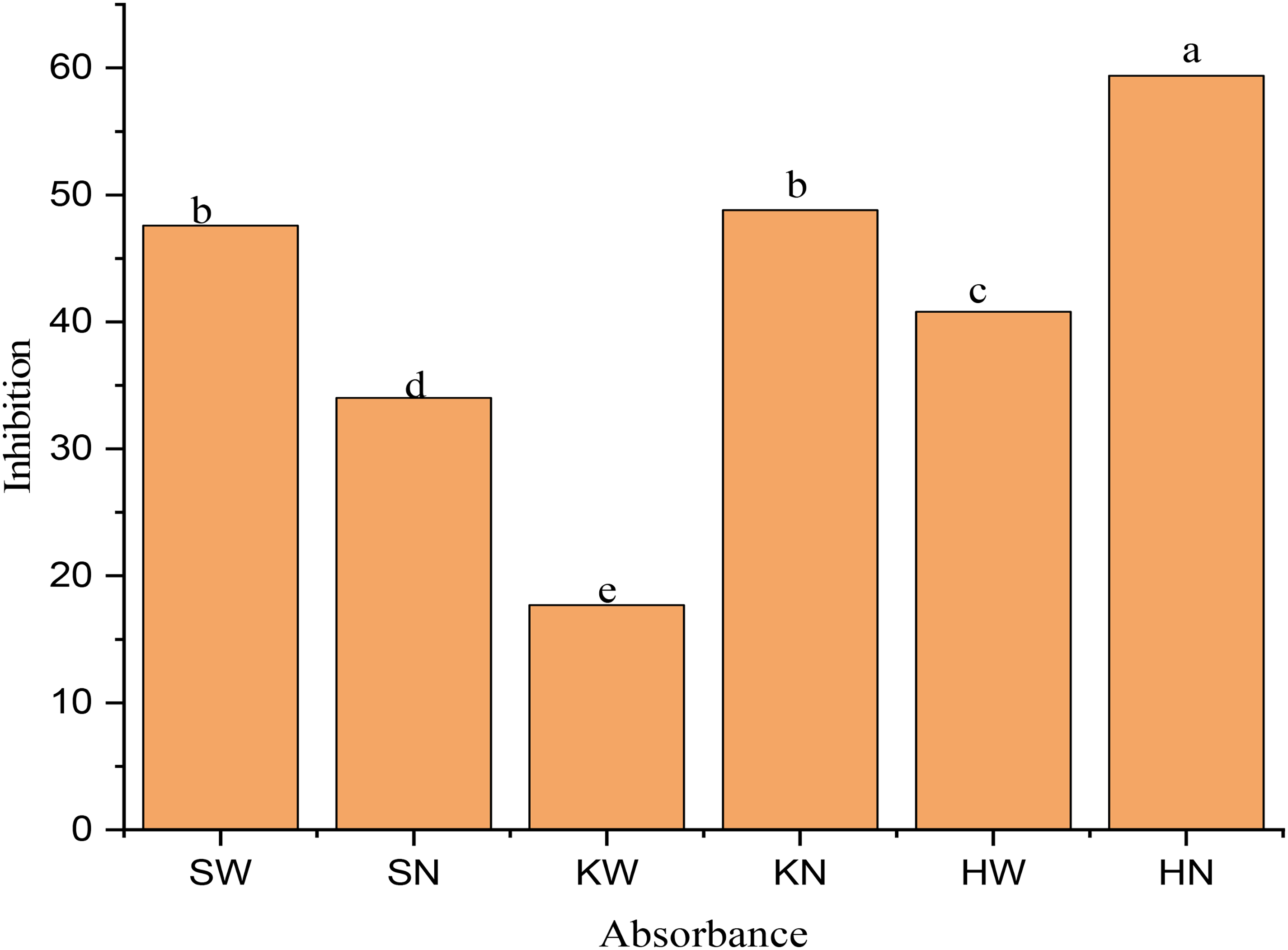
Antioxidant activities of water and NADES extracts of selected medicinal plants.
HPLC Analysis of Active Extracts
HPLC is a technology employed to separate, identify and quantify bioactive components in plant matrices. These phenolic compounds are responsible for various biological activities like antibacterial, anti-cancerous, antiviral activities. 9 HPLC results of all samples showed that polyphenols and flavonoids having anti-oxidant properties were present in the extracts. The qualitative and quantitative analysis of water and NADES extracts recovered from selected medicinal plants was represented in Tables 2 and Figures (3, S1) while their standard curves of gallic, p-coumaric, ferulic, chlorogenic, synergic and caffeic acids were presented as Figure S2.

Typical HPLC chromatogram of sample extract.
HPLC Analysis of Phenolic Compounds Extracted Using Water and NADES as Solvents from Citrullus Colocynthis, Peganum Harmala, Senna alexandrina.
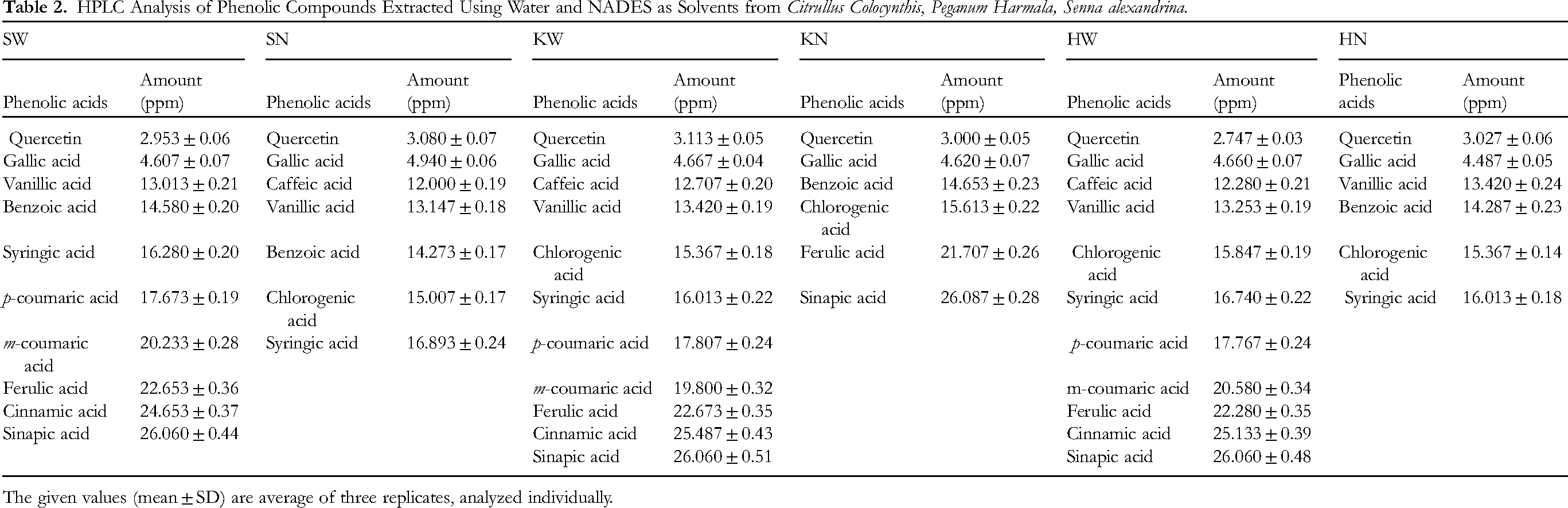
The given values (mean ± SD) are average of three replicates, analyzed individually.
Similar amount of certain phenolic acids including quercetin, gallic, chlorogenic acids were found in both water and NADES extracts of the plants. However, ferulic and sinapic acid were present in slightly higher concentration in NADES extract of C. colocynthis compared to its water extract. Conversely, phenolic acids including syringic, p-coumaric, m-coumaric acids were extensively detected in the aqueous extract of this plant. In P. harmala, the levels of gallic, vanillic, chlorogenic and syringic acids were consistent between the water and NADES extracts. However, certain phenolic acids including p-coumaric, m-coumaric, ferulic and cinnamic acids were only found in the aqueous extract were and were not extracted by NADES. Higher amount of sinapic acid was recovered using NADES in comparison to that of water. In case of S. alexandrina, the levels of gallic, vanillic and benzoic acids were approximately the same in both NADES and aqueous extracts. Chlorogenic acid, however, was only extracted using NADES and was absent in the water extract. Additionally, p-coumaric, m-coumaric, ferulic, cinnamic and sinapic acids were exclusively extracted using water from S. alexandrina.
For P. harmala and S. alexandrina, NADES extracts yielded higher amounts of gallic and quercetin compared to water extracts. Benzoic acid was extracted using NADES from C. colocynthis and P. harmala, but both water and NADES extracts of S. alexandrina contained benzoic acid. The results of presence of these bioactive compounds in extracts of selected plants was in line with literature. 21
FTIR Analysis
The FTIR was employed for the identification of functional groups of the active components by analyzing the peak in infra-red (IR) region. According to FTIR results, different chemical constituents were found to be present in water and NADES extracts of selected medicinal plants. Each extract displayed distinct peak values corresponding to different stretching frequency, indicating the presence of various functional groups.
FTIR spectra of S. alexandrina
The FT-IR spectroscopy results exhibited the occurrence of different chemical constituents including alcohol, alkanes, aromatic carboxylic acid and alkyl halide in both water and NADES extract of S. alexandrina (Figure 5). Prominent peaks were identified at 3492.45 cm− 1 which corresponded to O-H stretching, N-H stretching, O-H stretching vibration of –CO2H. This indicated that carbonyl compounds were present in water extract but not in NADES extract. Additionally, other functional groups including carboxylic acids, terminal alkynes, ketones, aromatic compounds, phenols or tertiary alcohols and acids were recorded at 3629.37 cm−1, 3492.45 cm−1, 3409.53 cm−1, 3315.03 cm−1, 1625.70 cm−1, 723.18 cm−1, 825.38 cm −1 and 559.26 cm−1, respectively in water extract in comparison to NADES extract. The IR stretching frequency (1720.19 cm−1) was attributed to C = O stretching frequency, indicating the presence of carbonyl groups linked with conjugated aromatic compounds. Bands at 1222.65 cm−1, 1108.87 cm−1, 1074.16 cm−1 and 1025.94 cm−1 were corresponded to the C-O stretch and C-N stretch groups, respectively, although the C-N stretch was often weak and hard to detect. The intense bands at 1222.65 cm−1 and 1108.87 cm−1 stretching suggested the occurrence of esters and secondary alcohols. Therefore, the FTIR spectrum verified the occurrence of phenols, amino acids, amides, carboxylic acids, alkanes, aliphatic esters, and secondary alcohols in NADES extracts of S. alexandrina. The results of present analysis were found to be in line with the literature. 22
FTIR spectra of P. harmala
The findings of FT-IR spectroscopy of tested plant exhibited the identification of six functional groups in its both extracts. The Figure 4 displayed the occurrence of different chemical constituents including alcohol, alkanes, aromatic carboxylic acid and alkyl halide in both the aqueous and NADES extracts. However, halogen compounds were only detected in the NADES extract. Prominent peaks were identified at 3492.45cm−1, corresponding to O-H stretch, N-H stretch and O-H stretching vibrations of–CO2 H, indicating the presence of carbonyl compounds in both extracts. Other functional groups were also identified at various absorption frequency; carboxylic acids at 3629.37cm−1, terminal alkynes at 3492.45cm−1, ketones at 3409.53cm−1, aromatic compound at 3315.03cm−1, phenol or tertiary alcohol at 1625.70 cm−1, acids at 723.18, 825.38 and 559.26 cm−1. The bands at 1222.65 cm−1, 1108.87cm−1, 1074.16cm−1 and 1025.94cm−1 were attributed to C-O and C-N stretches, with the latter often being weak and hard to detect. The peaks at 1222.65cm−1 and 1108.87cm−1 indicated the existence of esters and secondary alcohols. Consequently, FTIR authenticated the existence of phenols, amino acids, amides, carboxylic acids, alkanes, aliphatic esters, secondary alcohols in NADES extract. The findings of this analysis aligned with the literature. 23

FTIR Spectrum analysis of water and NADES extracts of medicinal plants.
FTIR spectra of C. colocynthis
Figure 4 represented FT-IR spectroscopy, confirming the existence of different compounds including alcohol, alkanes, aromatic carboxylic acid and alkyl halide in both the aqueous and NADES extracts of C. colocynthis, where halogen compounds were only detected in NADES extract. The prominent peaks detected at 1783.83cm−1 were attributed to the C = O stretch (conjugated) and aromatic overtones of ring bends, while in the NADES extract, this peak was noted at 1720.19cm−1, indicating the existence of carbonyl compounds in both extracts. Additionally, other functional groups were also detected at different absorption frequency: carboxylic acids at 3654.44, terminal alkynes at 3357.46 cm−1, ketones at 2952.48 cm−1, aromatic compounds at 1783.83 cm−1, phenol or tertiary alcohol at 1629.55 cm−1, acids at 723.18, 831.17and 622.89 cm−1. The absorption bands in NADES spectrum at 1218.79 cm−1, 1106.91 cm−1, 1074.16 cm−1 and 1029.80 cm−1 were linked to C-O and C-N stretches, where latter was found to be weak and hard to detect. The absorption bands at 1218.65 cm−1 and 1106.91 cm−1 confirmed the existence of esters and secondary alcohols. These findings aligned with the literature. 24
Antimicrobial Activity
Medicinal plants are believed to be abundant in antimicrobial ingredients, offering a cost-effective alternative for treating bacterial infections.
16
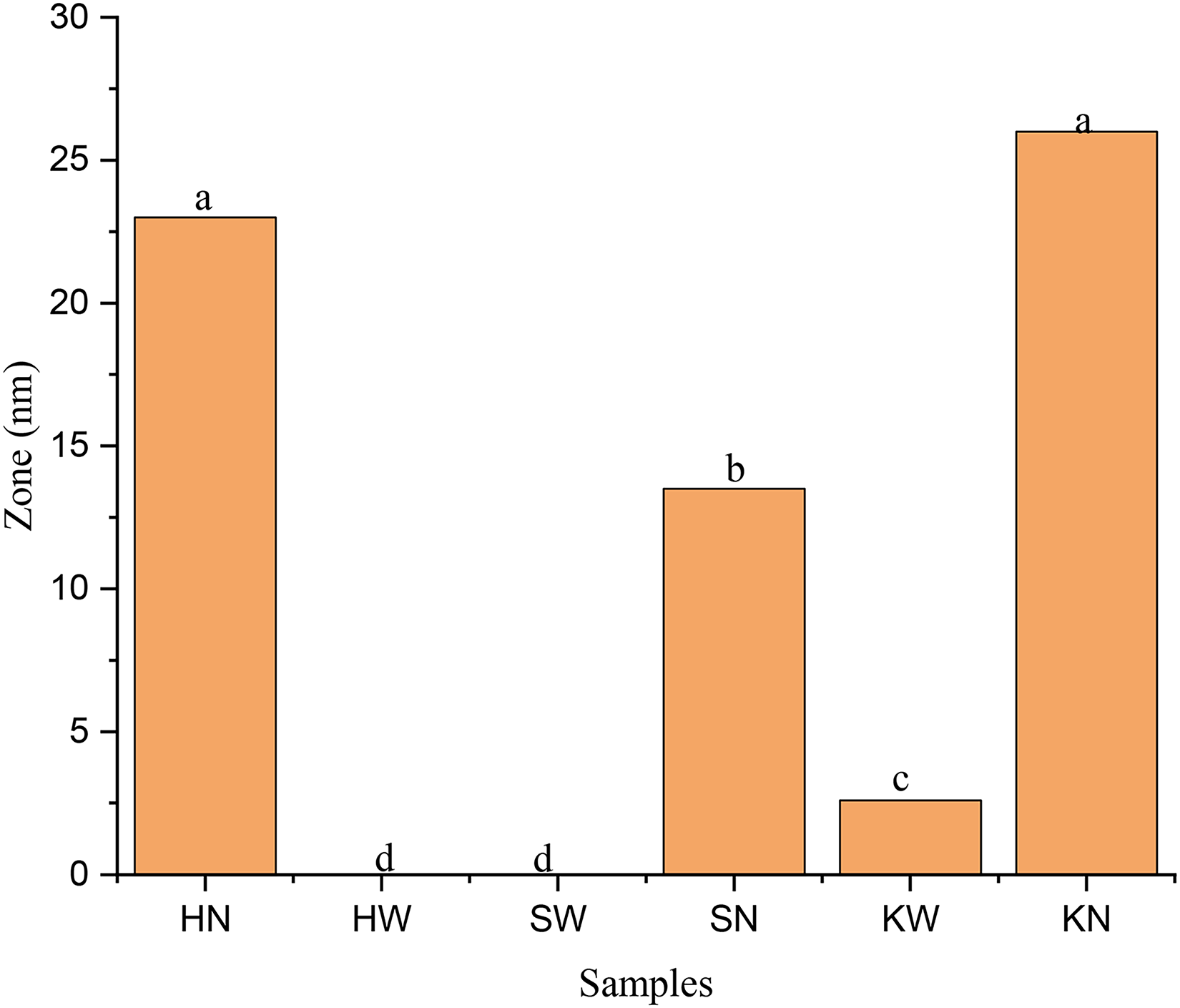
Antimicrobial potential of extracts, recovered from selected medicinal using water and NADES, were evaluated against a set of bacterial strains including S.aureus and E.coli, where ciprofloxacin was used as control. The results revealed that NADES extract of P. harmala showed highest bactericidal effect against S.aureus with a zone of inhibition (25 mm). In contrast, the water extract of P. harmala and S. alexandrina exhibited no inhibition against S.aureus, indicating their ability to inhibit the growth of tested bacteria. The NADES extract of C. colocynthis demonstrated a 23 mm inhibition zone against S.aureus followed by NADES extract of S. alexandrina with a inhibition zone (20 mm) and water extract of C. colocynthis with a inhibition zone (6 mm). The antimicrobial activity of all samples decreases in order: HN > KN > SN > KW. The Figure 5 showed the comparison of antimicrobial activity of tested samples

Antimicrobial activities of water and NADES extracts of selected medicinal plants against S. aureus.
Antimicrobial activities of water and NADES extracts of selected medicinal plants against E.coli.

Zone inhibition showing antimicrobial activities of water and the NADES extracts of selected medicinal plants against E.coli and S. aureus.
Antiviral Activity
To assess the antiviral potential of water and NADES extract of selected medicinal plants, the Cotton Leaf Curl Multan virus was used to infect Nicotiana benthamiana plants grown under optimal conditions. Plants infected with virus and treated with NADES extract of S. alxendrina did not survive after two weeks of analysis under these conditions (Figure 8), indicating that the NADES extract of S. alxendrina could not inhibit the viral growth. In contrast, when infected plants were treated with the water extract of S. alxendrina, the plant survived after 2 weeks of exposure under these conditions (Figure 9), showing that the water extract of S. alxendrina has antiviral potential, acting as an inhibitor of tested virus. The NADES extract of C. colocynthis also failed to inhibit the virus, leading to the death of the infected plant (Figure 10). Furthermore, these inhibitors were toxic to the plants, rendering them unable to inhibit the DNA of tested virus. The roots and stems of plants exhibited brittleness, leading to plant death. Infected plants treated with water extract of C. colocynthis survived after two weeks of exposure under optimum conditions, indicating its effectiveness as an inhibitor of the virus (Figure 11). This suggested that the water extract of C. colocynthis was potent inhibitor of Cotton Leaf Curl Multan virus and exhibited antiviral potential. HPLC analysis confirmed the presence of quercetin, p-coumaric acid, syringic acids, benzoic acids and other polyphenolic components and flavonoids in SW and KW extracts. These compounds primarily behaved as markers that exhibited antiviral activity.

NADES extract of S. alxendrina as a Cotton Leaf Curl Multan virus inhibitor.

Water extract of S. alxendrina as a Cotton Leaf Curl Multan virus inhibitor.

NADES extract of C. colocynthis as a Cotton Leaf Curl Multan virus inhibitor.

Water extracts of C. colocynthis as a Cotton Leaf Curl Multan virus inhibitor.
The water extract of P. harmala was not found to exhibit potential to inhibit the Cotton Leaf Curl Multan virus, resulting in plant death after 2 weeks of analysis (Figure 12). The roots and stems of plants exhibited brittleness, which led to plant death. Thus, it was concluded that water extract of P. harmala could not act as inhibitor of the tested virus. Likewise, plants infiltrated with the NADES extract of P. harmala as an inhibitor of the virus, could not survive after 2 weeks of analysis under optimums condition (Figure 13). In this case, the roots and stems of the plants showed brittleness, leading to plant death, indicating that the NADES extract of P. harmala could not act as virus inhibitor. The only active compound against viruses in the extract of P. harmala was harmine, an alkaloid that directly inhibited the viral growth by inhibiting the protein replication process in virus. This compound was not extracted using either water and NADES, resulting in no antiviral activity being shown by P. harmala. 25 Other phenolic and flavonoids compounds that were extracted (Table 2), however, these compounds were found to be inactive against Cotton Leaf Curl Leaf Multan Virus.

Water extract of P. harmala as a Cotton Leaf Curl Multan virus inhibitor.

NADES extract of P. harmala as a Cotton Leaf Curl Multan virus inhibitor.
Emerging viral or infectious diseases have become a significant challenged for human populations. Traditionally medicinal plants have been utilized for various types of viral and infectious diseases. According to recent study, 3 25% of modern medicines currently in use consist of bioactive compounds primarily extracted from plant source, as these plants are rich reserves for the discovery, especially for viral diseases. Therefore, in this study, NADES and water extracts of S. alexandrina, P. harmala and C. colocynthis were evaluated for their anti-viral potential against Cotton Leaf Curl Multan virus. The results showed that the water extract of C. colocynthis and S. alexandrina exhibited the maximum antiviral potential by inhibiting the viral growth, leading to plant survival. The higher antiviral activity of the water extract of C. colocynthis and S. alexandrina was attributed to the presence of quercetin and p-cumaroyl, which induced autophagy and inhibited the synthesis of viral mRNA. This conclusion was supported by the higher values of TPC, TFC and antioxidant activity of water extracts of C. colocynthis and S. alexandrina.
Phenols are the phytochemical present in plant extracts that exhibit antioxidant activity due to the presence of hydroxyl groups. C. colocynthis and S. alexandrina have been reported to be rich sources of secondary metabolites. 26 Furthermore, recent studies indicate that flavonoids also possess antiviral potential, with various mechanisms of action to inhibit viral growth. They attached themselves to the surface proteins of viruses, thereby prohibiting the entrance of virus into the host cell. They can also interfere with the protein translation and DNA replication of viruses. 27 FTIR and HPLC analysis of water extracts of these two plants showed an increased number of phenolic acids as well as quercetin, which have been reported in literature as inhibitor of viruses.28,29 In contrast, NADES extracts of C. colocynthis and S. alexandrina could not exhibit antiviral potential by inhibiting the growth of virus, resulting in plant death which was attributed to their lower values of TPC, TFC and antioxidant activity. The results of FTIR and HPLC analysis also confirmed their lower antiviral potential, as fewer compounds were extracted by NADES from these medicinal plants. This variation might be due to several factors including difference in harvesting time of plants, season, geographical area, extraction techniques, solvent polarities and genotype. 18
Water and NADES extract of P. harmala were also evaluated for their antiviral potential and the results showed that they could not inhibit growth of the virus, leading to plant death. However, their TPC, TFC and anti-oxidants results were quite considerable. The antiviral properties of P. harmala are primarily due to the presence of alkaloids, mainly β-carbolines, which include harmine, harmalol and harmaline. These β-carbos lines have a broad pharmacological range, functioning as antiviral and antimicrobial agents. 30 In this study, solvents including water, citric acid and glucose based NADES failed to extract alkaloids from P. harmala, resulting in no observed antiviral activity. FTIR and HPLC results also confirmed the absence of alkaloids in both water and NADES extracts. The antimicrobial analysis of NADES extracts from all three plants showed significant zone of inhibitions against S.aureus and E.coli compared to water extracts. Specifically, the NADES extract of P. harmala demonstrated greater antimicrobial activity against S.aureus with a 59.4% inhibition and 25 mm zone of inhibition. A study has reported strong antimicrobial activities of NADES extract against both gram-positive and gram-negative bacteria. 31
The higher antimicrobial activity of P. harmala has also been reported, where its ethanolic extract inhibits the growth of micro-organisms. While previous studies showed that the ethanolic and water extract of P. harmala seeds exhibited limiting inhibitory effects against micro-organisms, the present study found that P. harmala showed higher antimicrobial activity. 32 The NADES extract of C. colocynthis showed significant antioxidant and antimicrobial activity with a 27 mm zone of inhibition against E.coli and 23 mm zone of inhibition aganist S.aureus. This higher antimicrobial and antioxidant activity of NADES extract of C. colocynthis was attributed to its higher phenolic and flavonoids contents. In previously study, the methanolic extract of C. colocynthis roots also showed greater antioxidant activity (62.1%). 33 Water extracts of S. alexandrina and P. harmala showed intermediate antioxidant activity (47.6% and 40.8%, respectively) but exhibited no antimicrobial activity against both S. aureus and E.coli strains. This lack of antimicrobial activity was due to the resistance of both Gram-positive and Gram-negative organisms, which prevent the penetration of hydrophilic compounds through their outer cell membranes. 34 Another reason for their lack of antibacterial activity was the low phenolic and flavonoid contents in these extracts. The NADES extract of S. alexandrina demonstrated antioxidant activity (upto 34%) and antimicrobial activity with a zone of inhibition measuring upto 20 mm. In contrast, the water extract of C. colocynthis showed lower antioxidant and antimicrobial activity, which was attributed to its low phenolic and flavonoid contents.
Overall, the results of this study have shown that water extract of C. colocynthis and S. alexandrina suppressed Cotton Leaf Curl Multan viral infection. These extracts possessed bioactive compounds that could be inhibit the viral infections. However, the NADES extracts were found to contain insufficient concentrations of these compounds to effectively inactivate the tested virus. The results of this study indicated that bioactive compounds of C. colocynthis and S. alexandrina could be utilized to develop new therapeutic antiviral agents. This study employed in vitro model to analyze the antiviral capacity potential of tested samples; however, it is recommended to evaluate their antiviral capacity in in-vivo models for further validation and application as antiviral agents.
Conclusions
In the present study, C. colocynthis, P. harmala and S. alexandrina have been evaluated for the presence of potent bioactive components responsible for antiviral activity. Various compounds extracted from these plants have the ability to inhibit different mechanisms of viruses through diverse modes of action including replication, inhibition of the intracellular interaction of virus particles, inhibition of virus infectivity, RNA polymerase, DNA polymerase. The water extracts of C. colocynthis and S. alexandrina have shown potential to inhibit growth of Cotton Leaf Curl Multan virus as compared to their NADES extract. But on the other hand, water and NADES extract of P. harmala did not show antiviral potential. The antiviral activity of C. colocynthis and S. alexandrina is attributed to their higher levels of antioxidant activity and phenolic compounds. This indicates that these herbs have the potential to be used in pharmaceutical industries to enhance endogenous antioxidant system of body by reducing free radical levels. Further clinical studies and experiments are required to validate their use in controlling viral diseases.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241274986 - Supplemental material for Analytical Characterization, Antioxidant, Antiviral and Antimicrobial Potential of Selected Medicinal Plants
Supplemental material, sj-docx-1-npx-10.1177_1934578X241274986 for Analytical Characterization, Antioxidant, Antiviral and Antimicrobial Potential of Selected Medicinal Plants by Faiza Nazir, Shah Nawaz-ur-Rehman, Sumayya Khadim, Sania Amber, Fuad Ameen, Naveed Ahmad, Srimathi Priya Lakshminarayanan and Munawar Iqbal in Natural Product Communications
Footnotes
Acknowledgements
The research work in the present study has been done in the Department of Chemistry, Government College Women University Faisalabad and University of Agriculture Faisalabad, Pakistan. The author would like to thank the lab members of these universities. This research was funded by Researchers Supporting Project number (RSP2024R364), King Saud University, Riyadh, Saudi Arabia
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
This article does not contain any studies with human or animal participants.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Researchers Supporting Project number (RSP2024R364), King Saud University, Riyadh, Saudi Arabia.
Statement of Human and Animal Rights
This article does not contain any human or animal study.
Statement of informed consent and conflict of interest statement:
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. The Corresponding author is submitting this manuscript on the behalf of all other authors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
