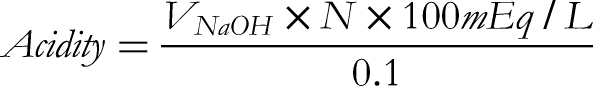Abstract
Introduction
Gastric ulcer is the most prevalent gastrointestinal disorder worldwide, as indicated by Helicobacter pylori infection. 1 Administration of certain types of medications, specifically non-steroidal anti-inflammatory, prominent tobacco consumption, alcohol, and stress, cause interruptions in gastric mucosal barrier defence.2‐4
Ethanol is a necrotizing substance that generates highly reactive free radical species, causing oxidative stress and lesions in the stomach mucosa. 5 These oxygen-derived free radical species are the most common invasive factors responsible for gastric ulceration through oxidative damage to gastric mucosal cells. 6 The gastric mucosa of experimental animals exhibited functional and morphological alterations upon ethanol administration, including aberrant gastric hypersecretion, reduced mucus secretion, and hemorrhagic necrotic foci.7,8
Omeprazole, a proton pump inhibitor (PPI), is a standard medication for gastric ulcers. 9 Despite the anti-ulcer effect, omeprazole has serious adverse effects, posing risks for subclinical hypernatremia, hypocalcemia, and hypomagnesemia. 10 Despite synthetic medications being predominant in treating many human disorders, including gastric ulcers, a substantial fraction of the global population currently opts for medicinal plants or natural substances. 11 Consequently, abundant research on medicinal plants with pharmacological effects is conducted to identify alternative bioactive phytochemicals. 1
Elettaria cardamomum (known as cardamom), a member of the Zingiberaceae family, is indigenous to Sri Lanka and India. Cardamom essential oil (CEO) and extract have several advantageous effects on the gastrointestinal tract. Numerous bioactive substances, including limonene, sabinene, cineol, and terpinene, have been identified in cardamom, which is responsible for its anti-bacterial, antioxidant, anti-inflammatory, and anti-ulcerogenic agent activities.12,13 However, the beneficial effects of cardamom essential oils are limited by their low solubility, low permeability, low bioavailability, uncontrolled volatility, and low storage stability. The integration of essential oils into nanoemulsions can solve these issues. The unique characteristics of nanoemulsions, such as their stability, increased surface area, and nanometric size, improve the effectiveness of pharmaceutical dosage. 14
Nanostructured lipid carriers (NLCs) stand out as highly desirable systems due to their superior encapsulation efficiency, increased colloidal stability (attributed to the higher solid lipid density), elimination of the necessity for organic solvents in their production, reduced likelihood of alterations in particle shape, and robust physicochemical stability. This stability results from the decreased mobility of bioactive substances within their solid matrix compared to a liquid matrix, consequently minimizing the occurrence of bioactive material expulsion during storage. 15 Thus, this study aimed to assess the effects of CEO-NLC supplementation, either as a prophylactic or therapeutic agent, on biochemical variables, redox balance, inflammatory responses, and gastro-protective effects in ethanol-induced gastric ulcers in a rat model.
Materials and Methods
Preparation of CEO-NLC
We followed the method developed by Nahr et al 15 to prepare CEO-NLC. Briefly, CEO-NLC was synthesized using the low-energy nano-emulsification approach in conjunction with high-shear homogenization and sonication. This CEO mixed with olive oil and melted solid lipid (cocoa butter). Then, tween-80 was added dropwise into the lipid phase, while high shear homogenization proceeded at 20000 rpm for 45 min. Consequently, sonication was implemented ten times for one minute each, separated by a one-minute interval. Throughout these procedures, the suspension's temperature was maintained at 50 ± 5 °C. The hot oil-water nanoemulsion mixture was frozen at - 4.0° C to recrystallize the lipid phase.
Characterization of CEO-NLC
The produced CEO-NLC was characterized to determine the particle size, zeta potential (ZP), and polydispersity index (PDI) using Zetasizer Nano ZS (Malvern, UK). The nanocomposites were diluted with Milli Q water (1:10) at 25 °C, and the measurements were done in triplicates by dynamic light scattering (DLS). Additionally, encapsulation efficiency (EE) and drug loading capacity (DLC) of lyophilized CEO-NLC were determined using the ultrafiltration method to measure the amount of free CEO in the NLC that was quantified later using UV-Vis spectrophotometer (Beckman-Coulter, Fullerton, CA, USA) at 280 nm. Then, the EE and DLC of CEO-NLC were calculated using the equations below: 16
EE (%) = [Total amount of CEO − Free amount of CEO] ÷ Total amount of CEO
DLC (%) = Total amount of CEO encapsulated into NLC ÷ [Total amount of lipid used in NLC − CEO formulation].
Experimental Animals
Forty adult male Wistar rats weighing 185–250 g were acquired from the College of Pharmacy, University of Sulaimani, Sulaimaniyah, Iraq and the rats were allowed to acclimatize for one week at a temperature of 25 ± 2° C, humidity of 55 ± 5.0%, and under controlled conditions with 12 h’ light/dark cycles with free access to tap water and a standard pellet diet.
Experimental Design
The animals were randomly assigned to eight groups of five rats. Group 1 rats received 1.0 mL of distilled water (DW) daily for 14 days (negative control). Group 2 rats received a single oral dose of 1.0 mL ethanol 80% to induce gastric ulcer after overnight fasting on day 15 (disease control), while Group 3 received omeprazole (20 mg/kg) for 14 days, while received single oral dose of 1.0 mL ethanol 80% to induce gastric ulcer after overnight fasting on day 15. Group 4 rats received omeprazole (20 mg/kg) with concomitant 1.0 mL ethanol 10% for 14 days (control positive). Group 5 rats received CEO-NLC (300 mg/kg) for 14 days (prophylactic low dose), and Group 6 rats received CEO-NLC (600 mg/kg) for 14 days (prophylactic high dose). Group 5 and 6 were received single oral dose of 1.0 mL ethanol 80% to induce gastric ulcer after overnight fasting on day 15. At the same time, Group 7 rats received CEO-NLC (300 mg/kg) with concomitant 1.0 mL of 10% ethanol for 14 days (therapeutic low dose). Group 8 rats received CEO-NLC (600 mg/kg) concurrent with 1.0 mL of 10% ethanol for 14 days (therapeutic high dose). On the 15th day, the overnight fasted rats were deeply anaesthetized and dissected, and blood was collected from the caudal vena cava while the stomach was removed to examine the test parameters.
Assessment of Gastric Mucosal Lesions
The rat's stomach was isolated and opened along the greater curvature; then, a buffered saline solution was applied to wash gastric tissues. The gastric epithelium revealed stomach lesions in the form of long, reddish bands of wounds. The area of the stomach ulcer was determined by taking images of the ulcers, which typically seemed aligned to the stomach's long axis. The inhibition percentage was estimated for the negative control group:
17
as follows
Measurement of Gastric pH and Volume
Gastric juice was obtained from the rat's stomach and drained into a graduated tube, centrifuged at 4000 rpm for 10 min, and the volume of the supernatant was measured. Then, gastric juice was diluted with 1.0 mL of DW, and pH was measured using a pH meter. 18
Determination of Free and Total Acidity
Titration of diluted gastric juice content with 0.01N sodium hydroxide (NaOH) utilizing Topfer's reagent as an indicator for free acidity and 1% phenolphthalein for total acidity of
Biochemical Parameters
Blood samples were centrifuged at 4000 rpm for 10 min; serum was obtained and used to estimate gastrin, inflammatory biomarkers (IL-6 and IL-10), lipid peroxidation (MDA), total antioxidant capacity, and TNF-α using an ELISA kit (Bioassay Technology Laboratory, UK), corresponding to the manufacturer's guidelines.
Histotechnique Protocol
Collected gastric tissues were cleaned and washed with standard saline solution and fixed for 48–72 h in 10% buffered formalin. Afterwards, tissue sections were immobilized in tissue cassettes and then dehydrated in xylene in three steps. Next, a paraffin-embedded tissue block was prepared and sectioned to 5.0 µm using a rotary microtome, then dried and fixed on glass slides. Later, the slides were deparaffinized for 30 min with xylene and dehydrated for 5 min with a hot plate. Then, sections were stained with Harris's hematoxylin and Eosin and read under a light microscope (NOVEL XSZ-N107 T, China).
Semi-Quantitative Lesion Scoring
Image analyzer software assessed lesion scoring semi-quantitatively (AmScope, 3.7). Overall, mucosal erosion was evaluated and estimated in the percentage of calculated erosional length and depth from erratically selected 10 fields in µm from the given mucosal section. In contrast, inflammatory exudate and the area of edema were assessed in µm and evaluated as a mean percentage. Necrotic debris and inflammatory cells were enumerated in ten fields under elevated power magnification (100×), and the mean percentage was assessed. Eventually, the mean of morphometric values was exhibited as a lesion scoring-grading system (0-10% as no lesions; 10-25% as mild; 25-50% as moderate; 50-75% as severe; and 75-100% as critical).
Statistical Analysis
The data was expressed as mean ± standard deviation (SD), and statistical analysis was accomplished using GraphPad Prism software (Version 8). One-way ANOVA was used to compare between groups, followed by Tukey's multiple comparison tests. Unpaired t-tests were used to compare each group with the positive control group. The results were considered statistically significant when the p-value was <0.05.
Results
Preparation and Characterization of CEO-NLC
The prepared CEO-NLC appeared as translucent, light yellow color due to the nature of the CEO (dark yellow). Then, characterized CEO-NLC had a mean PS of 150.13 ± 15.25 nm, ZP of −12.30 ± 1.9 mV, and PDI of 0.449 ± 0.45 (Table 1). Meanwhile, the EE and DLC of CEO-NLC were 91.95 and 4.94% respectively.
The Summary of Characterization of Blank and Loaded Nanoparticles.

Abbreviations: CEO, Cardamom essential oil; NLC, Nanostructured lipid carrier.
Prophylactic/Therapeutic Effects of CEO-NLC on Gastric Juice Parameters/Gastrin Level
The gastric volume significantly decreased in the low and high doses of CEO-NLC for prophylactic and therapeutic groups compared to the positive control group. Nevertheless, the pH of the gastric juice significantly increased in the omeprazole group (p < 0.001), prophylactic low dose group (p = 0.002), prophylactic high dose groups (p < 0.000), therapeutic low dose group (p < 0.000), and the therapeutic high dose group (p < 0.000) in comparison with the positive control group. CEO-NLC administration, either at prophylactic or therapeutic doses, causes a significant decrease in gastric free and total acidity compared to the positive control group (p < 0.01). Concerning the effect of CEO-NLC on serum gastrin levels, the results declare a significant reduction in the therapeutic low-dose group (p = 0.001), therapeutic high-dose group (p = 0.000), prophylactic low-dose group (p = 0.000), and prophylactic high dose group (p = 0.002). Furthermore, the omeprazole group exhibits a reduction in gastrin level (p = 0.005) compared to the positive control group (Table 2).
Prophylactic and Therapeutic Effects of CEO-NLC on pH, Volume, Free Acidity, Total Acidity, and Gastrin Levels in the Studied Animals.

Values are expressed as mean ± SD. There are 5 samples in each group (n = 5).
Abbreviations: CEO-NLC, Cardamom essential oil-loaded nanostructured lipid carrier; DW, Distilled water; EtOH, Ethanol.
*: Significant difference (p < 0.05), **: Significant difference (p < 0.01), ***: Highly significant difference (p < 0.001), and ****: Very highly significant difference (p < 0.0001) compared to the positive control group.
Prophylactic/Therapeutic Effects of CEO-NLC on serum Inflammatory Biomarkers
Treatment with CEO-NLC significantly decreases the levels of IL-6 in the prophylactic low-dose group (p = 0.002), prophylactic high-dose group (p = 0.036), therapeutic low-dose group (p = 0.005), and the therapeutic high-dose group (p = 0.044) (Figure 1A). The TNF-α levels were also significantly lower in the prophylactic high-dose group (p = 0.043), the therapeutic low-dose group (p = 0.010), and the therapeutic high-dose group (p = 0.006) (Figure 1B). Conversely, IL-10 levels were elevated significantly in the prophylactic high dose group (p = 0.026), therapeutic low dose group (p = 0.009), and the therapeutic high dose group (p = 0.000) compared to the positive control group (Figure 1C).
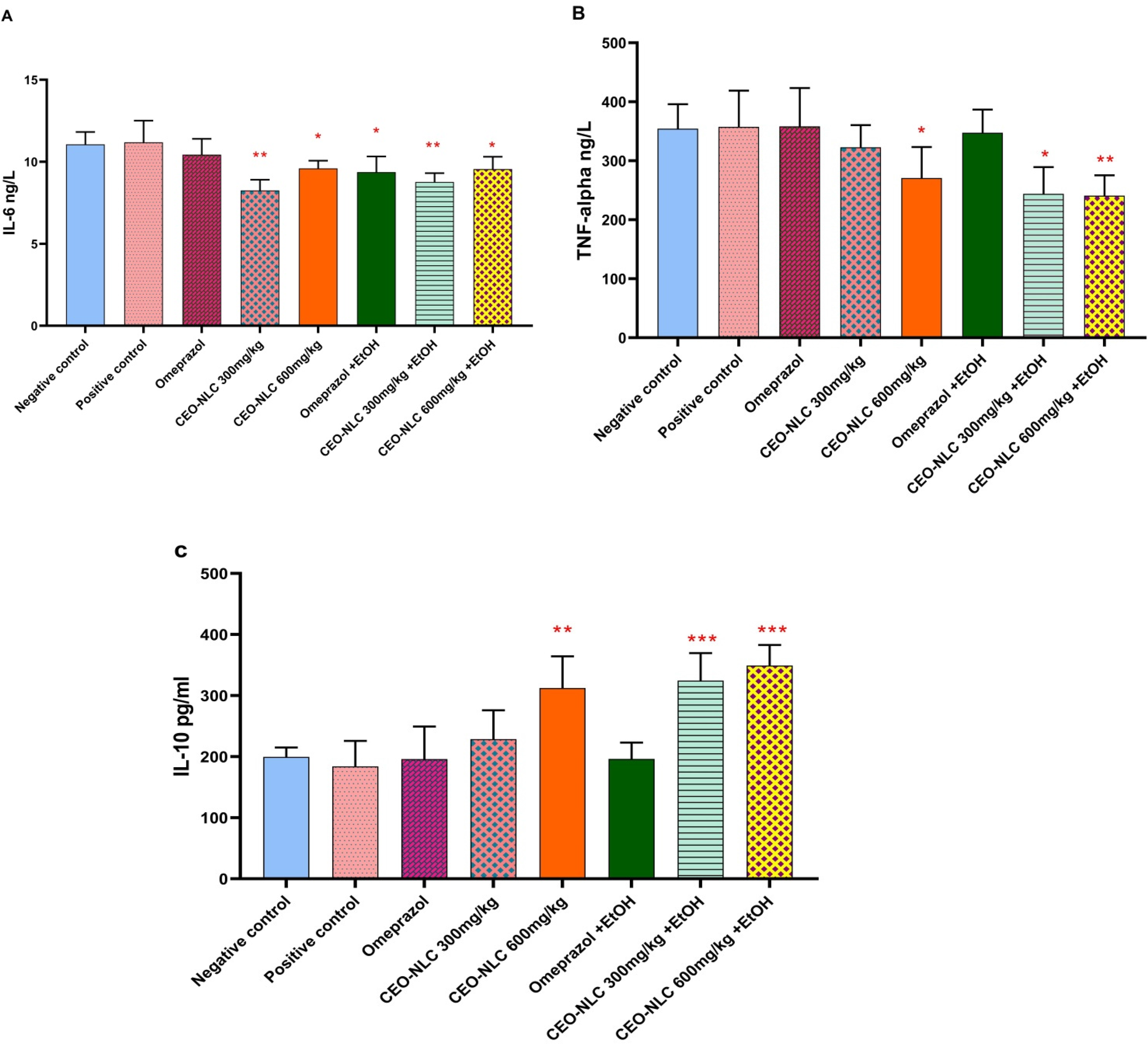
Prophylactic and therapeutic effects of CEO-NLC on serum levels of inflammatory biomarker
Prophylactic/Therapeutic Effects of CEO-LC on Oxidative Stress/Antioxidant Capacity
Administration of CEO-NLC alleviated antioxidant status at both low and high doses, either as prophylactic or therapeutic agents. Total antioxidant capacity exhibited a significant augmentation in the negative control (p < 0.030), prophylactic low dose (p = 0.003), prophylactic high dose (p = 0.003), therapeutic low dose (p = 0.001), and therapeutic high dose (p < 0.000) groups compared to the positive control group. In addition, MDA levels exhibited a significant regression in the prophylactic low dose (p = 0.005), prophylactic high dose (p = 0.005), therapeutic low dose (p = 0.001), and therapeutic high dose (p = 0.001) groups compared to the positive control group (Figure 2B).
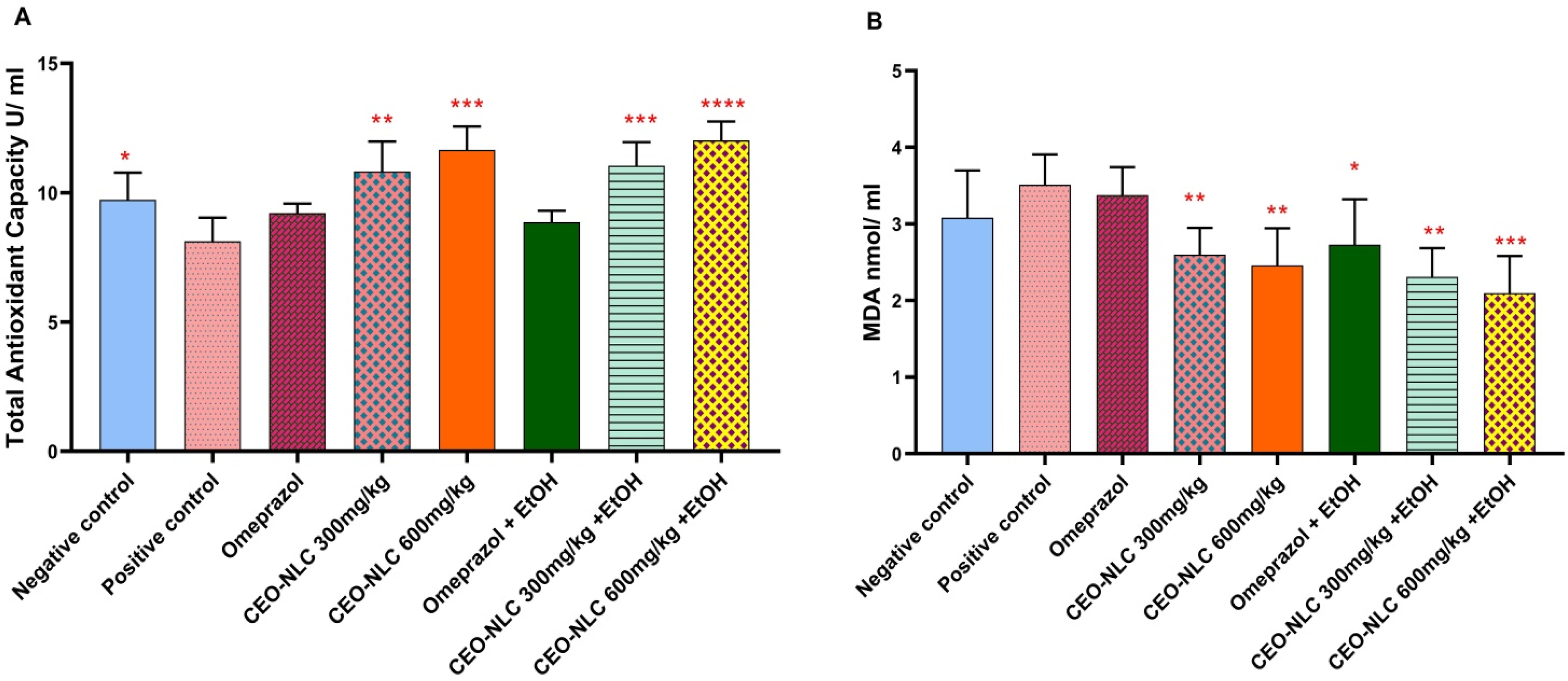
Prophylactic and therapeutic effects of CEO-NLCs on serum levels of A: Total antioxidant capacity, B: Malonaldehyde (MDA). CEO-NLC: Cardamom essential oil-loaded nanostructured lipid carrier, EtOH: Ethanol. *: Significant difference (p < 0.05), **: Significant difference (p < 0.01), ***: Highly significant difference (p < 0.001), and ****: Very highly significant difference (p < 0.0001) compared to the positive control group.
Gross Evaluation of Gastric Lesions
By macroscopic analysis of the stomach, administration of the CEO-NLC as prophylactic or therapeutic doses demonstrated a significant reduction in the gastric ulcerated area compared to the positive control group (Figure 3).

Photograph of the animal stomach reveals preventive effect of CEO-NLC on gastric ulceration induced by ethanol (etOH). (A) Negative control, (B) Positive control, (C) Omeprazole group, (D) Omeprazole + EtOH, (E) CEO-NLC (300 mg/kg), (F) CEO-NLC (600 mg/kg), (G) CEO-NLC (300 mg/kg) + EtOH, and (H) CEO-NLC (600 mg/kg) + EtOH.
Histopathological Evaluation
The semi-quantitative morphometric evaluation of lesion scoring protocols in prophylactic and therapeutic measures shows favour of the first agenda over the second one. Within both protocols and concerning the positive control group, oral feeding of 1.0 mL 80% ethanol induced severe damage and critical erosion to the gastric mucosa within the first few hours of administration, evident by acute mucosal sloughing, accumulation of edematous fluid mixed with inflammatory exudates and necrotic debris together with substantial infiltration of inflammatory cells (Figure 4 and Figure 5). Furthermore, all treatment groups showed significant (p < 0.05) mitigation and improvement in the lesion severity according to the doses and experimental groups. Animals in the prophylactic protocol prove a significant alleviation in the lesion severity from critical to moderate in both omeprazole (20 mg/kg) and the high dose CEO-NLC, respectively, compared to the positive control group, described by a significant reduction in the number of inflammatory exudates along with apparent regeneration to the severed mucosal area. Meanwhile, gastric sections of CEO-NLC (low dose) reveal a significant reduction in lesion severity compared to the positive control (Table 3).

Photomicrograph of stomach from negative control group (G1), received DW daily for 14 days, demonstrate no prominent morphological modifications, apparent by intact and usually organized gastric mucosal layers (M) with classically settled gastric glands (G), regular submucosal layer (SM) with inner and outer layers of muscularis smooth muscle fibers (ME). Positive control group (G2), received daily DW for 14 days, then treated with 1.0 mL of 80% ethanol at day 14, display the presence of strong and significant mucosal erosion and ulceration (ME and MU) together with the presence of deep purple necrotic debris combined with diffusely distributed pinkish inflammatory exudates (yellow arrows). Diffuse and haphazardly infiltration of inflammatory cells (IC) within the submucosal layer (SM) which contain clear light pink edematous fluid. G3 received 20 mg/kg omeprazole for 14 days, then 1.0 mL of 80% ethanol at day 14, demonstrate the existence of deep purplish necrotic debris (ND) at the area of erosion blended with profound and mixed diffuse inflammatory cells (yellow arrows) and inflammatory exudates. The submucosal layer (SM) and the muscle layers of muscularis externa (ME) show low grade of exudate and hyperplasia respectively. G4 received 300 mg/kg of CEO-NLC for 14 days, then 1.0 mL of 80% ethanol at day 14 show significant mucosal erosion (ME) with the presence of light-purplish inflammatory exudates (yellow arrows) mixed with friable mucosal necrotic debris (ND). The inflammatory exudates together with light pinkish edematous fluid also perceived within the submucosal layer (SM), together with obvious thickening of the muscular externa (ME). G5 received 600 mg/kg of CEO-NLC for 14 days, then 1.0 mL of 80% ethanol at day 14 show the existence of significant inflammatory exudate within the mucosal surface (yellow arrows) mixed with moderately graded necrotic debris. Areas of erosion (ME) can be clearly observed within the mucosal surface, the submucosa (SM) show low grade of inflammatory exudate with many dilated blood vessels. H&E staining. Scale bar: 4.0 mm.

Photomicrograph of the animals’ stomach from Negative control group (G1) that received DW daily for 14 days, exhibits no seeming lesions, evident by undamaged and typically appeared gastric mucosal layers (M) with generally organized gastric glands (G), normal submucosa loose connective tissue (SM) with slight and non-significant edema, and normally looked doubled layers of muscularis external smooth muscle (ME). Positive control group (G2) received daily DW for 14 days, then treated with 1.0 mL of 80% ethanol at day 14, demonstrate the presence high-graded and significant mucosal ulceration (MU), in addition to a diffuse infiltration of inflammatory cells (yellow arrows) combined with profound eosinophilic inflammatory exudates (IE) within an expanded-edematous submucosal layer (SM). The section also shows significant area of angiogenesis together with evidently dilated blood vessels (BV). G3 received 20 mg/kg of omeprazole together with 0.1 mL ethanol concomitantly for 14 days, reveal the presence of clear mucosal ulceration (MU) together with the existence of basophilic necrotic debris (ND) combined with inflammatory exudates (yellow arrow) at the mucosal surface. The submucosal connective tissue (SM) appears pinkish with many dilated blood vessels and the muscularis externa (ME) show significant muscular hyperplasia. G4 received 300 mg/kg of CEO-NLC together with 1.0 mL of 10% ethanol concomitantly for 14 days demonstrate the presence of significant mucosal ulceration (MU) with clear inflammatory exudates (yellow arrows) deposited at the superficial layers, moreover, the collagenous layers of submucosa (SM) appear deep pinkish due to a significant proliferative feature, together with a significant hyperplasia and thickening of the muscularis externa (ME). G5 received 600 mg/kg of CEO-NLC together with 1.0 mL of 10% ethanol concomitantly for 14 days. Area of mucosal ulceration (MU) mixed with purplish inflammatory exudates (yellow arrows) are seen. H&E staining. Scale bar: 4.0 mm.
Morphometric Semi-Quantitative Assessment of Gastric Tissue Sections (Prophylactic Measure).

Values are expressed as mean percentage (%). #: Statistical comparison among groups: Mean with different capital letters has significant differences at p < 0.05.
On the other hand, daily administration of ethanol as an ulcer induction protocol, together with a therapeutic medication for 14 days with both low and high doses of CEO-NLC in addition to ordinary antacid omeprazole (20 mg/kg), demonstrates a significant reduction in lesion scoring severity compared to control positive group. However, the improvement needs to be marked as shown in preventive measures. Thus, treatment groups in the preventive experiment show a significant improvement in lesion scoring compared to other treatment groups in the therapeutic design. This indicates that daily supplementation of CEO-NLC significantly protects the gastric mucosa from ulcerative damage after a single dose of ethanol. The prophylactic regimen has a better protective effect than the concomitant administration with ethanol for 14 days (Table 4).
Morphometric Semi-Quantitative Assessment of Gastric Tissue Sections (Therapeutic Measure).

Value expressed as mean percentage (%). #: Statistical comparison among groups: Mean with different capital letters has significant differences at p < 0.05.
Discussion
In this study, oral administration of ethanol was used to induce gastric ulcer that causes expansive disruption of the stomach mucus barrier manifested by hemorrhage and erosion.20,21 Various mechanisms may be responsible for this induction, including reduced mucus production and endogenous enzyme concentrations. Furthermore, ethanol promoted lipid peroxidation and enhanced microvascular permeability. Additionally, ethanol increased the stomach epithelium's capacity to generate free radicals, which severely damaged the stomach epithelium.22,23
The positive control group treated with ethanol detects an immense ulceration grade. The macroscopic and histopathological analysis declared severe bleeding, which is manifested as severe vascular congestion and red blood cell infiltration extended between mucosal and submucosal layers of the stomach tissues. These results describe the toxicity of ethanol in reducing the coagulopathy mechanisms that cause continued bleeding. 24
Ethanol consumption might cause blood flow stagnation and disruption of the gastric micro-vessels, resulting in spacious gastric damage.25,26 The ulcer index was significantly decreased by pretreatment of the animal's group with CEO-NLC at both doses (300 and 600 mg/kg) in contrast to the positive control group (ulcerated group). Furthermore, the pre-treated animals group with CEO-NLC exhibited a moderate reduction in ulcer index compared to the omeprazole standard drug, suggesting that CEO-NLC could be relevant in gastric ulcer treatment. This result is in line with this study. 27
This study illustrates that treatment with CEO-NLC in two different doses results in a significant increase in gastric pH levels and concomitant decreases in total acidity and gastric volume as compared to the positive control group, indicating a protective effect of CEO-NLC on gastric mucosa by hindering gastric acidity. This finding is in line with these studies.28‐30 A decreased gastric juice pH and excessive hydrogen ion concentration are acidic factors that accelerate stomach injury. 31
Our finding demonstrated that treatment with CEO-NLC had comparable effects on gastric pH as the omeprazole reference drug, the most often used PPI for reflux esophagitis, duodenal ulcers, gastric ulcers, and gastro-esophageal hyperacidity, which has a great potential to reduce stomach acidity. 32
Furthermore, our study showed that the pretreatment of the animal group with CEO-NLC resulted in a substantial decline in serum gastrin levels, which has an analogous effect to omeprazole in contrast to the positive control group. Gastric ulcer and H. pylori infection typically elevated serum gastrin levels. 33 The polypeptide hormone gastrin, generated by gastrin cells, is essential for regulating acid secretion and cell proliferation. 34 Increased gastrin levels cause the stomach to secrete more acid than usual. Additionally, the elevated gastrin causes hyperplasia of the parietal cell through trophic action due to increased acid secretion, leading to gastric metaplasia of the duodenal bulb and, ultimately, the development of ulceration. 35
One of the distinctive characteristics of gastric ulcers is the inflammatory reaction, which induces damage to the gastric mucosal by causing macrophages and leukocytes to migrate into the ulcerated area and the surrounding areas. 36 TNF-α, IL-6, and IL-10 are the dominant inflammatory cytokines released during inflammation. In the current study, Pretreatment with CEO-NLC expressed a significant decrease in the level of TNF-α and IL-6, with an increase in IL-10 levels compared to the positive control group. This finding agrees with medicinal plant studies.29,30,37,38
Gastric ulcers emerge when the balance between protective and aggressive factors is interrupted. 39 Oxidative stress is essential in causing numerous diseases, particularly gastric ulcers. Another crucial outcome of reactive oxygen species (ROS)-induced oxidative stress is lipid peroxidation. 40 The gastrointestinal damage caused by ethanol is partially attributed to lipid peroxidation and ROS. MDA is considered a lipid peroxidation biomarker that evaluates and identifies oxidative stress. 41 Previous studies declared a marked elevation in MDA concentration and reduced total antioxidant capacity levels in rats’ administrated ethanol. Nevertheless, the current study's findings demonstrated that CEO-NLC administration substantially increased total antioxidant activity and decreased MDA levels, critical in preventing gastric ulcers. Comparably, numerous researchers have published many studies utilizing medicinal plants, revealing decreased MDA levels.29,30,37,38
The current study had several limitations, including a small animal house, sample size, and facility shortage. Therefore, further studies using a larger sample size and advanced laboratory tests are required to corroborate gene expression.
Conclusions
Treatment with different doses of CEO-NLC exhibited gastro-protective benefits against ethanol-induced gastric mucosa injury, which may be related to its antioxidant properties and capacity to raise mucus secretion, lower MDA levels, and modify inflammatory cytokines levels. Considering these findings, CEO-NLC might have been utilized as a potential prophylactic and therapeutic intervention for managing gastric ulcers.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study protocol was approved by the Ethical Committee of the College of Pharmacy at the University of Sulaimani, Sulaimaniyah, Iraq (No. PH92-23 on April 10, 2023).
Statement of Human and Animal Rights
The animals were managed according to the guidelines of the Canadian Council on Animal Care (CCAC).
Statement of Informed Consent
Not applicable