Abstract
Introduction
Dental caries, one of the most prevalent infectious diseases in the world, is a biofilm-mediated, sugar-driven, multifactorial dynamic disease.
1
Biofilm is a single or multispecies microbial community attached to the surface of teeth and wrapped in the self-produced matrix.
2
The formation of dental plaque biofilm is one of the important causes of dental caries.
3
The biofilm formation process will produce anaerobic and acidic environment, leading to the formation and development of dental caries.
4
Therefore, the eradication of dental plaque biofilm and the control of acid production by bacteria are effective ways to control dental caries.
5
Natural products, especially plant essential oils, are suitable leads for antimicrobial drugs that can inhibit bacterial biofilms.
Materials and Methods
Bacterial Strain and Growth Condition
Determination of Minimum Inhibitory Concentration (MIC)
The MIC was calculated in triplicate on 96-well microtiter plates using the broth-based microdilution technique described before. 30 In a 96-well microtiter plate, 10 stages of 2-fold successive dilutions of the OEO were carried out before sucrose-supplemented BHI medium had been added to the wells. The microtiter dishes were then inoculated with a predetermined amount of cultured bacteria and left to grow for 24 h.
Growth Curves Assay
As previously published,
31
a growth curves experiment was performed to investigate
Bacteria Colony Forming Units Count
After varied incubation periods (0, 12, and 24 h) with the OEO ranging from 0.0625 to 0.5 mg/mL, the bacteria colony-forming unit (CFU) was determined. A total of 100 μL of the bacterial sample was grown onto BHI agar plates for a further 2 days using 10-fold successive dilutions of both untreated and treated samples. CFUs were determined and recorded, to ascertain the amount of viable bacteria under the bactericidal action of the OEO.
Fluorescence Staining
The bacterial membrane integrity was assessed by 4′,6-diamidino-2-phenylindole (DAPI), and Nile Red (NR) staining.
Transmission Electron Microscopy (TEM)
TEM was used to explore the influence of OEO on bacterium cellular morphology, as reported by Duan et al. 32 Regarding the mature biofilm experiment, confocal dishes (BS-20-GJM, Biosharp) were first filled with overnight cultured bacteria diluted in BHI broth, and the plates were subsequently left to incubate at 37 °C for 24 h in order to generate mature biofilms. In brief, OEO was introduced into the BHI medium in increments of 0.0625 to 0.5 mg/mL for 24 h at 37 °C. As controls, groups with no OEO were employed. The microorganisms were gathered in a 1.5 mL centrifuge container and fixed for 6 h at 4 °C in 200 to 400 μL of 2.5% (v/v) glutaraldehyde. Following fixation, samples were washed 3 times with PBS and centrifugation after each wash. The supernatant was eventually removed after being centrifuged (8000 r/min for 3 min). Gradient dehydration was performed for 10 min using ethanol concentrations of 30%, 60%, 70%, 80%, 90%, and 100% (v/v). Subsequently, the mesh copper grid was filled with 2 or 3 drops of the suspensions and allowed to dry. Using a field emission electron microscope with transmission (Talos F200S, Fei), the sample morphologies were studied.
Scanning Electron Microscopy (SEM) Analysis
Following the method presented,
33
SEM was used to study the physical characteristics of
Confocal Laser Scanning Microscope (CLSM) Analysis
The impacts of OEO on constructed
Crystal Violet Assay
Following biofilm development, the biomass of the biofilm was estimated using crystal violet staining. 35 Regarding the mature biofilm experiment, 24-well plates were first filled with overnight cultured bacteria diluted in BHI broth (Guangdong Huankai Microbial Sci. & Tech. Co., Ltd), and the plates were subsequently left to incubate at 37 °C for 24 h in order to generate mature biofilms. In short, the biofilm was allowed to air dry and the supernatant was disposed of after the biofilm was fixed on 24-well plates using methanol for 15 min. After that, each well received 100 μL of 0.1% crystal violet, which was then incubated. The biofilm was then washed with sterilized ionized water and left to air dry. In order to do a quantitative study, the biofilm's crystal violet stain was dispersed in a 33% acetic acid solution, and the absorbance at 595 nm was quantified by a Flash microplate reader (SuPerMax 3100).
Bacteria Metabolic Assay
The MTT (methyl thiazolyl tetrazolium) assay is effective for determining bacterial metabolic activity by using the colorimetric technique. Mature biofilms were grown as described in the above section. The procedure was carried out as previously reported, with various adjustments. 36 Following OEO incubation, the growth medium was gently removed, washed 3 times with sterile PBS, and replaced with 100 L of MTT (0.5 mg/mL, Biosharp) for 3 h in the dark. After washing the wells with PBS, the tetrazolium precipitates in the biofilm were extracted using 150 L dimethylsulfoxide (DMSO). The absorbance of the sample was determined at 450 nm using visible spectrophotometers (721, Shanghai Jinghua Technology Instrument Co., Ltd) after 10 min on an orbital shaker.
Extracellular Polysaccharides (EPS) Production Assay
The approach used to assess the influence of the OEO on EPS was described in the published research. 37 Mature biofilms were grown as described in the above section. In brief, 100 L of the suspension of bacteria and 1 mL BHI solution were combined and cultivated for 24 h at 37 °C with or without various doses of the OEO ranging from 0.0625 to 0.5 mg/mL. The supernatant dissolved in NaOH (0.1 M) was collected by centrifugation. These supernatants were mixed, and overnight at 4 °C, on 3 separate occasions, a volume of 95% ethanol was added. The mixture was centrifuged for 30 min at 4 °C at 12 000×g, and the resultant precipitate was insoluble in water EPS. To assess the content of the precipitate, it was dissolved in NaOH. The phenol-sulfuric acid technique was used to detect the concentration of water-insoluble EPS at the wavelength of 492 nm by an ultraviolet-visible (UV–vis) spectrophotometer (TU-1950, Purkinje General Instrument, Beijing).
Acid Production Measurement
The pH measurement represents the capacity of the
Extracellular DNA Isolation and Interactions
Since EPS and eDNA are crucial for bacterial biofilm defense, the interaction of OEO with eDNA is a useful technique for breaking down mature biofilm. eDNA was isolated using a procedure described in a published study.
39
Sonication is in a bath of ice water for 5 min dispersed the mature
Chemical Components
Using a mass spectrometer (5977B, Agilent) and gas chromatography (7890A, Agilent), the chemical composition of OEO was examined using the methodology outlined. 40 The following were the chromatographic conditions: The temperature program ramps from 60 °C to 300 °C with a gradient of 5 °C/min (holding the beginning and final temperatures for 5 min); the injector and detector temperatures are 260 °C and 280 °C, respectively. The sample preparation is 1 mL in 1 mL of ethanol; the injection volume is 0.4 μL; the split ratio is 120:1; the helium flow rate is 1.6 mL/min. By comparing volatile molecules’ retention indices (RI) to those of n-alkanes (C6-C19), volatile chemicals were found. To qualitatively examine unknown substances, the chemical workstation's NIST 14 standard mass spectrometry library was searched for the spectrum peak. Peak area normalization was used to compute each component's relative content.
Cytotoxicity
The cytotoxicity of OEO on Human Oral Epithelial Cells (HOEC, NEWGAINBIO) was determined using the CCK-8 assay according to the manufacturer's directions (Cell-Counting-Kit 8, Biosharp). The cells were grown in DMEM (Gibco, USA) with 15% FBS (Excell Bio, Uruguay) at 37 °C in 5% CO2. The cells were grown at a concentration of 5000 cells per well on a 96-well plate. The OEO concentrations ranged from 0.0625 to 0.5 mg/mL. The group serving as the negative control received the same supply of PBS as the positive control group. After 1 h of incubation at 37 °C in 5% CO2, the cells were rinsed with sanitized PBS, and CCK-8 solution was applied to each well. After 1 h of incubation at 37 °C, the absorbance at 450 nm was measured with a reader for micro-plates (Labserv K3, Fisher Scientific). The OD of pretreated control cells was deemed to represent 100% vitality.
Statistical Analysis
Three duplicates of each experiment were run, and the results were reported as means ± deviations from the average. The IBM SPSS 25.0 software (Armonk, NY) was used for the statistical analyses. One-way ANOVA with Waller-Duncan's multiple range test (
Results
Antibacterial Activity
When compared to the control group, the OEO demonstrated substantial inhibition at doses > 0.125 mg/mL and dose-dependent antibacterial action against
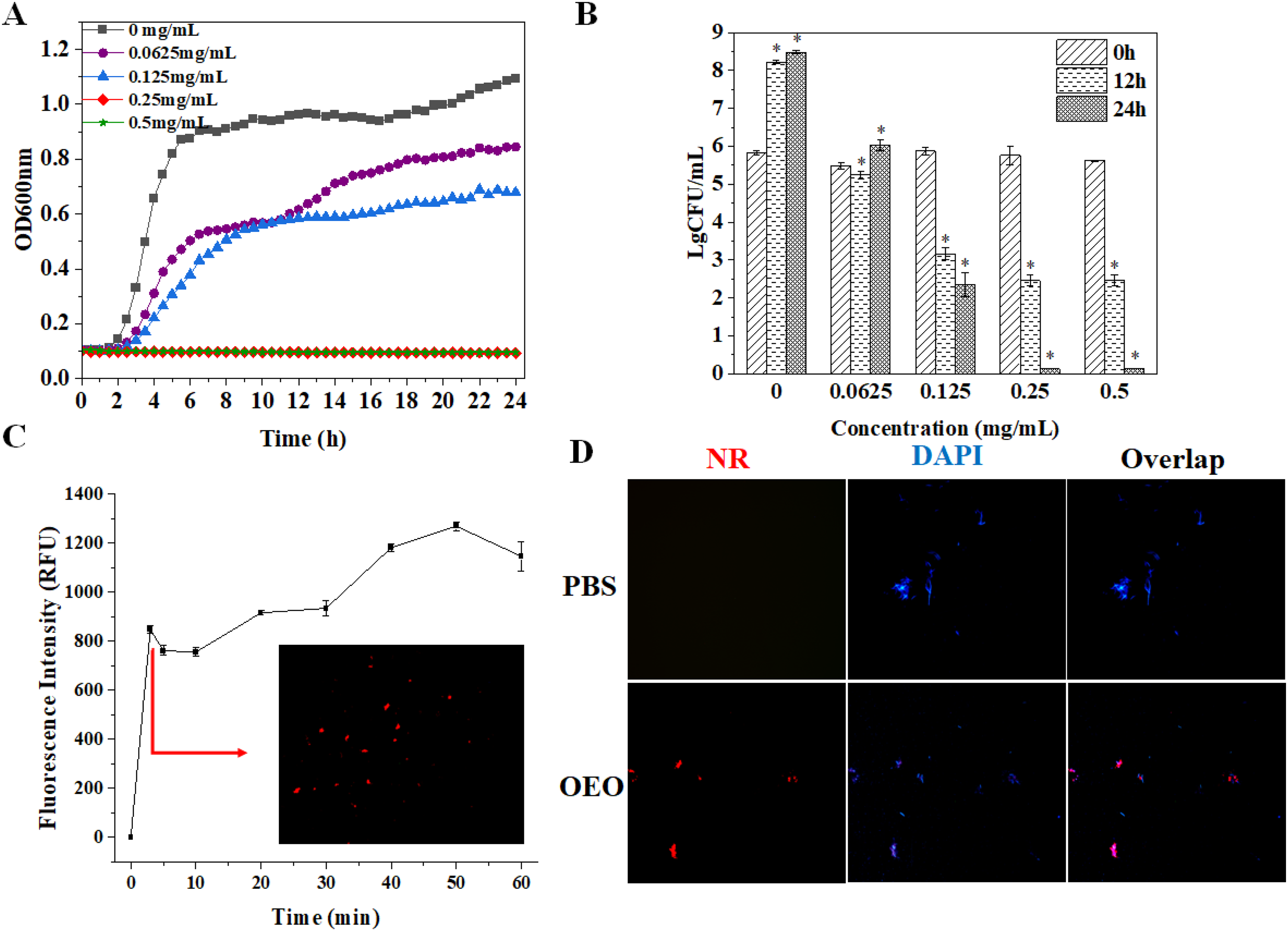
Antibacterial activity with different concentrations of OEO: (A) growth curves; (B) CFU of
Transmission Electron Microscopy
Figure 2A demonstrates that without the OEO treatment, the cell surface of

Transmission electron microscopy (TEM) micrograph of
Scanning Electron Microscopy
SEM was used to assess how the OEO affected the amounts of bacteria present and the surface shape. The biofilm produced by

Scanning electron microscopy (SEM) micrograph depicting the effect of the OEO on biofilm formation: (A) 0 mg/mL, (B) 0.0625 mg/mL, (C) 0.125 mg/mL, (D) 0.25 mg/mL, and (E) 0.5 mg/mL.
Confocal Laser Scanning Microscope
As shown in Figure 4, a series of dimensional micrographs were created by merging the pictures using Zeiss Zen software. The living bacteria were colored green and dead organisms were labeled red to further ascertain the impact of the OEO on mature biofilm. The CLSM analysis supported the experimental findings on the OEO's ability to suppress biofilm formation, as shown in Figure 4. When the quantity of OEO was increased from 0.0625 to 0.5 mg/mL, the amounts of dead bacteria in the groups that were treated increased significantly in comparison to the control group. Additionally, as indicated in Figure 4F, the percentage of living and dead bacteria in the

The CLSM images of
EPS Production and pH Decrease
Figure 5A depicts the inhibitory impact of OEO on water-insoluble EPS. In comparison to the control group, the water-insoluble EPS produced by

The effect of OEO on
Oregano Essential Oil of Anti-Biofilm Mechanism Analysis
The developed biofilm by
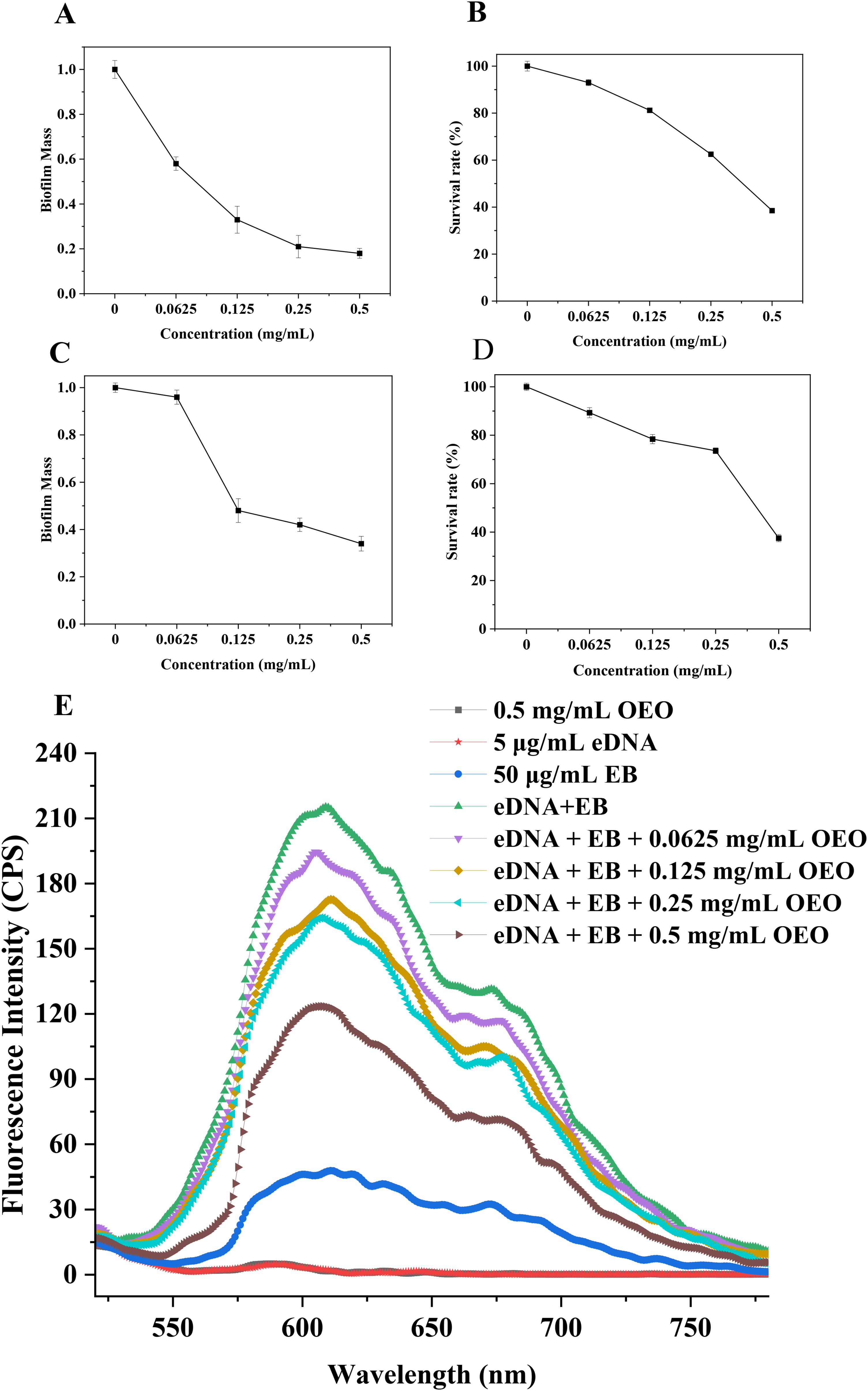
Anti-biofilms of the OEO to inhibit
Chemical Components Analysis
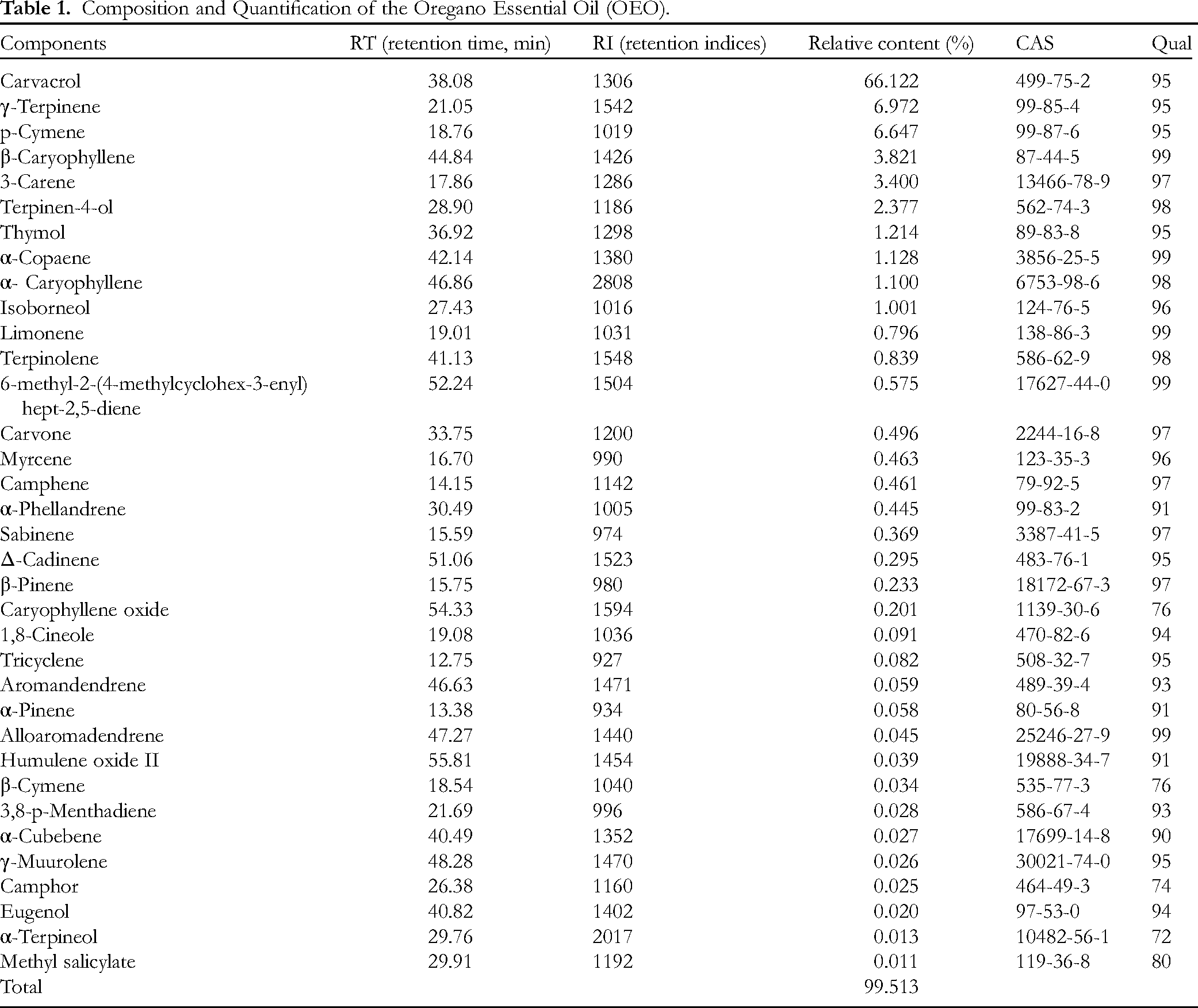
Forty volatile chemicals were found in the OEO using GC-MS technology in conjunction with the retention indices; these compounds made up 99.629% of the entire contents. Table 1 shows that the most common chemical by identification and quantification was carvacrol (66.121%), followed by γ-terpinene (6.972%), p-cymene (6.647%), β-caryophyllene (3.821%), 3-carene (3.400%), and terpinen-4-ol (2.377%). Other chemicals were detected in trace amounts, including isoborneol (1.001%), α-copaene (1.128%), thymol (1.214%), and α-caryophyllene (1.100%). In OEO, there are still 30 volatiles with extremely low contents, each with < 1% of substance.
Composition and Quantification of the Oregano Essential Oil (OEO).
Cytotoxicity
HOEC were subjected to OEO dosages that varied from 0.0625 to 1 mg/mL to investigate the cytotoxicity of OEO (Figure 7). The toxicological potential rose with increasing dosage during our incubation period, although OEO did not significantly limit HOEC growth from 0.0625 to 0.5 mg/mL. On the other hand, HOEC viability was damaged by OEO at 1 mg/mL, and this was different from the control group. However, it's worth noting that 0.5 mg/mL demonstrated strong bactericidal activity to

Cytotoxicity of the OEO to the HOEC. Cell viable rate of the HOEC under the OEO based on CCK-8. Data represent the mean ± standard deviation of 3 independent tests performed in triplicate. Significant differences (*
Discussion
Tests were conducted on
The OEO often damages cellular membranes, which interferes with the bacteria's ability to function normally. This causes the bacterium to lose cytoplasm and vacuolate, which finally results in its death. The OEO can permeate the cell wall and alter the cytoplasmic membrane's structure since it is hydrophobic.
42
According to the TEM results, the OEO changed the morphology of the cell surface and obstructed the cell membrane. In accordance with earlier studies, changes to the shape of the cell membrane may have an impact on the physiological and metabolic functions of the cell, perhaps halting the growth or death of the cell.
Usual bacteria had intact, uniform surfaces and a catenulate shape, while the OEO-treated cells rapidly lost their usual morphology and were lysed. However, notable surface structure alterations were seen in both OEO-treated and -untreated bacteria, with the changes being more pronounced at increasing the OEO concentrations. These surface structure changes coincided with significant variations in the ultimate biomass of the bacterial biofilm. In terms of CFU and growth curve, this result confirmed the antibacterial activity of OEO and is consistent with the previous study's findings (Figure 1). Higher OEO concentrations were associated with an increase in dead bacteria. These pictures showed that the OEO-treated group's inhibition of planktonic bacterial proliferation reduced the amounts of bacterial cells that accumulated in the biofilm. At the same time, after treatment with OEO, the biofilms seemed looser and more scattered in contrast to the control group, which showed a thick and compact structure (Figure 4A).
The water-insoluble extracellular polymer generated by
To provide more insight into how OEO affects other cariogenic traits, we also investigated the effect of OEO on acid production in
Besides regulating EPS and acid production, OEO exerted anti-biofilm effects through multiple mechanisms. The intensity of the fluorescence was greatly reduced with the introduction of OEO, and this reduction was contingent upon the concentration of OEO. According to these findings, OEO may interact with the eDNA of the biofilm by integrating into its duplex area and upsetting the structure of the double helix. The anti-biofilm results corresponded with the CLSM and SEM pictures (Figures 3 and 4). SEM and CLSM measurements of the OEO treatment showed a considerable decrease in the number of microbial cells sticking to the substratum, indicating that it had an impact on biofilm inhibition. Furthermore, a slower rate of population growth in biofilm-adhered cells may be the cause of the reduced biofilm matrix in OEO-treated circumstances. These findings showed that OEO can effectively limit biofilm formation by reducing bacterial viability. 46 OEO might prevent biofilm development by lowering EPS and acid generation during the first adhesion period. OEO might weaken the stiff structures of mature biofilms and eventually destroy them.
The antibacterial action of the OEO was related mostly to the presence of volatile chemicals, particularly carvacrol and thymol, which were shown to be responsible for the OEO's antimicrobial function as separate molecules. 47 However, many intrinsic and extrinsic factors such as agricultural factors, species, harvest time, plant part, season, climate, geography, extraction methods and storage care influence the composition of the OEO and concentration of each volatile compound, majority, or minority. Because OEO has a variety of antibacterial substances, it has a scavenging effect on mature biofilm in this paper. The anti-biofilm mechanism of OEO is probably caused by the binding of small molecular substances to the proteins related to the synthesis of biofilm, which can be investigated by computer molecular simulation software in the future. 48 In the meantime, the cytotoxicity experiment of the OEO showed that OEO was well-biocompatible when used orally in humans, suggesting that it may be turned into anti-carcinogenic therapies and eventually eliminated.
In terms of the limitation of the study, there is no in-depth target research on the interaction between OEO and virulence factor-related enzymes in the anti-biofilm of
Conclusion
To summarize, the current study discovered that OEO is anti-microbial to
Footnotes
Acknowledgments
The Guangdong Provincial Key Laboratory of Plant Resources Biorefinery (2021GDKLPRB01), the Science and Technology Planning Project of Guangzhou City (202103000078, 202206010181), and the Guangdong Province Science and Technology Plan Project (22070510471437) provided funding for this work. Thank Dr Wang Pingping for her valuable contributions to this research.
Authors Contributions
Kegang Wu: conceptualization, methodology, writing – review and editing, supervision, and funding acquisition. Yuqiang Huang: writing – original draft, writing – review and editing, methodology, data curation, formal analysis, software, validation, investigation, and visualization. Tong Zhang: methodology, investigation, and supervision. Xianghua Chai: project administration, supervision, and resources. Xuejuan Duan: resources, validation, and project administration.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Guangdong Provincial Key Laboratory of Plant Resources Biorefinery, Guangdong Province Science and Technology Plan Project, Science and Technology Planning Project of Guangzhou City (grant numbers 2021GDKLPRB01, 22070510471437, 202103000078, and 202206010181).
Ethical Approval
Ethical approval is not applicable to the article.
Informed Consent
Not applicable to the article.
Statement of Human and Animal Rights
Not applicable to the article.
