Abstract
Two new geranylated biphenyl esters, 2,3′,4,4′-tetrahydroxy-3-geranyl-5-carboxyethyl-biphenyl methyl ester (
Moraceae is a large family comprising 40 genera, with over 1200 species widely distributed in the tropical and subtropical regions and to a lesser extent in temperate regions worldwide.
1
The genus
Results and Discussion
The butanol-soluble fraction of the methanol extract of the leaves of

Compounds isolated from
1H (600 MHz) and 13C (150 MHz) NMR Data of Compounds 1 and 2 in DMSO-D6.

Abbreviations: DEPT, distortionless enhancement by polarization transfer; DMSO, dimethyl sulfoxide; NMR, nuclear magnetic resonance.
Acetylation of
Acetylation of
Biological Activity
The 2 new geranylated biphenyl esters were tested for cytotoxicity against 3 human cancer cell lines (breast carcinoma MDA-MB-231, lung cancer A549, cervical carcinoma HeLa) using the standard 3-[4,5-dimethylthiazol-2-yl]−2,5-diphenyltetrazolium bromide (MTT) method with doxorubicin as a positive control. The results are summarized in Table 2. Compound
Cytotoxicity of Compounds 1 and 2 Against Human Cancer Cell Lines.
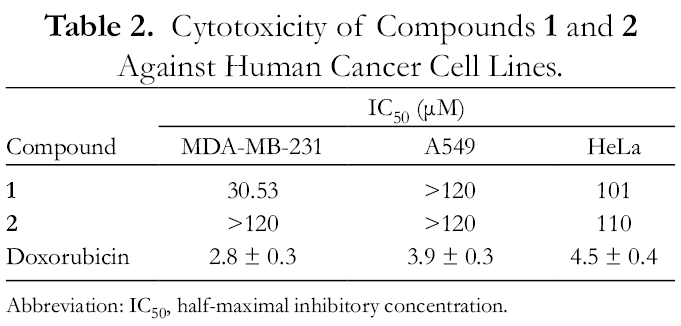
Abbreviation: IC50, half-maximal inhibitory concentration.
Compound
Antimicrobial Activity of Compounds 1 and 2.
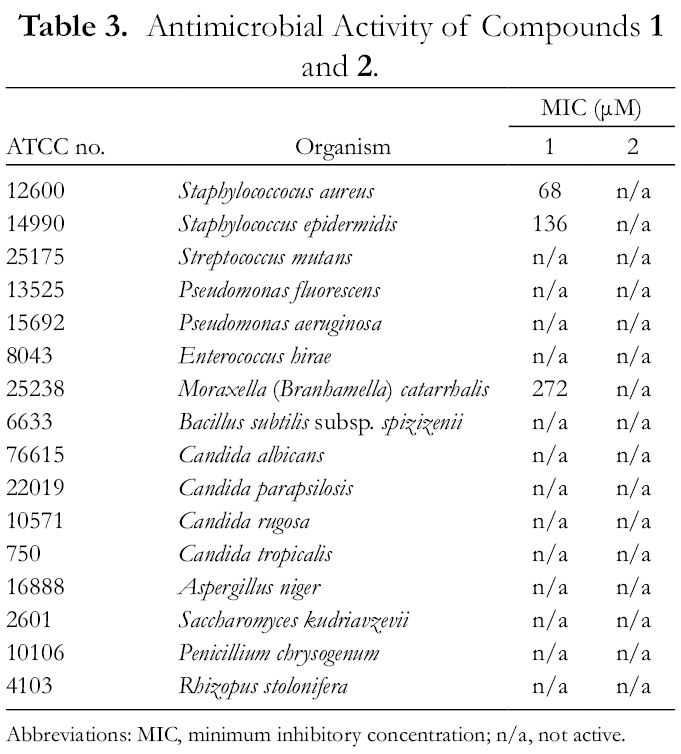
Abbreviations: MIC, minimum inhibitory concentration; n/a, not active.
Experimental
General
All solvents used throughout these studies were HPLC grade (Concord Technologies, Tianjin, China). CC was performed on Diaion HP-20 (Mitsubishi Chemical Corp., Tokyo, Japan). Analytical and semipreparative HPLC separations were performed on an Agilent 1260VL quad gradient system (G1311C pump, G1329B autosampler, G1316A thermostatted column compartment and G1315D photodiode array detector, Agilent Technologies, Santa Clara CA, USA), using a Hypersil GOLD C18 analytical column (3 µm, 2.1 × 150 mm, ThermoFisher Scientific, Waltham MA, USA) or a Hypersil GOLD C18 semipreparative column (5 µm, 10 × 250 mm). Preparative separations were performed on an Agilent 1260VL quad gradient system (G1311C pump, and G1315D photodiode array detector) equipped with a Rheodyne 7725i manual injection valve (IDEX Health & Science, Middleboro, MA, USA) and Shimadzu CTO-20A column oven (Shimadzu Scientific Instruments, Kyoto, Japan). Preparative separations were conducted using a Hypersil GOLD C18 column (5 µm, 21.2 × 250 mm). Flash chromatography was performed on an ISCO Combiflash Rf+ instrument (Teledyne ISCO, Lincoln, NE, USA) using prepacked columns of spherical C18 bonded silica 20‐45 µm (SepaFlash, Santai Technologies, Changzhou, China). Mass spectra were determined on a Q-Exactive HF Orbitrap LCMS with electrospray ionization coupled to an UltiMate 3000 RSLC nano HPLC system (Thermo Fisher Scientific, Waltham, MA, USA). Ultraviolet (UV) spectra were acquired using a HITACHI U3900 spectrophotometer (HITACHI, Kyoto, Japan). NMR spectra were recorded on Bruker Avance III spectrometers (Bruker Biospin Corp., Billerica, MA, USA) operating at 400 or 600 MHz (1H) and 100 or 150 MHz (13C). Residual solvent resonances were used as internal reference, and chemical shifts are reported in PPM (δ). Optical densities on 96-well plates were measured with a Spectramax M2 plate reader (BioTek Instruments, Winooski, VT, USA). MTT was obtained from Solarbio Science Technology Co. Ltd (Beijing, China). 2,3,5-Triphenyltetrazolium chloride (TTC) was obtained from Biotopped Science Technology Co. Ltd. (Beijing, China).
Plant Material
Samples comprising the leaves of
Extraction and Isolation
Dried and powdered leaves of
The
Fraction 5 (7.47 g), eluted from Diaion HP-20 with 100% MeOH, was analyzed by HPLC (Hypersil GOLD C18, 10%-100% acetonitrile containing 0.1% formic acid, 0.2 mL/min, 40 ℃), which showed this fraction to be a very complicated mixture of components. Fraction 5 was rechromatographed on a 220-g column of Diaion HP20, eluting with a step gradient of MeOH–water (60% MeOH, 80% MeOH, 100% MeOH, 700 mL each). Recovered weights of 3 subfractions were 2.47 g (fraction 5‐1), 2.03 g (fraction 5‐2), and 2.95 g (Fraction 5‐3), respectively.
Fraction 5‐3 was analyzed by HPLC (Hypersil GOLD C18, 47% acetonitrile containing 0.1% formic acid, 0.2 mL/min, 40 ℃) and then purified by preparative HPLC (40 ℃, Hypersil GOLD C18, 47% acetonitrile, 15 mL/min) to obtain compound
Fraction 5‐2 was analyzed by HPLC (Hypersil GOLD C18, 37% acetonitrile containing 0.1% formic acid, 0.2 mL/min, 40 ℃) and then purified by preparative HPLC (Hypersil GOLD C18, 47% acetonitrile, 15 mL/min, 40 ℃) to obtain compound
Fraction 4 (20.56 g), eluted from Diaion HP-20 with 80% MeOH, was analyzed by HPLC (Hypersil GOLD C18, 10%-100% acetonitrile containing 0.1% formic acid, 0.2 mL/min, 40 ℃). Fraction 4 was separated by repeated flash chromatography on a C18-bonded silica column (330 g) eluting with a step gradient consisting of 60% MeOH (720 mL) 80% MeOH (360 mL) and 100% MeOH (540 mL) at 18 mL/min to give 7 subfractions (each 180 mL).
Fraction 4‐4 (1.39 g), eluting from HP-20 with 60% MeOH, was analyzed by HPLC (Hypersil GOLD C18, 47% acetonitrile containing 0.1% formic acid at 0.2 mL/min, 40 ℃) and then fractionated by preparative HPLC (Hypersil GOLD C18, 47% acetonitrile at 15 mL/min, 40 ℃) to yield compound
Subfraction 4‐5 was analyzed by HPLC (Hypersil GOLD C18, 24% acetonitrile containing 0.1% formic acid at 0.2 mL/min, 40 ℃) then purified by semipreparative HPLC (Hypersil GOLD C18, 24% acetonitrile at 3 mL/min, 40℃) to yield compound
2,3′,4,4′-tetrahydroxy-3-geranyl-5-carboxyethyl-biphenyl methyl ester
Brown oil. 1H and 13C NMR, see Table 1. UV (MeOH)
2,3′,4,4′-tetrahydroxy-3-geranyl-5-carboxymethyl-biphenyl methyl ester
Yellowish oil. 1H and 13C NMR, see Table 1. UV (MeOH)
Acetylation of 1 and 2
Five milligrams of compound
2,3′,4,4′-tetraacetoxy-3-geranyl-5-carboxyethyl-biphenyl methyl ester (
HRESIMS [M+NH4]+
2,3′,4,4′-tetraacetoxy-3-geranyl-5-carboxymethyl-biphenyl methyl ester (
HRESIMS [M+NH4]+
Cytotoxicity Assay
Three human cancer cell lines including MDA-MB-231 (breast carcinoma cell line), A549 (lung cancer cell line), and HeLa (cervical carcinoma cell line) were used for cytotoxicity evaluation. Cells were grown in RPMI 1640 medium supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 µg/mL streptomycin at 37 °C in a humidified atmosphere (95% air and 5% CO2). Exponentially growing cells were used throughout the experiments. The inhibitory effects of compounds on the growth of human cancer cell lines were determined by measuring metabolic activity using a MTT assay as previously described. 10
Briefly, human cancer cell lines (4 × 103 cells/mL) were treated for 2 days with a series of concentrations (0, 10, 20, 40, 60, and 80 µM) of the compounds dissolved in dimethyl sulfoxide (DMSO; 10 µL). After incubation, 0.5 mg/mL MTT solution (100 µL) was added to each well, and the cells were then incubated at 37 °C for 4 hours. The medium was then carefully aspirated. DMSO (100 µL) was then added to each well to dissolve the formazan crystals. The plates were read immediately at 490 nm on a microplate reader. All experiments were performed 3 times, and the mean absorbance values were calculated. The results are expressed as the percentage of inhibition that produced a reduction in the absorbance by the treatment of the compounds compared to the untreated controls. A dose–response curve was generated, and the IC50 was determined for each compound against each cell line.
Antimicrobial Assay
Well diffusion antimicrobial assay
Crude extracts, fractions, and purified compounds were all evaluated for antimicrobial activity against a panel of bacteria and fungi using a standard well diffusion assay. 11 The microorganisms used and the culture conditions are shown in Table 4. Nutrient agar, brain–heart infusion agar, yeast malt agar, and potato dextrose agar media were prepared according to the manufacturer’s instructions, sterilized at 120 °C for 15 minutes, allowed to cool to room temperature, and then incubated at 37 °C for 24 hours to ensure sterility prior to use.
Microbial Culture Conditions.

Abbreviations: BHI, brain-–heart infusion agar; NA, nutrient agar; PDA, potato dextrose agar; YM, yeast malt agar.
Hexane-, dichloromethane-, butanol-, and water-soluble fractions of
Minimum inhibitory concentrations
MICs were determined for the purified compounds which showed activity in the well diffusion assays using a broth dilution method with TTC as the chromogenic agent as previously described. 12,13 Pure compounds were dissolved in DMSO to obtain sample solutions at a concentration of 1000 µg/mL. Positive and negative controls were also applied on each 96-well plate. Control antibiotics for each organism (Table 3) were prepared at 1 mg/mL, and DMSO was used as negative control.
The chromogenic reagent, TTC, was used in this study as a redox indicator measuring microbial respiration. In the presence of living microorganisms, the colorless TTC is converted to the red 1,3,5-triphenylformazan. TTC dissolved in sterilized water was added to liquid media to a final concentration of 12.5 mg/L.
Microbial cultures were suspended in sterile distilled water and adjusted to a concentration equivalent to McFarland standard No. 0.5 and then diluted 100-fold with culture medium to afford a microorganism suspension (5 × 105 CFU/mL).
Compounds were tested using 2-fold serial dilutions starting at 100 µg/mL performed in 96-well microtiter plates. After 20‐24 hour incubation at the preferred temperature for each organism, microbial growth/viability was assessed by measuring the optical density at 570 nm using a microplate reader. The results were expressed as % reduction of microorganism growth/viability compared with control, and MIC values were obtained using GraphPad Prism software.
Supplemental Material
Supplementary Material 1 - Supplemental material for Two New Geranylated Biphenyl Esters From Artocarpus altilis
Supplemental material, Supplementary Material 1, for Two New Geranylated Biphenyl Esters From
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Basic Research Program (973) of China (2015CB856500), and by the Hawaii EPSCoR program under NSF (USA) award EPS-0903833.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
