Abstract
Background
Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) is well known to selectively induce apoptotic cell death in cancer cells, not in normal cells, with death receptors (DRs)—DR4 and DR5. In consequence of this specialty, this cytokine and its receptors are considered for candidates of target therapy in clinic. SH003, a new traditional medicine-based polyherbal preparation, consists of
Methods
Cell proliferation and cytotoxicity were measured by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), clonogenic assay, and, trypan blue exclusion staining, protein expression by western blot analysis, and apoptosis by fluorescence-activated cell sorting analysis.
Results
We found that SH003-induced apoptosis in NSCLC cells through several mechanisms. First of all, MTT and colony formation assay confirmed the growth-inhibitory effect of SH003 in H460 cells. Second, SH003 upregulated the expression of DR4 and DR5. Third, it activated caspase-8, caspase-7, and caspase-3 cascades, which are essential for DR-mediated extrinsic apoptosis. The effect of SH003-induced apoptosis was significantly abolished by inhibition of caspases enzymes. And also, SH003 cleaved caspse-9. Fourth, SH003 reduced AKT kinase phosphorylation, and overexpression of AKT abrogated the caspase-dependent apoptosis by SH003. Fifth, SH003 inactivated ERK, but, constitutive ERK expression did not completely reduce SH003-mediated growth inhibition and apoptosis.
Conclusions
SH003 potentiates caspase-dependent apoptosis of NSCLC through the upregulation of DRs, activation of caspase cascades and downregulation of AKT cell survival pathways.
Introduction
Unlimited cell progression, dedifferentiation, and resistance of apoptosis are essential characteristics of tumors. The resistance against cell death is one of the 6 hallmark capabilities of cancer. 1 Apoptosis, known as programmed cell death, is a major type of regulated cell death in human bodies, however, cancer cells have ability to escape from this apoptosis mechanism.2,3 The protease enzyme activity of caspases is essential for the biochemical hallmarks of apoptosis.4,5 In brief, caspase cascades can be activated through 1 of the 2 pathways—the extrinsic (also called death receptor) pathway and the intrinsic (also called mitochondrial) pathway. In extrinsic pathway, death receptor (DR5; also called as TRAIL-R2 or Killer/DR5), is a death domain-containing transmembrane protein, is the most important role in apoptosis. DR5 is well known to induce apoptosis through ligation with its ligand such as tumor necrosis factor (TNF)-related apoptosis-inducing ligand (TRAIL). This cell death mechanism involves that activated DR5 interactions with the adaptor protein, Fas-associated death domain (FADD), consequently recruits and activates caspase-8 and the activation of downstream caspase-3, and caspase-7 cascades, leading to apoptosis. The intrinsic apoptotic pathway is activated by a variety of stimuli that are sensed intracellularly, including DNA damage or endoplasmic reticulum (ER) stress.6,7 This pathway involves mitochondrial disruption, release of cytochrome c, and consequent activates caspase-9, caspase-3, and caspase-7 cascades, leading to apoptosis as well.
The Ak strain transforming (AKT)/protein kinase B (PKB), a serine/threonine kinase, has been reported as a central regulator in cell growth, proliferation, apoptosis, and survival and is frequently deregulated in cancer.8,9 In tumors, AKT kinase is dysregulated by several mechanisms: by overexpression; by constitutive activation; or by inactivation of the inhibitory phosphatase, PTEN.10,11 In anti-apoptotic signaling pathway, AKT phosphorylates pro-apoptotic Bcl-2 family protein, Bad and blocks apoptosis by preventing Bad from binding to Bcl-XL, which is making a mitochondrial pore. 12 Eventually, this kinase induces phosphorylation of caspase-9, and decreases apoptosis by inhibiting protease activity directly. 13 Therefore, regulation of AKT is considered to be a therapeutic target for the treatment of malignant tumors.
Mitogen-activated protein kinases (MAPKs) are key signaling pathways that regulate various cellular events, such as progression, differentiation, cell death, and stress responses. 14 However, hyperactivation, or overexpression of the ERK/MAPK pathway plays a major role in the survival and development of tumor cells, among all MAPKs family. Overexpressed ERK has been reported in human tumors, including breast, colon, lung, and ovarian cancer.15-18
Herbal products have been used as medicinal remedy for centuries and many drugs approved for cancer treatment, prevention, or adjuvant were natural products or based on natural products.19-22 SH003, one of traditional medicine-based herbal mixture, consists of
Despite aggressive surgical and chemotherapeutic treatments, lung cancer is still the leading cause of cancer-related death worldwide. 26 Therefore, the objective of the current study was to determine whether SH003 has anticancer effects on human non-small-cell lung cancer (NSCLC) cells.
Results
The objective of this study was to investigate whether SH003 has anticancer potential in lung cancer.
SH003 Suppresses Proliferation of Non-small-Cell Lung Cancer Lines
We first determined the
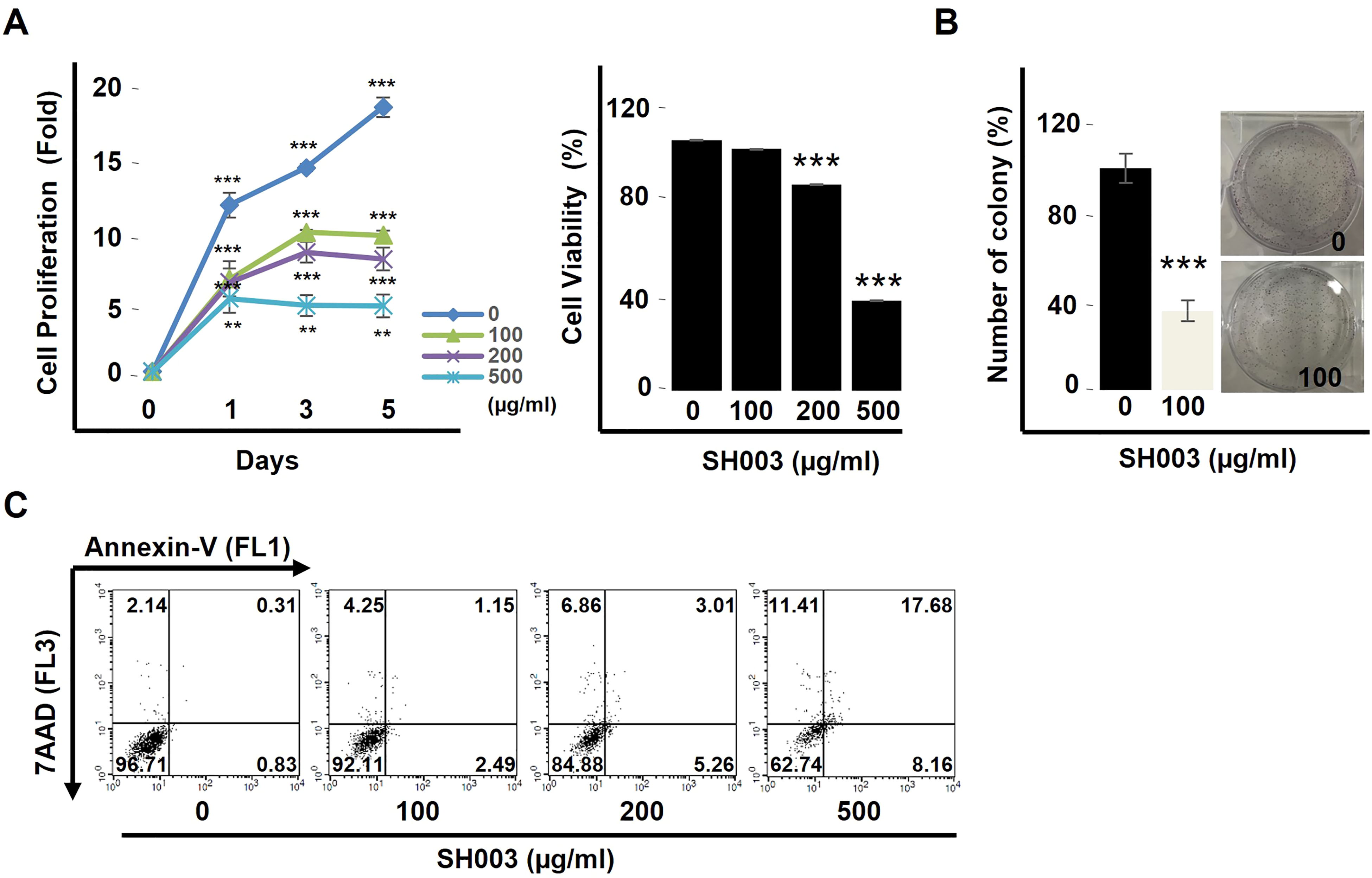
SH003 inhibits cell growth in NSCLC cells. (A) Cell proliferation (left) and viability (right) were measured by MTT assay. H460 cells were treated with SH003 for the indicated concentrations and times. The values are defined as the mean and SD from the triplicated experiments. *
SH003 Induces DR5-Mediated Caspase-Dependent Apoptotic Cell Death on H460 Cells
Next, we performed experiments to determine whether the SH003-induced inhibition of cell proliferation was due to apoptotic cell death on H460 cells. Since caspase-3, and caspase-7 are the main effectors of the intrinsic apoptotic signals,2,5 we investigated by western blot analysis for the expressions and activations of caspase-3 and caspase-7. Incubation of H460 cells with SH003 resulted in cleavage of caspase-3, caspase-7, subsequent cleavage of caspase-9, and poly(ADP-ribose) polymerase (PARP), their substrate. We also observed the proteolytic processing of caspase-8 (Figure 2A). To confirm the previous result, we conducted experiments whether the SH003-induced apoptosis was due to caspase-dependent apoptotic cell death on H460 cells. As shown in Figure 2B, treatment of H460 cells with Z-VAD-FMK (a pan-caspase inhibitor), significantly increased in cell viability (to 87% from 51% for SH003 alone), and moreover, thoroughly restrained PARP degradation, and caspase cascades activation (Figure 2C) with western blot analysis, implying a role of caspases activation in the SH003-induced cell death. In order to investigate whether the apoptosis of H460 cells was dependent on death receptors, we conducted the western blotting for those receptors. When treated SH003 to H460 cells, the expressions of DR5, not DR4 proteins were significantly increased (Figure 2D). Thus, these results suggest that SH003 induces DR5-mediated apoptotic cell death through caspase-dependent manners.

SH003 induces apoptotic cell death in H460 cells. (A) Expression of apoptotic signaling proteins as determined by western blotting assay. Representative images of PARP, cleaved-PARP, caspase-3, cleaved caspase-3, caspase-7, cleaved caspase-7, caspase-9, cleaved caspase-9, caspase-8, cleaved caspase-8, and internal control tubulin. (B) The pan-caspase inhibitor Z-VAD-FMK blocks caspase-dependent apoptotic cell death induced by SH003 treatment in H460 cells. Cells were pretreated with or without Z-VAD-FMK (50 µM) for 1 h and then incubated with SH003. Cell morphology was photographed under the microscope, and cell viability was assessed by MTT assay. ***
AKT Overexpression Abolishes SH003-Induced Apoptosis in H460 Cells
The serine/threonine kinase AKT/PKB is maybe the central regulator in cellular survival, growth, proliferation, and apoptosis, and frequently deregulated in a wide spectrum of human cancers, including lung cancers. 27 We found that AKT/PKB was phosphorylated in the H460 lung cancer cells and inactivated by SH003 in a dose-dependent manner (Figure 3A). We next conducted experiments whether AKT plays a role in the SH003-induced apoptosis on H460 cells. As shown in Figure 3B, AKT overexpression significantly reduced SH003-mediated growth inhibition based on Trypan blue staining (to 30.59% from 61.22% for SH003 alone), and moreover, completely suppressed PARP degradation, and caspase cascades activation (Figure 3C) with western blot analysis, indicating a critical role of AKT (inactivation) in the SH003-induced cell death.

AKT overexpression abrogates SH003-induced apoptosis in H460 cells. (A) Cells were treated with different concentrations of SH003 for 30 min. Cell lysates were analyzed for p-AKT, AKT, and GAPDH by western blotting assay. (B) Cells were transiently transfected with 5 µg of constitutively active AKT (AKT-CA) plasmid for 24 h by using Lipofectamine 3000 reagent and treated with 500 µg/mL SH003 for another 24 h. Cell morphology was photographed with microscope, and cell viability was measured by MTT assay. (C) Control and plasmid transfected cells were analyzed by western blotting assay. Whole cell lysates were resolved on SDS-PAGE for the analysis of cleaved PARP, cleaved caspase-3, caspase-9, caspase-8, p-AKT, total AKT, and GAPDH.
ERK Dephosphorylation is Not Involved in SH003-Induced Apoptosis in H460 Cells
MAPKs signaling pathways are also one of the key signaling pathways that regulate a wide variety of cellular processes, including proliferation, and apoptosis. 14 So, we next sought to examine whether SH003 regulates these signaling pathway as well. As shown in Figure 4A, SH003 reduced phosphorylation of both extracellular signal-regulated kinase (ERK), and p38, not c-Jun N-terminal kinase (JNK). However, constitutive ERK expression did not completely reduced SH003-mediated growth inhibition based on Trypan blue staining (to 57.84% from 66.13% for SH003 alone), and moreover, partially suppressed PARP degradation, and caspase cascades activation (Figure 4B and C) with Western blot analysis. These results indicate that the inactivation of ERK pathway could be mediated by SH003, but it is not associated with SH003-induced of apoptosis in H460 cells.
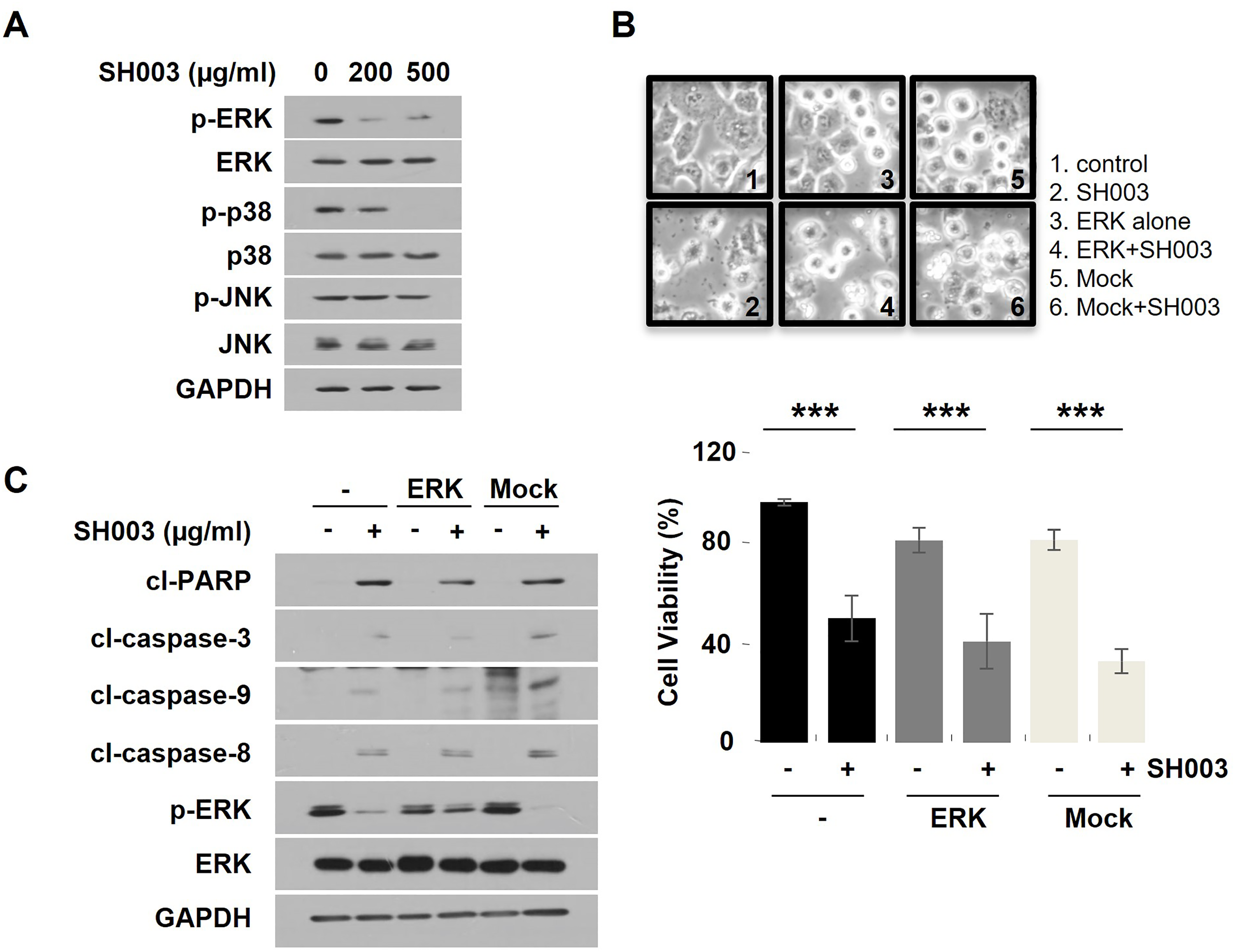
ERK overexpression is not involved in SH003-induced apoptosis in H460 cells. (A) Cells were treated with SH003 (0, 200, and 500 µg/mL) for 30 min. Western blot analysis of p-ERK, ERK, p-p38, p38, p-JNK, JNK, and GAPDH expression in H460 cells. (B) Cell viability and morphological change in 500 µg/mL SH003-treated H460 cells following transient transfection of ERK-CA plasmid or empty vector. (C) Western blotting of apoptosis-related proteins, p-ERK, total ERK, and GAPDH.
Discussion
In this study, we showed that SH003 induced DR5-mediated apoptotic signaling pathways, as indicated by intracellular metabolic modulation, and activation of caspases cascades. It is well reported that the DR-induced apoptosis requires the FADD and caspase-8 recruitments, which result in subsequent activation of its downstream mechanism. 28 Our studies found that SH003 induced DR-mediated apoptosis. Activation of caspase cascades- caspase-8, -9, and -3/-7 activation and cleavage of PARP, their substrate, is the hallmark of apoptosis. The intrinsic apoptosis pathway involves mitochondrial disruption by pro-apoptotic Bcl-2 family members such as Bax, Bad, release of cytochrome c, and activation of caspase-9 cascades. 29 Our result showed that SH003 activated caspase-9. Therefore, SH003 activated both the extrinsic and intrinsic apoptosis pathways.
We explored how SH003 could induce apoptosis of NSCLC cells in detail. We found that SH003 significantly upregulated the expression of the DR5 in a dose-dependent manner but did not have much effect on DR4. The extrinsic pathway requires an external signal; this stimulates through DR family members, such as TNF receptor 1 (TNFR1, also known as TNFRSF1A), DR4, DR5, or Fas cell surface death receptor (FAS, also known as CD95), located at the plasma membrane. It was reported that TRAIL selectively induces apoptosis in cancer cells
We found that the important intracellular signal linked to the modulation of intrinsic apoptosis signaling is AKT. AKT phosphorylates pro-apoptotic Bcl-2 family protein, Bad and blocks apoptosis by preventing Bad from binding to Bcl-XL, which is making a mitochondrial pore 12 and decreases apoptosis by inhibiting protease activity by phosphorylation of caspase-9 directly. 13 In many tumors, AKT kinase is dysregulated by several mechanisms: by overexpression; by constitutive activation; or by inactivation of the inhibitory phosphatase.10,11 Therefore, dysregulated AKT is considered to be a major therapeutic target in cancer treatment. In the current study, AKT was activated in the H460 lung cancer cells and SH003 was found to reduce AKT phosphorylation, and overexpression of AKT abolished the caspase-dependent apoptosis by SH003. The potential components of SH003 acting as AKT inhibitor are decursin, 33 nodakenin, 34 and cucurbitacin. 35 Although it is not easy to conclude accurately, it is considered that pro-apoptotic ability of SH003 is from all these components.
In addition to upregulation of DRs and downregulation of AKT, our results also showed that SH003 inactivates ERK. Hyperactivation of the ERK pathway plays a major role in the survival and development of tumor cells, among MAPKs signal pathways. Overexpressed ERK has been reported in human tumors.15-18 While our previous study also found that SH003 induces apoptotic cell death by inhibiting ERK mediated signaling in DU145 prostate cancer cells. 24 However, ERK dephosphorylation is not involved in SH003-induced apoptosis in H460 NSCLC cells. Therefore, SH003 may inhibit the growth and induce the death of tumor cells through organ-specific mechanisms.
SH003 is a new polyherbal remedy based on 3 different plant extracts-Am, Ag, and Tk Maximowicz—used for traditional medicine. The main components present in SH003 are decursin,
33
nodakenin,
34
and cucurbitacin D.
35
In the previous study, our group have been reported High Performance Liquid Chromatography (HPLC) and Ultra Performance Liquid Chromatography (UPLC) analysis to confirm the major phytochemicals of SH003 and each plant extract as a supportive information (Supplemental data; S1).
36
The greater part of these natural compounds has been reported to have antiallergic inflammatory, antiangiogenetic, and anticancer effects. These studies suggest that the compounds from SH003 mediate their activities through an additive or synergistic manner. However, there was no report about cell death through DR5-mediated mechanism. Therefore, future studies should focus on demonstrating roles of its phytochemicals in terms of DR5-mediated caspase-dependent apoptosis. And, there are a few studies about the efficacy, and safety of SH003 in
Conclusions
Our results overall suggest that SH003 can inhibit the growth of human NSCLC cells and induce caspase-dependent apoptosis through the upregulation of DRs, activation of caspase cascades, and downregulation of AKT cell survival pathways (Figure 5). However, there are some limitations to develop SH003 as a substitute for chemotherapeutic agents as a part of NSCLC cancer therapy. In this polyherbal preparation, it is unclear which active chemical moieties are responsible for the induction of apoptosis. Therefore, further studies should focus on demonstrating its active component.

A schematic diagram showing the mechanisms by SH003.
Material and Methods
Chemicals, Reagents, and Preparation of SH003
SH003, an herbal mixture, was provided from Hanpoong Pharm & Foods Co., Ltd (Republic of Korea, K-GMP) in the form of freeze-dried powder. SH003 is composed of Am, Ag, and Tk at a 1:1:1 ratio and was extracted with 30% ethanol. Most of antibodies were purchased from cell signaling technology (Danvers, Massachusetts, USA).
Cell Lines and Cell Culture Conditions
H460 cells were purchased from Korean Cell Line Bank (KCLB, Seoul, Republic of Korea) and cultured in RPMI1640 medium (Welgene, Gyeongsan, Republic of Korea). These media were supplemented with 10% heat-inactivated fetal bovine serum (FBS, JR scientific Inc., USA) and 1% penicillin-streptomycin (Roche, Switzerland). Cells were incubated at 37°C in a humidified atmosphere with 5% CO2.
Cell Viability Assay
Cell viability was assessed using MTT assay. Cells were seeded at a density of 2 × 103 per well into 96-well plates. Cells were treated with SH003 at indicated concentrations (0, 100, 200, and 500 µg/mL) for the indicated hours. At the end of time, 20 µL of MTT solution (Sigma) was added to each well and incubated for 2 h, and then was dissolved in dimethyl sulfoxide (Sigma). The measurement was performed by enzyme-linked immunosorbent assay plate reader (Merck, Germany) at 590 nm wavelength.
Trypan Blue Exclusion Assay
Cell cytotoxicity was identified with Trypan blue assay. Cells (5 × 105/100pi-dish) were seeded in 100pi-dish and treated with SH003 or inhibitor at indicated concentrations for the indicated hours. At the end of time, cells were trypsinized, diluted in a 0.4% Trypan blue solution (1:1 dilution), and incubated the cells for 1 to 2 min at room temperature. Cells were counted under the microscope in hemocytometer.
Clonogenic Assay
Cells (5 × 102/well) were plated in 6-well plates and exposed to SH003 (0 and 100
Cell Apoptosis Analysis
Cells were seeded in 6-well plates and incubated with SH003 for 48 h. Thereafter adherent cells were gently detached by using trypsin-EthyleneDiamine Tetraacetic Acid (EDTA) (Welgene, Republic of Korea) and cells were stained with Annexin V-FITC (BD Biosciences, USA) and 7-AAD for 30 min at room temperature. Cells were measured by using FACSCalibur (342973, BD FACSCalibur™, San Jose, CA, USA).
Western Blotting Assay
The cells were lysed with RIPA buffer (Biosesang, Republic of Korea) containing protease inhibitor cocktail (Sigma), and incubated in ice for 30 min. The protein quantification was performed by using Bradford protein assay dye (Bio-Rad Laboratories, USA). Equal amounts of protein were loaded onto a sodium dodecylsulfate polyacrylamide gel electrophoresis (SDS-PAGE) gel, along with a molecular weight marker (Thermo Fisher Scientific, USA), and then transferred to nitrocellulose membrane (Amersham, UK). The membranes were blocked with 3% skim-milk (BD Biosciences) and 2% bovine serum albumin (BSA, Sigma) solution for 1 h and washed 3 times in Phosphate Buffered Saline-Tween (PBS-T,containing 0.1% tween-20, Sigma) for 5 min each. The membranes were visualized by EZ-Western kit (Dogen, Republic of Korea). Antibodies against PARP, cleaved-PARP, caspase-3, cleaved caspase-3, caspase-7, cleaved caspase-7, caspase-8, cleaved caspase-8, caspase-9, cleaved caspase-9, DR5, DR4, FAS, p-AKT, AKT, and GAPDH were from cell signaling technology (USA). Antitubulin antibody was purchased from Sigma, and all other antibodies were from Santa Cruz Biotechnology (USA).
Transient Cell Transfection
All plasmids were purchased from Addgene (Addgene Inc., USA). Transient transfection was performed using Lipofectamine 3000 reagent according to the manufacturer's instructions (Invitrogen, USA). Briefly, diluted plasmid DNA was added to diluted transfection reagent and incubated for 15 min at room temperature. Then, DNA-liposome complexes were dispensed to each well and incubated at 37°C for 24 h. Transfection efficiency was determined using western blotting assay 48 h following cell transfection.
Statistical Analysis
All data are expressed as means ± standard deviation. Data were analyzed by the 2-tailed Student's
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241265216 - Supplemental material for SH003 Induces DR5-Mediated Caspase-Dependent Apoptosis of NSCLC Through Inhibition of AKT Survival Pathway
Supplemental material, sj-docx-1-npx-10.1177_1934578X241265216 for SH003 Induces DR5-Mediated Caspase-Dependent Apoptosis of NSCLC Through Inhibition of AKT Survival Pathway by Ji Hye Kim, Sooyeon Kang, Gyu-Ri Lee and Seong-Gyu Ko in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X241265216 - Supplemental material for SH003 Induces DR5-Mediated Caspase-Dependent Apoptosis of NSCLC Through Inhibition of AKT Survival Pathway
Supplemental material, sj-docx-2-npx-10.1177_1934578X241265216 for SH003 Induces DR5-Mediated Caspase-Dependent Apoptosis of NSCLC Through Inhibition of AKT Survival Pathway by Ji Hye Kim, Sooyeon Kang, Gyu-Ri Lee and Seong-Gyu Ko in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X241265216 - Supplemental material for SH003 Induces DR5-Mediated Caspase-Dependent Apoptosis of NSCLC Through Inhibition of AKT Survival Pathway
Supplemental material, sj-docx-3-npx-10.1177_1934578X241265216 for SH003 Induces DR5-Mediated Caspase-Dependent Apoptosis of NSCLC Through Inhibition of AKT Survival Pathway by Ji Hye Kim, Sooyeon Kang, Gyu-Ri Lee and Seong-Gyu Ko in Natural Product Communications
Supplemental Material
sj-docx-4-npx-10.1177_1934578X241265216 - Supplemental material for SH003 Induces DR5-Mediated Caspase-Dependent Apoptosis of NSCLC Through Inhibition of AKT Survival Pathway
Supplemental material, sj-docx-4-npx-10.1177_1934578X241265216 for SH003 Induces DR5-Mediated Caspase-Dependent Apoptosis of NSCLC Through Inhibition of AKT Survival Pathway by Ji Hye Kim, Sooyeon Kang, Gyu-Ri Lee and Seong-Gyu Ko in Natural Product Communications
Footnotes
Acknowledgments
The authors would like to thank Hanpoong Pharm and Foods Company (Republic of Korea, K-GMP) providing the herbal mixture and Korea Institute of Oriental Medicine providing pictures of each herbal medicine.
Authorship Contribution Statement
JHK designed conceptualization and performed the validation, formal analysis, investigation, data curation, writing—original draft preparation, writing—review and editing, and visualization. SK provided methodology and writing—original draft preparation. GRL designed conceptualization. SGK provided supervision, project administration, and funding acquisition the study. All authors have read and agreed to the published version of the manuscript.
Availability of Data and Materials
The data used and/or investigated during the present study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (no. 2020R1A5A2019413) and the Korean Medicine R&D Project of the Ministry of Health and Welfare (grant number: HI18C2382).
Ethics Approval Statement of Human and Animal Right
Not applicable.
Informed Consent
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
