Abstract
Objective:
To analyse the relationship between male infertility and chromosomal translocations, and the influence of different types of chromosomal translocations on semen quality, testicular volume and hormone levels.
Methods:
A retrospective cohort of infertile men was recruited for chromosomal analysis using standard Giemsa stain banding. Physical examinations, semen analysis, hormonal analysis and the detection of azoospermia factor (AZF) microdeletions were carried out. Men with normal fertility were used as controls.
Results:
Among the 1056 infertile men, 22 had chromosomal translocations (2.1%), including seven with Robertsonian translocations (0.7%), 11 with autosome- autosome reciprocal translocations (1.0%) and four with gonosome-autosome reciprocal translocations (0.4%). Left and right testicular volumes of patients with chromosomal translocations were significantly smaller than those in the fertile control group. There were no significant differences in hormone levels between patients with chromosomal translocations and fertile controls, except for significantly lower testosterone levels in patients with Robertsonian and gonosome- autosome reciprocal translocations compared with the controls. All AZF microdeletion analyses showed normal results.
Conclusions:
Chromosomal translocations may cause reductions in testicular volume and testosterone level, which may impact spermatogenesis, resulting in azoospermia or oligozoospermia and male infertility.
Introduction
Infertility, affecting about 10% – 20% of couples, is a major health problem.1,2 Male-associated issues are identified as the cause of infertility in ∼50% of infertile couples. 2 Proper evaluation of male infertility includes taking a careful patient and family history, physical examination, and semen, hormone, chromosomal and molecular analyses. Infertile men with chromosomal translocations have shown very variable sperm counts, ranging from normal counts to oligospermia or even azoospermia. 3 Thus, semen analysis is not a perfect test to investigate chromosomal translocation in men. Normal spermatogenesis requires complex interactions between germ cells and various somatic cells, and plasma follicle-stimulating hormone (FSH), luteinizing hormone (LH) and testosterone. Failure of spermatogenesis can result from impaired secretion or action of FSH, LH and testosterone, or from intrinsic defects in spermatogenesis within the testes. 4
Based on the findings of cytogenetic analysis, physicians tend to categorize infertile patients on a genetic basis. 5 Chromosomal analysis has become increasingly important for characterizing a number of causes of human infertility, 6 and has shown that male infertility might be associated with autosomal and gonosomal chromosome abnormalities. Some chromosomal abnormalities, such as Klinefelter's syndrome, are associated with male infertility. 7 In men with sperm counts < 10 × 106/ml, the rates of chromosomal abnormalities are estimated to be 5 – 7%, increasing to 10 – 15% in those with azoospermia. 8
As early as 1976, it was reported that, in six azoospermic patients with a long-arm deletion of the Y chromosome, the azoospermia factors (AZF) essential for spermatogenesis resided within the euchromatic region of the Y chromosome. 9 Within the Yq region, the AZF region is divided into three subregions: AZFa, AZFb, and AZFc. 10 Microdeletions of the three subregions result in spermatogenic arrest, and are related to azoospermia and oligozoospermia at different phases of spermatogenesis. 11 Thus, to analyse the relationship between male infertility and chromosomal translocations, the impact of AZF microdeletions on male infertility should be excluded.
To the best of our knowledge, to date, no systematic studies of the impact of chromosomal translocations of infertile men on sperm concentration, testicular volume and hormone levels have been performed. The present study analysed the relationship between male infertility and chromosomal translocations, and determined the influence of different types of chromosomal translocations on semen quality, testicular volume and hormone levels.
Patients and methods
Study Population
Between August 2008 and June 2011, consecutive male patients who sought help for infertility and ordered chromosomal analyses from the Centre for Reproductive Medicine, The First Hospital of Jilin University, Changchun, China, were recruited to this study. Infertility was defined as the inability to produce living offspring after attempting to do so for 1 year. 3 Each patient was interviewed about his family background and the underwent physical examinations and investigations described below. Fertile men (who were volunteers from Changchun City, with an age range of 24 – 40 years, without a history of reproductive failure and who had fathered at least one child) were used as the control group and underwent the same examinations.
All the participants provided written informed consent and the study was approved by the Ethics Committee of the First Hospital, Jilin University, Changchung, China.
Physical Examination
Bilateral testicular volume was measured using standard clinical techniques when the scrotum was relaxed, and compared with Prader orchidometer testis models. All examinations were carried out by the same physician.
Semen Analysis
Samples of semen were obtained from all infertile male patients and fertile control men, at least three times, after a 3 – 7 day period of ejaculatory abstinence. Semen samples were analysed immediately after collection. If the samples were not collected in the hospital, they were kept at 25 – 35 °C and transferred to the hospital within 1 h, otherwise they were excluded from the study. Semen analysis was performed according to the World Health Organization guidelines 12 using the WeiLi Color Sperm Quality detection system WLJY-9000 (WeiLi New Century Science and Techology Development Co. Ltd, Beijing, China).
Three instances of complete absence of spermatozoa in the semen ejaculate, with an ideal interval of 1 – 2 weeks between each sample collection, was considered as azoospermia. If the interval between sample collection was < 1 week or > 3 months, samples were excluded from the analysis. Severe oligozoospermia was diagnosed when sperm concentration was < 5 × 106/ml. Oligozoospermia was diagnosed when the the sperm concentration was < 20 × 106/ml. All consecutive sperm analyses were included in the study. Only data from the first sperm analysis are listed in Table 1.
Age, semen analysis and abnormal karyotypes In 22 Infertile male patients with chromosomal translocations

Patient was diagnosed with oligozoospermia 8 years previously, but was diagnosed with azoospermia during the present study.
Patient was diagnosed with oligozoospermia (sperm concentration, 0.57 x 106/ml), but 1 year later was diagnosed with azoospermia.
A, azoospermia (absence of sperm); O, oligozoospermia (sperm concentration < 20 x 106/ml); SO, severe oligozoospermia (sperm concentration < 5 x 106/ml).
Blood Collection, Preparation and Storage
Peripheral venous blood samples (9 ml from each patient) were collected into three different commercial collection tubes. The first contained 30 IU/ml heparin (2 ml of blood) and was used for chromosome karyotype analysis. The second contained 2.25 mg/ml ethylenediamine tetra-acetic acid (2 ml of blood) and was used for the detection of Y-chromosome microdeletions. The third tube contained UR040SG coagulant and separation glue (5 ml of blood) and was centrifuged at 2095
Analysis of Reproductive Hormone Levels
Reproductive hormones, including prolactin, FSH, LH and testosterone, were measured in all infertile patients and fertile controls by electrochemiluminescence immunoassay, using a Elecsys® 2010 Serum Chemistry Analyzer (Roche Diagnostics, Mannheim, Germany) according to the manufacturer's instructions. Normal reference ranges for men are: prolactin, 86 – 324 μIU/ml; FSH, 1.5 – 12.4 mIU/ml; LH, 1.7 – 8.6 mIU/ml; testosterone, 2.8 – 8.0 ng/ml.
Karyotype Analysis
Karyotype analysis of all patients and control subjects was performed. Briefly, 0.8 ml of heparinzed peripheral blood was cultured in commercial lymphocyte culture medium (RPMI-1640 containing phytohaemagglutinin, fetal bovine serum, penicillin and streptomycin (at concentrations of 5%, 12%, 0.5% and 0.48%, respectively; Guangzhou Baidi BioTechnology Co. Ltd, Guangzhou, China) at 37 °C for 72 h, 1h before the termination of cell culture, adding 50 μg/ml colchicine (Guangzhou Baidi Bio-Technology Co. Ltd) to stagnate the chromosome on metaphase. Surplus blood was stored at 4 °C in case experiments needed to be repeated.
Harvesting of cultured peripheral blood lymphocytes was performed by hypotension, fixation, trypsinization and Giemsa banding (GTG-banding). Briefly, cultured cells were transferred to a 15-ml centrifuge tube and centrifuged at 524
Y-Chromosome Microdeletion Analysis
The detection of Y-chromosome micro -deletions was carried out in any patients who were found to have chromosomal translocations in the karyotype analysis, and in all the control subjects. Genomic DNA was isolated from peripheral blood lymphocytes using the Tiangen blood DNA extraction mini kit (Beijing Tiangen Biotech Co., Ltd). If the blood samples were not analysed immediately, they were stored at 4 °C for a maximum of 7 days before discarding. The screening for AZF microdeletions was performed by multiplex polymerase chain reaction (PCR). A series of nine specific sequence-tagged sites, including SY84 and SY86 for the AZFa region, SY27, SY134 and SY143 for AZFb region, SY152, SY157, SY254 and SY255 for AZFc region were mapped. Detailed experimental procedures have been described previously. 14
Statistical Analyses
Statistical analysis was performed with SPSS® version 17.0 statistical package (SPSS Inc., Chicago, IL, USA) for Windows®. The Mann– Whitney U-test was used to compare hormone levels and testicular volume between the patients with chromosomal translocations and the fertile control group. A P-value < 0.05 was considered to be statistically significant, and all P-values were two-sided.
Results
The data set consisted of 1056 infertile men (mean age 29.5 y, range 23 – 50 y) and 50 fertile control men (mean age 26.4 y, range 23 – 35 y). Among the infertile men, there were 16.1% (170/1056) with chromosomal abnormalities and 22 (2.1%, 22/1056) with chromosomal translocations. Among these translocations, the most frequent translocations were autosome–autosome reciprocal translocations (1.0%, 11/1056), followed by Robertsonian translocations (0.7%, seven of 1056) and gonosomal– autosome reciprocal translocations (0.4%, four of 1056). The incidence of Y-autosomal translocations in the infertile men was 0.28% (three of 1056 patients). The results revealed that for the seven cases of Robertsonian translocations, there were three cases of azoospermia (42.9%), two cases of severe oligozoospermia (28.6%) and two cases of oligozoospermia (28.6%). For the 11 cases of autosome–autosome reciprocal translocations there were two cases of azoospermia (18.2%), eight cases of severe oligozoospermia (72.7%) and one case of oligozoospermia (9.1%). For the four cases of gonosomal–autosome reciprocal translocations there was one case of azoospermia (25.0%), two cases of severe oligozoospermia (50.0%) and one case of oligozoospermia (25.0%). The average semen volume for the patients with chromosomal translocations was 2.8 ml, which is within the normal range (2 – 6 ml), but the average sperm concentration was 2.57 × 106/ml, which is much lower than normal levels (> 20 × 106/ml). Data for all 22 patients with chromosomal translocations are shown in Table 1.
Comparison of the Robertsonian translocations group, the autosome– autosome reciprocal translocations group, the gonosome–autosome reciprocal translocations group and the fertile control group showed that the left and right testicular mean ± SD volumes of all the three translocation groups were significantly smaller than the fertile control group (P < 0.05), whereas there were no significant differences between the three translocation groups (Table 2).
Testicular volumes, measured by comparison with Prader orchidometer testis models, in infertile male patients with different types of chromosomal translocation (n = 22) and in fertile controls (n = 50)
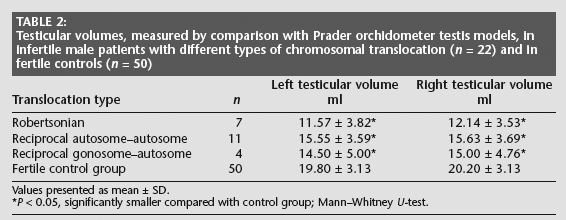
Values presented as mean ± SD.
P < 0.05, significantly smaller compared with control group; Mann–Whitney U-test.
Analyses of the reproductive hormones prolactin, FSH and LH in patients with chromosomal translocations showed that levels were slightly higher than those in the fertile controls, although the differences were not statistically significant. Testosterone levels in patients with Robertsonian and gonosome–autosome reciprocal translocations were significantly lower than those in the fertile control group (P < 0.05) (Table 3).
Reproductive hormone levels in infertile male patients with different types of chromosomal translocation (n = 22) and in fertile controls (n = 50)

Values presented as mean ± SD.
FSH, follicle-stimulating hormone; LH, luteinizing hormone.
P < 0.05, significantly different compared with control group; Mann–Whitney U-test.
Y-chromosome microdeletion analyses, including AZFa (sY84, sY86), AZFb (sY127, sY134, sY143), and AZFc (sY152, sY157, sY254, sY255), did not show any microdeletions among the patients with chromosomal translocations and the fertile controls.
Discussion
Chromosomal abnormalities have been described as important causes of male infertility for decades. 15 The incidence of chromosomal abnormalities is ∼0.6% in general populations, 16 whereas the present study showed a chromosomal abnormality rate of 16.1% among patients with infertility. Van Assche et al. 3 reported that structural reorganizations, including Robertsonian translocations and reciprocal translocations, were about 10 times more frequently found among infertile men than in general populations. The present study showed that the frequency of chromosomal translocations was 2.1% in infertile men.
The incidence of Robertsonian translocations has been estimated to be 0.1% in newborns; 17 the frequency in infertile men in the present study was seven times higher (0.7%). Robertsonian translocations can cause various degrees of sperm alteration, 18 as was seen in the present study, where the seven patients with Robertsonian translocations had either azoospermia, severe oligozoospermia or oligozoospermia.
Reciprocal translocations, which consist of mutual exchange of chromosomal segments between autosomal and gonosomal chromosomes, are found with a frequency of 0.09% in newborns. 18 The frequency in the present study among infertile men was 1.4%, which was 15 times higher than that reported in newborns. The incidence of Y-autosomal translocations among infertile men in the present study (0.28%) was higher than that in other previously reported oligozoospermic groups (0.2%) and intracytoplasmic sperm injection groups (0.09%). 19 In such cases it is possible that spermatogenic cells might not overcome meiosis I, and become blocked and degenerate at the primary spermatocyte stage, leading to oligozoospermia; 20 which is a possible explanation for cases 19 – 21 in the present study. In the patients with X-autosomal translocations, azoospermia is the most frequent aberration, although a few cases of such translocations have been reported in men with severe oligozoospermia. 21 Case 22 in the present study was azoospermic, but this was the only case with an X-autosome translocation that we identified. In a review of 26 men with X-autosomal translocation, a marked preference for involvement of a specific autosome was not shown, but it appeared that the break point was more common in the critical region of Xq13-26. 21 The break point of case 22 in the present study was Xp22, which may be another region of spermatogenic arrest.
Meiotic research in two human reciprocal translocations have shown that different translocations produce different quadrivalent or trivalent structures, 22 and that these structures may easily produce unbalanced gametes, where the risk of this meiotic imbalance is primarily determined by characteristics of the chromosomes involved and by the break-point positions. 15 In one study, at least two different meiotic checkpoints were present in a t(13;20) carrier; 22 in such a case, an association may occur between the sex body and quadrivalent structure, which could also increase during the pachytene stage, such that decreased numbers of cells reach the later pachytene stages. In essence, this could be an early–mid pachytene checkpoint. Another pachytene checkpoint occurred later at a postsynaptic stage. The second breakdown of the meiotic process could be caused by an inactivation of genes located on the regions associated with the sex body. 23 This gene inactivation would block transcription of some of these genes, which in turn could trigger an apoptotic response. If spermatogonia of patients with translocations could cross at least one meiosis checkpoint, sperm would be produced, but with a high risk of oligozoospermia. If both checkpoints are crossed, then the semen might be normal, whereas if neither is crossed there is presumably a high risk of azoospermia. Furthermore, translocations may change the gene location and undermine the structural integrity, leading to the improper spermatogenetic function of genes located in the autosome and gonosome, and resulting spermatogenic failure. Moreover, phosphatidylserine externalization, which is a typical marker of apoptosis, has significantly higher DNA fragmentation rates in ejaculated spermatozoa from translocation carriers than in spermatozoa from donors, 24 providing another explanation for translocation being a high risk factor for azoospermia or oligozoospermia.
Azoospermia factor microdeletions, which are believed to result in spermatogenic arrest, are related to azoospermia and oligozoospermia at different phases of spermatogenesis. 11 None of the 22 infertile men with translocation abnormalities in the present study had microdeletions in the AZF subregions, suggesting that the cause of spermatogenesis failure may not be associated with AZF microdeletions.
A review of published literature using the key words ‘testicular volumes' and ‘chromosomal translocations' identified very few studies. Only one paper, which reported that testicular volume was reduced in seven of eight men with autosomal translocations, was found. 25 The left and right testicular volumes of three types of translocation patients were significantly smaller than those in the fertile control group, in the present study. Thus, it is possible that patients with chromosomal translocations may have slightly damaged testes, leading to spermatogenesis failure.
To our knowledge, this was the first time that the relationship between levels of the reproductive hormones (prolactin, FSH, LH and testosterone) and different types of chromosomal translocations has been studied. Levels of prolactin, FSH and LH in patients with different chromosomal translocations (Robertsonian, autosome– autosome reciprocal and gonosome– autosome reciprocal) were slightly higher than those in the fertile control men but were not significantly different, which may indicate that testicular function was not seriously damaged or that changes in hormone levels were not obvious at that time. Testosterone levels in patients with Robertsonian and gonosome–autosome reciprocal translocations were significantly lower compared with those in the fertile control group; however, most other studies have shown that hormone levels in patients with chromosomal translocations were normal.26 – 30 One possible reason for the difference is testicular damage at the time of analysis; in disorders of spermatogenesis, the human body has the potential to activate compensatory mechanisms that help to regulate hormone levels. 31 There are likely to be varying degrees of damage to the structure and function of seminiferous tubules in different patients, such that they may or may not be able to produce normal levels of reproductive hormones. 32 If further follow-up studies on hormone levels are carried out, it is possible that different results may be observed. In addition to hormone levels, multiple factors impact on spermatogenic function. Another reason for the difference in results may be that too few cases were collected to study the different types of translocations and hormone levels.
In summary, the present study in infertile men with chromosomal translocations but normal phenotypes showed that chromosomal translocations may have a devastating impact on spermatogenesis during meiotic division, leading to azoospermia, oligozoospermia and infertility. Physicians should consider chromosomal translocation as an important factor in infertility, and carry out chromosomal karyotype analyses in infertile men with low (or even normal) hormone levels, reduced testicular volumes, azoospermia or oligozoospermia. Microdeletions of genetic material cannot be excluded unless comparative genomic hybridization is used. Furthermore, larger studies, with more cases, should be conducted to analyse the frequency of different chromosomal translocations. In addition, detailed meiotic analysis should be performed for each translocation, to obtain better diagnosis and prognosis, and to provide genetic counselling to the patient. Preimplantation genetic diagnosis is a valuable reproductive option for these translocation carriers, in addition to in vitro fertilization treatment. 33
Footnotes
Acknowledgements
We thank all staff of the Andrology Laboratory for their excellent work. We also thank Drs Trejaut and Elfateh for their constructive comments. This work was kindly supported by funds from the National Population and Family Planning Commission of China (no. 2011-GJKJS-07).
Conflicts of interest: The authors had no conflicts of interest to declare in relation to this article.
