Abstract
The edible Blaps rynchopetera Fairmaire is widely used for its various medicinal effects. From its ethyl acetate fraction, three new hydroxytyrosol dimers, rynchopeterine H (
Introduction
Like plants, many animals, including insects, have unique nutrients and pharmacological activities and can be used as food or medicine. Medicinal insects have been utilized by humans for a long time, and they played an important role in the prevention and cure of various diseases by providing antibacterial, anti-inflammatory, antitumor effects.1,2 Besides, they could also enhance immunity and act as sedative and analgesic agents. 3 Investigations have found that alkaloids, steroids, fatty acids, aromatic species, and glycosides are the main components of medical insects.4-6 Blaps rynchopetera Fairmaire, a member of the family Tenebrionidae has been used in traditional Yi medicine in Yunnan Province as a regular treatment for tumors, gastritis, fever, cough, whitlow, and rheumatoid arthritis. 7 Its main chemical components are polyphenols, 8 amino acids, 9 cyclic dipeptides, 10 fatty acids, 11 saccharides 12 and so on, these compounds exhibit extensive pharmacological activities, including antitumor, analgesic, anti-inflammatory, bacteriostasis, and anti-oxidation activities.13,14 Previous researchs show that the defensive secretion of B. rynchopetera has cytotoxicity against a variety of tumor cell lines. 15 Moreover, phenolic components show significant antioxidant activity and antitumor activity.10,14
Hydroxytyrosol (HT) is a kind of polyphenolic compounds which mainly exist in the fruits and branches of olive oil, and acts as a natural antioxidants.
16
It is reported that HT has various physiological effects, including lipid-lowering,
17
anti-inflammatory,
18
and antitumor,
19
these effects are usually related to their antioxidant capacity. The HT monomer and a number of dimer derivatives have previously been isolated from the B. rynchopetera,14,20 further chemical constituents provided three new HT dimers, rynchopeterine H (

Structure of compounds
Results
Structural Determination of Compounds 1-7
Compounds
Based on 1H-1H COSY and HMBC correlation signal analysis, the main structure is 2-(3,4-dihydroxy phenyl) ethoxy group (unit a) and 2-(3,4-dihydroxy phenyl)-2-hydroxy hemiacetal group (unit b). δ 5.33 (H-3) and δ 4.68 (H-2) correlate with C-4a and C-8a in 2-(3,4-dihydroxyphenyl) ethanol, respectively, Figures 2 (A and B) and 3 (compounds
For another two compounds, only δ 5.56 (H-8) is correlated with C-1’, and δ 4.90 (H-7) is uncorrelated with C-2’. Figure 2(C), Figure 3 (compounds

Partial correlation signals in HMBC of compounds

Key COSY ( ) and HMBC (
) and HMBC ( ) correlations of compounds
) correlations of compounds

Intramolecular hydrogen bonds and dominant configuration of compounds
NMR Spectroscopic Data of Compounds

NMR Spectroscopic Data of Compounds

Compound
Compound
Compound
Cytotoxic Activity of Compounds
The cytotoxicity of mixture
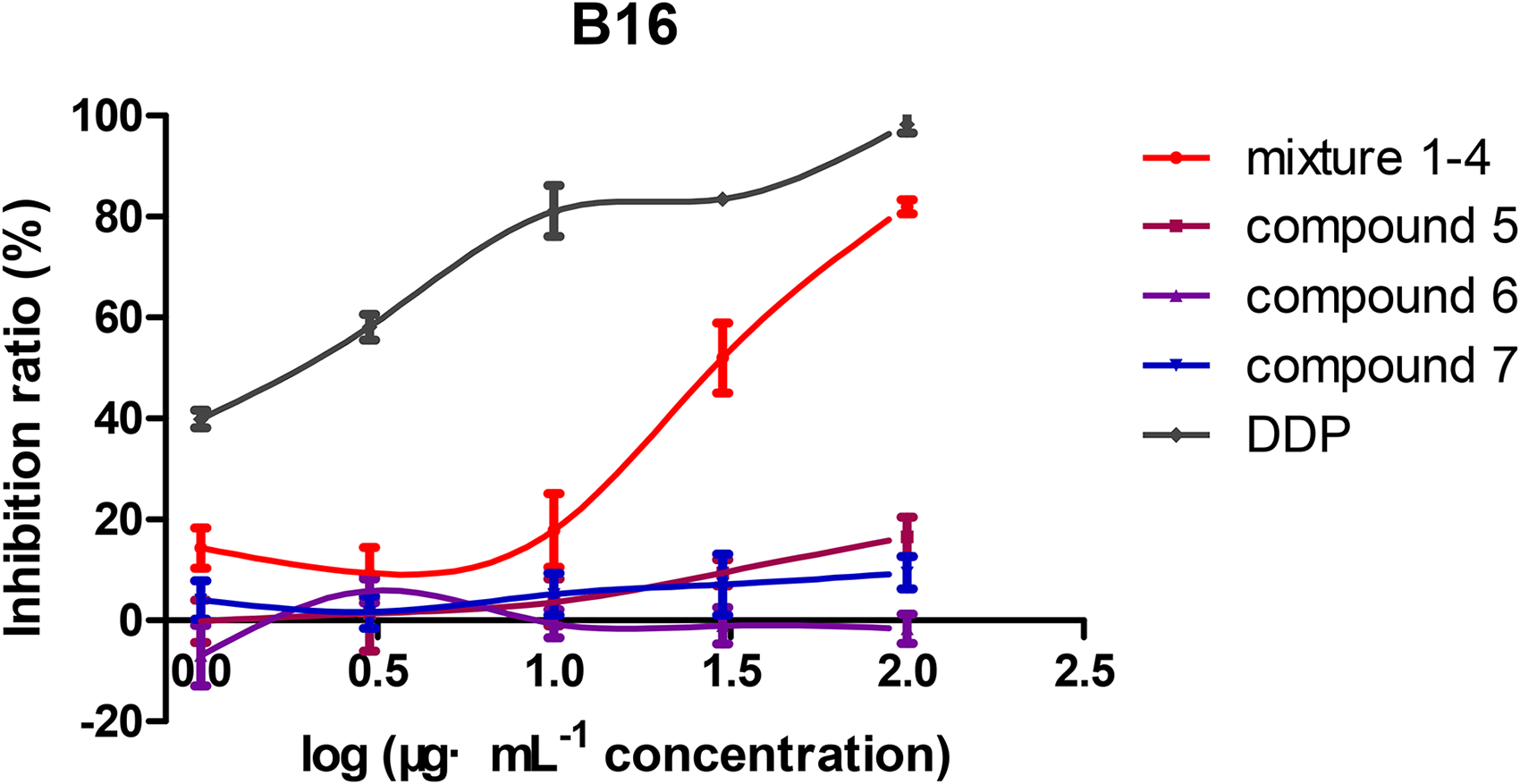
Proliferation inhibition curves of mixture
Discussion
Studies have shown that HT dimers could bring beneficial biological outcomes in neurodegenerative diseases, 26 nervous system diseases, 27 cardiovascular diseases, 28 metabolic syndrome 29 ; these effects are often related to its anti-inflammatory activities and antioxidant capacities. Besides, the anticancer properties of HT dimers were confirmed in vitro on different cell lines, predominantly breast, thyroid, liver and digestive cancer cell lines. B. rynchopetera has been used in many areas of Yunnan Province in China for the treatment of tumors; the phenolics are responsible for its therapeutic effect. Xiao Huai 14 isolated a variety of antioxidant phenolics from the ethyl acetate fraction of B. rynchopetera, and HT dimers are the main phenolics and exhibited significant antioxidant activities, rynchopeterines B and rynchopeterines C inhibited the proliferation of tumor cell Caco-2 with IC50 values of 119.7 μg·mL−1 and 158.7 μg·mL−1. Xu Fa isolated the anti-inflammatory active ingredient HT from the B. rynchopetera, increase cell apoptosis by inhibiting tumor growth and enhancing immune surveillance against cancer cells. 18
In this report, seven hydroxytyrosol dimers were isolated from the EtOAc fraction of B. rynchopetera. The cytotoxicity of mixture
Conclusions
In summary, three new and four known hydroxytyrosol dimers were isolated and identified from B. rynchopetera. The mixture of compounds
Materials and Methods
General Procedures
NMR spectra were performed on a Bruker AV-400 spectrometer (Karlsruhe, Germany) with TMS as an internal reference. Mass spectra analyses were measured on an Agilent G3250AA LC/MSD TOF spectrometer. Cooling water circulation device (EYELA, Tokyo, Japan). Sephadex LH-20 (GE Healthcare, USA) were used for column chromatography.
The specimen were collected from Dali, Yunnan Province, China. Compared with the voucher specimen (No. 2008071001), it was identified as Blaps rynchopetera Fairmaire by Prof. Guo-Dong Ren of Hebei University and preserved in Special Medicinal Insect Development National Engineering Research Center of Dali University.
Cell lines of human HepG2 hepatocellular carcinoma, human Caco-2 colorectal adenocarcinoma, human U251 glioma, human AGS gastric adenocarcinoma, mouse melanoma cells B16 and human Bel-7402 hepatocellular carcinoma cell lines, were all purchased from Shanghai Cell Institute, Chinese Academy of Sciences, China. Cisplatin was purchased from Qilu Pharmaceutical Co., Ltd (A1A1004007, Shandong, China).
Extraction and Isolation
B. rynchopetera adult body was extracted with 95% EtOH for three times. The crude extract were obtained by decompressing the solvent and then fractionated with Petroleum ether, EtOAc and butyl alcohol successively to obtain three soluble parts. The EtOAc fraction (295 g) was separated into eleven fractions (Fr.A-Fr.K) by gradient elution of petroleum ether and EtOAc on silica gel (10:1 to 0:1). Fr.F was further purified by eluting silica gel with CHCl3-CH3OH to afford compounds
Cytotoxic Activity Assay
Antiproliferative activity was detected by modified MTT reduction assay
30
on B16, U251, Caco-2, AGS, Bel-7402 and HepG2. The mixture
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221086906 - Supplemental material for Hydroxytyrosol Dimers from Medicinal Insect Blaps Rynchopetera and the in Vitro Cytotoxic Activity
Supplemental material, sj-docx-1-npx-10.1177_1934578X221086906 for Hydroxytyrosol Dimers from Medicinal Insect Blaps Rynchopetera and the in Vitro Cytotoxic Activity by Xiu-Qin Pang, Xiu-Mei Wu, Qi Wang, Di Meng, Yan-Ming Huang, Jing-Lei Xu, Yue Li, Heng Liu, Huai Xiao and Zhong-Tao Ding in Natural Product Communications
Footnotes
Acknowledgments
We are appreciated for the NMR testing from the Analysis and Testing Center of Dali University and the technical support from National-Local Joint Engineering Research Center of Entomoceutics, Dali University.
Author Contributions
Zhong-Tao Ding and Huai Xiao conceived and designed the experiments; Xiu-Qin Pang, Xiu-Mei Wu, Yan-Ming Huang, Jing-Lei Xu, Yue Li, and Heng Liu performed the experiments; Qi Wang and Di Meng analyzed the data; Xiu-Qin Pang, Huai Xiao and Xiu-Mei Wu wrote the manuscript; and Zhong-Tao Ding polished it. Zhong-Tao Ding and Huai Xiao received a research grant.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, the Special Program of Science and Technology of Yunnan Province, Special Fund Project for the Development of TCM Decoction Pieces Industry of Yunnan Province (grant number 81860742, 81960755, 82160822, 202002AA100007, 2019-YG-067).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
