Abstract
Walnut green husk (WGH) is the immature exocarp of Juglans regia L., a famous traditional herbal medicine that has long been used in China, Korea, Iran, and other places for the treatment of psoriasis, baldness, type II diabetes mellitus, intestinal worms, promotion of wound healing, and other diseases. With the advancement of modern scientific research, many studies have identified that WGH contains quinones, diarylheptane, triterpenoids, and flavonoids, and has excellent antitumor, anti-inflammatory, antibacterial, and antioxidant properties. These findings support the ethnopharmacological application of WGH and highlight the need for further investigation of the plant's potential uses in pharmaceutical fields. In this article, we made a comprehensive review of the traditional applications, botany, chemical components, pharmacological effects, and factors affecting the therapeutic properties of WGH based on a scientific literature review with a view to provide the latest information and research direction for further study and potential use of WGH. All data are gathered from scientific databases including Google Scholar, Pubmed, Sci-Finder, Web of Science, ScienceDirect, and China National Knowledge Infrastructure.
Introduction
The walnut tree originated before Greek time and now is widely distributed throughout the world. 1 It is also called Persian walnut, White walnut, English walnut, or Common walnut. 2 It is native to Iran in western Asia, and was introduced into China during Zhang Qian's diplomatic mission to the Western Regions during the Han Dynasty. It can be seen that the walnut tree has a long history of cultivation in China. Walnut is a high-quality nut that contains fat, protein, sugar, dietary fiber, and vitamins. 3 It is highly nutritious and has a high food value. Due to its rich nutritional value, it has been widely appreciated worldwide. 4
In addition to this, all parts of walnut are important. As in Figure 1, walnut leaves are a rich source of natural antibacterial agents and antioxidants 5 ; walnut septa have a positive effect on cough 6 ; walnut brown husk have an effect of lowering blood pressure in hypertensive people 7 ; walnut bark was found to have good antioxidant activity in vitro and a certain positive effect on human tumor-leukemia 8 ; walnut green husk (WGH) has excellent antitumor, anti-inflammatory, antibacterial, and other pharmacological activities. However, most studies have mainly focused only on the walnut fruit (the edible part) and ignored the important role of other parts of walnut. Thankfully, with the increasing demand for natural products in nutrition, food, cosmetics, pharmaceuticals, and industry, other parts of walnut are expected to be used in a variety of industries. 3

The various parts of the walnut.
According to the theory of traditional Chinese medicine (TCM), WGH has the effects of pungency, bitterness, astringency, calmness, clearing away heat and detoxification, expelling wind and treating ringworm, relieving pain and stopping dysentery. 9 WGH has long been used as a folk medicine for the treatment of various diseases, including type II diabetes mellitus (T2DM), superficial gastritis, uterine prolapse, warts, psoriasis, vitiligo, and cancer. 10 Due to its long history of therapeutic use, researchers are increasingly interested in the biological activity of the herb. Recently, WGH has made gratifying achievements in phytochemistry, bioactivity, agriculture, and industry. Some recently published studies have reported various health benefits of WGH extracts, including its anti-inflammatory and antibacterial activities. 11 At the same time, WGH extracts have also been shown to have strong antioxidant activity, 12 and even a positive effect on breast cancer, colon cancer, ovarian cancer, lung cancer, and other cancers. 13 All these studies provide scientific support for the use of WGH as a TCM in the treatment of these diseases.
In this article, we make a review of the phytochemistry and pharmacology of WGH by searching PubMed, Web of Science, Google Scholar, and China National Knowledge Infrastructure, and analyze the current research achievements, future research challenges and scientific gaps, hoping that this review could provide references and inspirations for future research and clinical application of WGH.
Botany
Juglandaceae, a dicotyledonous plant, is a family in the subclass Hamamelis. Walnut family plants are deciduous, semi-evergreen, or evergreen trees with pinnate compound leaves, uni-sexual flowers, double fruit skin and ovaries below. 14 There are about 60 species of 8 genera in the world, mainly distributed in Asia, Europe, North America, Central America, West Indies, and South America. 15 Walnut plants are suitable for growing in tropical and subtropical areas, and now there are 27 species in 7 genera in China. They are Platycarya, Engelhardia, Cyclocarya, Pterocarya, Juglans, Annamocarya, and Carya. In China, they are mostly distributed in northwest, northeast, north, and central areas such as Pinggu, Miyun, and Huairou in Beijing; Danfeng, Shangluo, and Mianxian in Shaanxi; and Yinchuan and Guyuan in Ningxia. Some species are distributed in Xinjiang and Tibet. 16 A map of the distribution of walnut cultivation in China is shown in Figure 2.

Map of walnut planting in China.
WGH is a dry fleshy exocarp of the walnut, which is semispherical or flaky and curled inward vertically, with a diameter of 3∼5 cm and a thickness of 6∼10 mm. The surface is smooth, looking dark green with black spots, with a broken stem at one end. The inner surface is yellowish white and uneven. WGH tastes pungent, bitter and astringent, and have the actions of clearing heat and toxins, driving away wind, treating tinea, relieving pain, and dysentery. 9
Traditional Uses
In China, the medicinal value and traditional practices of WGH have been documented in many famous ancient classics. For example, in the 14th year of the Qianlong era, the “Fangmai Zhengzong” (The Authentic Square Vein) recorded that pounded and stir-fried WGH in an iron pot and oral administration of 15g every morning could be used to treat diarrhea. “Ben Cao Gangmu” (Compendium of Materia Medica) written by Li Shizhen of the Ming Dynasty, record application of the mixture of WGH and sulfur powder to the skin could treat vitiligo. 17 In addition to the literature, WGH is also used in Chinese folklore for various diseases. Table 1 provides a list of several ancient texts and traditional uses which not only prove the long history of WGH use but provide documentary references for the modern application of WGH.
The Ancient Literatures and Traditional Uses of Walnut Green Husk (WGH).

Harvesting Period, Pretreatment, and Extraction of WGH
The period from June to September is the season for harvesting WGH. Although both immature and mature walnut pericarps can be used as medicine, this harvesting period is a bit too broad, knowing that the content of active substances in WGH may undergo changes over time. To study the dynamic accumulation rule of naphthoquinone in WGH with different harvesting time, Huo Jinhai used high performance liquid chromatography to determine the content of juglone (
Similarly, drying of medicinal herbs after extraction and different drying processes can affect the content of bioactive compounds in the herbs. Therefore, drying is a key step in preserving the bioactive components in herbs. To investigate the effect of different drying methods on the quality of WGH, 5 drying methods, including shade drying, sun drying, drying, microwave drying, and freeze drying, were evaluated by Hong Xiaoqin. The effects of different drying methods on the quality of WGH were comparatively analyzed based on the indexes of drying time, rehydration rate, water content, alcohol extract content, the content of effective components juglone (
Extraction conditions and solvents also have a significant effect on the bioactive strength and chemical composition of the extract. The extraction solvent and extraction time are the key influencing factors of the extraction process, which are not only related to the cost of extraction, but need to consider the harm of organic solvents to the human body. A study reported the antimicrobial and antioxidant properties of WGH extracted from 5 different solvents (methanol, cyclohexane, ethyl acetate (EtOAc), n-butanol, and water), and the results showed that the EtOAc extract had the highest antimicrobial activity against Staphylococcus aureus SG511 compared to the other 4 extracts, with a minimal inhibit concentrations (MICs) of 3.2 mg/mL, and that its total flavonoids and polyphenols content were 42.48 mg quercetin equivalent (QE)/g and 223.25 mg of gallic acid equivalents (GAE)/g respectively. The sample extracted with EtOAc had the highest antioxidant potential (EC50 = 13.43 μg/mL for 1,1-diphenyl-2-picrylhydrazyl and EC50 = 41.83 μg/mL for 2,2'-azinobis-(3-ethylbenzthiazoline-6-sulphonate) radical scavenging activity assay).
29
In addition, some scholars used the common method, reflux method, and ultrasonic method to verify the influence of different extraction methods on the extraction rate of total flavonoid and juglone (
Many factors have a certain impact on the quality and activity of WGH. Although the selection of harvest time, optimization of pretreatment, screening of the extraction solvent, and extraction method increased the extraction amount of effective active substances in WGH, the preservation of WGH from the fresh product of the harvest to the drying process has not been studied. WGH is known to suffer water loss, blackening, and oxidative browning during storage. Browning reduces the content of polyphenols in WGH and decreases its antioxidant activity. 31 Therefore, exploring the causes of WGH browning and suggesting the correct preservation method are concerns that need to be addressed in future scientific research. The research framework for WGH is listed in Figure 3.
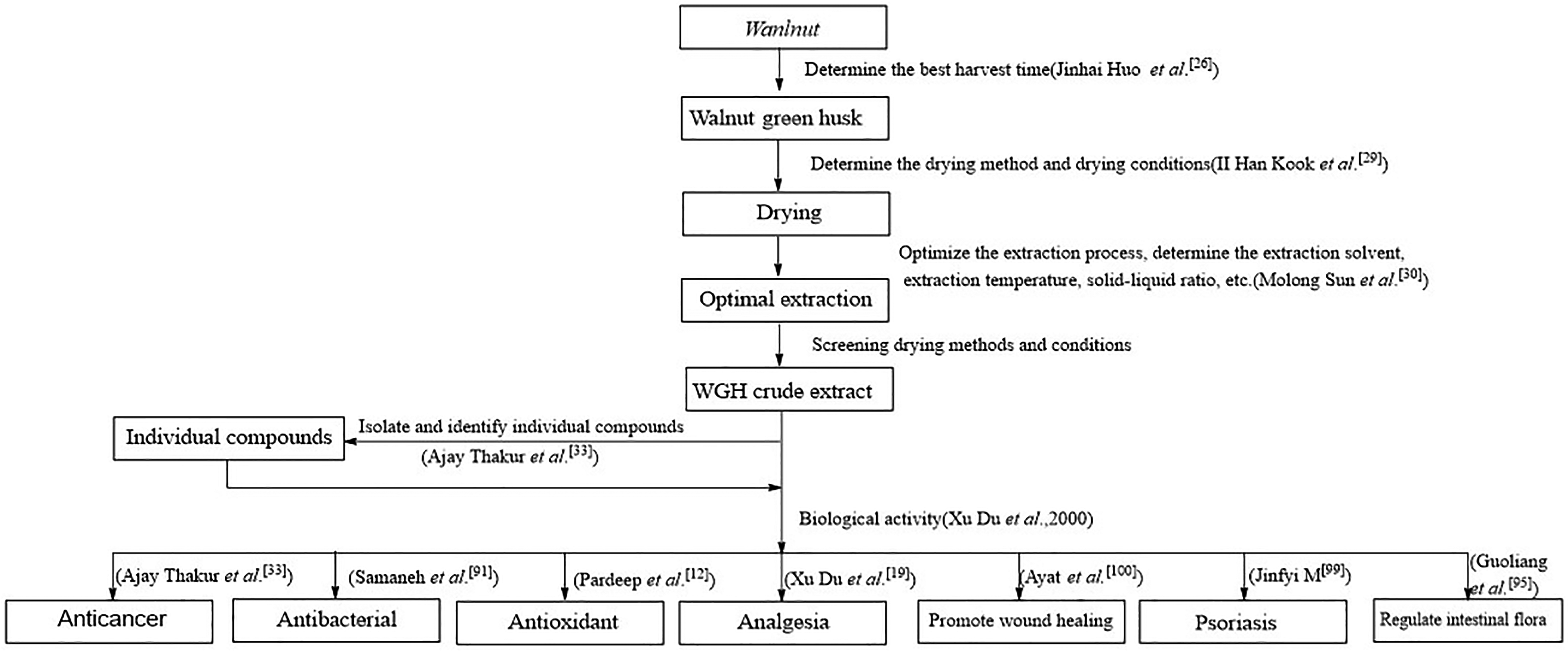
Research framework of Walnut Green Husk (WGH).
Phytochemicals Identified in WGH
WGH has yielded over 200 chemicals, including 52 quinones (

Part of quinones in walnut green husk.
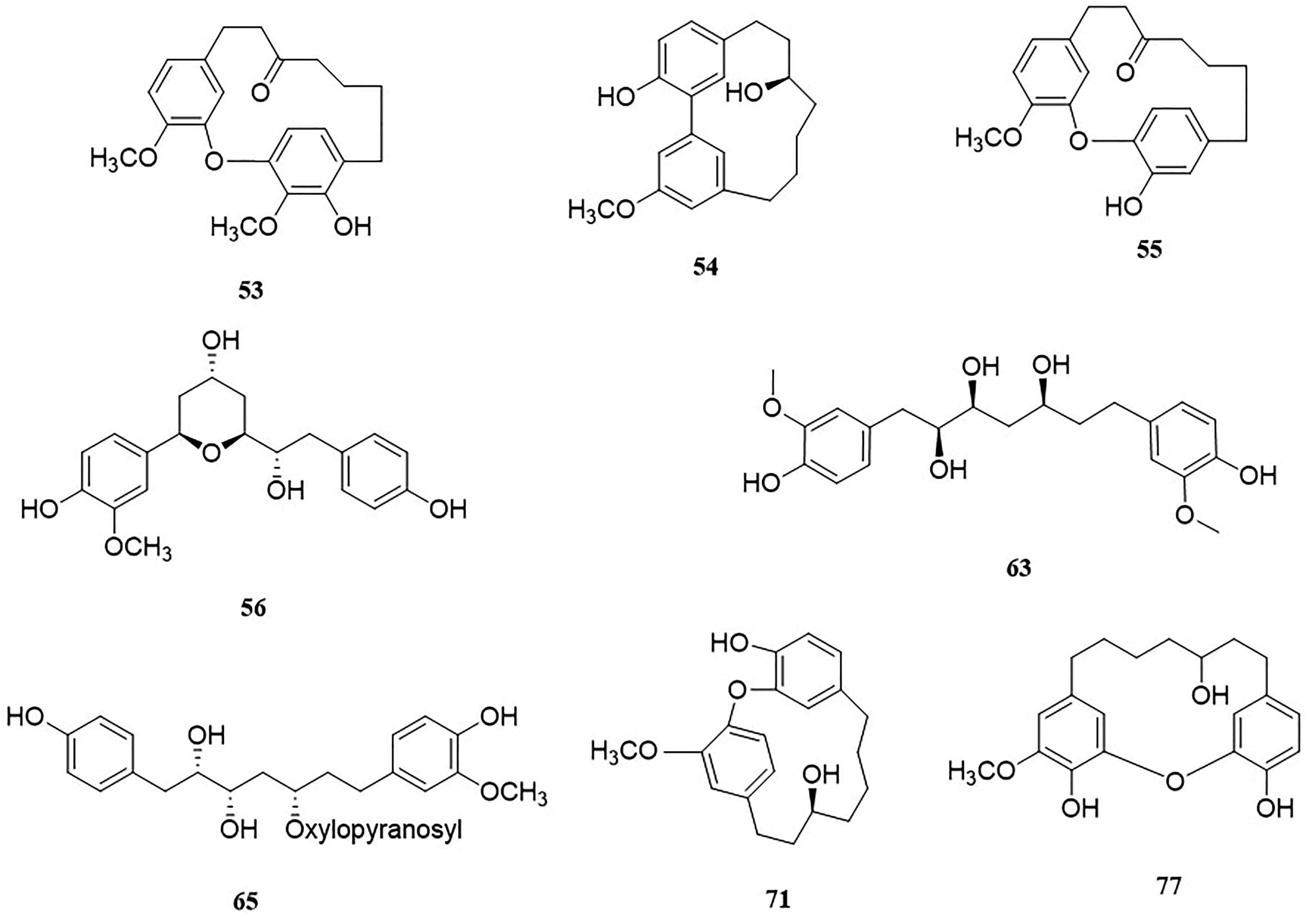
Part of diarylheptanes in walnut green husk.

Part of triterpenoids and steroids in walnut green husk.
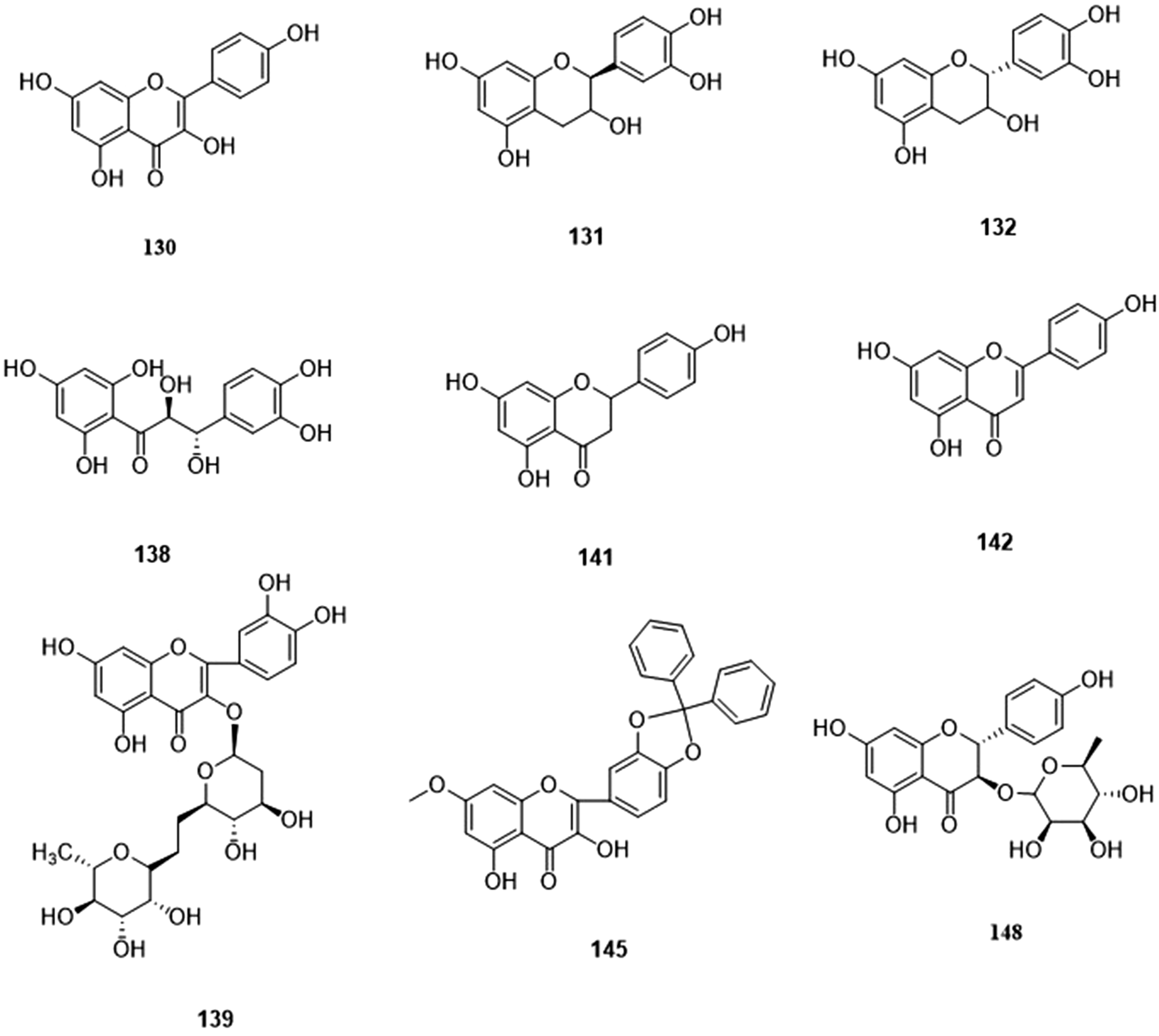
Part of flavonoids in walnut green husk.

Part of phenolic compounds in walnut green husk.
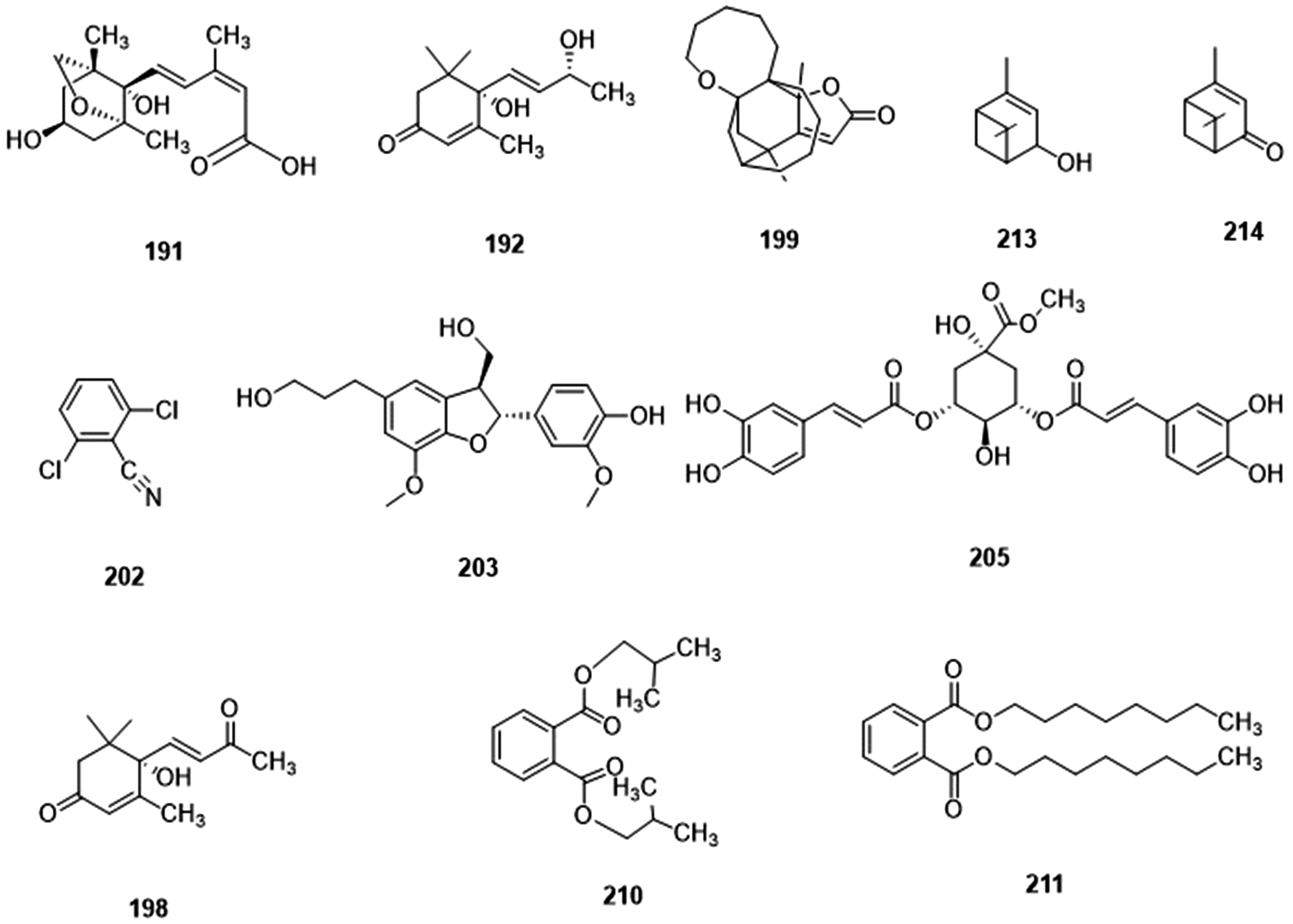
Part of other compounds in walnut green husk.
the Quinone Chemistry of Walnut Green Husk (WGH).

The Diarylheptanoids Chemistry of Walnut Green Husk (WGH).

The Terpenoids Chemistry of Walnut Green Husk (WGH).

The Flavonoids Chemistry of Walnut Green Husk (WGH).

The Polyphenols Chemistry of Walnut Green Husk (WGH).

The Other Compounds of Walnut Green Husk (WGH).

Quinone Compounds
Quinones are widely found in plants and can be classified into 4 groups: benzoquinone, naphthoquinone, phenanthrenequinone, and anthraquinone. Currently, 52 such compounds are found in WGH (
Such compounds exhibit significant biological activity, which may be related to their conformational relationship. It is worth mentioning that (4R)-3,4-dihydro-4-butoxy-5-hydroxy-naphthalen-1-(2H)-one (
Diarylheptanoids
About 25 different types of diarylheptanes were isolated from the WGH, most of which belong to the diphenyl ether structural type (
Terpenoids and Steroidals
Terpenes in WGH are mostly pentacyclic triterpenes, which account for a large proportion in WGH. Due to its structural diversity and biodiversity, extensive studies have demonstrated that they have good cytotoxic activities, such as Gerasimia Tsasi isolated the triterpene 3β,21α-dihydroxyurc-12-en-28-oic acid (
A frequent steroidal component in WGH is β-sitosterol (
Flavonoids
Flavonoids are not only widely distributed in the plant world but have many biological activities. Most flavonoids exist in the form of glycosides combined with sugar in plants, and some exist as a free form. At present, there are about 27 flavonoids have been isolated from WGH (
Polyphenols
Polyphenols are compounds
Other Compounds
Compounds
Pharmacology
Antitumor Activity
With the deepening of the research on WGH, more and more new chemical components have been found, and great progress has been made in antitumor aspects. Among the compounds isolated from WGH, juglone (
Antibacterial Activity
Numerous studies have shown that WGH extracts have good antibacterial effects against most pathogenic microorganisms such as bacteria and fungi. Meihua Xi investigated and analyzed the antioxidant and antimicrobial activities of petroleum ether, EtOAc, dichloromethane, and n-butanol extracts of WGH. The results showed that the EtOAc fraction contained the highest total flavonoids (65.74 ± 1.01 mg rutin equivalents [RE]/g dry weight [DW]) and total phenols (48.73 ± 1.09 mg gallic acid equivalent [GAE]/g DW) compared to the other fractions. Moreover, the EtOAc also showed good antibacterial activity against Escherichia coli and Bacillus cereus trophoblasts with inhibitory diameters of 33.5 and 37.6 mm, respectively, and the lowest inhibitory concentration of 31.25 mg/mL. The mechanism of the antibacterial activity is that the WGH disrupts the integrity of the cell membranes, increases the permeability of the cell membranes, and triggers the leakage of intracellular substances. 89 Parvin Abedi compared the effects of WGH extract and clotrimazole on Candida albicans in female rats. The research finding that the average colony-forming units were significantly reduced after 1 week of intervention with 2% WGH extract. After 2 weeks of intervention, the average number of colony forming units in all groups was 0 except for positive control (P < .001). After 1 week of treatment with 4% WGH extract, C. albicans colony-forming unit could be eliminated, and the effect was similar to clotrimazole. The results indicated that WGH was expected to be a kind of natural antibacterial agent which could inhibit C. albicans. 90 Samaneh Dolatabadi determined the bacteriostatic effect of WGH methanolic extract using microtitre plate method. The results of the study showed that different concentrations of WGH methanolic extract had a significant effect on the growth inhibition of Pseudomonas aeruginosa (P ≤ .05), with a mean MIC value of 16 mg/mL. This suggests that WGH can be used as an alternative treatment for P. aeruginosa infection. 91
Besides its use in the pharmaceutical industry, WGH can also be used in agriculture as an antibacterial agent. For example, the naphthoquinone compounds isolated from WGH can retard fungal germination. High-concentration WGH can completely inhibit the growth of Aspergillus flavus, but low-concentration WGH has a catalytic effect on aflatoxin synthesis, in which the 5-hydroxy or 2-methyl structural feature of the naphthoquinones is the key to the inhibition of aflatoxin. However, it is interesting to find that there was no significant additive effect when both substituents were present simultaneously. 35 For the first time, WGH was chelated with trace elements to make a fungicide against gray mold of grapes with good results. 5-Fold dilution of the WGH inhibitor was comparable to 50% carbendazim for the control of gray mold of grapes. The results showed that WGH had a good fungicidal effect and the fungicides made from WGH not only had a protective effect on the agro-ecological environment but could effectively control grape diseases, which has high economic benefits. 91
Regulatory Effect of WGH on Intestinal Flora
High-fat diets can lead to obesity, intestinal microbiome imbalance and related diseases. 92 Recent studies have shown that WGHP can reverse obesity by regulating the intestinal flora. WGHP not only reduced the abnormal weight gain, lipid metabolism disorders, inflammation, oxidative stress, and colonic tissue damage induced by high-fat diet in rats but also it improved intestinal flora dysbiosis in rats by increasing bacterial diversity in the colon and reduced the abundance of potentially pathogenic bacteria.93,94 It is suggested that WGHP may play a protective role in high-fat diet-induced metabolic inflammation by regulating intestinal flora and short-chain fatty acids. At the same time, the protective effect of WGHP on hepatic steatosis and vascular endothelial disorder has also been confirmed. 95
In addition to WGHP, WGH extract also showed beneficial effects on intestinal flora. It was found that it could reduce weight gain and reverse hepatic total cholesterol (TC), triglyceride (TG), low-density lipoprotein cholesterol (LDL-C), high density lipoprotein cholesterol, malondialdehyde (MDA), alanine aminotransferase, aspartate aminotransferase and serum TC, TG, LDL-C, MDA, leptin, insulin resistance, and glucose tolerance levels in high-fat diet-induced obese rats. Thereby reducing fat accumulation, fatty liver, and adipose tissue hypertrophy. 16S rRNA sequencing demonstrated that WGH could significantly reverse HF-induced destruction of intestinal flora, reducing the relative abundance of Fusobacteria and Firmicutes at the phylum level, increasing the relative abundance of Bacteroidetes, reducing the relative abundance of Peptonococcaceae at the family level, reducing the relative abundance of Ronalbacteria and Lactobacillus, and increasing the relative abundance of Bacteroides at the genus level. 94 This result confirmed that the beneficial effects of WGH in reducing obesity and lipid disorders may be related to the regulation of intestinal flora. Qionglian Fang and Yang Yang also back up this finding.96,97
Treat Psoriasis
WGH has long been used in traditional Chinese folklore for the treatment of psoriasis with a high cure rate. 98 However, its action mechanism has not been clearly defined. Recent studied explored the action mechanism by comparing the effects of different concentrations of WGH extract on the skin barrier function in mice with psoriasis caused by imiquimod cream. The results showed that different concentrations of WGH extract reduced the redness and swelling of the skin lesions, decreased the area of silver flakes, smoothened the skin, reduced skin thickening and relieved epidermal hyperplasia in psoriatic mice. They believed that these actions were associated with the increased expression of 4 related moisturizing factors (AQP3, Cer, Filaggrin, and Caspase14) in the skin tissue, and the therapeutic effect was linearly correlated with the dosage. These findings provide experimental and documentary support for the ancient documentation and folk applications of WGH for the treatment of psoriasis. 99
Promote Wound Healing
In Iranian traditional medicine, WGH is considered as a good remedy for would healing. This wound-healing effect of WGH has also been confirmed by experimental studied. The researchers divided 48 male Wistar albino rats into 4 groups of 12 rats each. An incision was made at the back of each rat. WGH extract (20% w/w), WGH burnt residues (20% w/w), Eucerin, and Phenytoin ointments were used in each group. Wound length, shrinkage, and histopathological evaluations were recorded on days 3, 7, 10, and 14, respectively. The result showed that the physiopathological indexes were improved significantly in rats of the WGH extract group as compared with the control group (P ≤ .05), and the effect of WGH extract was better than that of WGH slag, demonstrating that WGH has a promising application potential in promoting wound healing and suggesting that WGH may be a valuable resource for developing new trauma healing drugs. 100
Analgesic Activity
According to the Compendium of Materia Medica written by Li Shizhen more than 1200 years ago, WGH had the effect of “relieving heavy pain in the low back and legs” and “relieving pains in the heart and abdomen.” In recent years, some scholars have demonstrated through experiments that potassium salt is the main analgesic component in WGH. 19 They found that that pain could be relieved within 10 min after WGH application as an analgesic in more than subjects, and believed that this analgesic effect was attributed to the potassium salt in WGH under the action of the central nervous system by reducing the Ca2+ content and increasing the K+ content in the brain. At the same time, it stimulated certain neurons to release neurotransmitters to modulate the nociceptive response. These experimental results are consistent with the clinical observations, indicating that there is great potential for the clinical application of WGH as an analgesic. 101
Other Pharmacological Activities
Some studies observed the effect of WGH Astragalus Root decoction by WGH, astragalus root, rhizoma dioscoreae, radix trichosanthis, radix rehmanniae, lycium chinense miller, rhizoma coptidis, salvia miltiorrhiza for the treatment of T2DM and found that it had a good effect. 102 Also, WGH has good antibacterial and analgesic effects against gastric and duodenal ulcer, pain from gastritis, ringworm sores, and favus of the scalp. The pharmacological effects and mechanism of WGH are listed in Table 8.
Pharmacological Effects of Walnut Green Husk (WGH).

Conclusion
WGH is a famous traditional herb widely used in China, North Korea, Iran, and other places. At present, 218 compounds have been isolated and identified from WGH. There were 52 quinones, 25 diarylheptane compounds, 51 triterpenoids and steroids, 27 flavonoids, 35 polyphenols and 28 other compounds. Among them, quinone compounds are the main chemical components of WGH, and also the potential pharmacodynamic material basis. With the development of modern pharmacology, the research of WGH has been carried out from the aspects of antitumor, anti-inflammatory, and antibacterial. Unlike previously, in recent years, pharmacological studies on WGH have gradually leaned toward traditional folk applications such as psoriasis, analgesia, and regulation of intestinal flora, rather than being limited to modern pharmacological applications such as antitumor, as shown in Figure 10. Although there are few reports on the traditional treatment effect of WGH at present, we can see the trend of basic research toward traditional application, which is necessary to further characterize the relationship between traditional application of WGH and modern pharmacology.

Number of literatures on pharmacological effects of walnut green husk before and after 2015.
Footnotes
Author Contribution Statement
BL collated documents, wrote, and finalized the manuscript; CC reviewed the literature, discussed the layout, finished the artworks (figures and tables), and finalized the manuscript; CZ directed the pharmacology section; FH retrieved the relevant literature and discussed the layout. LH and CB designed the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Research & Development Program of Ningxia (grant number No. 2021BEG03102).
