Abstract
Objectives
The herbal product corilagin is renowned for its liver-protecting benefits and antioxidant properties. However, the protective role of corilagin in alcoholic liver injury remains unexplored. The objective of this study is to delve into the protective effects of corilagin on alcoholic liver injury, both in HepG2 cells and a mouse model.
Methods
To assess the antioxidant capacity of corilagin in vitro, DPPH and ABTS assays were conducted. HepG2 cells were pretreated with 800 mM ethanol for 24 hours, followed by corilagin treatment for another 24 hours, to evaluate its liver-protecting activity. For in vivo studies, ICR mice, with and without corilagin treatment, were given ethanol for 4 weeks to induce alcoholic liver injury. At the end of the study, serum and liver tissue samples were collected to analyze liver function, enzymatic antioxidants, reactive oxygen species (ROS) levels, alcohol dehydrogenase, and liver tissue histology.
Results
The findings reveal that corilagin exhibits remarkable free radical scavenging ability and alleviates alcohol-induced cell damage in vitro. In vivo, corilagin significantly reduced liver index and serum levels of aminotransferase aspartate, alanine transaminase, total cholesterol, and triglyceride in mice with alcoholic liver injury. Additionally, corilagin decreased ROS levels in liver tissue and increased the activities of antioxidant enzymes CAT, SOD, and GSH-Px. GSH content increased, while lipid peroxide MDA decreased in liver tissue.
Conclusion
Our data strongly suggests that corilagin possesses protective effects on alcoholic liver injury, both in vitro and in vivo, which is likely attributed to its exceptional antioxidant capacity. In summary, corilagin holds promise as a potential preventive or therapeutic agent for alcoholic liver injury.
Introduction
Alcoholic liver disease (ALD) is defined as liver damage caused by long-term or excessive drinking. 1 ALD can be classified into alcoholic fatty liver disease, alcoholic hepatitis, alcoholic liver fibrosis, alcoholic liver cirrhosis, and hepatic cancer depending on the severity of the lesions.2,3
Oxidative stress is one of the key driving factors for the occurrence and development of ALD. Alcohol is metabolized in the body into acetic acid through a process involving alcohol dehydrogenase (ADH) and aldehyde dehydrogenase, with the assistance of nicotinamide adenine dinucleotide (NAD+) as a cofactor. 4 Reactive oxygen species (ROS) will be formed as a result of the decrease in the NAD+/NADH ratio. Typically, these ROS are tolerable as their elevated levels can activate defense mechanisms and trigger the transcription of antioxidant genes, thereby enhancing the body's natural resistance against oxidative stress. 5 However, excessive alcohol consumption disrupts the balance of redox reactions. To counter this imbalance in redox homeostasis, Nrf2 plays a crucial role. Nrf2 can activate the transcription and expression of downstream antioxidant enzymes and phase II detoxification enzymes such as HO-1, NQO-1, GST, NAD(P)H, GCL, and UDP to regulate the decrease of ROS levels in the body and promote the transformation and detoxification of exogenous toxins.6–8 CYP2E1 is another important enzyme in alcohol metabolism. CYP2E1 is activated when excessive drinking and ADH is insufficient to metabolize alcohol. CYP2E1 catalyzes the oxidation of ethanol to acetaldehyde and produces a large amount of ROS, which will lead to lipid peroxidation, enzyme inactivation, DNA mutation, cell membrane destruction, and acceleration of ALD. 9
Current research demonstrates that numerous nature-sourced foodstuffs, plant extracts, and antioxidants exert beneficial therapeutic effects on ALD. For example, methanolic extract of Phyllanthus amarus, 10 total phenol extracts of Amaranthus hypochondriacus seeds, 11 polysaccharides of Hovenia dulcis, 12 green tea, 13 olive oil, 14 betaine 15 exerted significant protective roles against alcohol-induced liver injury in animal models. These substances protect biological systems from oxidative stress through their antioxidant activities, thus mitigating the harmful effects of alcohol consumption on the liver.
Corilagin is a tannin polyphenol with a chemical formula of C27H22O18. It is mainly distributed in Euphorbiaceae and is the main active ingredient in P. amarus16,17 and Phyllanthus emblica L. 17 Corilagin possesses beneficial characteristics, such as antitumor, antimicroorganism, antioxidant activity, antiinflammatory, hepatoprotective activities.18–21 Corilagin has been shown to possess strong scavenging activities against DPPH, hydroxyl, and superoxide radicals, similar to that of EGCG. 22 Additionally, it exhibits NO-scavenging and β-carotene-linoleic acid bleaching activities, and it protects against the degradation of BSA by ClO−, lipid peroxidation, and LDL oxidation.23,24 Moreover, corilagin has various protective effects on the liver, including against liver cancer,25,26 antiviral activity against HCV, 27 prevention of liver fibrosis,28–30 treating NAFLD, 31 managing acute liver failure. 32 The antioxidant and liver protective effects of corilagin may also contribute to the treatment and prevention of ALD.
Currently, there is a lack of pertinent research exploring the effect of corilagin on alcoholic liver injury. This study evaluated the protective effect of corilagin on alcoholic liver injury in vitro and in vivo. HepG2 cells and a mouse model of alcoholic liver injury by consuming alcohol (ethanol) and treatment with corilagin were constructed to evaluate the hepatoprotective effect of corilagin on alcoholic liver injury in mice. This study will contribute to the development and research of hepatoprotective drugs and the application of corilagin in the treatment and/or prevention of ALD.
Materials and Methods
Reagents
Corilagin (≥98% purity) was procured from Chendu Ruifensi Biotechnology Co. Ltd (Chendu, China). HepG2 was procured from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). Dulbecco's Modified Eagle Media (DMEM) was obtained from Hyclone (Grand Island, NY, USA). Methyl thiazolyl tetrazolium (MTT) was obtained from Sigma (St. Louis, MO, USA). Edible ethanol (alcohol) was purchased from Kezhanhuabo Instrument Co. Ltd (Xiamen, China). ADH and ROS reagent kits were purchased from Shanghai Jianglai Biotechnology Co. Ltd (Shanghai, China). Alanine transaminase (ALT), aminotransferase aspartate (AST) and the other biochemical reagent kits were purchased from Nanjing Jiancheng Biotechnology Co. Ltd (Nanjing, China).
Assay of DPPH Scavenging Activity
Two milliliter of the sample solution with different corilagin concentrations was added into 2 mL 0.1 mmol/L DPPH solution, the mixture was shaken vigorously and kept in light-deprived conditions for 30 min at room temperature. The absorbance was measured at 517 nm. Ascorbic acid (Vc) was used as a positive control set. Anhydrous ethanol was used as a blank control instead of the sample.
Assay of ABTS Scavenging Activity
An equal amount of 7.9 mg/mL ABTS was added into 1.3 mg/mL potassium persulfate solution, to yield ABTS mix solution. The solution was kept in light-deprived conditions for 12 hours at room temperature and diluted 4 times before use. Two milliliter of the sample solution with different corilagin concentrations was added into 2 mL ABTS solution, the mixture was shaken vigorously and the absorbance was measured at 734 nm within 6 min. Ascorbic acid (Vc) was used as a positive control set. Water was used as a blank control instead of the sample.
Cell Culture and Corilagin Treatment
Human hepatoblastoma cells HepG2 were cultured in DMEM medium with 10% FBS and 1% penicillin/streptomycin at 37 °C in a humidified incubator (5% CO2, 95% air). Corilagin was dissolved in DMSO to make a stock solution of 50 mM and further diluted to final concentrations with a serum-free culture medium. The amount of DMSO added to the cell culture was less than 0.5% in all cases.
To determine the concentration of corilagin used in the experiment, HepG2 cells were treated with increasing concentrations of corilagin for 24 hours and cell viabilities were assayed. In the following experiments, cells were left untreated or treated with ethanol at a concentration of 800 mM for 24 hours; then, cells were again left untreated or treated with corilagin at 6, 12.5, 20, and 50 μM for 24 hours.
MTT Assay
Cells were seeded at a density of 1 × 105 cells/mL in 96-well polystyrene culture plates at 37 °C with 5% (v/v) CO2 for 24 hours. Corilagin was added to cells at a predesigned concentration and cultured for 24 hours; cells were treated with DMSO as control, and 3 replicates were set for each group. Subsequently, 100 μL of 0.5% (w/v) MTT dissolved in phosphate-buffered saline was added to each well and incubated for 4 hours. The medium was removed from the 96-well plates, and 100 μL of DMSO was added to each well to dissolve the formazan crystals. After shaking the plates for ten minutes, a microplate reader was used to calculate the absorbance at 570 nm.
Animal Experiments
Male ICR mice (6-week-old, 20 ± 2 g) were procured from the Shanghai SLAC Laboratory Animal Co., Ltd (SCXK2017-0005). All animal experiments were approved by the Experimental Animal Ethics Committee of Xiamen University (No. XMULAC20210054). According to pertinent rules established by the Chinese Academy of Medical Sciences, experiments were carried out, animals were cared for, and the study was reported in compliance with ARRIVE standards. The mice spent a week getting used to the experimental environment before the study. Mice were randomly divided into the following 6 groups (n = 8/group): control, model (ethanol), positive (ethanol + bifendate), and experimental group (ethanol + corilagin). The control group was not treated with any drugs or alcohol. The positive group and the experimental group were gavaged with 200 mg/kg bifendate and 50, 100, and 200 mg/kg corilagin 1 hour in advance respectively. One hour after daily dosing, the mice in the model, positive and experimental groups were provided gavage with a single dose of 50% ethanol (12 mL/kg b.w.) for 4 weeks. After 4 weeks, the mice were euthanized, and their blood and liver tissue were collected for subsequent analyses.
Determination of Liver Index of Mice
The body weight and the liver of mice were measured 12 hours after the last gavage. The liver index was calculated by the following formula: liver index (%) = liver weight/mouse body weight × 100%.
Determination of Serum ALT, AST, triglyceride, and total cholesterol in Mice
Blood was collected and serum was separated by centrifugation (4 °C, 3000 r/min, 15 min). The levels of serum ALT, AST, triglyceride (TG) and TG were determined according to the manufacturer's instructions.
Determination of ROS Level, CAT, SOD, GSH-Px Activity, GSH, and MDA Contents in the Liver of Mice
Liver tissue was homogenized to 10% liver homogenate in 0.86% NaCl. The homogenate was centrifugated (4 °C, 3000 r/min, 20 min) and the supernatant was collected. The protein concentration in the liver was measured with a BCA protein assay reagent kit. ROS, CAT, SOD, GSH-Px, GSH, and MDA were determined according to the requirements of the instructions provided in reagent kits.
Determination of ADH Activity in the Liver of Mice
Liver tissue was homogenized to 10% liver homogenate in 0.86% NaCl. The homogenate was centrifugated (4 °C, 3000 r/min, 20 min) and the supernatant was collected. The protein concentration in the liver was measured with a BCA protein assay reagent kit. ADH were determined according to the requirements of the instructions provided in reagent kits.
Histopathology
The liver tissues of mice were fixed in a 10% neutral phosphate-buffered formalin solution and embedded in paraffin. The sections were cut into cross sections 5 μm thick and stained with hematoxylin and eosin (H&E). Paraffin-embedded tissues were deparaffinized and were then subjected to H&E staining. The specific staining was observed under an optical microscope.
Statistical Analysis
All values are expressed as the M ± SD The SPSS 13.0 software was used for statistical analysis. The significant analysis was performed using a 2-sided unpaired Student's t-test. P < .05 was considered statistically significant.
Results
Effects of Corilagin on Antioxidant Capacity In Vitro
The DPPH and ABTS free radical scavenging experiments are internationally recognized methods for assessing antioxidant capacity. As shown in Table 1, corilagin exhibited significant scavenging effects against DPPH and ABTS free radicals, with IC50 values of 4.80 ± 0.12 and 0.99 ± 0.02 µg/mL, respectively. The antioxidant effects of corilagin were superior to the positive control Vc, with IC50 values of 10.53 ± 0.15 and 7.27 ± 0.06 µg/mL, respectively.
Evaluation of Antioxidative Ability of Corilagin.

Corilagin Prevents Ethanol-Induced Toxicity and Injury in HepG2 Cells
In the experiment, 100 µM corilagin was tested to be noncytotoxic to HepG2 cells (Figure 1A). Consequently, a dose range of 5-50 μM was chosen to assess the protective effect of corilagin on alcoholic liver injury in HepG2 cells. As shown in Figure 1B, corilagin dose-dependently reduced cell damage caused by ethanol. Fifty micromolar corilagin significantly improved HepG2 cell viability. The protective effect of corilagin on alcoholic liver injury in vitro was observed.

The effect of corilagin on cell viability and alcoholic liver injury. (A) Cytotoxicity of corilagin on HepG2 cells; (B) Protective effect of corilagin on ethanol-induced damage in HepG2 cells.
Changes of Behavior and Liver Index in Experimental Mice After Drinking
The control group mice exhibited normal hair condition, appetite, and fecal consistency. In contrast, the experimental group mice displayed yellowing of their hair, looser feces, and a significant reduction in body weight compared to the control group. Notably, the mice treated with corilagin and those in the positive control group showed improved fecal, dietary, and hair conditions. Furthermore, mice in the 200 mg/kg corilagin treatment group exhibited a significant increase in body weight compared to the model group. The changes in mice’s body weight during the experimental process were detailed in Supplemental Material 1.
Compared with the control group, the liver index of the mice in the experimental model group increased significantly (P < .001). Corilagin (50, 100, 200 mg/kg) treatment significantly reduced the liver index of alcoholic liver injury mice(P < .01, P < .01, P < .001). The mouse liver index of the high-dose corilagin group (200 mg/kg) basically returned to normal levels. The results are illustrated in Figure 2.
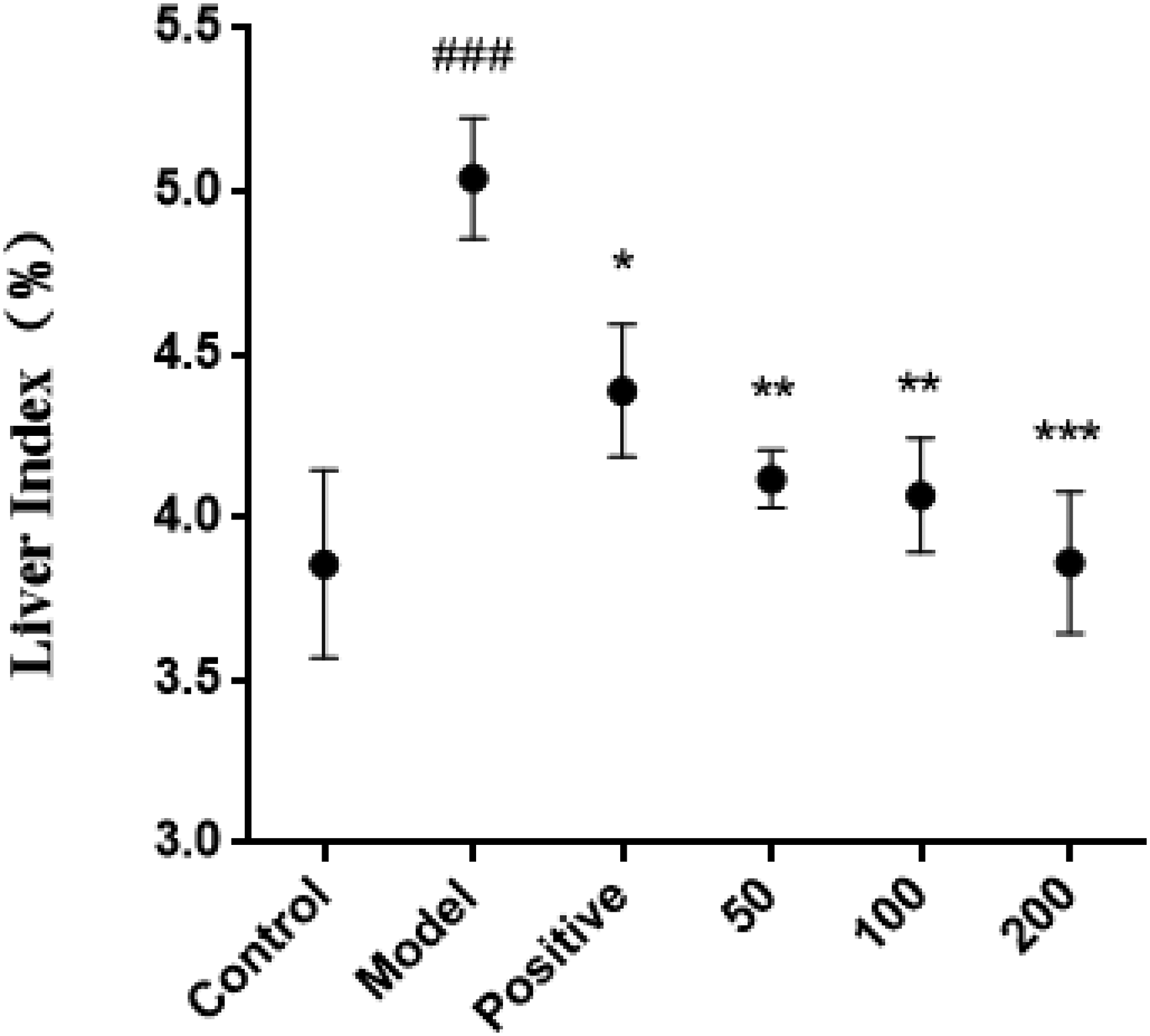
The effect of corilagin on liver index in mice with alcoholic liver injury (x ± s, n = 8). *P < .05,** P < .01, and ***P < .001 versus Model group, ###P < .001 versus control group.
Effects of Corilagin on Liver Function in Mice With Alcoholic Liver Injury
As shown in Figure 3, the liver function markers ALT and AST were significantly increased in the model group compared to the control group (P < .001, P < .01). Compared with the model group, treatment with corilagin (50, 100, and 200 mg/kg) significantly decreased the serum ALT (P < .01, P < .001, P < .001) and AST (P < .05, P < .01, P < .01) levels. These findings suggested that corilagin protected the liver and decreased the transaminase in mice with alcoholic liver injury.
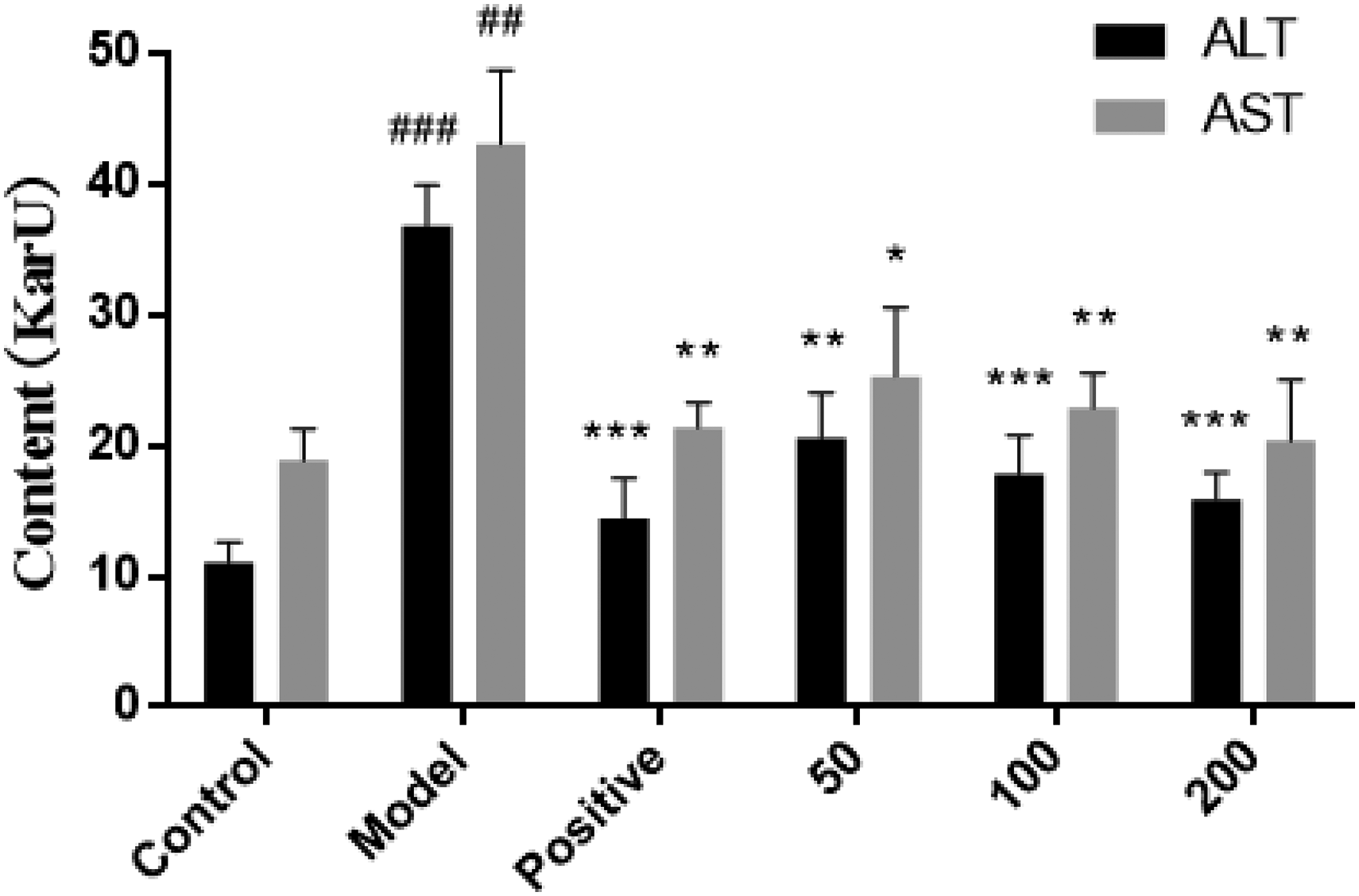
The effect of corilagin on serum ALT, AST in mice with alcoholic liver injury (x ± s, n = 8). *P < .05,** P < .01, and ***P < 0.001 versus Model group, ##P < 0.01 and ###P < 0.001 versus Control group.
Effects of Corilagin on Lipid Metabolism in Mice With Alcoholic Liver Injury
As illustrated in Figure 4, the total cholesterol (TC) and TG contents in the model group were significantly increased compared to the control group (P < .001, P < .001). Compared with the model group, 50 mg/kg corilagin treatment significantly reduced the serum TG content of alcoholic liver injury mice (P < .05). 100 and 200 mg/kg corilagin treatment significantly reduced the contents of TC (P < .05, P < .001) and TG (P < .01, P < .001). These findings showed that corilagin could effectively mitigate the elevation of blood lipids caused by alcohol consumption, potentially preventing lipid accumulation in the liver during alcoholic liver injury and thus contributing to the prevention of alcoholic fatty liver disease.

The effect of corilagin on serum TC and TG in mice with alcoholic liver injury (x ± s, n = 8). *P < .05,** P < .01, and*** P < .001 versus model group, ###P < .001 versus control group.
Effects of Corilagin on Oxidative Stress in Mice With Alcoholic Liver Injury
The results of ROS detection showed that alcohol consumption significantly increased ROS levels in the liver of mice in the model group compared to the control group (P < .001). However, 100 and 200 mg/kg corilagin treatment significantly reduced the level of ROS in the liver tissue (P < .05, P < .01) (Figure 5A). Additionally, alcohol intake caused a decrease in the activities of antioxidant enzymes SOD, CAT, and GSH-Px, while corilagin treatment reversed this change to varying degrees (Figure 5B, 5C, 5D). The effect of corilagin was more pronounced for SOD and CAT, while just 100 mg/kg of corilagin can significantly increase GSH-Px activity, which may be related to drug targeting. To further study, the changes in GSH and MDA contents were confirmed in the liver tissue. Alcohol intake depleted GSH and increased MDA. Compared with the model group, 100 and 200 mg/kg corilagin treatments significantly increased the content of GSH (P < .01, P < .01) (Figure 5E), and the content of MDA in the liver of the mice in the model group was significantly higher than each corilagin treatment group. (P < .01) (Figure 5F). These findings showed that corilagin could decrease ROS levels, enhance the activities of antioxidant enzymes and antioxidant levels, and decrease lipid peroxide formation in mice with alcoholic liver injury. These effects suggested that corilagin may play a protective role in the liver by mitigating oxidative stress and lipid peroxidation caused by alcohol consumption.

The effect of corilagin on oxidative stress in the liver tissues of mice with alcoholic liver injury (x ± s, n = 8). (Levels of expression in the liver tissues of mice with alcoholic liver injury: (A) ROS; (B) SOD; (C) CAT; (D) GSH-Px; (E) GSH; (F) MDA. *P < .05,** P < .05, and ***P < .001 versus model group, #P < .05, ##P < .01, ###P < .001 versus control group.
Effect of Corilagin on Liver Histopathology in Mice With Alcoholic Liver Injury
In histopathological examination, intake of ethanol-induced severe pathological changes in the liver. As depicted in Figure 6, the liver tissues of the control group exhibited a healthy and intact appearance, with clearly defined hepatic lobules and normally distributed hepatic cords. Additionally, there was no evidence of inflammatory infiltration (Figure 6A). However, in the model group, significant pathological changes were observed (Figure 6B). The hepatic cords appeared disorganized, and numerous lipid vacuoles formed within the cytoplasm, indicating the progression of the liver tissue toward alcoholic fatty liver disease. In contrast, corilagin treatment significantly reduced the signs of steatosis. The pathological changes of liver histopathology were normalized with increasing doses (Figure 6D, 6E, 6F). These results indicate that corilagin can protect the liver and reduce steatosis in mice with alcoholic liver injury.

Pathological observation of liver tissue in mice with alcoholic liver injury. (A) Control group; (B) model group; (C) positive group; (D) 50 mg/kg corilagin; (E) 100 mg/kg corilagin; (F) 200 mg/kg corilagin.
Effects of Corilagin on Alcohol Metabolizing Enzymes in Mice With Alcoholic Liver Injury
The results of alcohol metabolizing enzyme detection (Figure 7) revealed a marked decrease in ADH activity in the liver tissue of mice in the model group, in comparison to the control group (P < .01). Notably, treatment with corilagin significantly enhanced ADH activity in comparison to the model group. The increase in ADH activity may contribute to accelerate alcohol metabolism, which may indicate a decrease in dependence on CYP2E1 and a decrease in levels of oxidative stress and liver damage caused by CYP2E1 metabolism.

The effect of corilagin on ADH in the liver tissues of mice with alcoholic liver injury (x ± s, n = 8). **P < .01 and ***P < .001 versus model group, ##P < .01, ##P < .001 versus control group.
Discussion
ALD is a serious liver condition resulting from long-term excessive alcohol consumption and its treatment remains a significant clinical challenge. Currently, despite the existence of some therapeutic strategies, effective treatment options for ALD are still limited, making abstinence the most effective means of controlling the pathological process of the disease. However, changing lifestyle habits is not an easy task for chronic alcoholics, making the search for substances that can assist or treat ALD particularly important.
ROS are significant factors that contribute to the occurrence and progression of ALD. The metabolism of alcohol generates a large amount of ROS. Excessive ROS will deplete the antioxidants (such as GSH, vitamin A, vitamin C, and vitamin E) and lead to the imbalance of redox homeostasis in the body. This imbalance further triggers lipid peroxidation, DNA damage, cell membrane damage and apoptosis,33–35 ultimately contributing to the occurrence and progression of ALD. Therefore, reducing oxidative stress will contribute to mitigating the occurrence and progression of alcoholic liver injury.
Polyphenols, a class of naturally occurring compounds widely found in fruits, vegetables, and beverages such as red wine and green tea, have demonstrated remarkable antioxidant and antiinflammatory properties that hold great promise in the prevention of ALD. 36 Compounds such as ellagic acid, 37 anthocyanin, 38 dihydromyricetin, 39 polyphenolic extracts of Tartary buckwheat, 40 green tea extract, 41 , curcumin 42 have been shown to effectively mitigate oxidative stress and modulate alcohol metabolism, offering a natural and effective supportive measure for those striving to abstain from or control their alcohol consumption.
In this study, we investigated the protective effects of a natural polyphenol, corilagin on alcoholic liver injury in vivo and in vitro. In vitro experiments, corilagin showed an excellent ability to scavenge DPPH and ABTS free radicals. It means that corilagin is a promising potential antioxidant. Further in vitro experiments have confirmed the protective effects of corilagin on alcoholic liver injury. In a model of alcohol-induced HepG2 cell damage, 50 µM corilagin was able to significantly improve the viability of damaged cells. Furthermore, results from animal experiments have demonstrated that treatment with corilagin significantly improved liver function indicators (AST, ALT) in mice with alcoholic liver injury, while also reducing serum lipid levels (TC, TG). The improvement in these biochemical markers is consistent with the recovery and alleviation of pathological changes in fatty liver observed with corilagin treatment. Similar changes have been observed in the treatment of mice with a high-fat diet, where corilagin has been shown to prevent and alleviate fatty liver lesions by improving lipid metabolism. 43 This suggested that corilagin may also be beneficial in the early fatty lesion stage of the development of ALD.
It is noteworthy that we also observed corilagin to upregulate the activities of antioxidant enzymes (CAT, SOD, and GSH-Px) and the levels of the antioxidant GSH while downregulating the levels of lipid peroxide MDA in the animal experiments. These results revealed the protective effects of corilagin against oxidative stress in the livers of mice with alcoholic liver injury, and the mechanism may be closely related to its ability to scavenge free radicals. The decrease in ROS levels in liver tissue further supports the antioxidant capabilities and antioxidant role of corilagin. Moreover, corilagin may also be involved in the regulation of oxidative stress signaling pathways. Recent studies have shown that corilagin could attenuate acetaminophen-induced liver oxidative stress injury and acute liver failure through the regulation of Nrf2-related signaling pathways. 44 Additionally, corilagin has been found to alleviate histopathological damage, oxidative stress, and apoptosis in rat intestinal tissue through the regulation of the AMPK/Sirt1-autophagy pathway. 45 Moreover, corilagin improved oxidative stress injury in the hippocampus of the brain by inhibiting NOX2 and activating Nrf2. 46 These discoveries suggested that the antioxidant stress capabilities of corilagin may not solely originate from its antioxidant properties, but rather, it might also participate in the regulation of various intricate antioxidant mechanisms.
Another noteworthy discovery was that corilagin treatment can revive ADH activity in the liver tissue. The liver metabolizes around 90% alcohol that is consumed by the body and the metabolism of alcohol mainly depends on ADH and CYP2E1. ADH is necessary for the primary alcohol metabolism in the liver. Previous studies have shown that ADH is the enzyme with the highest catalytic efficiency of alcohol, but its binding efficiency is far inferior to CYP2E1. A small amount of alcohol can be quickly metabolized by the body into acetaldehyde through ADH, but only around 10% of ethanol is converted to acetaldehyde by CYP2E1. 47 Excessive drinking causes the body to express CYP2E1, which is involved in the metabolism of alcohol. 48 Oxidative stress and the degree of lipid peroxidation caused by alcohol induction are directly connected to CYP2E1 levels.49,50 It is possible that corilagin could improve ADH activity and accelerate alcohol metabolism, which may decrease the induction of CYP2E1 by alcohol metabolism and more ROS generation. However, further research is still necessary to delve deeper into the relevant mechanisms.
Taken together, our findings indicated that corilagin may play a beneficial role in the early stages of ALD development, particularly in the context of fatty liver injury and oxidative stress. Its ability to modulate oxidative stress and scavenge free radicals suggested that it could be a promising candidate for the treatment of ALD and related liver conditions. However, given that ALD is a complex chronic condition with distinct characteristics at different pathological stages, this study has primarily focused on its early phase. Future research is warranted to conduct additional experiments to explore other liver-protective mechanisms, as well as the effects and mechanisms of corilagin in various pathological stages of ALD.
Conclusions
The study findings reveal the remarkable antioxidant capabilities and protective effects of corilagin against alcoholic liver injury in HepG2 cells. In mice with alcoholic liver injury, corilagin treatment significantly reduced serum levels of ALT, AST, TC, and TG, while enhancing the activities of antioxidant enzymes such as CAT, SOD, and GSH-Px, as well as antioxidant GSH content in liver tissue. Additionally, corilagin decreased lipid peroxide MDA content and ROS levels in liver tissue. These results suggested that corilagin can improve liver function, reduce lipid accumulation and counter oxidative stress to prevent alcoholic liver injury. Both in vitro and in vivo, corilagin exhibited protective effects against alcoholic liver injury. The relevant mechanism may be related to the antioxidant capacity of corilagin and the regulation of oxidative stress. Overall, our research underscores the hepatoprotective effects and potential of corilagin in inhibiting the progression of ALD. Hence, corilagin holds promise as a potential drug or dietary supplement for treating or preventing ALD in the future.
Supplemental Material
sj-doc-1-npx-10.1177_1934578X241257089 - Supplemental material for Protective Effect of Corilagin on Alcoholic Liver Injury
Supplemental material, sj-doc-1-npx-10.1177_1934578X241257089 for Protective Effect of Corilagin on Alcoholic Liver Injury by Xudan Li, Xuan Li, Yuan Deng, Lianghua Chen, Zhizhong Zheng and Yanlin Ming in Natural Product Communications
Footnotes
Authors’ Notes
Xudan Li and Xuan Li contributed equally to this work.
Author Contributions
Xuan Li and Yuan Deng performed experiments. Lianghua Chen, Xuandan Li, and Zhizhong Zheng were involved in data analysis. Yanlin Ming designed the outline of the whole experiment. All authors agreed to be accountable for all aspects of the work, ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Experimental Animal Ethics Committee of Xiamen University (No. XMULAC20210054).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Xiamen major Science and Technology Plan Project (3502Z20211006, 3502Z20211005).
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Experimental Animal Guidelines of Xiamen University and approved by the Experimental Animal Ethics Committee of Xiamen University, Xiamen City, China.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
