Abstract
Diabetes mellitus is a major health problem that affects the general population. Commiphora gileadensis (C gileadensis) is a small tree that grows in the Middle East.
Introduction
Diabetes mellitus is a group of diseases associated with elevated blood glucose levels resulting from defects in the body's ability to produce and/or use insulin. It is a metabolic condition that appears with changes in the metabolism of carbohydrates, fats, and proteins. Diabetes mellitus is largely characterized by the degree of hyperglycemia, which increases the risk of microvascular damage (retinopathy and neuropathy). 1 It is linked to a shorter life expectancy, considerable morbidity from macrovascular complications (ischemic heart disease, stroke, and peripheral vascular disease), and a lower quality of life.2,3 Although there are many oral hypoglycemic medications available along with taking insulin, there is still no effective treatment for diabetes. 4 In the twenty-first century, diabetes mellitus has become a major healthcare issue. From the current 150 million cases, it is anticipated that there will be 300 million cases in 2030. 5 T1DM is an autoimmune disease characterized by the progressive invasion of pancreatic β-cells by T-cells destroying these cells. There is strong evidence that both CD4+ helper and CD8+ cytotoxic T lymphocytes play a crucial role in type 1 diabetes mellitus progression. 6 A rapidly expanding field of medicine that needs attention is herbal medicine. For thousands of years, plants have been essential to preserving human health and enhancing the quality of human existence, and they have also been used by people as useful medicinal ingredients.7,8 According to the World Health Organization (WHO), almost 80% of people on Earth rely on traditional medicine for their main healthcare requirements. 9 The majority of this therapy uses plant extracts or their active components. This is justified by the fact that medicinal plants often have advantages in terms of cost and lack of side effects over commonly used pharmaceuticals. 10 More than 1200 plant species are used to treat diabetes mellitus worldwide; many of these plants have been tested in the laboratory and found to have effective hypoglycemic activity.11,12 These medicinal herbs serve as both a dietary supplement to already prescribed treatments as well as a valuable source of oral hypoglycemic substances for the creation of novel pharmaceutical products.13,14 One of those plants that have long been used to treat a variety of ailments is C gileadensis.15,16 A member of the Burseraceae family, C gileadensis is found throughout the Arab world, including Saudi Arabia.17,18 This plant can be found in western Saudi Arabia near the Sarawat Mountains. C gileadensis is known by a number of traditional names in Arab nations, including balm of Makkah, basin, and bisham. In the Hellenistic and Roman eras, C gileadensis balsam was used as a fragrance.19,20 An aqueous extract of this plant was previously employed in a few studies as an analgesic, diuretic, and antihypertensive agent. 18 Moreover, the sap of C gileadensis was used as an antibacterial agent in both in vivo and in vitro studies. 21 Furthermore, a previous study found that the methanolic extract of C gileadensis has antibacterial effects and assists in wound healing. 22 This study aimed to investigate the effect of different extracts of C gileadensis on blood glucose levels, lipid profile, and CD3+, CD4+, CD8+, and CD25+ subsets in the T1DM model.
Materials and Methods
The current study was done at Taif University from March to June 2023 (16 weeks).
Commiphora gileadensis Collection
C gileadensis was collected from a high mountain area called the Alaab Valley, located near the Al-Madinah region of western Saudi Arabia. Leaves and fallen branches were collected in March 2023.
Preparation of Commiphora gileadensis Sap
The growing tips of C gileadensis branches were lopped off 5 mm from the ends, and the running sap was immediately collected upon wounding. Following dilution in an equal volume of ethanol, the sap was centrifuged for 10 min at 10 000 rpm after being agitated for 15 min at room temperature. Then the supernatant was kept at −20 °C until analysis. 23
Preparation of Commiphora gileadensis Acetone Extract
The C gileadensis leaves and branches were dried at 60 °C for 6 hours in a vacuum oven before being broken into small bits with a razor blade to create a powder. Ten grams of raw C gileadensis was immersed in 200 mL of acetone for 3 days at room temperature, with the acetone being replaced every day on a magnetic stirrer. The acetone extract sample was dried using a rotary evaporator to remove the acetone residues. The sample was then stored at −20 °C for subsequent analysis. 24
Experiment Design
Forty male BALB/c mice, 2 months old with 20 to 25 g body weight, were obtained from the Umm Al-Qura University animal house. The mice were housed in a standard rodent cage with woodchip bedding in a large, well-ventilated room with a 12-h light/dark cycle and a temperature of 25 °C. A standard rodent diet and tap water were provided to all mice included in the present study throughout the experiment's duration. After 2 weeks of acclimatization, 40 mice were randomly classified into four groups.
The first group was assigned as a control group. Mice in this group received no treatment. The second group was the untreated diabetic group. The mice in this group were injected with 55 mg/kg body weight of streptozotocin intraperitoneally for 5 consecutive days to induce T1DM. The third group was assigned as the diabetic C gileadensis acetone extract-treated group. T1DM was induced in the mice as in the second group. The mice consumed the same food and water as the negative control group. In addition, they received 200 mg/kg body weight per day of C gileadensis acetone extract via intragastric gavage after 1 week of streptozotocin administration. The fourth group was assigned as the diabetic C gileadensis sap-treated group: all mice in this group, after the induction of T1DM, were treated with C gileadensis sap at a dose of 200 mg/kg per day via intragastric gavage after 1 week of streptozotocin administration.
The mice in all four groups were fed a standard diet for rodents and had unrestricted access to tap water. Random blood glucose levels were estimated at 5-day intervals for all mice included in this study. At the end of the experiment (16 weeks), blood samples were collected to estimate HbA1c and lipid profiles.
Body Weight Measurement
The body weight of each mouse included in this study was measured for 16 weeks using a digital balance (OHAUS, Model: Scout Pro SPU601, made in China). The body weight was estimated for each mouse in the four groups on the first day before streptozotocin administration (0 days), then 2, 4, 6, 8, and 16 weeks after treatment with different C gileadensis extracts.
Blood Collection
Blood samples were collected from the retro-orbital venous plexus of all mice in non-heparinized tubes before and after six days of streptozotocin administration for blood glucose level estimation. Moreover, blood glucose levels were estimated at 3-day intervals after treatment with different extracts of C gileadensis. For HbA1c and lipid profile estimation, a blood sample was collected from each mouse in all groups at the end of the experiment (16 weeks after C gileadensis treatment). All blood samples were immediately centrifuged at 2500 rpm for 15 min, and blood sera were stored at −80 °C until analysis. Moreover, for flow cytometry, a blood sample was collected in an EDTA tube from each mouse by cardiac puncture under anesthesia at the end of the experiment and the blood was kept on ice until further processing.
Estimation of Random Blood Glucose Levels and Glycated Hemoglobin (HbA1c)
Random blood glucose levels were estimated by colorimetric assay. Furthermore, HbA1c was measured for all mice included in this study at the end of the experiment (16 weeks) using a glycohemoglobin kit (POINTE Scientific Inc., USA). 25
Lipid Profiles Estimation
At the end of the experiment, the triglycerides, total cholesterol, and high-density lipoprotein (HDL) were estimated by colorimetric assay for each mouse included in the present study, while the low-density lipoprotein (LDL) was calculated.
Estimation of CD3+, CD4+, CD8+, and CD25+
Preparation of the Samples
The EDTA blood samples were layered over a Ficoll–Paque density gradient medium in a 15-mL conical tube. Without break, the samples were centrifuged at 400 × g for 30 min at a cold temperature (4 °C). The mononuclear cell interface layer was carefully aspirated and transferred to a new tube. The cells were washed twice with PBS and resuspended in staining buffer (PBS containing 2% fetal bovine serum and 0.1% sodium azide).
Flow Cytometry Staining and Analysis
The cell suspension was aliquoted into 100 μL per tube and stained with the following fluorochrome-conjugated monoclonal antibodies:

CD, cluster of differentiation; PE, phycoerythrin; BV510, brilliant violet 510; FITC, fluorescein isothiocyanate; APC-Cy7, allophycocyanin cyanine. Microgram per milliliter (μg/mL)
The staining was performed for 30 min at 4 °C in the dark. After staining, the cells were washed with staining buffer and fixed with 1% paraformaldehyde. Then all samples were left for 30 min at 4 °C for the incubation period, protected from light, washed, and resuspended in FACS buffer. Then, the samples were analyzed on the flow cytometer. 26
Statistical Analysis
For blood glucose levels, HbA1c, and lipid profile statistical analysis was done using SPSS software version 17 (SPSS Inc., Chicago, IL, USA). All data were expressed as mean ± SD. The comparisons of weight, blood glucose levels, and HbA1c between all groups were made with a t-test. The level of significance was set at P < .05. For Flowcytometry FlowJo software (Tree Star, Ashland, and Oregon) analyzed the flow data. For graphing the data, GraphPad Prism 8 (Mac version 8.4.2) was used. The Shapiro–Wilk normality test was performed to ensure the data was normally distributed.
Results
Body Weight
Table 1 represents the weight changes of all groups throughout the 10 weeks. For 10 weeks, the diabetic C gileadensis sap-treated group had the highest weight compared with all groups. Furthermore, the diabetic C gileadensis sap and acetone extract-treated groups had significantly higher weights compared with the untreated diabetic group in the eighth and 16th weeks (P < .05).
Body Weight of Mice in All Groups Throughout 16 Weeks.

* Significant <0.05.
Blood Glucose Levels
Blood glucose levels for all groups throughout the 21st day after streptozotocin administration are represented in Table 2. The diabetic C gileadensis sap-treated group had the lowest blood glucose levels versus all other diabetic treated and untreated groups throughout the 21 days after treatment with C gileadensis extracts. Compared with the untreated diabetic group, the diabetic C gileadensis sap and acetone extract-treated groups had significantly lower blood glucose levels in the 6th, 9th, 12th, 15th, 18th, and 21st days of treatment (P < .01). In the diabetic C gileadensis sap-treated group, normal levels of blood glucose were achieved after 6 days of treatment while diabetic C gileadensis acetone extract-treated group achieved normal blood glucose levels after 15 days of treatment.
Blood Glucose Levels (mg/dL) of Mice in all Groups Throughout 21 Days.

* Significant <0.05.
** Significant <0.05.
HbA1c Percentages
The HbA1c percentages of all groups are represented in Table 3. The diabetic C gileadensis sap and acetone extract-treated groups had significantly lower HbA1c values versus the untreated diabetic group (P < .01).
HbA1c of Mice in all Groups After 16 Weeks.
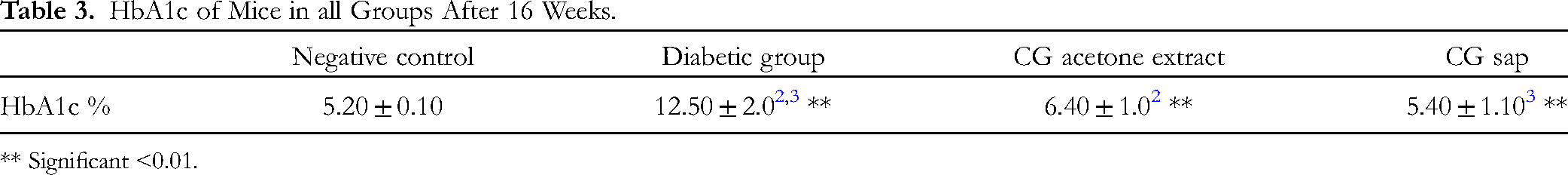
** Significant <0.01.
Lipid Profiles
Lipid profiles for all groups are represented in Table 4. The lowest triglyceride levels were present in the diabetic group treated with C gileadensis sap. The triglyceride levels in the diabetic mice treated with C gileadensis sap and acetone extract were significantly lower compared with the untreated diabetic group (P < .01). According to total cholesterol levels, the diabetic C gileadensis sap-treated group had the lowest level among all groups. Compared with the diabetic group, those diabetic mice treated with C gileadensis sap and acetone extracts had significantly lower total cholesterol levels than those in the untreated diabetic groups (P < .01). LDL levels were significantly decreased in the diabetic C gileadensis sap and acetone extract-treated groups compared with the untreated diabetic group (P < .01). Regarding HDL, the diabetic C gileadensis sap and acetone extract-treated groups had significantly higher levels than the untreated diabetic group (P < .01).
Serum Triglyceride, Total Cholesterol, LDL, and HDL of Mice in all Groups After 16 Weeks.
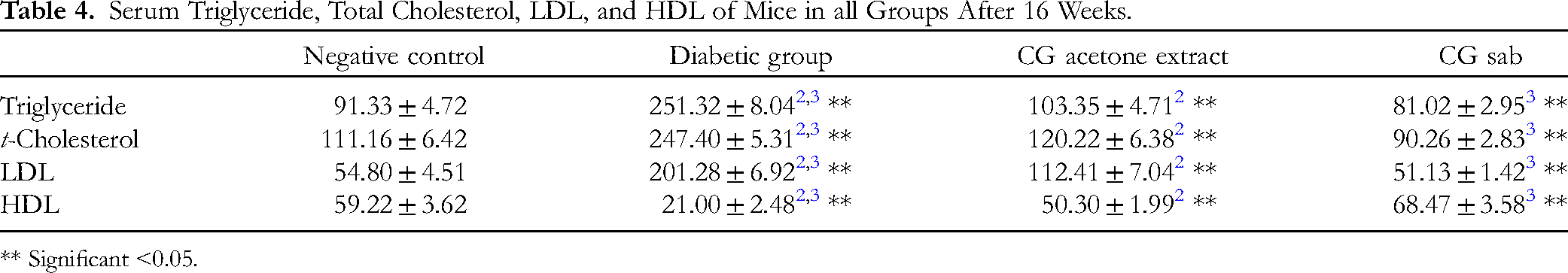
** Significant <0.05.
Peripheral CD3+, CD4+, CD8+, and CD25+ Subsets
Data showed that total lymphocytes were decreased in diabetic groups treated with C gileadensis sap and acetone extract compared with the untreated diabetic group (P < .05) (Figure 1). Data also showed that CD3+ was lower in diabetic mice treated with C gileadensis sap and acetone extract compared with the untreated diabetic groups (P < .05) (Figure 2). CD4+ T cells decreased significantly in the diabetic mice treated with C gileadensis sap and acetone extract compared with the untreated diabetic group (P < .05) (Figure 3). Also, CD8+ T cells decreased in the diabetes groups treated with C gileadensis sap and acetone extract compared with the untreated group (P < .05) (Figure 4). CD4+ CD25+ cells were decreased in all diabetic groups compared with the control group (P < .05) (Figure 5). Furthermore, CD8+ CD25+ cells were increased in diabetic mice treated with C gileadensis sap and acetone extract compared with the diabetic untreated group (P < .05) (Figure 6).

Effects of C gileadensis on total lymphocytes. The percentage of total lymphocytes was measured by flow cytometry.

Effects of C gileadensis on CD3+. The percentage of total lymphocytes was measured by flow cytometry.

Effects of C gileadensis on CD4+. The percentage of total lymphocytes was measured by flow cytometry.

Effects of C gileadensis on CD8+. The percentage of total lymphocytes was measured by flow cytometry.

Effects of C gileadensis on CD4+ CD25+ The percentage of total lymphocytes was measured by flow cytometry.

Effects of C gileadensis on CD8+ CD25+ The percentage of total lymphocytes was measured by flow cytometry.
Discussion
The purpose of the present experimental study was to find out the effect of different extracts of C gileadensis on blood glucose levels in T1DM male mice and the way by which it decreased them. The β-cells in the pancreas of animals that produce the hormone insulin are particularly sensitive to the naturally occurring chemical streptozotocin, which is generated by the bacterium Streptomyces achromogenes. Streptozotocin has particular effects on pancreatic β-cells. Streptozocin is specifically toxic to pancreatic β-cells due to the presence of large levels of glucose transporter 2 (GLUT2) proteins present in these cells. Because of its structural resemblance to glucose, streptozotocin can be uptake by pancreatic β-cells. It is a nitrosourea alkylating agent that induces injury in β-cells through the generation of free radicals and DNA destruction. This is how streptozotocin induces T1DM in mice. C gileadensis sap and its acetone extracts are non-toxic for mice. 18 A previous study found that C gileadensis extract had antioxidant activity due to its content of free radical scavengers such as saponins, flavonoids, volatile oils, sterols, and triterpenes. 27 Elrabey et al found that an aqueous extract of C gileadensis ameliorated altered liver and kidney function to nearly normal levels due to its content of antioxidant compounds that reduced oxidative stress by scavenging free radicals. 28 Moreover, the same study found that the aqueous extract of C gileadensis reduced blood glucose levels in diabetic rats. The present study is in accordance with these studies in that C gileadensis sap and its acetone extracts reduced blood glucose levels. C gileadensis sap had the greatest effect in ameliorating blood glucose levels in the shortest duration compared with the acetone extracts. Furthermore, HbA1c percentages were reduced by C gileadensis sap and its acetone extracts, but the sap gave a lower value. T1DM starts in two steps; antigen-presenting cells (APCs) displaying β-cell antigens trigger autoreactive T lymphocytes in pancreatic lymph nodes. At this stage, effective activation of CD8+ T cells requires CD4+ T cells. 29 These activated T lymphocytes (CD8+) invade the pancreas and destroy β-cells. Furthermore, CD4+ T cells also contribute to β-cells death via activating macrophages.30,31 In the present study, C gileadensis sap and acetone extract reduce total lymphocytes and CD3+ in diabetic mice. More specifically, these extracts reduce CD4+ and CD8+ in these mice. This anti-inflammatory property of C gileadensis may give us evidence that this herb ameliorates β-cells function and lowers blood glucose levels besides its antioxidant activity. The sap of C gileadensis exhibited more efficacy compared to its acetone extract in reducing CD4+ and CD8+ activity. Consequently, it is more active than the acetone extract in reducing blood glucose levels and HbA1c, as indicated by the results. Pancreatic lymph node-derived CD4+ CD25+ T regulatory (Treg) cells reduce diabetes progression by inhibiting islet-reactive CD8+ T cell differentiation into cytotoxic T lymphocytes (loss of tolerance). All diabetic mice showed a reduction in CD4+ CD25+ and these indicate loss of tolerance in all T1 diabetic mice. CD8+ CD25+ T cells prevent T1DM progression and normalized glycemic index. 32 In this study, the number of these cells is higher in diabetic mice received treatment with C gileadensis sap and acetone extract compared to the group of untreated diabetic group. This provides additional evidence of the therapeutic efficacy of C gileadensis in normalizing the glycemic index. Regarding the lipid profile, a previous study found that an aqueous extract of C gileadensis reduced the triglyceride, total cholesterol, and LDL in diabetic rats to normal levels. Regarding HDL, the C gileadensis aqueous extract increased its level in rats. The present study showed that C gileadensis sap and acetone extract reduced triglyceride, total cholesterol, and LDL to their normal levels while increasing HDL levels.
Conclusion and Recommendation
The present study found that C gileadensis sap and acetone extracts lower blood glucose, HbA1c, and lipid profiles in diabetic mice. The study found that this herb has anti-inflammatory properties by lowering CD4+ and CD8+, potentially preventing β-cell death. Additionally, C gileadnesis increased the CD4+ CD8+ population, which may regulate the glycemic index and prevent T1DM progression. The C gileadensis sap is more potent than acetone extract in antidiabetogenic activity which may be due to the high concentration of effective materials (more viscous). A human-based study is recommended to evaluate the antidiabetogenic effect of C gileadensis, as well as an in vitro study of C gileadensis's effect on β-cell cultures to determine the pathway by which this plant reduces blood glucose levels.
Limitation of the Study
The study focused on the peripheral lymphocyte subsets only. Moreover, the study investigates a few lymphocyte subsets (CD4+, CD8+, and CD25+).
Footnotes
Acknowledgments
The authors extend their appreciation to Taif University, Saudi Arabia, for supporting this work through project number (TU-DSPP-2024-270).
Authors’ Contributions
All experiments, data presentation, and writing were done by the authors.
Availability of Data and Material
The datasets utilized and/or examined in the present work can be obtained from the relevant authors upon a reasonable request.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The animal study protocol was accredited by the National Committee for Bioethics at Taif University (protocol code HAO-02-T-105) and the Committee considered that the proposal fulfills the requirements.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Taif University, Saudi Arabia, Project No. (TU-DSPP-2024-270).
Statement of Human and Animal Rights
The corresponding Author declares that the procedures followed were in accordance with the ethical standards of the responsible committee on animal experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000.
Statement of Informed Consent
Not applicable.
