Abstract
Objectives/Background
Natural products are preferred over synthetic drug as fertility enhancers as they are cost effective. Infertility among men of reproductive age keeps rising. Synthetic drugs ameliorate the problem but leave adverse side effects as men advance in age, hence the need for alternative medicine. Tom brown weaning diet is a processed cereal-based powdery blend which is cooked into a semi-liquid food for children from 6 months and contains good nutrient levels. This work focused on the effects of Tom brown weaning diet on testes of Wistar rats.
Method
Fifteen Wistar rats were divided into 3 groups namely: Control group, 50% tom brown group and 100% tom brown group. Control received only rat chow, 50% Tom brown group received tom brown diet and normal rat chows at 12 h cycle per time. This cycle was repeated till the end of the experiment. 50% Tom brown group received only tom brown diet throughout the experimental period. The testes were excised, measured, and processed for morphological and histological analysis.
Results
Average testicular weight and relative weight in the 50% tom brown, 100% tom brown and control groups were not significant at alpha level of P ˂ .05. This was also supported by the normal histological features seen in sections of the testes of rats in all groups as there were no shrinking and denser spermatogenic cells in the seminiferous tubules. Hormonal analysis showed a non-significant difference in the level of FSH and LH at alpha levels of P˂0.05. However, testosterone level increased significantly in 100% group compared to 50% and the control group at P˂ .05. Also, GC-MS study on the diet revealed 30 compounds in the diet.
Conclusion
This study revealed that Tom brown diet has no detectable damaging effect(s) on the testes but could improve the cytoarchitecture and enhance fertility.
Introduction
Infertility in males is often associated with certain factors such as Reactive oxygen species (ROS). As a matter of fact, there exist an association between ROS and varococoele, a condition characterized with abnormal enlargement of the pampiniform venous plexus in the scrotum and a possibly resulting in infertility. 1 According to Shookoohi et al and Alsaikhan et al, it has been reported that about one-third of men with primary infertility and 80% of men with secondary infertility are afflicted with varicocoele.1,2 This further leads to an increase rate of infertility in males. Following the cost of treating certain diseases with orthodox measure, tropics have resulted to herbal measures of handling infertility and its associated disorders. 3 This could be the case of trying locally made diets like tom brown as the disease continues to be on the rise and the cost of treatment using orthodox measure are becoming expensive on a daily. Certain medications that portrays possible favourable outcomes in handling infertility related cases such as Hesperidin as put forward by Shookoohi et al tend to be expensive in the tropical region of the world. 1 Hence, there is a need to find possible herbal or natural remedies in handling infertility related cases. It is on this premise that investigation having to do with the effect of a naturally prepared weaning diet, Tom brown which is popularly consume and affordable in Africa was done on some male reproductive parameters. Again, certain researches has confirmed the efficacy of some natural products in protecting against infertility. However, this protective effect might occur indirectly as they do not have the direct effect of reversing infertility. Abadi et al in their research showed that natural flavonoid such as Chrysin protects against Testicular Apoptosis induced by torsion/detorsion in adult rats. 4 In their research, it was concluded that administration of Chrysin, a natural product of flavonoid existing in plants impedes ischemia/reperfusion damages in testis tissue and improved sperm quality. Tom brown weaning diet contains soybeans, a source of certain form of flavonoids called isoflavones. 5 Therefore, there exists a possibility of the diet being useful in treating infertility.
Diet might affect male reproductive potential, but the biochemical mechanisms involved in the modulation of sperm remain poorly understood. 6 Researches have shown that while certain diets offer protection against infertility, others might possess detrimental effect on fertility. Ferramosca et al claim that western diet is considered a risk factor for male infertility while the Mediterranean diet seems to protect against male infertility. 6 However, little or nothing is known of the effect of most locally prepared African diet on fertility. Tom brown, an African diet used to wean children and often consume by adult is said to be rich in nitrogen and protein. 7 There exist certain controversies regarding the use of protein rich diet on reproductive function in experimental animals. Nassan et al suggested that high protein from Fish consumption is related to a higher probability of live birth following infertility treatment with assisted reproductive technology (ART) 8 while Ferguson et al showed in their research that feeding diets with high concentrations of crude proteins decreased reproductive efficiency in dairy cows. 9 Variations in these researches could be linked to the source of protein used in each case. Again, Proteins of vegetable and grain seems to be more beneficial in increasing fertility rate. Chavaro et al claim that replacing animal protein diets with vegetable sources of protein may reduce ovulatory infertility risk. 10 This further implies that protein rich meals especially those from grains will likely be a remedy for infertility in females even though this fact has not been established for male genders. Tom brown, a diet rich in protein and Nitrogen might be beneficial in improving reproductive function in male animals.
Again, Tom Brown is a popular breakfast cereal in Nigeria that has become a staple in many households. The history of Tom Brown in Nigeria dates back to colonial times when it was introduced by the British. During the colonial era, the British brought various food items to Nigeria, including breakfast cereals like Tom Brown. The cereal quickly gained popularity due to its convenience and nutritional value. It provided a quick and easy breakfast option for both the colonial administrators and the local population. Over the years, Tom Brown evolved and adapted to local tastes and preferences. Nigerian manufacturers started producing their own versions of the cereal, adding flavors like chocolate and vanilla to appeal to the Nigerian palate. Today, Tom Brown is widely consumed across the country and Africa at large. It is often enjoyed with milk, sugar, and additional toppings like nuts or fruits. 11 Its popularity as a weaning meal for children makes it a diet of concern as little or no effect is known on many physiological system. Also, aside the fact that it is been used to wean children, adults also consume Tom brown even when they know little or nothing of its effect on various systems in the body. It is on this premise that this research seeks to find out the effect of Tom brown on some reproductive parameters in males with the aim of assessing the morphology of the testis and certain testicular function. Testicular disorders have long been associated with infertility. 12 Certain dysfunctions associated with infertility such as Klinefelter's syndrome present an endocrine profile of hypogonadism (low –normal testosterone, high Follicle stimulating hormone (FSH) and luteinizing hormone (LH) levels, and undetectable inhibin B consistent with testicular failure). Testicular histology of same Klinefelter's syndrome may exclude extensive fibrosis and hyalination of seminiferous tubules with loss of spermatogonial stem cells. 13 The alteration in the morphology of testis and other functions is associated with all types of infertility showing that testicular function enhances fertility. As a matter of fact, all infertility associated syndromes and diseases usually alters testicular function and to a greater extend, the morphology of the testis.
The testes otherwise known as testicles are two oval shaped organs in the male reproductive system which is homologous with the ovary in female. They are located in the scrotum which is a sac of skin located outside the body anterior to the pelvic region. The testis is made up of seminiferous tubules that are lined by cells (spermatogenic cells) that produce spermatozoa. The testes also produce a hormone called testosterone. This hormone is responsible for sex drive, fertility, and the development of muscle and bone mass. Studies on the effect of ingesting tom brown on the testes function are becoming interesting to be conducted since Structures within the testes are important for the production and storage of sperm until they are mature enough for ejaculation and children has tendencies to develop testicular dysfunction that could cause infertility in the future. Gate et al have shown the precedent of undescended testis in children and possible ways of managing such condition In order to prevent infertility. 14
Again, the role of cytoarchitecture of organs is significant in determining the healthy status of such organs. Cytoarchitecture, the study of cellular composition of the physiological system's tissues under the microscope is one way to parse the brain, by obtaining sections of the brain using microtome and staining them with chemical agents which reveal where different neurons are located. 15 This procedure is not only restricted to the brain but other tissues with the genitals inclusive. Marettova et al revealed the effect of pyrethroids on female genital system via assessment of certain hormonal activity and cytoarchitecture of reproductive organs via staining procedures. 16 This further goes to show that alteration of the cytoarchitectures of organ could be an additional way of assessing the functionality of such organs rather than doing only an assessment of the physiological secretions from such organs. Intra peritoneal salinomycin administration at 1, 3 or 5 mg/kg for 28 days affects histoarchitecture of testis and epidydmis, induces testicular dysfunction, fails spermatogenesis, suppresses steroidogenesis and leads to infertility in mice. 17 This particular research was carried following the assessment of histological presentations of the testis and assessment of hormonal secretion, further showing how important cytoarchitectural study is in assessing the function of organs.
Materials and Methods
Materials
The materials used for this experiment includes;
Fifteen (15) adult male albino wistar rats, wooden cages with iron netting, saw dust, animal feed, tom brown, distilled water, weighing scale, dissecting set and board, measuring cylinder and specimen bottles, syringes and orogastric, cotton wool, 10% formalin, Bouin's fluid, graded alcohol, ethanol, xylene, paraffin wax, Haematoxyllin and eosin stain (H & E), cover slips, light microscope, water bottles, calibrated beakers, laboratory coat and gloves, isoflurane (Escain ®), graded alcohol(70%, 95% and absolute), hot plate, glass slides and rack, & Disterene Plasticizer Xylene (DPX), desiccator, hand gloves, methylated spirit, nose mask, feeding plates.
Breeding/Grouping of Animals
Fifteen (15) adult male albino wistar rats were purchased from the Department of Biochemistry Animal House, University of Calabar, Calabar. These animals were kept in the animal house of the Department of Human Anatomy, Faculty of Basic Medical Sciences, Arthur Jarvis University. The animal house was brightly lighted, aired (Ventilated) and kept at a conducive environmental temperature between 28 °c to about 33 °c. Apart from being always swept, dusted and disinfected, the animal house was well protected.
The wistar rats were fed with grower's mash from Premier Feed Mills Co. Ltd, with the aid of feeding through (plates) and water bottles and were allowed to acclimatize for a period of 14 days. After the two weeks (14 days) of acclimatization, the rats weighed between 93.4 and 163.0 g. They were then randomly selected into three groups each with five rats in well labeled cages. The groups were labeled Control, 50% tom brown group and 100% tom brown group. Control group received only normal rat chow, 50% tom brown group received tom brown weaning diet for 12 h daily and normal rat chow for the remaining 12 h of the day interchangeably while 100% tom brown group were fed with only tom brown throughout the period of administration.
Test Substances (Tom Brown)
The edible substances used in the preparation of Tom brown were purchased from Watt market in Calabar, Cross River State, Nigeria. They include; Zea mays (yellow corn), Sorghum bicolour (guinea corn), Glycine max (soya beans), Pennisetum glaucum (millet), Arachis hypogeal (groundnut), Phoenix dactylifera (date), Cambrus species (crayfish).
The preparation was done in a percentage (%) ratio of 29:30:14:14:6:5:2 respectively. The grains were carefully selected in order to separate dirt. Thereafter, they were washed and air dried. Yellow corn, guinea corn, soya bean, millet, groundnut and crayfish were roasted at temperature of 70 °C for a period of ten (10) minutes, grinded into particulate form using the Chester Edu Smart 820 CNC milling machine and used as feed for the experimental animals. The weight of the different food substances was weighed before and after roasting. Then, the animals were divided into three groups namely; Control, Combined (50% Tom brown and 50% rat-chow) and Tom brown group. Control group received Normal rat-chow, combined group received 50% Tom brown and 50% rat-chow mixed together while Tom brown group received 100% of Tom brown feed for a period of one month.
GC-MS Analysis of Tom Brown
Analysis of ligand composition of Tom brown feed was done as reported previously by Ebana et al, 18 using a GCMS instrument. Exactly 10 g of the milled Tom brown feed was added to 20 ml of methanol and the resulting mixture was placed on an orbital shaker for 15 min to shake it. Thereafter, the mixture was allowed to stand for another 15 min. The mixture was then filtered and the filtrate was concentrated to 5 ml using a rotator evaporator. From the concentrate, bioactive compounds were screened using Agilent 5890N gas chromatography fitted with an auto sampler connected to an Agilent mass spectrophotometric detector. All the operating conditions were as previously reported. The bioactive compounds in the Tom brown feed were identified and interpreted using the database of National Institute of Standards and Technology (NIST). 18
Experimental Design, Animal's Groups and Maintenance
Fifteen albino wistar rats were used for this research. The rats were weighed at the start of the experiment, and were randomly distributed into their metabolic cages. The animals were separated into 3 groups (Control, 50% tom brown group, and 100% tom brown group). Each group had 5 animals each. Control group received only normal rat chow, 50% tom brown group received tom brown weaning diet for 12 h daily and normal rat chow for the remaining 12 h of the day interchangeably while 100% tom brown group was fed with only tom brown throughout the period of administration. The animals were maintained in the animal house of the Department of Human Anatomy, Arthur Jarvis University, at a temperature of 28 degrees Celsius for 12 h’ light and dark cycles. The cages were always kept clean. Food and water intake was determined daily with the aid of an electronic weighing balance.
Determination of Body Weight
The body weight of the animals was determined by using weighing balance. The initial weight of each animal was taken after separating them into the three (3) respective groups before commencement of the administration. The rats were subsequently weighed daily until the end of the study.
Termination of Experiment
The animal sacrifice was done a day after the last dose of administration was carried out. Under isoflurane (Escain ®) at a concentration of 5% with airflow used as a carrier gas for 1 min, animals were anaesthetized. The anterior abdominal wall was incised using surgical blade, scissor and scalpel. The testes were accessed, removed immediately and fixed in (10%) formalin for histological analysis. The samples were collected based on their groups.
Morphological Examination of Testis
Weighing of Testes and Collection of Samples
The testes were dissected and cleaned of fat and connective tissue. They were then weighed to the nearest 0.001 g with an electronic scale (Mon Scientific Electronic scale MS) and their morphology was grossly evaluated.
Tissue Processing
The tissues were fixed in 10% Formalin fluid for 24 h. The main aim of fixing the tissue was to preserve the tissue and to protect the tissue during subsequent histological processes.
At the end of the 24 h, the tissues were transferred through increasing grades of dehydrating agent. The dehydrating agent used was alcohol (75, 90% and 100%).
The procedure involves;
Two changes of 75% alcohol for 1 h each
Two changes of 90% alcohol for 1 h each
Two changes of absolute (100%) alcohol for 1 h each
Clearing was done to remove the alcohol from the tissue using xylene.
The dehydrated tissues were placed in xylene, 2 changes, one hour each.
The tissues were then infiltrated in an oven at a temperature of 60 degrees Celsius using molten paraffin. The tissues were thereafter embedded in a trough contain molten paraffin wax. This was done twice each lasting for 1 h each. The tissues were then transferred from the final bath into Aluminum moulds filled with molten paraffin wax, with the tissues being inverted so as to face the section to be cut and to free the surface of air bubble. The tissue was trimmed leaving the paraffin wax, 3 mm thick around the tissue to ease sectioning.
A rotary microtome was used to make sections of the tissues. The ribbons of the sections were gently lowered onto the surface of a warm water bath. The floated sections were mounted on slides and dried in the oven at 60 degrees Celsius, for one hour before staining.
Tissue Staining
The staining of tissue sections was done using Haematoxylin and Eosin method. The slides were warmed with hot plate and treated with xylene to remove the wax for 5–10 min. Dehydration of tissues was done through descending grades of alcohol which were two changes of absolute alcohol for 5 min and then transferred to 95% alcohol for 5 min and subsequently to 70% alcohol for another 5 min.
The tissue sections were washed in running tap water for 10 min after which they were stained in Haematoxylin for 15 min. Excess Haematoxylin was rinsed off and differentiation was done quickly using acid alcohol. This differentiator helps the nucleus absorb the stain.
Bluing of sections was done under running tap water for 30 min. It gives the stain its characteristic background. The slides were then counter stained with 1% aqueous Eosin for 5 min. Sections were passed through ascending grades of alcohol (70%, 95% and then two changes of absolute alcohol) for dehydration.
Finally, sections were cleared in two changes of xylene and mounted on DPX and then covered with a cover slip. The slides were left to dry before viewing under the microscope and photomicrograph were taken.
The slides were carefully studied under low (x100) and high (x400) magnification of the light microscope. Comparisons were made with the slides of the control group for detection of histological changes.
Serum Hormonal Assessment
FSH, LH, and Testosterone were determined through method of ELISA. Blood was collected from the animals through Cardiac Puncture. The blood was introduced into a sample bottle which was further spun in a hematocrit centrifuge at 3000 revolution per minute (3000RPM) to get the serum. The serum was used for the hormonal assessment. Thereafter, LH, FSH, and testosterone levels were examined in the serum using commercially available ELISA kits (Elabscience) in line with the protocol of the manufacturer.
Ethical Approval
The research was approved by the Medical Research Ethics Committee, Faculty of Basic Medical Science, Arthur Jarvis University with number 000345 (MREC-FBMS-000345). Animal welfare was taken care while performing the experiment.
Statistical Analysis
Data are presented as mean ± SEM. One-way analysis of variance (ANOVA) was used for accessing differences among groups. This was followed by post hoc paired comparison using Window SPSS version 20.00. P < .05 was taken as indicating statistically significant differences.
Results
Analysis of Phytochemical Ligands in Tom Brown Extract
The phytochemical ligands found in Tom brown extract from GC-MS instrument were thirty (30) in numbers. The ligands are shown below (Table 1). Figure 1 shows the GC-MS chromatogram (abundance vs retention time) of the bioactive compounds from Tom brown extract. The bioactive compounds are represented by the various peaks. The peaks were 30, representing a total of 30 bioactive compounds. The retention times of the various bioactive compounds as well as their concentrations are also shown in Table 1. The table shows the retention time (min) of the 30 bioactive compounds, their molecular weights, peak areas (%) and development (min). The retention time ranged from 8.358 to 26.761 for compounds 4-methyl itaconate and OctadecaNoic acid. On the other hand, the molecular weights of the various compounds ranged from 112 to 660.04 g/mole for Butanedioyl dihydrazide and phytic acid, respectively. Peak area shows that lupeol was the most abundant with a peak area of 25.69 while Beta-caryophyllene had the least peak area with a value of 0.03.

Comparison of initial and final weight of the rats in the different experimental groups. Values are expressed as mean ± SEM, n = 5.
Phytochemical Ligands of Tom Brown Feed Extract.

(Key: R.T, Retention time; M.W, Molecular weight; Dev, Development).
Average Body Weight of Animals
Percentage body weight change was observed to increase in all groups. There was no significant (P > .05) difference in the weight changes in both the control and treated groups. The initial weight, final weight, change in weight and percentage change in weight for the control and treated groups are indicated in Table 2 and Figure 1 respectively.
Mean Body Weights (Initial, Final and Change) in the Different Groups.

Values are expressed as mean ± SEM, n = 5.
The p-value for the change in body weight is 0.285. Therefore, there is no significant difference in the change in body weight at P < .05, for all the groups.
Morphological Observation
Average Testicular Weight and Relative Weight
Average testicular weight is shown in Table 3 and Figure 2. The result showed a non-significant difference (P > .05) in the average testicular weights of rats in the 50% T.B. group (2.46 ± 0.29) and 100% T.B. (2.24 ± 0.35) compared to the control group (2.10 ± 0.27).

Comparison of average testicular weight and relative weight. Values are expressed as mean ± SEM, n = 5.
Mean Testicular Weights and Relative Weight of the Different Groups.

Values are expressed as mean ± SEM, n = 5.
The average testicular relative weight was calculated by dividing the weight of the testis of an animal by the body weight of the animal. The result showed a non-significant difference (P > .05) in the average testicular relative weights of rats in the 50% T.B. group (0.015 ± 0.002) and 100% T.B. (0.015 ± 0.003) compared to the Control group (0.014 ± 0.002).
Testicular Dimensions
Results of testicular dimension are shown in Table 4 and Figure 3. From the measurement of the testis, the results also showed a non-significant difference (P > .05) in the length (cm) of the testis between the various groups. The lengths of the testis in the various groups are; control group (1.68 ± 0.20). 50% T.B. group (1.68 ± 0.20) and 100% T.B. (1.93 ± 0.21)

Comparison of length, breadth and volume of testis in the different experimental groups. Values are expressed as mean ± SEM, n = 5. A (100 Magnification). B (400 Magnification).
Testicular Dimensions of Experimental Groups.
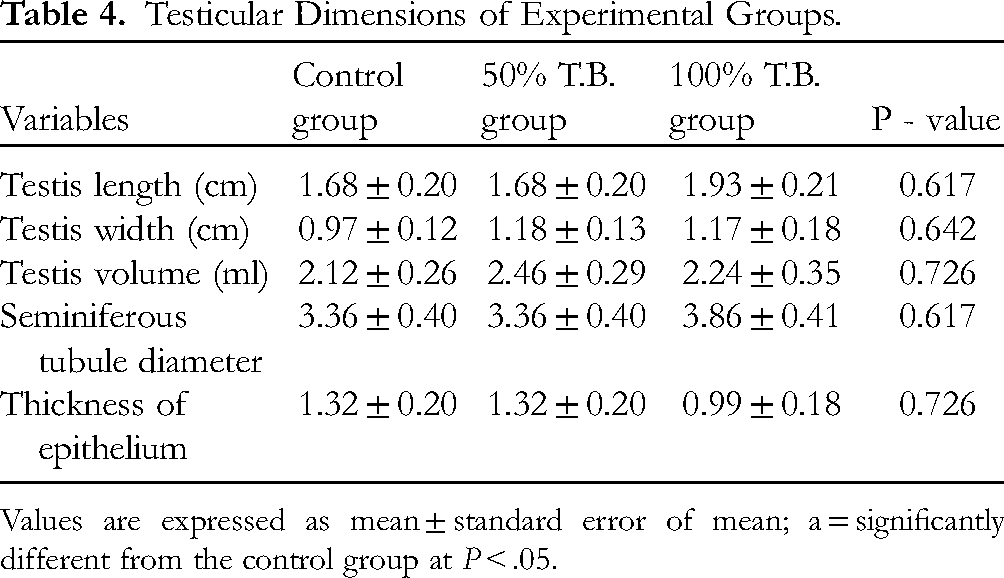
Values are expressed as mean ± standard error of mean; a = significantly different from the control group at P < .05.
From the result, the width (cm) of the testis for the control group is 0.97 ± 0.12. There was no significant difference in the width of the testis of the 50% T.B. group (1.18 ± 0.13) and the 100% T.B. group (1.17 ± 0.18) compared to the control.
The volume (ml) of the testis, from the result, showed a non-significant difference between the 50% T.B. (2.46 ± 0.29) and 100% T.B. (2.24 ± 0.35) groups compared with the control group (2.12 ± 0.26).
Histological Staining
H&E staining of the Control's testis at 100 and 400 magnification (Figure 4) shows seminiferous tubules with many cell layers of germinal epithelium (G), numerous Sertoli cells (S), numerous spermatozoa (SP), basement membrane (M), and interstitial space with Leydig cell (L) and normal capillary (C).

Histological presentation of testis in the control group. [The scale bar equals 10 µm in A and B. A shows 100 magnification while B shows 400 magnification of the testis in the control group) G = germinal epithelium, S = Sertoli cells, SP, Spermatozoa, M, Basement membrane, L, Interstitial cell of Leydig and C, Normal capillary]. A (100 Magnification) B (400 Magnification).
In the 50% T.B group, H&E staining of the testis at 100 magnification showed Seminiferous tubules with mild proliferation of cells of the germinal epithelium (G), numerous Sertoli cells (S), numerous spermatozoa (SP), disruption of basement membrane (M), and interstitial space with Leydig cell (L) and moderate enlargement of capillary (C). However, 400 magnification examination of the tissue under the microscope showed Seminiferous tubules with mild proliferation of cells of the germinal epithelium (G), numerous Sertoli cells (S), numerous spermatozoa (SP), and interstitial space with Leydig cell (L) and moderate enlargement of capillary (C) (Figure 5).

Histological presentation of testis in the 50% T.B group [the scale bar equals 10 µm in A and B. 100 magnification showed Seminiferous tubules with mild proliferation of cells of the germinal epithelium (G), numerous Sertoli cells (S), numerous spermatozoa (SP), disruption of basement membrane (M), and interstitial space with Leydig cell (L) and moderate enlargement of capillary (C). However, 400 magnification examination of the tissue under the microscope showed Seminiferous tubules with mild proliferation of cells of the germinal epithelium (G), numerous Sertoli cells (S), numerous spermatozoa (SP), and interstitial space with Leydig cell (L) and moderate enlargement of capillary (C)]. A (100 Magnification) B (400 Magnification).
In the 100% T.B group, H&E staining of the testis at 100 magnification showed Seminiferous tubules with marked proliferation of cells of the germinal epithelium (G), numerous Sertoli cells (S), numerous spermatozoa (SP), disruption of basement membrane (M), dilations (D), and interstitial space with Leydig cell (L) and marked enlargement of capillary (C). However, 400 magnification examination under the microscope showed Seminiferous tubules with marked proliferation of cells of the germinal epithelium (G), numerous Sertoli cells (S), numerous spermatozoa (SP), dilation (D), and interstitial space with Leydig cell (L) and moderate enlargement of capillary (C) (Figure 6).

Histological presentation of testis in the 100% T.B group [the scale bar equals 10 µm in A and B. H&E staining of the testis at 100 magnification showed Seminiferous tubules with marked proliferation of cells of the germinal epithelium (G), numerous Sertoli cells (S), numerous spermatozoa (SP), disruption of basement membrane (M), dilations (D), and interstitial space with Leydig cell (L) and marked enlargement of capillary (C). However, 400 magnification examination under the microscope showed Seminiferous tubules with marked proliferation of cells of the germinal epithelium (G), numerous Sertoli cells (S), numerous spermatozoa (SP), dilation (D), and interstitial space with Leydig cell (L) and moderate enlargement of capillary (C)].
Hormonal Analysis
The result for the hormonal assay showed a non-significant (P > .05) difference in the level of testosterone (ng/mL) in the 50% T.B. group (0.91 ± 0.012) and 100% T.B. group (0.94 ± 0.010) compared to the control group (0.89 ± 0.019) (Figure 4 and Table 5).
Hormonal Analysis.
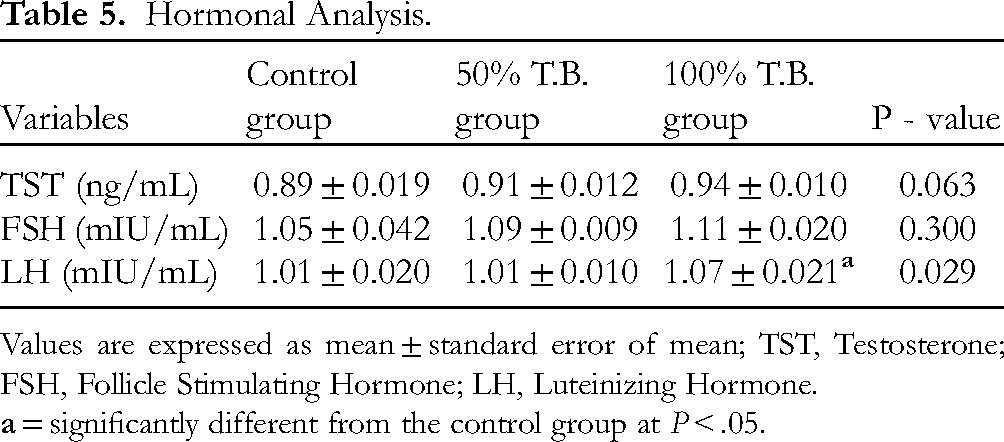
Values are expressed as mean ± standard error of mean; TST, Testosterone; FSH, Follicle Stimulating Hormone; LH, Luteinizing Hormone.
The result also showed shows a non-significant (P > .05) difference in the level of follicle stimulating hormone (mIU/mL) in the 50% T.B. group (1.09 ± 0.009) and 100% T.B. group (1.11 ± 0.020) compared to the control group (1.05 ± 0.042) (Figure 4 and Table 5).
For the serum level of luteinizing hormone (mIU/mL), the result also showed a non-significant (P > .05) difference for 50% T.B. group (1.01 ± 0.010) compared to the control group (1.01 ± 0.020). But there was a significant (P < .05) increase in 100% T.B. group (1.07 ± 0.021) compared to the control group (Figure 4 and Table 5).
Linear Correlation Between the Hormones
The linear relationship or correlation of Testosterone, Follicle Stimulating hormone and Luteinizing hormone among the rats of the various groups was analyzed using Pearsońs correlation. The result of the analysis showed that only Testosterone had a significant (P < .05) positive correlation with Follicle Stimulating Hormone (FSH) and Luteinizing hormone (LH) with Pearson’s correlation of 0.769 (P = .003) and 0.765 (P = .004), respectively (Table 6).
Correlations.

Correlation is significant at the 0.01 level (2-tailed).
Bioavailability of Tom Brown Diet
Calcium, nitrogen, iron, and sodium bioavailability for Tom brown weaning diet (TB) and normal rat chow (NC) according to Amah et al are presented in Tables 7 to 10. 19
Calcium (Ca) Bioavailability of Tom Brown (TB) and Normal rat Chow (NC) in the Food Consumed (FC) and the Fecal Matter (FM) of Experimental Animals.

Different alphabet (
Nitrogen (N) bioavailability of Tom brown (TB) and Normal rat chow (NC) in the food consumed (FC) and the fecal matter (FM) of experimental animals.

Same alphabet (
Iron (Fe) bioavailability of Tom brown (TB) and Normal rat chow (NC) in the food consumed (FC) and the fecal matter (FM) of experimental animals.

Same alphabet (
Sodium (Na) Bioavailability of Tom Brown (TB) and Normal rat Chow (NC) in the Food Consumed (FC) and the Fecal Matter (FM) of Experimental Animals.

Different alphabet (
Biological Value of Tom Brown
The biological value of Tom brown is such that it contains 21.8 g of protein, 0.02 g of salt, 11.7 g of fibre, 46.2 g of carbohydrate of which 2.4 g is made of sugar, 2.3 g of fats of which 2.1 g is mainly saturated fats and 1699KJ/406Kcal of energy. This was assessed using 100 g of Tom brown diet. 20
Discussion
The knowledge of the effect of certain meal is essential for the maintenance of health. This notwithstanding, the component of such meal should be considered in order to ascertain what constituent is essential in maintaining health. Reactive oxygen species and free radicals levels are often linked to disruption of cellular activities involving male reproduction. 21 As such, antioxidant and free radicals scavengers become a necessary remedy to fight off this disruption in cellular activities and further restore fertility and sperm production levels in men. 21 There exist several diets that portray detrimental effect on various systems of the body, yet they are still been consumed on a daily. The possible mechanism of this diet which could likely cause the detrimental effect is a function of its constituent. Results from this study have shown the abundance of certain fatty acid in Tom brown with tendencies to play an antioxidant function in the body. Pentadecanoid acid as proposed by Chowdhury et al is found not to be associated with any cardiometabollic disease or dysfunction in any system of the body. 22 Rather, it enhances the proper functioning of various system following its antioxidant property. Again, Fuhrman exposes the hidden dangers of fast processed food. 23 These dangers often start with obesity and lead to other disorders of various body system affecting even the reproductive system. Reproduction is an important aspect of physiological system as it is concerned with procreation. Adler et al have emphasized in their research the changes in the frequency and type of barriers to reproductive health between 2017 and 2021 further showing how important Reproductive health is in daily biological processes. 24 As applicable in every system of human body, a disorder often leads to pathological conditions. Most of these disorders are a result of diets, lifestyle or certain ligands which could be in form of drugs and herbs used by individuals. In as much as drugs, herbs and diets could cause certain disorders, they also have a potential of curing certain disorders depending on the usage. A case study is that of magnesium which is said to be an effective measure for managing COVID-19. 25 However, other researches claims that magnesium might be a potential cause of discomfort in gastrointestinal tract following its decreased motility effect on the intestine. 26 Herbs like ginger have shown possible beneficial effect in treating inflammatory disorder and liver injuries when compared to its counterpart medicinal agent, aspirin.3,27 In Reproduction, drugs especially those which contains nicotine could have serious effect on fertility rate of women. This agent can cause adverse effect on pregnancy and birth. Also, drugs and medication may impair male fertility through basic mechanisms: direct gonadotoxic effects, alteration of the hypothalamic-pituitary-gonadal axis, impairments in ejaculation and erectile function, adverse effects on libido 28 and release of free radicals, reactive oxygen species and apoptosis.21,29 Therefore, one mechanism in which drugs, herbs or diet could restore the earlier damage could be by lowering reactive oxygen levels, which will in turn reduces the oxidative stress and testicular damage. A case study is seen in a research by Dolatkhah et al. 29 In the research, Fumaria parviflora was found to alleviate varicocele, possibly by lowering oxidative stress and testicular damage. Often times, the alterations in reproductive functions are seen when assessment of reproductive hormones such as testosterone, FSH, estrogen and LH etc. Harringtom et al 30 have categorically specified that reproductive hormone are essential for health and well-being of a mare or stallion, it could be referred to the hallmark of assessing reproductive functions in animals. Cases where there are alterations in these hormones will usually point to disorders of the reproductive system. In this research, Tom brown experimental groups showed mostly non-significant difference in these hormones compared to the Control. However, the 100% Tom brown experimental animals showed a significant increase in LH compared to the control group. LH is synthesized and secreted by the anterior pituitary gonadotrophs in response to the pulsatile secretion of hypothalamic gonadotropin releasing hormone (GnRH). It binds to specific receptors localized primarily in the ovary and testes. It is further required for synthesized of androgens in the ovary by follicular theca cells further resulting in ovulation. However, in males, LH is the key factor regulating androgen production by interstitial Leydig cells. 31 LH is used as part of a treatment plan for infertility in women. 32 The use of LH in treating infertility supports the fact that Tom brown diet which showed increased LH level in this research work will serve the function of ameliorating infertility in both male and female gender if this result is applicable to humans. Further evidence is shown in the H&E staining results of this research. Numerous spermatozoa and mild changes in the testis of the Tom brown experimental groups also points to the fact that Tom brown will enhanced fertility rather than disrupt it. Nallella et al have shown that sperm motility and concentration provide more accurate information than morphology during infertility evaluation. 33 The concentration of sperm is also affected by the number of spermatozoa present following examination. Cases of numerous spermatozoa enhances fertilization unless in deformed spermatozoa. 33 Morphological examination of Tom brown experimental groups also showed no significant difference compared to the control. This further shows the possible effect of Tom brown in enhancing fertility. Also, (Z)-octade-9-enoic acid, Pentadecanoic acid, Tetra-decanoic acid and 15-Hydroxypentadecanoic acid may be the possible constituent of Tom brown that enables it plays an important role in enhancing male fertility as studies have shown that fatty acids of omega −3 may be essential in improving male fertility. 34 Recently, branched fatty acids esters of hydroxyl fatty acids have been discovered to have anti-inflammatory, anti-diabetic and even antioxidant functions. 35 It is further suggested that these fatty acids mediates their antioxidant actions following induction of cytoprotective proteins against oxidative stress and induced cellular damage. 35 (Z)-octade-9-enoic acid, Pentadecanoic acid, Tetra-decanoic acid and 15-Hydroxypentadecanoic acid constitute some branched fatty acids found in Tom brown. It is therefore worthwhile to conclude that Tom brown's protective effect on the testis is a function of these fatty acids following their various antioxidant properties as they induce the release of certain cuytoprotective proteins against oxidative stress and induced cellular damage following the release of free radicals and reactive oxygen species. Therefore, the use of Tom brown can be recommended as a diet for patients who under certain infertility dysfunction following an increase reactive oxygen species. This is because this diet will rather offer an antioxidative effect on their system following its rich nutrient and antioxidant property. However, there is room for further investigation such as docking procedures of various ligands seen in Tom brown with certain proteins and reactive oxygen species to ensure which of these ligands of Tom brown is most effective in enhancing fertility in males (Figure 7).

Comparison of Serum Hormone levels in the different experimental groups. Values are expressed as mean ± standard error of mean; TST, Testosterone; FSH, Follicle Stimulating Hormone; LH, Luteinizing Hormone. a, significantly different from the control group at P < .05.
Limitation of the Study
The study was limited to parameters of male reproductive system. Further studies should be done on fertility parameters of the female reproductive organ. Docking studies could also be performed to find out specific phytochemicals of Tom brown that causes its effect on reproduction. More molecular studies will be needed to determine the mechanism of action of Tom brown as pertaining to the signaling pathway or gene expression. The efficacy of the diet will also need to be determined.
Conclusion
Tom brown diet has no detectable damaging effect(s) on the testes but could improve the cytoarchitecture and enhance fertility. However, its effect can be said to be dose-related and usage should be controlled. (Z)-octade-9-enoic acid, Pentadecanoic acid, Tetra-decanoic acid and 15-Hydroxypentadecanoic acid may be the possible constituent of Tom brown that enables it play an important role in enhancing male fertility.
Footnotes
Author Contributions
Victor Kanu Uno: Conceived the experiment and supervised the work
Justina Nwandimma Nwangwa: Co supervised the work and made provision of resources
Ekementeabasi Aniebo Umoh: Conceptualization and manuscript writing
Atim-Ebim Michael Raymond: Data analysis and Contributed reagents, materials, analysis tools or data
Joseph Okon Asuquo: Contributed reagents, materials, analysis tools or data
Effiom-ekaha Otu Otu: Contributed reagents, materials, analysis tools or data
Maxwell Inawerebua Beniangba: Contributed reagent, fed the animals and administered
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The research was approved by the Medical Research Ethics Committee, Faculty of Basic Medical Science, Arthur Jarvis University with number 000345 (MREC-FBMS-000345).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Animal Rights
All the experimental procedures involving rats were conducted in accordance with the International Animal Care guidelines of Arthur Jarvis University, Nigeria and were approved by the Medical Research Ethics Committee, Faculty of Basic Medical Sciences, Arthur Jarvis University.
