Abstract
Introduction
Malaria continues to be one of the most significant global health challenges, posing a serious threat to human populations worldwide. Despite global efforts to eradicate it, malaria remains endemic in many parts of the world, particularly in the undeveloped tropics and subtropics.1–5 Based on the latest Malaria Report from the World Health Organization, the disease caused an estimated 627 000 fatalities and 241 million cases worldwide in 2020, making malaria one of the most serious life-threatening infections. 6
Recently, a breakthrough in the fight against malaria occurred with the approval of Mosquirix, a vaccine developed to combat this infectious disease. Mosquirix, also known as RTS,S/AS01, represents a pioneering milestone as the first and currently the only licensed malaria vaccine. This vaccine targets the Plasmodium falciparum parasite, the most prevalent and lethal species of malaria-causing parasites affecting humans.1–3 In this context, while the approval of Mosquirix marks a significant advancement in the fight against malaria, there remains a continued need for complementary approaches and the development of additional effective vaccines or therapeutic interventions to further combat this pervasive disease.1–5
Parasites of the genus Plasmodium are responsible for human malaria. The most lethal strain among several species of obligate intracellular protozoa is P. falciparum, which is primarily transmitted by female Anopheles mosquitoes..7,8 During the intra-erythrocytic phase of the parasite life cycle within the human host, the parasite digests the host hemoglobin as a resource of amino acids. This digestion occurs to be in the food vacuole of the parasite, where the pH is typically maintained between 5.0 and 5.4.9,10 As a result, significant levels of ferriprotoporphyrin (IX) (FePPIX), a highly active monomeric free heme, build up and can cause cellular oxidative stress that kills parasites.11,12 The ferric heme, a byproduct of hemoglobin digestion by the parasite, undergoes polymerization within the parasite to form hemozoin or malaria pigment. This insoluble, stable, inert, crystalline pigment appears as a black-brown substance. This process serves as a survival mechanism against the harm posed by the heme to the parasite.10,13,14 A synthetic crystalline counterpart of hemozoin, known as β-hematin, is utilized as a tool in the search for potential new antimalarial drugs. The need for such drugs is crucial due to the emergence of drug-resistant strains of Plasmodium, including resistance to widely used drugs like chloroquine.15,16 Moreover, the improper use of antimalarials has contributed to common malaria parasite resistance to many drugs on the market. 17
Throughout history, plant-based medications have been utilized for medical purposes, and many pharmaceutical therapies still use them today. The history of antimalarials is closely linked to the history of herbal medicines. 18 Achillea species, including Achillea fragrantissima (A. fragrantissima), have a long history of use in traditional medicine for the cure of various ailments. These plants have been employed to address respiratory diseases, smallpox, eye infections, fever, headache, gastrointestinal disturbances, diabetes, dysmenorrhea (painful menstruation), as well as fatigue. A. fragrantissima, specifically, is renowned for its medicinal properties and is commonly utilized in traditional healing practices. A. fragrantissima is a biologically active plant that exhibits therapeutic benefits that include antioxidant, anti-inflammatory,19–21 and antimicrobial activity. 22
The genus Achillea L. contains the flowering plant A. fragrantissima, which is in the Asteraceae family. A. fragrantissima is a small shrub characterized by its white-woolly appearance and strong aromatic fragrance. This A. fragrantissima is native to North Africa and the Middle East. It can only be found in deserts and semideserts. 22 It is rich in various bioactive compounds, including phenolics, flavonoids, tannins, essential oils, sterols, and triterpenes such as achillolide A, cirsimaritin, chrysoplenol, swertisin 2-arabinosideafroside, cirsiliol, eupatilin-7-methyl ether, and isovitexin 4-methyl ether. It also contains unsaturated amides and sesquiterpene lactones.20,23 Notably, achillolide A, a sesquiterpene lactone present in A. fragrantissima, exhibits strong anti-inflammatory properties, acts as a scavenger of reactive oxygen species, as well as inhibits the formation of nitric oxide (NO). 24 A variety of water-soluble polyphenols were identified in water-alcohol extracts of A. fragrantissima's above-ground parts. 22
The ethanolic and aqueous extracts of A. fragrantissima are well-tolerated substances with a large margin of safety. The various extracts from A. fragrantissima were repeatedly administered over an extended period of time with no adverse effects being observed, 25 or toxicity. 26 The presence of polyphenolic compounds, including flavonoids, phenolic acids, and tannins, is prominent in various Achillea species. Furthermore, in a previous study, 27 it was found that hydro-alcoholic extracts had higher total phenol content compared to aqueous extracts. A similar trend was observed with flavonoid content. 27 The major objective of the current study is to examine the potential of antimalarial activity of water and alcoholic extracts from A. fragrantissima in inhibiting the formation of β-hematin using an in vitro semiquantitative assay.
Materials and Methods
Plant Material
Leaves or aerial, stems, and flowers of A. fragrantissima were collected from Bani Naim wilderness in Hebron, Palestine, during the period between May and June 2021 with voucher number (Pharm-PCT-272). After separating the leaves as well as flowers from the stems, tap water was used to clean the A. fragrantissima plants. The samples were dried naturally at room temperature, away from direct sunlight, for approximately 9 days. After drying, they were ground into a powdered form and stored in a refrigerator.
Chemicals
Glacial acetic acid was obtained from Fluka, while ethanol (EtOH) solvents were purchased from Merck (Germany). Dimethyl sulfoxide (DMSO) with a purity of 99.5% chloroquine diphosphate salt, sodium acetate with a purity of 99%, and hemin chloride were all obtained from Sigma-Aldrich.
Extract Preparation
To prepare the extracts, 2 g of dried powder obtained from A. fragrantissima leaves, stems, and flowers were separately soaked in 150 mL of distilled water, ethanol, and sodium bicarbonate (1%) as extraction solvents for a duration of 20 min. The mixture was then left at room temperature for approximately 12 h to facilitate further extraction. Subsequently, the extracts were filtered and concentrated using an IKA WEREKRV06-ML rotary evaporator. Once the desired concentrations were achieved, the extracts underwent freeze-drying using a Labconco freeze dryer until a constant weight was reached. Finally, the dried plant extracts were stored in a refrigerator until they were needed for future applications.
Screening for Antimalarial Activity: In Vitro Semiquantitative Test
In the study conducted by Deharo et al, 28 a methodology for screening the antimalarial activity of compounds was described. The experiment involved the incubation of a specific combination in a 96-well plate. The combination consisted of 50 μL of freshly dissolved hemin chloride (0.5 mg/mL) in the DMSO, 100 μL of the sodium acetate buffer (0.5 M, pH 4.4), as well as 50 μL/mL of either the potential antimalarial solution being tested or the control. The order in which these solutions were combined to the plate was crucial for the accuracy of the results. The plate was then introduced to centrifugation at 4000 rpm for only 10 min to separate the supernatant from the reaction mixture. The pH of the reaction was determined after removing the supernatant, with the desired pH range being between 5.0 and 5.2. To eliminate any unreacted hemin, each well had been washed with 200 μL of DMSO. Following the second centrifugation, the supernatant was removed from the plate. Subsequently, the hematin was solvated in 200 μL of 0.1 M NaOH to form the alkaline hematin, which could be tested via spectrophotometrically. Furthermore, the absorbance of the alkaline hematin was then read at a wavelength of 405 nm via an enzyme-linked immunosorbent assay (ELISA) reader. Then, the negative control used was ultrapure water, where chloroquine (CQ) as well as 2-mercaptopyrimidine (2-MP) were solvated in ultrapure water as well as used as positive controls. By following this methodology, the researchers were able to assess the antimalarial activity of the tested compounds based on the absorbance measurements at 405 nm, providing valuable insights for further analysis.
HPLC-PDA Analysis of Phytochemicals
To prepare the standards for analysis, various compounds including chlorogenic acid, gallic acid, 3,4-dihydroxyphenylacetic acid, 3,4-dihydroxybenzoic acid, vanillic acid, syringic acid, 4-hydroxyphenylacetic acid, ferulic acid, caffeic acid, verbascoside, isovanillic acid, p-coumaric acid, quercetin, sinapic acid, rutin, trans-cinnamic acid, as well as kaempferol were used. These standards were prepared by dissolving them in a solvent consisting of 20% ethanol at a concentration of 25 mg per 100 mL. To create a standard mixture, 1.0 mL of every standard solution was combined in a 25 mL volumetric flask as well as then diluted to the mark with the identical solvent. Regarding sample concentration of A. fragrantissima, 0.1 mg/mL was used for high-performance liquid chromatography (HPLC) analysis. For the analysis, a mobile phase (Table 1) composed of two components was used, with (A) representing 0.5% acetic acid and (B) representing acetonitrile. The detection of the main components was performed using a RP BDS Hypersil C18 column (Thermo Scientific) with dimensions of 250 × 4.6 mm as well as a particle size of 3 µm. Moreover, the flow rate was set at 0.6 mL/minute. In addition, the photodiode array (PDA) detection range was put from 210 to 400 nm, and the column temperature has been maintained at 25 °C. An injection volume of 20 µL was used for each sample. Before injection, all samples underwent filtration using a 0.45 µm disposable filter to eliminate any particulate matter or impurities.
Composition of the Mobile Phase.

A: 0.5% acetic acid and B: acetonitrile.
Statistical Analysis
The reported results are presented as means ± standard deviation (SD). All experimental data underwent one-way analysis of variance (ANOVA) with Tukey's post hoc test, using the Statistical Package for the Social Sciences software (SPSS, version 16). Statistical significance was considered at P < .05.
Results and Discussion
Beta-Hematin Inhibitory Effects
Potential antimalarial herbs can be evaluated for their effectiveness against the parasite by using the model of β-hematin inhibition.29–31
The findings from the semiquantitative in vitro experiments conducted on various parts of the A. fragrantissima plant are presented in Figures 1–5. Figure 1 specifically illustrates the antimalarial effectiveness of water and alcoholic extracts collected from the leaves, flowers, as well as stems of the plant, as indicated by the absorption of solvated β-hematin at 405 nm. The results are compared to a negative control (water) as well as a positive control (CQ-0.1 mg/mL). The absorption values are inversely correlated with the extract efficiency as well as hemozoin content, meaning that lower absorption indicates higher extract efficiency and a stronger inhibitory effect on hemozoin formation. As seen in Figure 1, the water flower extract showed promising results that are comparable to that of CQ positive control. The lowest inhibiting activity was that of the stem, water, and alcoholic extracts. Although the precise mode of action of A. fragrantissima as an antimalarial extract is not yet understood, it is clear that some of the plant extracts strongly prevented the synthesis of β-hematin.
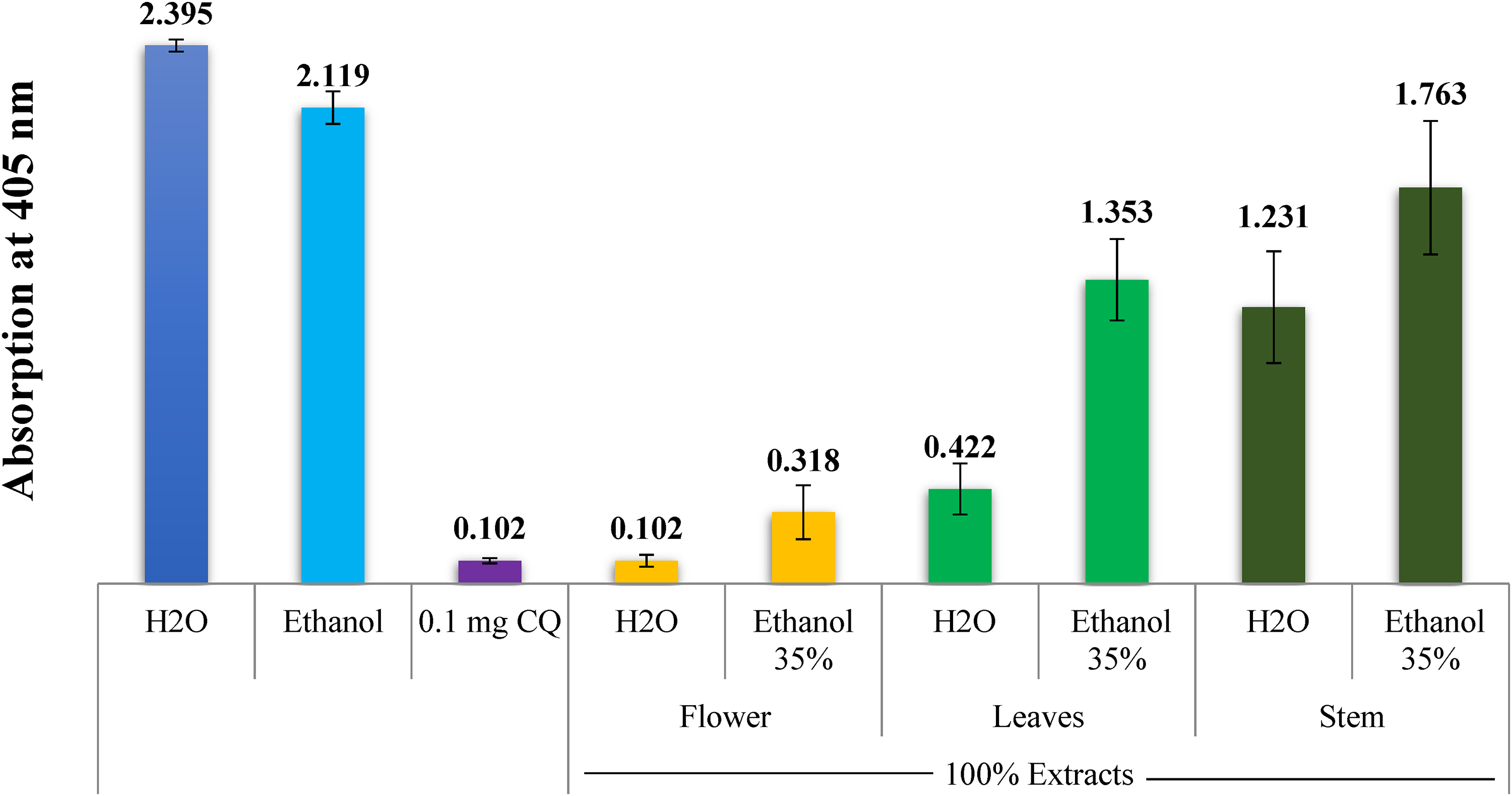
Column diagram showing the antimalarial effectiveness of water as well as alcoholic extracts of Achillea fragrantissima's leaves, flowers, and stems when compared to the negative (water as well as ethanol) as well as positive (CQ-0.1 mg/mL) controls (absorption of solvated β-hematin at 405 nm). An average of 16 different experiments is represented by each result.

Column diagram showing possible antimalarial effectiveness (absorption of solvated β-hematin at 405 nm) by various aqueous extracts of Achillea fragrantissima stems (water infusion as well as water with bicarbonate) when contrasted to negative (water and ethanol) as well as positive (CQ-0.1 mg/mL) controls. Each outcome is the average of 16 different experiments.

A column diagram showing potential antimalarial effectiveness (absorption of solvated β-hematin at 405 nm) utilizing several aqueous extracts (water infusion as well as water with bicarbonate) of Achillea fragrantissima leaves as compared to negative (water as well as ethanol) and positive (CQ-0.1 mg/mL) controls. Each result is the average of 16 different experiments.
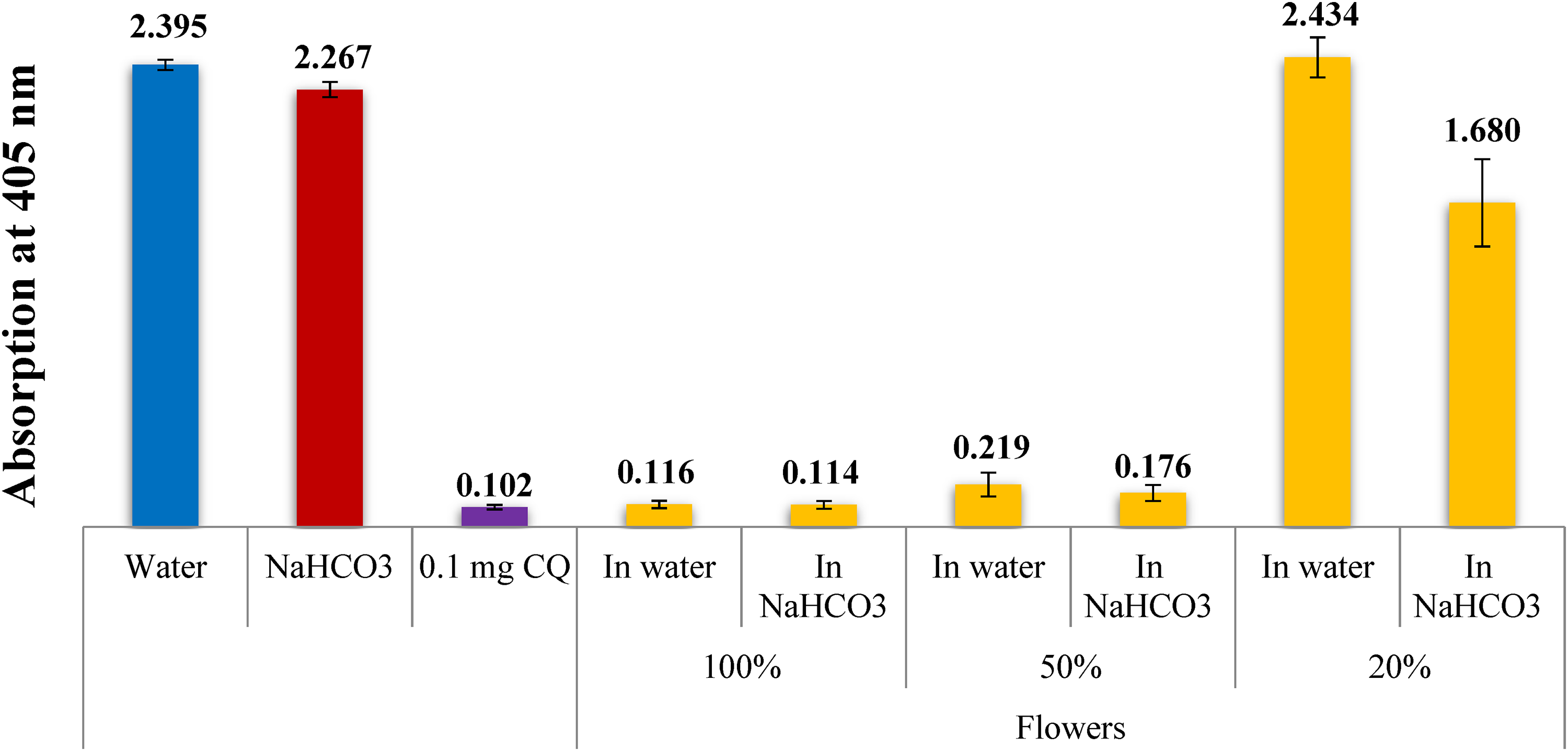
A column diagram displaying the potential antimalarial effectiveness (absorption of dissolved β-hematin at 405 nm) among various aqueous extracts of Achillea fragrantissima flowers with regard to the negative (water and ethanol) as well as positive (CQ-0.1 mg/mL) controls. An average of 16 different experiments are represented by each result.
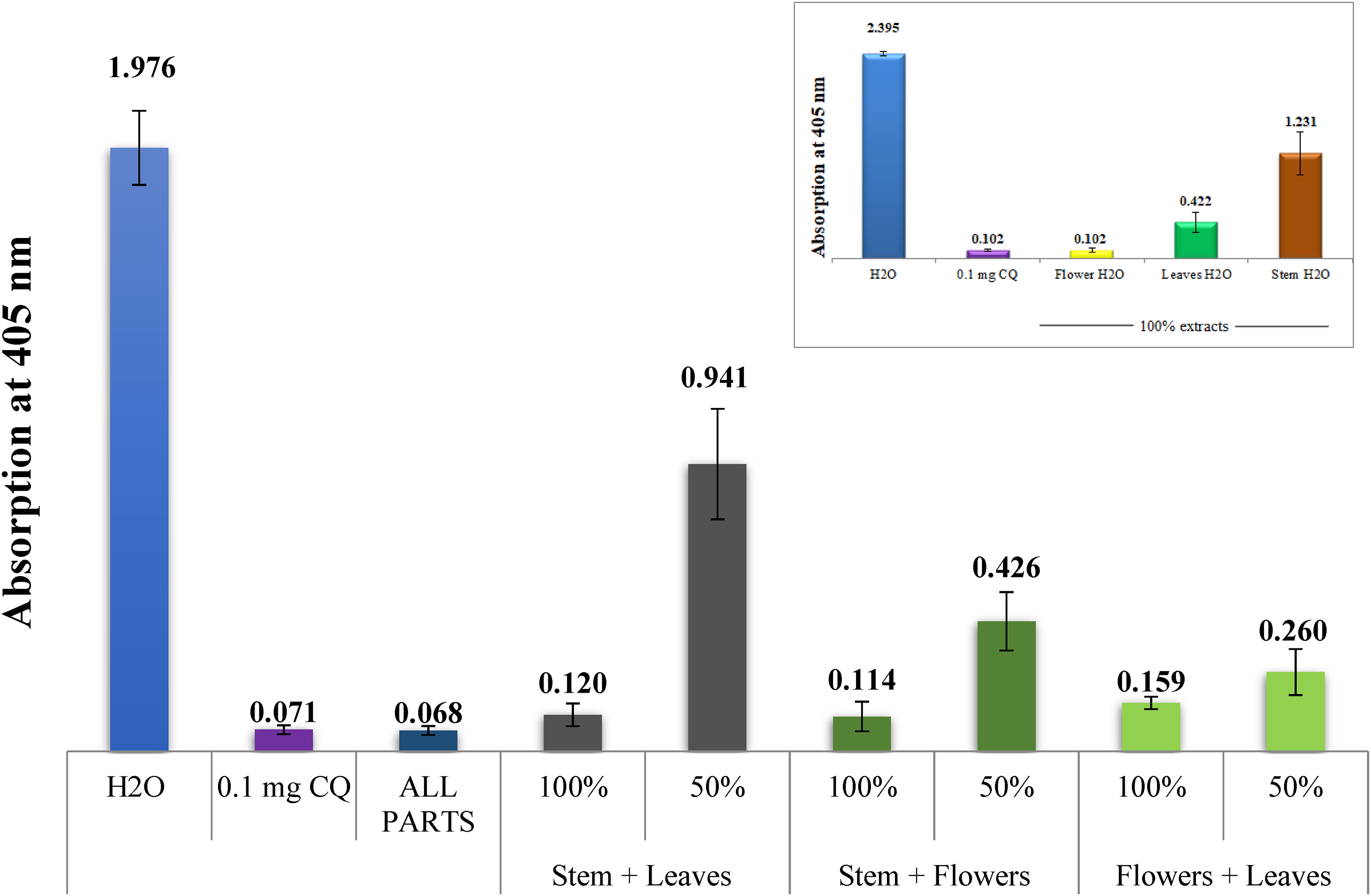
Comparing various combinations of flower, leaf, as well as stem water extracts to the negative (water) as well as positive (CQ-0.1 mg/mL) controls, a column diagram indicates the potential antimalarial efficacy (absorption of solvated β-hematin at 405 nm). The result of every experiment is the average of 16 different experiments.
In previous studies, it was discovered that the addition of sodium bicarbonate to the extraction water enhanced the inhibitory effect. 12 In this paper, we conducted various experiments using diluted sodium bicarbonate solutions, which resulted in a notable increase in the inhibitory effect on β-hematin, as depicted in Figures 2–4. It is important to note that the concentration of extract utilized in every experiment shows a crude concentration, suggesting that the concentration of active components may be much lower. Moreover, the most probable mechanism of inhibition involves the generation of a complex between the active compounds present in these extracts and ferriheme, thereby impeding the formation of β-hematin.
Figure 2 presents the test results comparing stem water injection to bicarbonate infusion. Furthermore, the semiquantitative procedure employed in this study indicates that absorption is oppositely proportional to drug effectiveness. Thus, lower absorption values indicate greater drug efficiency. The impact of bicarbonate is particularly evident in Figure 2, where there is essential increase in the inhibitory activity of β-hematin formation in the bicarbonate infusion sample, with an absorbance value of 0.185, compared to a value of 1.231 for the stem water infusion. The effects of bicarbonate are also observed in the infusions of flowers and leaves, as depicted in Figures 3 and 4, respectively.
To investigate the potential for synergy, different random combinations of aqueous extracts from different parts of A. fragrantissima were tested for their in vitro inhibitory impact on β-hematin generation. Furthermore, the results of these combinations can be seen in Figure 5. An improvement in the activity was found when mixing both the stem and leaves water extracts than of each alone, or when each of the stem and leaves were mixed with flowers extract. A clear synergistic effect is seen when all three parts of the plant were mixed, yielding activity comparable to the positive control.
HPLC-PDA Detection of Phytochemicals
Seventeen flavonoids as well as phenolic compounds standards have been separated as well as assigned specific numbers according to their retention times, as can be seen in Figure 6. The chromatogram shown in Figure 6 was selected as the best chromatogram due to how it effectively visualized every one of the standards at a wavelength of 280 nm, despite the fact that each standard enjoyed a different maximum wavelength. Figure 7 illustrates the chromatogram obtained from the ethanolic extract of A. fragrantissima leaves at a wavelength of 280 nm. The identification of compounds in both standards and samples was achieved by comparing their retention times and wavelengths. Specifically, rutin was identified at a retention time of 46.16 min. These findings demonstrate the successful separation as well as identification of flavonoids as well as phenolic compounds in the A. fragrantissima leaves extract, with rutin being one of the identified components at a specific retention time.
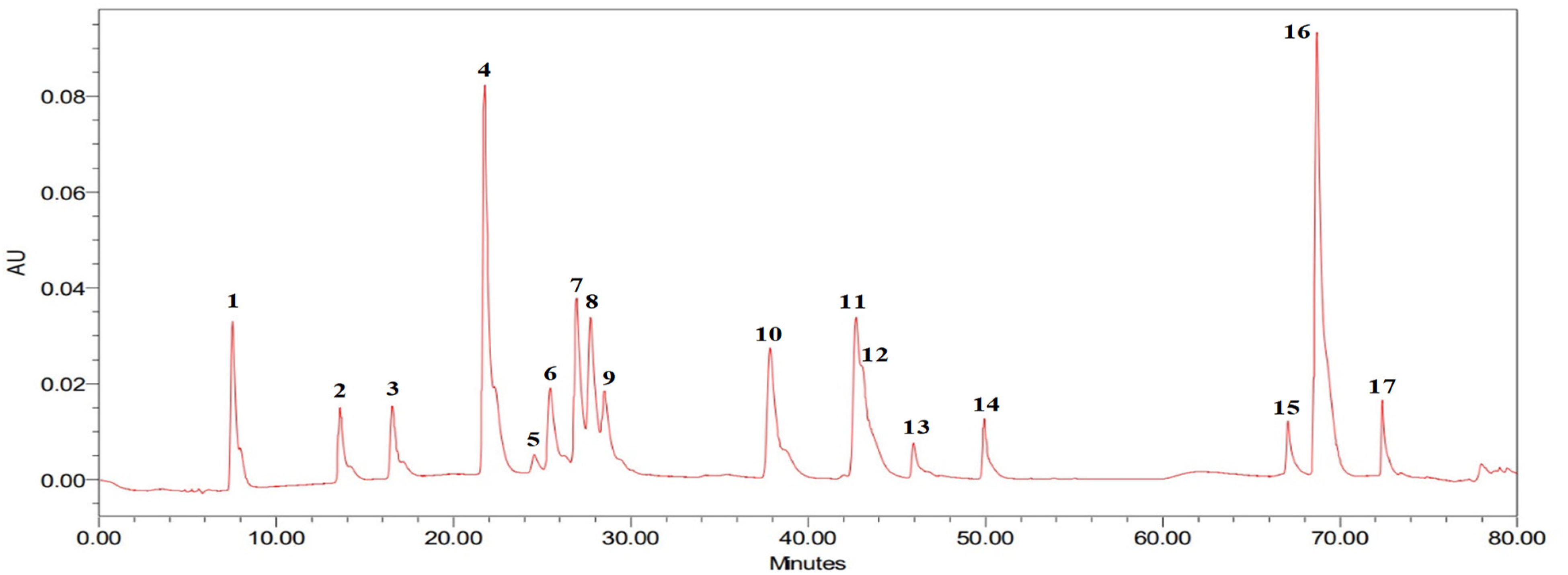
HPLC chromatogram at 280 nm presents a comprehensive analysis of various standards used in the study. Abbreviation: HPLC, high-performance liquid chromatography.

HPLC chromatogram for Achillea fragrantissima leaves extract at 280 nm (a) and UV spectrum for rutin detected at 46.16 min. Abbreviation: HPLC, high-performance liquid chromatography; UV, ultraviolet.
In Figure 6, the chromatogram clearly demonstrates the separation and detection of 17 distinct compounds. These compounds include gallic acid (1), 3,4-dihydroxybenzoic acid (2), 3,4-dihydroxyphenylacetic acid (3), chlorogenic acid (4), 4-hydroxyphenylacetic acid (5), vanillic acid (6), caffeic acid (7), syringic acid (8), isovanillic acid (9), p-coumaric acid (10), ferulic acid (11), sinapic acid (12), rutin (13), verbascoside (14), quercetin (15), trans-cinnamic acid (16), and kaempferol (17). Each compound is assigned a specific number to facilitate their identification. Remarkably, despite having different maximum wavelengths, all the standards are successfully visualized when the chromatogram is analyzed at a wavelength of 280 nm. This meticulous analysis provides valuable insights into the composition and distinctive characteristics of these flavonoids and phenolic compounds.
Strong antimalarial activity exhibited by both aqueous and ethanolic extracts of A. fragrantissima. These extracts effectively hindered the synthesis of β-hematin, a compound linked to the survival of the malaria parasite within the human body. Notably, the addition of sodium bicarbonate to the extraction water intensified this inhibitory effect, suggesting a potential optimization method for extracting or enhancing the active antimalarial compounds from the natural extracts and paving the way for potential advancements in malaria treatment or prevention strategies. It's noted that the stem's aqueous and ethanolic extracts demonstrated lower inhibitory activity in contrast to the extracts obtained from the flowers and leaves of the same plant.
Intriguingly, when combinations of extracts were tested, a synergistic effect emerged. Mixing the stem and leaves’ water extracts together or combining each of these extracts with the flower extract resulted in heightened antimalarial activity. The most significant synergistic effect was observed when all three parts of the plant—stem, leaves, and flowers—were mixed, leading to antimalarial activity similar with the positive control, suggesting a potent combined effect from the different plant parts.
The utilization of an analytical technique, HPLC-PDA chromatography, confirmed the presence of rutin specifically in the leaves extract. Rutin is a flavonoid compound known for its antioxidant properties and has been associated with various potential health benefits. The identification of rutin in the leaves extract provides insight into the potential active compounds contributing to the observed antimalarial activity, emphasizing the importance of a holistic approach when exploring the therapeutic potential of plant extracts.
Conclusion
The aqueous and ethanolic extracts of A. fragrantissima showed promising antimalarial activity by effectively inhibiting the synthesis of β-hematin. The inhibitory effect was further enhanced by adding sodium bicarbonate to the extraction water. Consequently, future research will prioritize isolating the active compounds responsible for the plant's antiplasmodial activity.
Footnotes
Acknowledgments
The authors would like to extend their sincere gratitude and acknowledge the unyielding support of the Deanship of Scientific Research at their respective universities throughout this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Funding
This study did not receive any dedicated funding from public, commercial, or not-for-profit sectors.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
