Abstract
The chemical structure of compounds identified in species of the genus Zaluzania is reported herein, highlighting the contributions by Professor Pedro Joseph-Nathan to the chemistry of these plants. Although 36 names have been proposed for Zaluzania, only 14 species are currently accepted in the genus, and they are reported to be a rich source of sesquiterpene lactones and flavonoids. Their essential oils also contain several terpenoids, especially ent-kaurene-type diterpenes. In addition, we identified the guaianolides zaluzanin A and zaluzanin B, zaluzanin C, and zaluzanin D, and the eudesmanolides ivalin and ivalin acetate as the major constituents in infusions of aerial parts of Zaluzania augusta, Zaluzania parthenioides, and Zaluzania triloba, respectively. These sesquiterpene lactones were characterized by their physical and spectroscopic data, including 1H and 13C nuclear magnetic resonance (NMR). Likewise, the structures of zaluzanins C and ivalin were confirmed by X-ray diffraction analysis. The presence of guaianolides in Z. parthenioides and eudesmanolides in Z. triloba could be used as a biomarker to differentiate these 2 species, which were previously considered synonymous.
Introduction
The genus Zaluzania includes a small number of shrubby plants native to Mexico, except for Zaluzania grayana, which has also been found in Arizona and New Mexico. 1 According to the International Plant Names Index, 36 names related to Zaluzania have been recorded, but only 14 species are accepted in the genus (Table 1). 1 Several of these plant species are considered synonyms and, in some cases, there is uncertainty about their taxonomic identification.
Plant Names Related to Zaluzania. 1

Names that cannot be accepted or synonymized.
Some Zaluzania species have been used in traditional medicine in Mexico because of their antidiabetic and abortifacient effects; in particular, Zaluzania augusta and Zaluzania triloba have been reported to have such properties.2,3 Infusion of the roots of Z. augusta has been used to induce menstruation in some parts of the state of Hidalgo, while Z. triloba is used for stomach pain and wounds. 4 The branches of Z. augusta are used to fumigate poultry houses infested with Dermanyssus gallinae (red mite), and the infusion of aerial parts of Z. triloba is used to wash wormy wounds in sheep and goats to eliminate fly larvae. 5
Chemical studies on Zaluzania species have shown them to be valuable sources of sesquiterpene lactones, while flavonoids, essential oils, and lipophilic metabolites have also been identified. This work is aimed at describing the chemical structure of the compounds identified in Zaluzania species, highlighting the contribution of Professor Pedro Joseph-Nathan in the chemistry of this plant genus.
Results and Discussion
Sesquiterpene Lactones
The chemical composition of a Zaluzania species was first reported by Romo et al.
6
They studied the EtOH extract of the aerial part of Z augusta, which was partitioned with CHCl3 and the residue chromatographed on alumina. This procedure allowed the isolation of sesquiterpene lactones of the guaine series, zaluzanin A (
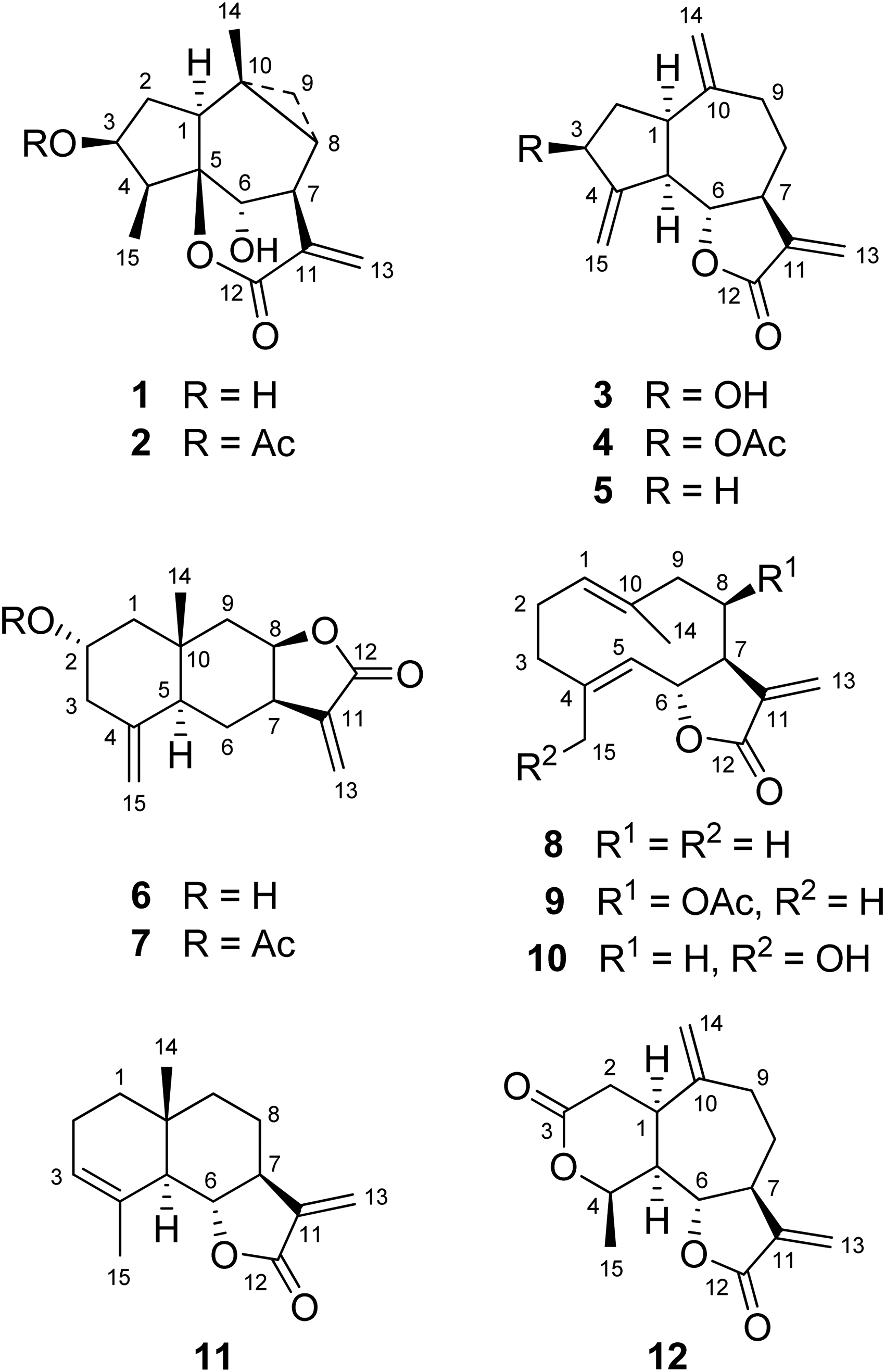
Chemical structure of sesquiterpene lactones isolated from Zaluzania spp.
Yabuta et al
12
reported the presence of sesquiterpene lactones in Z. augusta, Z. montagnaefolia (named by then as Z. montagnifolia), Z. parthenioides, Z. pringlei, and Z. triloba. The aerial part of each of the species was extracted with CHCl3 and direct crystallization or chromatography on silica gel or alumina of the extract led to obtaining
Some sesquiterpene lactones from Zaluzania spp. possess relevant biological activity. Thus, for example, zaluzanin A (
Flavonoids
In an interesting study on flavonoids in Zaluzania spp.,
20
Olsen and Mabry (1980) found that various species in this genus produce about 20 compounds, 13 of which were characterized as methyl ethers and/or glycosides of quercetin (
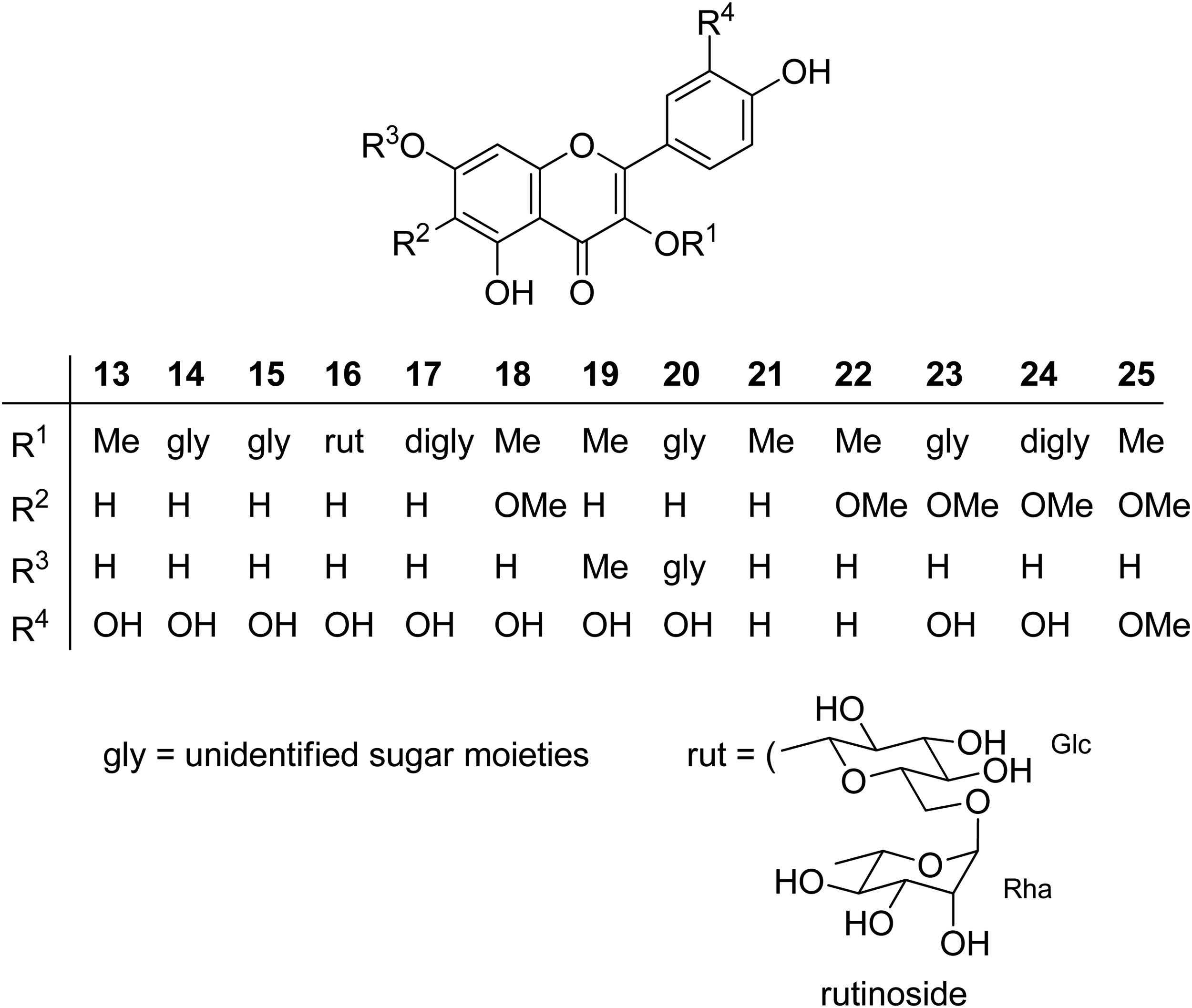
Chemical structure of flavonoids identified in Zaluzania spp.
Lipophilic Constituents and Essential Oils
Lipophilic constituents of Zaluzania spp. have been chemically characterized in Z. parthenioides,
11
Z. augusta,
21
and Z. montagnaefolia (Z. montagnifolia).22,23 On silica gel column chromatography of the petroleum ether extract from the aerial part of Z. parthenioides, octacosane, β-sitosterol (

Chemical structure of sterols and diterpenes identified in Zaluzania spp.

Chemical structure of monoterpenes identified in Zaluzania montagnaefolia.

Chemical structure of sesquiterpenes identified in Zaluzania montagnaefolia.
Sesquiterpene Lactones in Infusion of Z. augusta, Z. parthenioides, and Z. triloba
Since Z. augusta, Z. parthenioides, and Z. triloba have been traditionally used as infusions in the state of Hidalgo, Mexico,
4
the content of sesquiterpene lactones in the infusion of aerial parts of these species was investigated. Some years ago, P. Joseph-Nathan and 2 co-authors of this work reported the isolation and characterization of zaluzanin A (
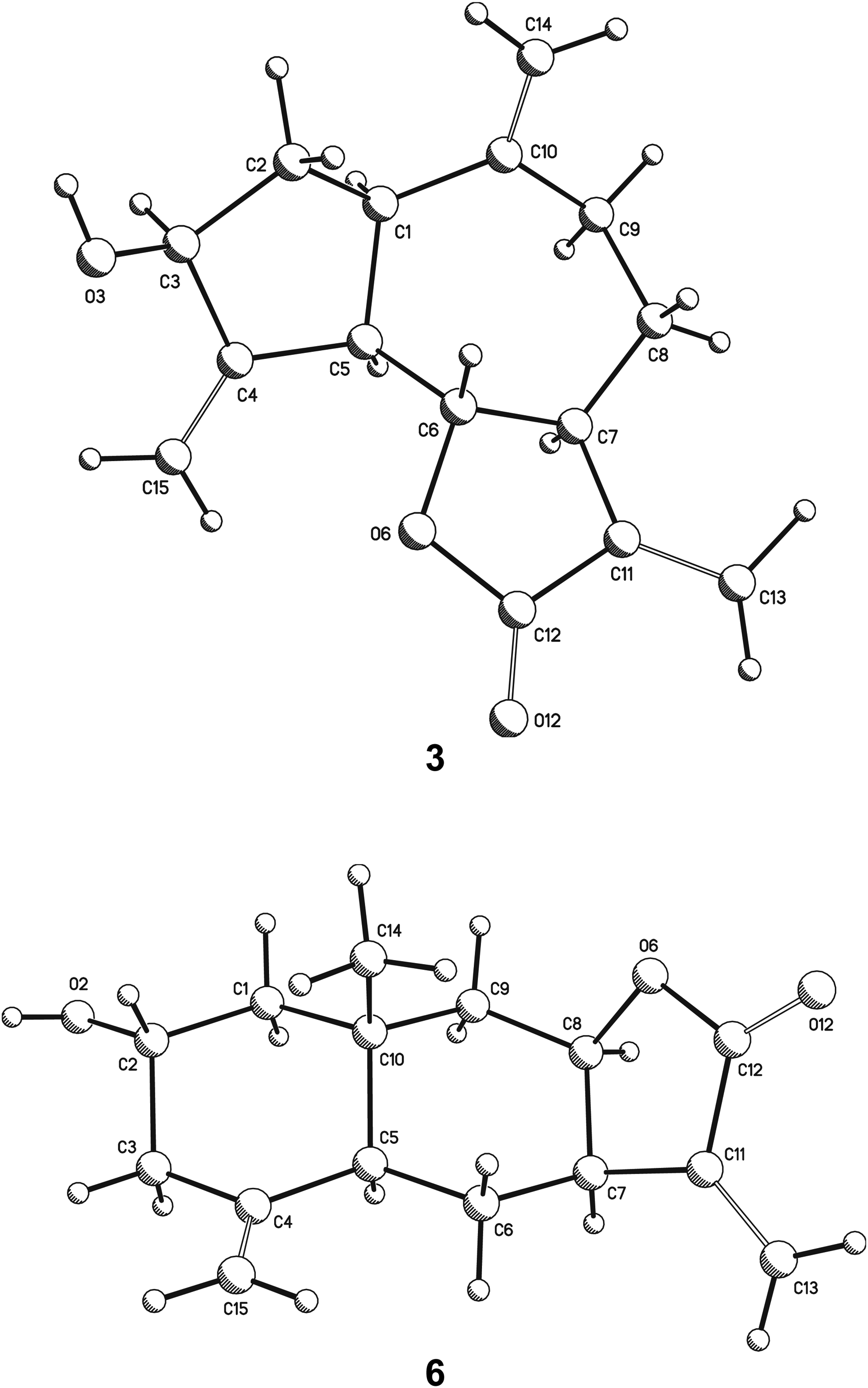
X-ray crystal structure of zaluzanin C (
Specimens of Z. parthenioides and Z. triloba were collected in the municipality of Ixmiquilpan and Mineral de la Reforma, State of Hidalgo, Mexico, respectively. Morphologically, both species are very similar, which may have caused them to be confused or considered synonyms. The sesquiterpene lactones identified in the infusion of their aerial parts suggested that Z. parthenioides biosynthesizes mainly guaianolides (zaluzanin C), whereas Z. triloba predominantly produces eudesmanolides (ivalin), so these compounds may serve as biomarkers to distinguish between both shrubs.
Conclusions
Chemical studies have been conducted on 10 out of the 14 species accepted in the genus Zaluzania. These studies have demonstrated the presence of 12 sesquiterpene lactones, 13 flavonoids, 5 diterpenes, 18 monoterpenes, 39 other sesquiterpenes, β-sitosterol, stigmasterol, and squalene. The infusion of aerial parts of Z. augusta contains zaluzanins A (
Experimental
General
Chromatographic separations were carried out on Merck silica gel 60 (230-400 mesh ASTM). TLC assays were performed on sheets of silica gel 60 F254, using ceric sulfate as developer and λ = 254 and 365 nm as fluorescence excitatory wavelengths. 1H and 13C nuclear magnetic resonance (NMR) spectra were obtained on a Bruker Ascend instrument (400 MHz for 1H; 100 MHz for 13C) from CDCl3 solutions, using TMS as an internal standard. Optical rotation was recorded in CHCl3 on a PerkinElmer 341 polarimeter. Melting points were measured on a Fisher–Johns apparatus and are uncorrected. Infrared spectra were obtained on a PerkinElmer FT-IR System 2000 spectrophotometer using cesium iodide (CsI) cells and diffuse attenuated reflectance.
Plant Material
Specimens of Zaluzania augusta (Lag.) Schultz Bip., Zaluzania parthenioides (DC.) Rzed., and Zaluzania triloba (Ortega) Pers. were collected during September–October 2019 from the municipality of Epazoyucan (98°36′54″W, 20°03′08″N), Ixmiquilpan (99°07′05″W, 20°23′47″N), and Mineral de la Reforma (98°42′13″W, 20°05′36″N), respectively, in the state of Hidalgo, México, and identified by Professor Manuel González Ledesma. Voucher specimens (JM Torres-Valencia 168, JM Torres-Valencia 131, and JM Torres-Valencia 130, respectively) are preserved in the Herbarium of the Universidad Autónoma del Estado de Hidalgo, Mineral de la Reforma, Hidalgo, Mexico.
Ethyl Acetate Extracts from Z. augusta, Z. parthenioides, and Z. triloba Infusion
Aerial parts (100 g) of Z. augusta, Z. parthenioides, and Z. triloba were extracted with hot water (750 mL) for 30 min, filtered and extracted with EtOAc (750 mL). The organic phase was dried over anhydrous Na2SO4 and concentrated to obtain the EtOAc extracts (2.13, 1.90, and 1.37 g, respectively).
Zaluzanins A (1) and B (2)
The EtOAc extract (1 g) from Z. augusta was subjected to chromatographic separation as described elsewhere
26
to obtain
Zaluzanins C (3) and D (4)
The EtOAc extract (1 g) from Z. parthenioides was passed through a silica gel column (138 g), using hexane-EtOAc (3:1, 2:1, 1:1, 1:2, and 0:1) as eluent. Fractions of each polarity (300 mL) were collected and labeled A–E. A second chromatographic separation of fractions B (105 mg), and E (87 mg) under the same conditions afforded
Ivalin (6) and Ivalin Acetate (7)
The EtOAc extract (1 g) from Z. triloba was subjected to separation under the conditions described above for Z. parthenioides. Fraction B (97 mg) was purified on a silica gel column, with hexane-EtOAc (2:1, 500 mL, and 1:3, 150 mL) as eluent, collecting 20 mL eluates. Compound
Quantification of Sesquiterpene Lactones in Infusion of Aerial Parts of Z. augusta, Z. parthenioides and Z. triloba
A portion of the EtOAc fraction (60 mg) of each infusion of Z. augusta, Z. parthenioides, and Z. triloba was analyzed by HPLC in triplicate to quantify zaluzanin A (
Single Crystal X-ray Diffraction Analysis of Compound 3
Data were acquired on a Bruker Smart 6000 CCD diffractometer, using Mo Kα radiation (λ = 0.7073 Å). A total of 1321 frames were collected, at a scan width of 0.3° and an exposure time of 10 s/frame. These data were processed with the SAINT software package, provided by the diffractometer manufacturer, by using a narrow-frame integration algorithm. An empirical absorption correction was applied. Crystal data were, C15H18O3, M = 246.29, monoclinic, space group P21, a = 11.927(5) Å, b = 12.998(5) Å, c = 13.102(5) Å, V = 2008.7(1) Å3, Z = 2, Z′ = 3, ρ = 1.22 mg/mm3, μ(MoKα) = 0.084 mm−1, total reflections = 13 495, unique reflections = 6719 (Rint 0.07%), observed reflections = 3025. The structure was solved by direct methods using the SHELXS-97 program. For structural refinement, nonhydrogen atoms were treated anisotropically, and hydrogen atoms, included in the structure factor calculation, were refined isotropically. Final R indices were [I > 2σ(I)] R1 = 4.8% and wR2 = 9.5%. Largest difference peak and hole, 0.105 and −0.135 e.Å3.
Single Crystal X-ray Diffraction Analysis of Compound 6
The data were collected on an Agilent Xcalibur Atlas Gemini diffractometer using the enhance CuKα X-ray source radiation (λ = 1.54184 Å) at 293(2) K in ω scan mode. Unit cell refinements using 5022 machine-detected reflections were made with the software CrysAlisPro, Agilent Technologies, v.1.171.34.49. Crystal data were C15H20O3, M = 248.31, orthorhombic, space group P212121, a = 6.89340(1) Å, b = 11.3914(3) Å, c = 34.7740(8) Å, V = 2730.7(1) Å3, Z = 4, Z′ = 2, ρ = 1.21 mg/mm3, μ(CuKα) = 0.666 mm−1, total reflections = 13 339, unique reflections = 5315 (Rint 0.01%), observed reflections = 4684. The structure was solved by direct methods using the SHELXS-97 program. For structural refinement, nonhydrogen atoms were refined anisotropically, and most hydrogen atoms were refined at geometrically idealized positions on their parent atoms. Final R indices were [I > 2σ(I)] R1 = 3.3% and wR2 = 7.9%. Largest difference peak and hole, 0.146 and −0.110 e.Å3. The software Olex2 v.1.1.5 31 allowed to calculate Flack parameter 32 x = 0.00(19) and Hooft parameter 33 y = 0.08(8). For the inverted structure, these parameters were x = 0.99(19) and y = 0.92(8), respectively.
Crystallographic data (excluding structure factors) of
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231184010 - Supplemental material for Chemical Composition of Plants of the Genus Zaluzania
Supplemental material, sj-docx-1-npx-10.1177_1934578X231184010 for Chemical Composition of Plants of the Genus Zaluzania by J. Martín Torres-Valencia, Reyna Zeferino-Díaz, Laura A. Ortiz-León, Miriam Téllez-Reyes and R. Inés Escamilla-Baños in Natural Product Communications
Footnotes
Acknowledgments
Partial financial support from CONACYT-Mexico (grant No. 238206) is acknowledged. RZD thanks CONACYT for fellowship No. 418442. We are grateful to Professor Manuel González Ledesma of the Área Académica de Biología (U.A.E.H.) for the identification of Z. parthenioides and Z. triloba, and to Angelina Hernández Barragán, Department of Chemistry, CINVESTAV-IPN, Mexico, for her invaluable support on X-ray diffraction analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Consejo Nacional de Ciencia y Tecnología (grant number 238206).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
