Abstract
Several dietary and medicinal herbs have been shown to be effective in the treatment and prevention of cancer. Although Piper nigrum has been shown to have anti-cancer activities against various cancer cells, its anti-pancreatic cancer properties have not been reported. In the present study, P. nigrum extract (PNE) inhibited proliferation of PANC-1 human pancreatic cancer cells. Flow cytometry showed G0/G1 arrest caused by PNE in PANC-1 cells. In addition, Western blot analysis showed that PNE suppressed the protein levels of cell cycle regulators such as cyclin B1, cyclin D1, survivin, and Forkhead box M1 (FoxM1). These findings suggested that the inhibitory activity of PNE against the growth of PANC-1 cells was correlated with cell cycle arrest and repression of cell cycle regulators. Wound healing and trans-well assays showed that PNE suppressed migration and invasion of PANC-1 cells. Piperine, a major alkaloid of Piper nigrum, was identified as the main component of PNE by HPLC analysis. Piperine also attenuated the cell growth, migration, and invasion of PANC-1 cells, suggesting its contribution to the anti-pancreatic cancer effects of PNE. These results demonstrate that PNE and its major constituent, piperine, have anti-pancreatic cancer properties such as growth-inhibition, anti-migration, and anti-invasion of cancer cells.
Introduction
Pancreatic cancer, one of the digestive system cancers, is still a fatal disease with a 5-year survival rate of less than 8%. Although cancer research has advanced significantly and cancer mortality is steadily declining, the pancreatic cancer death rate increases. 1 Late diagnosis and high metastatic potential cause the poor prognosis and high mortality of pancreatic cancer. 2 Surgical resection is the first and only potential curative treatment. Unfortunately, up to 80% of cases are unresectable due to early and extensive metastasis. Chemotherapy and radiotherapy have been unsatisfactory due to drug toxicity, chemo-resistance, and recurrence.3,4 This highlights the dire need for effective, therapeutic, and preventive non-toxic natural agents for pancreatic cancer. Vast evidence indicates that dietary herbs, medicinal plants and their constituents are potential options for the prevention of diseases, development of therapeutic agents, and functional products.5–7
Piper nigrum L. (Piperaceae family) has a long history of seasoning, flavoring, and preserving foods.
8
In addition, it has been used for the treatment of fever, influenza, and muscular pain.
9
Recent studies have demonstrated its digestion enhancement,
8
anti-oxidant,
10
and anti-inflammatory properties.
11
P. nigrum contains various alkaloids, flavonoids, phenols, steroids, and terpenes.
12
Piperine, a main alkaloid from P. nigrum, has been reported to have immunomodulatory, anti-oxidant, anti-asthmatic, antimicrobial, anti-ulcer, and anti-inflammatory activities.
13
It has also been reported to exhibit anti-tumor activities against colon cancer, breast cancer, and melanoma.
14
Extracts of P. nigrum have also shown anti-cancer activity in breast cancer, Ehrlich carcinoma, colon, cervical, and prostate cancer cells with different mechanisms.15–17 However, effect against pancreatic cancer of P. nigrum and piperine has not yet been reported. Hence, the present study was proposed to investigate the effects of P. nigrum extract (PNE) and piperine on proliferation, migration, and invasion of pancreatic cancer cells.
Results and Discussions
PNE Inhibited Cell Growth and Cell Cycle Progression of PANC-1 Human Pancreatic Cancer Cells
The growth of normal cells is tightly controlled by a coordinated cell cycle process. However, the cell cycle of cancer cells is often dysregulated due to changes such as mutations and malfunctions of cell cycle regulators resulting in uncontrolled cell growth.18–20 To examine the effect of PNE on cell growth, cell viability was assessed by MTT assay. As shown in Figure 1(a), PNE repressed the viability of PANC-1 cancer cells in a concentration-dependent manner at 72 h. The IC50 value of PNE for the viability of PANC-1 cells was 54.2 μg/mL. Since cell growth is controlled by cell cycle progression, cell cycle arrest is considered as one of the causes of repressed cellular growth. To evaluate the mechanism through which PNE interrupts cell growth, we examined the effect of PNE on cell cycle progression using flow cytometry. The cell populations of control at G0/G1 phase, S phase, and G2/M phase were 42.2%, 22.0%, and 35.7%, respectively. However, PNE treatment (100 μg/mL for 20 h) increased the cell population of the G0/G1 phase (60.6%), and decreased that of the G2/M phase (17.7%) (Figure 1(b)). These results indicate that interruption of cell growth by PNE in PANC-1 pancreatic cancer cells was correlated with the cell cycle arrest at the G0/G1 phase.

Effects of PNE on cell growth and cell cycle progression in PANC-1 human pancreatic cancer cells. (
PNE Suppreesed Cell Cycle-Related Protein Levels in PANC-1 Pancreatic Cancer Cells
Since PNE reduced the growth of PANC-1 pancreatic cancer cells by arresting the cell cycle, we thus examined the effect of PNE on the cell cycle regulatory proteins such as cyclin B1, cyclin D1, survivin, and Forkhead box protein M1 (FoxM1). The level of cyclin B1 was high throughout cell cycle progression in cancer cells, but was very low and accumulated only at the G2/M cell cycle transition phase in normal cells.
21
Cyclin D1, a representative biomarker in the transition of G1 to S phase, is highly accumulated in solid tumors.
22
Survivin, as an anti-apoptotic protein, regulates cell division and spindle formation, which is essential for cell cycle progression. High expression of survivin is frequently associated with poor prognosis and drug resistance in cancer.
23
FoxM1, an oncogenic transcription factor, is a key cell-cycle regulator for both transition from G1 to S phase and progression from G2 to M phase.
24
It is also an upstream regulator for the expression of cyclin B1, cyclin D1, and survivin,25–27 and is up-regulated in most human cancers. In addition, elevated expression of FoxM1 is associated with poor prognosis and low survival rate in most cancers, including pancreatic cancer.28,29
As shown in Figure 2, PNE significantly suppressed the protein levels of cyclin B1, cyclin D1, survivin, and FoxM1 compared to the high levels of untreated cells. Since cell cycle regulators such as cyclin B1, cyclin D1, and survivin are FoxM1 target genes, these regulators can be down-regulated by suppressing FoxM1 expression with PNE treatment. Taken together, PNE down-regulated the cell cycle regulators, thereby inhibiting the cell cycle progress and subsequently suppressed the cancer cell growth.
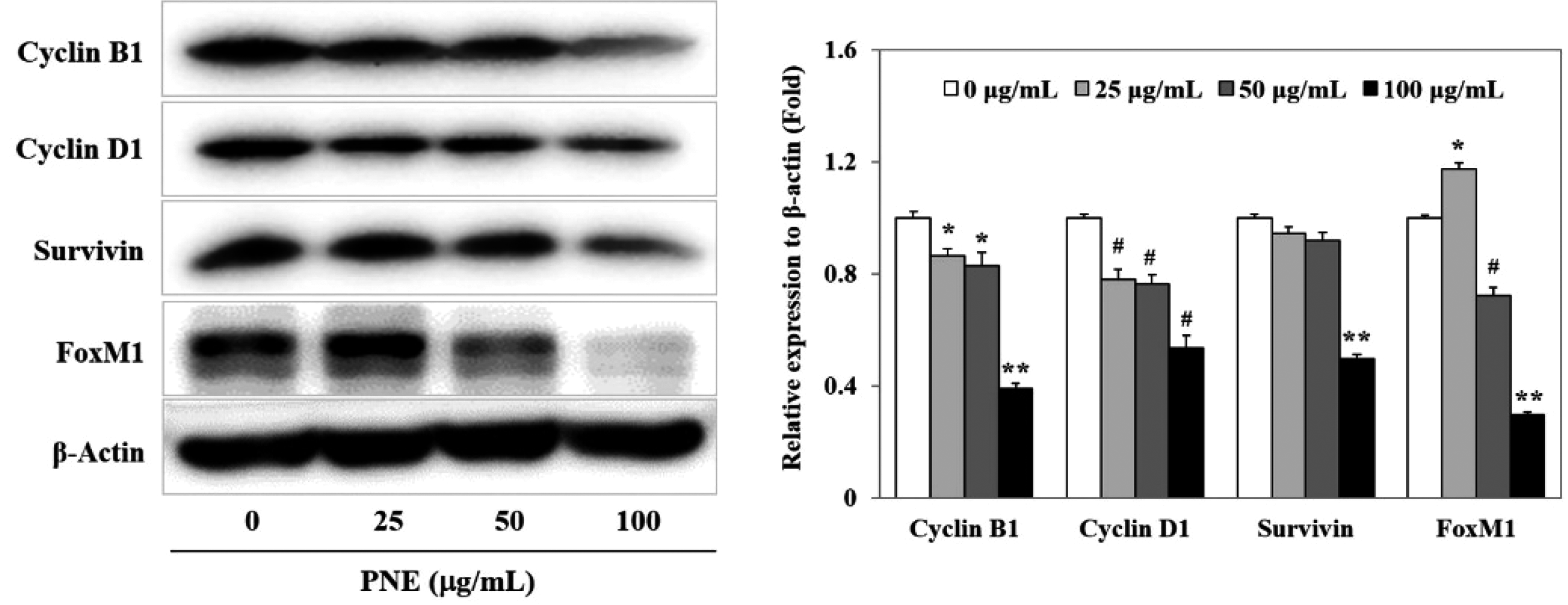
Effects of PNE on protein levels of cell cycle regulators. PANC-1 cells were treated with the indicated concentrations of PNE for 20 h. The protein levels of cyclin B1, cyclin D1, survivin, and FoxM1 were determined by Western blotting and the protein levels were quantified using ImageJ program. *p < .05, #p < .01, ** p < .001 indicate significant differences from the untreated control.
PNE Inhibited Migration and Invasion of PANC-1 Human Pancreatic Cancer Cells
Metastasis is the most malignant and complex cancer event. The metastatic cascade of cancer cells includes dissemination from the primary tumor, spreading through the circulatory and lymphatic systems, invasion across the basement membranes, and finally formation of new colonies in distant organs. Cell migration and invasion are fundamental steps in metastatic process.30,31 Metastasis of pancreatic cancer is a major cause of morbidity and mortality.2,3 It involves alteration of gene expression such as overexpression of FoxM1 that has been reported to have neoplastic and invasive potentials in various cancers, including pancreatic cancer.32,33 We previously reported that broussoflavonol B from Broussonetia kazinoki repressed the migration and invasion of PANC-1 pancreatic cancer cells through downregulation of FoxM1. 26
PNE decreased the level of FoxM1 protein expression (Figure 2), which led us to investigate the effects of PNE on migration and invasion of pancreatic cancer cells using wound healing and trans-well assays. As shown in Figure 3(a), the wound area of the control cells was diminished by 51.5% compared to that at 0 h, which means pancreatic cancer cells aggressively moved to adjacent spaces for 20 h. However, PNE dose-dependently sustained the wound area. Treatment with 100 μg/mL of PNE for 20 h showed 95.9% wound area compared with 0 h. PNE treatment decreased cell migration in a concentration-dependent manner, as confirmed by our trans-well assay results (Figure 3(b)). The migratory cancer cells of the control (100%) were reduced to 63.3%, 49.9%, and 14.3% at 25, 50, and 100 μg/mL of PNE, respectively. Furthermore, the effect of PNE on the invasion of PANC-1 pancreatic cancer cells was examined using a Matrigel-coated chamber. PNE treatment for 20 h reduced invasion of the cells into the Matrigel-coated lower chambers in a concentration-dependent manner, which is in accordance with decreased migration of pancreatic cancer cells by PNE (Figure 3(c)). These results suggested that PNE inhibited migration and invasion of PANC-1 pancreatic cancer cells. Further study could disclose detailed molecular mechanisms through which PNE inhibits migration and invasion of pancreatic cancer cells.

Effects of PNE on migration and invasion of PANC-1 human pancreatic cancer cells. (
High Performance Liquid Chromatography (HPLC) Analysis of PNE and Anti-Pancreatic Cancer Activities of Piperine in PANC-1 Human Pancreatic Cancer Cells
Piperine, an alkamide, is a major component of P. nigrum. Previously, piperine was reported to exert antioxidant, anti-inflammatory, anti-allergic, anti-microbial, anti-depressant, and anti-carcinogenic activities. 12 In addition, some studies have demonstrated in vitro and in vivo anti-cancer effects of piperine through triggering apoptosis, inhibiting cell proliferation, and deterring migration/invasion in various cancers including breast, prostate, colon, lung, melanoma, and ovarian cancer cells.14,34 However, the effects of piperine against pancreatic cancer cells have not been reported.
We confirmed that piperine was a major constituent of PNE through HPLC analysis (Figure 4(a)). Further, we investigated whether piperine could be responsible for the anti-pancreatic cancer activities of PNE. As shown in Figure 4(b), piperine treatment for 72 h reduced cell viabilities in a concentration-dependent manner. It has been reported that piperine inhibited the growth of cancer cells by arresting the cell cycle and suppressing cell cycle regulators such as cyclin B1 and cyclin D1 expression in human prostate cancer cells and osteosarcoma cells.35,36 However, further research is needed to elucidate the effects of piperine on the cell cycle and the expression of cell cycle regulators in pancreatic cancer cells.
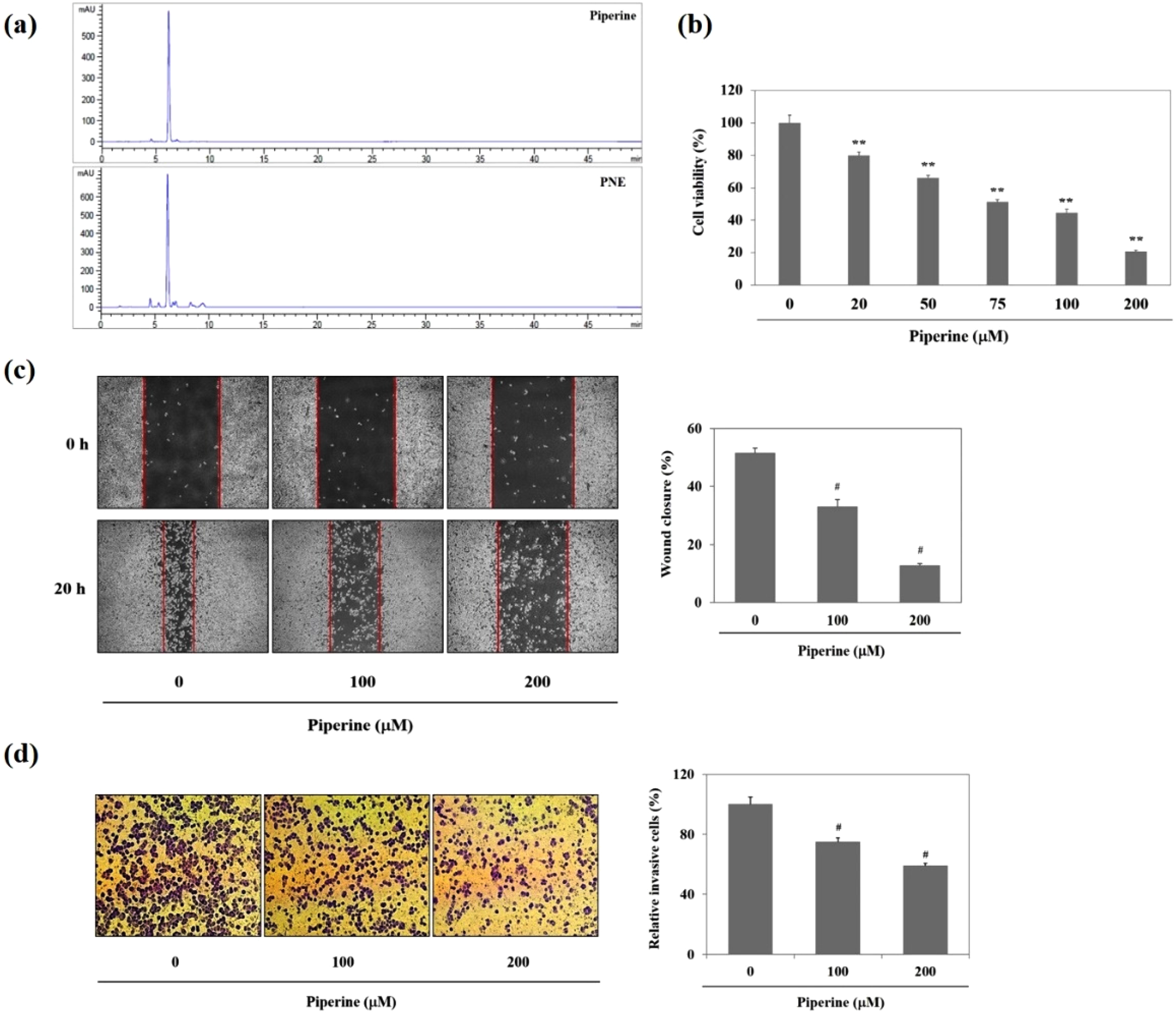
Identification of piperine as the main constituent of PNE by HPLC analysis and effects of piperine on growth, migration and invasion of PANC-1 pancreatic cancer cells. (was expressed as a relative value (%) of untreated control. #p < .01 indicates significant differences from the untreated control.
Treatment with 200 μM of piperine for 20 h showed an 87.3% wound area compared to 0 h whereas control cells showed a 51.4% wound area compared with 0 h. This means that piperine inhibits the cell movement of pancreatic cancer cells (Figure 4(c)). Furthermore, treatment with piperine (100 and 200 μM) for 20 h significantly decreased the number of invading cells by 25.1% and 41.0%, respectively, when compared to those (100%) of untreated cells, on a Matrigel-coated lower chamber (Figure 4(d)). These results show that piperine potently suppresses the invasion of pancreatic cancer cells.
Taken together, piperine, as a main component of P. nigrum, showed anti-proliferative, anti-migratory, and anti-invasive properties in PANC-1 human pancreatic cancer cells. These results suggest that the anti-pancreatic cancer activities of PNE can be partially attributed to piperine.
Conclusions
Piper nigrum extract (PNE) suppressed the growth of PANC-1 human pancreatic cancer cells through G0/G1 cell cycle arrest. PNE also down-regulated protein levels of cell cycle regulators such as cyclin B1, cyclin D1, survivin, and FoxM1, and inhibited invasion and migration of pancreatic cancer cells in vitro. Further, piperine, a major PNE constituent, inhibited the growth, migration, and invasion of PANC-1 pancreatic cancer cells. Collectively, PNE has anti-pancreatic cancer properties, and piperine is mainly responsible for these activities. These findings suggest that P. nigrum and its major alkaloid piperine may have preventive and therapeutic potential against human pancreatic cancer, and therefore warrant further animal experiments.
Experimental
Reagents
Antibodies to cyclin B1, cyclin D1, and survivin were from Cell Signaling Technology, Inc. (Danvers, MA, USA), Forkhead box protein M1 (FoxM1) antibody from Bethyl Laboratories, Inc. (Montgomery, TX, USA), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), piperine and propidium iodide from Sigma-Aldrich Co. (St. Louis, MO, USA), ethanol (Reagent grade) from Duk San Pure Chemicals Inc. (Ansan, Korea), and acetonitrile (HPLC grade) from TEDIA High Purity Solvents (Fairfield, OH, USA).
Preparation of PNE
Piper nigrum L. (unripe fruit) was purchased from a herbal medicine store (Seoul, Korea) and authenticated by Prof. Sun Young Park, College of Korean Medicine, Semyung University (voucher specimen No. PN201901F001). P. nigrum (100.3 g) was extracted three times with ethanol (1 L) at 40 °C for 1 h in an ultrasonic extractor. The solvents were removed using a vacuum rotary evaporator to generate the ethanol extract (PNE, 8.7 g). This was dissolved in dimethylsulfoxide (DMSO) and stored at 4 °C until use.
Cell Culture Condition
PANC-1 human pancreatic cancer cell lines (ATCC, Manassas, VA, USA) were cultured in DMEM comprising 10% fetal bovine serum (FBS), 0.1 mg/mL of streptomycin, and 100 units/mL of penicillin (Life Technologies Corporation, Carlsbad, CA, USA). Cells were incubated at 37 °C in a moist atmosphere with 5% CO2.
Cell Viability Assay
PANC-1 cells (3 × 103 cells/well) were treated with serial concentrations of either PNE (0, 25, 50, 100, and 200 μg/mL) or piperine (0, 100, and 200 μM) for 72 h. Then, MTT was added to the cells for 4 h, and the formazan crystals formed from MTT were solubilized with DMSO. Then, the absorbance of formazan dye at 540 nm was measured.
Cell Cycle Analysis
PANC-1 cells (4 × 105 cells/dish) were seeded in a 60 mm dish. After treatment with thymidine (2 mM) for 24 h for cell cycle synchronization, the cells were incubated with PNE (100 μg/mL) for 24 h. Cells were collected and then fixed with 70% ethanol at 4 °C. On the next day, RNase A solution was added to the fixed cells for 15 min at 37 °C, and propidium iodide (PI) was added for 20 min at 4 °C. Then the cells (1 × 104) were applied to a flow cytometer (BD Biosciences, San Jose, CA, USA), and the cell-cycle distribution was analyzed with BD CellQuesTM Pro software (version 6.0, BD Biosciences).
Western Blot Analysis
PANC-1 cells were treated with several concentrations of PNE for 20 h and then the cells were lysed with RIPA lysis buffer to extract the protein. The cell suspension was centrifuged for 20 min at 4 °C, and the resultant supernatant was collected. Protein lysates (20 μg) were subsequently applied to sodium dodecyl sulfate polyacrylamide gels and transferred onto polyvinylidene fluoride membranes (GE Healthcare, Chicago, IL, USA). The membranes were incubated with primary antibodies against cyclin B1, cyclin D1, survivin, and FoxM1 overnight at 4 °C following incubation with blocking solution containing skim milk in TBS-T solution. Then, the membranes were treated in horseradish peroxidase (HRP)-conjugated secondary antibody solution for 2 h and proteins on the membranes were detected by chemiluminescence using VersaDoc 3000 (Bio-Rad, Hercules, CA, USA).
Wound Healing Assay
PANC-1 cells in a 6-well plate were incubated until 90% confluent, and were scratched by a pipette tip to induce a wound. The detached cells were discarded by washing with PBS. The attached cells were incubated with media containing either PNE or piperine, and then permitted to migrate for 20 h. The scraped wound areas were observed and their images captured under a microscope (Olympus, Tokyo, Japan) at 0 h and 20 h. The wound area was measured using the ImageJ program (NIH, Bethesda, MD, USA). The wound area was considered as cell migration rate (wound closure), which was calculated as follows: 100 × {1-(WA20h)/(WA0h)}; WA0h and WA20h mean wound area at 0 and 20 h, respectively.
Migration and Invasion Assay
Cellular migration and invasion were measured using a BioCoatTM MatrigelTM Invasion Chamber (BD Bioscience, San Jose, CA, USA). Cell migration was detected by the trans-well chamber system, while cell invasion was measured by Matrigel-coated chambers. Cells (5 × 104 cells/200 μL of FBS free medium) were added to each insert with either PNE or piperine, and FBS containing medium (750 μL) was added to each lower chamber. Plates were incubated at 37 °C for 20 h in a 5% CO2 incubator. Cells in the plates were allowed to penetrate the pores of the membrane of the insert toward the lower chamber covering the serum-containing medium. Next, the residual cell suspension was discarded and the cells on the inserts were fixed in formaldehyde solution (3.7% in PBS) and then stained with 0.1% crystal violet for 15 min. The stained cells were counted and their images captured using a microscope (Olympus, Tokyo, Japan).
High Performance Liquid Chromatography (HPLC) Analysis
HPLC was performed with an Agilent HPLC 1260 system (Agilent, Santa Clara, CA, USA) with an UV detector. An Eclipse Plus C18 (5 μm, 4.6 × 250 mm, Agilent) column was used and the mobile phase consisted of water (A) and acetonitrile (B) with a gradient solvent system. The gradient elution was as follows: 0 to 30 min, 40%-80% B; 30 to 35 min, 80%-95% B; 35 to 45 min, 95%-95% B. The flow rate was 1.0 mL/min and detection for peaks in either PNE or piperine was accomplished at 345 nm.
Statistical Analysis
All the experiments were conducted three times. The data values are expressed as the mean ± standard deviation. Statistical difference between the two groups (untreated control and treatment groups) was analyzed by a two-tailed Student's t-test. A p value of less than .05 was considered as statistically significant.
Footnotes
Author Contributions
Conceptualization: Hwa Jin Lee, Methodology: Ji Hye Jeong and Hwa Jin Lee, Data analysis and investigation: Ji Hye Jeong, Jae-Ha Ryu, and Hwa Jin Lee, Writing-original draft preparation: Hwa Jin Lee, Writing-review and editing: Jae-Ha Ryu and Hwa Jin Lee. All authors have read and agreed to the published version of the manuscript.
Ethical Approval
Ethical approval is not applicable for this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Semyung University Research Grant of 2018.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
