Abstract
Introduction
Lavandula dentata L. (LD) is a perennial subshrub that can attain a height of 60 cm with toothed purple flowers. Its leaves are gray-green and linear. 1 The LD is native to the Mediterranean basin, southern Europe, and the Canary Islands.2,3 The leaves and flowers of LD are commonly used in traditional medicine in the form of infusions, decoctions, and essential oils (EOs) to treat various infections, such as respiratory infections, digestive problems, and diabetes.4–7 In addition, LD EOs are widely used in aromatherapy as a relaxant. 8 The therapeutic and aromatic use of LD is due to the variety of phytochemical compounds in organic extracts, such as polyphenols and flavonoids.9,10 LD EO is rich in terpene compounds such as linalool, camphor, and eucalyptol. 11 The diversity of LD's composition explains the biological properties of this species. Several studies have demonstrated the strong antibacterial activity of LD EOs against Gram-positive bacteria such as Staphylococcus aureus, and Streptococcus faecalis.12,13 In addition, LD has significant antifungal and insecticidal activity.14,15 Furthermore, the presence of linalool and camphor gives LD antispasmodic and antioxidant properties.16,17 The effectiveness of these biological properties differs according to the chemical composition of the LD EO, which changes from region to region due to different factors such as human activity, climate, and harvesting periods. The main objective of this study is to identify and compare the chemical composition of LD in 3 distinct periods. In addition, we aim to assess the biological properties of each period, in particular, their antioxidant and anti-inflammatory activities. In addition, this approach will enhance a better understanding of the variations in the chemical composition of LD over periods and their impact on its biological properties. Furthermore, in-silico studies were performed to substantiate the empirical observations, employing computational simulations to analyze and validate experimental data, thereby strengthening the overall scientific investigation. This study is taking place in the Al Hoceima region, in the municipality of Ajdir, where few studies have been conducted.
Experimental
Chemicals
DPPH (2,2-diphenyl-1-picrylhydrazyl) and BSA (bovine serum albumin) were purchased from Sigma-Aldrich Saint-Louis, L-ascorbic acid from Oxford Lab Fine Chem LLP India, and diclofenac from Novartis Pharma Moroc SA. All other chemicals were of analytical grade.
Plant Material
The plant was harvested in the province of Ajdir Al-Hociema, Morocco (35.20994°N, 3.91799°W), in the early hours of the morning, during the region's 3 seasons, namely the rainy, intermediate, and dry seasons, comprising December, April, and July 2022, respectively. The plant was identified at the Faculty of Science and Techniques Al-Hoceima, Morocco (FSTH), and an herbarium sample was deposited at the FSTH herbarium, voucher number L03L12/21.
Extraction of Essential Oils
LD leaves were dried in the dark for 6 days. Next, dried LD leaves (100 g) were hydrodistilled using a Clevenger-type apparatus for 3 h, EOs were obtained, dried with anhydrous sodium sulfate, and stored in the refrigerator at a temperature of 4 oC for further examination.
Gas Chromatography-Mass Spectrometry (GC-MS) Analysis
The analysis of LD EOs was carried out with GC-MS using a BPX25 capillary column with 5% diphenyl, 95% dimethylpolysiloxane (30 m × 0.25 mm internal diameter × 0.25 µm film thickness) from a Shimadzu GC system (Kyoto, Japan) coupled to a QP2010 MS, pure helium gas (99.99%) was used with a flow rate of 3 min/min. The injection, ion source, and interface temperatures were all set at 250 °C. The temperature program for the column oven was 50 °C (held for 1 min), heated to 250 °C at 10 °C/min, and had for 1 min. The LD EO sample components were ionized in EI mode (70 eV) and analyzed within the 40 to 300 m/z mass range. The identification of LD EO compounds and their retention times were compared with those of authentic standards, and the fragmentation patterns of their mass spectra were compared with those stored on the National Institute of Standards and Technology (NIST147). 18
Anti-Inflammatory Activity In Vitro
The anti-inflammatory potential of LD EO was evaluated using a standard protocol utilizing the BSA. Different concentrations (600-4000 µg/mL) of LD EO were prepared and the volumes were adjusted to 2.5 mL with 0.85% NaCl solution, and 500 µL of 0.2% BSA solution prepared with PBS (phosphate buffer saline) at pH 6.4 was added. The incubation temperature of the mixture was 37 °C for 20 min, after which it was placed in a water bath (72 °C) for 5 min. The protein inhibition percentage was measured using a spectrophotometer (Rayleigh; UV-1800) at 660 nm after the tubes had cooled,19,20 Diclofenac was a positive control, at concentrations 0.1, 0.2, 0.3, 0.4, 0.5, and 0.6 mg/mL.
Antioxidant Capacity In Vitro
DPPH Assay
To estimate the antioxidant capacity of various samples, we used the DPPH method. We determined the DPPH assay of different concentrations of LD oils using the method described by Labhar et al
21
and Alam et al
22
with some modifications. 1 mL of the sample's concentrations and 1 mL of 0.5 mM DPPH solution were mixed vigorously and incubated in a dark area for 30 min. The following formula was used to calculate the percentage of DPPH inhibition. A positive control was ascorbic acid, which was used at 0.6, 0.8, 1, 2, and 4 mg/mL concentrations.
Statistical Analysis
The experiments were performed in triplicate, and the results were presented as the mean value with the corresponding standard deviation (SD). These data were then analyzed using a one-way analysis of variance (ANOVA) in GraphPad Prism software (Version 8, GraphPad Inc). Statistically significant differences were determined at a significance level of P < .05. To explore variations in the chemical composition of EOs as a function of the harvesting period. A principal component analysis (PCA) was performed using OriginPro 2023b.
ADMET Analysis
This study predicted the selected compounds ADMET properties (absorption, distribution, metabolism, excretion, and toxicity) using the SwissADME webserver. 23 The compounds were subjected to ADMET analysis based on established rules outlined by Lipinski et al, 24 Muegge et al, 25 Ghose et al, 26 Egan et al, 27 and Veber et al. 28 Several other parameters were considered, such as the number of hydrogen bond donors and acceptors, rotatable bonds, total polar surface area and synthetic accessibility to assess the drug likeliness of investigated compounds.
Molecular Docking
To observe in-silico anti-inflammatory, and antioxidant activity, the X-ray crystal structure of cyclo-oxygenase 2 (PDBID: 3LN1) at 2.40 Å resolution, 23 and catalase compound II (PDB ID: 2CAG) at 2.70 Å resolution, 29 respectively, was fetched from RCBS protein data bank in PDB format. The protein was prepared for molecular docking studies using the protein preparation wizard from the Schrödinger suite (http://www.schrodinger.com). The process involved eliminating water molecules, rectifying missing side chains or loops with the prime module, and allocating bond orders. Protonation states for each heteroatom were determined based on the number of hydrogen bonds, and the Epik penalty score. Subsequently, the structure was refined, and partial atomic charges were accurately computed using the OPLS4 (Optimized Potential for Liquid Simulation-4) forcefield. Interactive optimization was performed for a cluster of hydrogen-bonded species, and the optimized structure was saved in .mae (Maestro) format within the specified working directory. 30
The two-dimensional (2D) representations of the investigated ligands were obtained from PubChem in .sdf format. Subsequently, the LigPrep module from Schrödinger Release 2016-4 was employed to generate, and optimize their three-dimensional (3D) structures, utilizing the OPLS-2005 force field. The receptor grid generation module within the Schrödinger suite was utilized to create a grid box, the dimensions of which were adjusted to encompass the crystallized ligand within the binding pocket of each protein. The docking procedure was executed using the Glide tool in standard precision (SP) mode. 31 The glide, and emodel scores were documented for each ligand's optimal conformational pose and were subsequently compared with the scores of the corresponding crystallized ligands.
Results and Discussion
Chemical Composition of LD EOs
The chemical composition, and yield of oils exhibit considerable variability, influenced by many factors. These factors encompass the timing of harvest in regions marked by distinct rainy, intermediate, and dry seasons, the micronutrient composition of the soil, the plant's age, and its genetic diversity. Moreover, solar radiation, and precipitation notably influence this compositional variability. 32 These external factors can modify the biosynthesis of volatile compounds in EO, resulting in significant variations in their chemical profile, which will influence the biological activity of the plant's EO through variations in their chemical composition.33,34 Despite their importance, limited research has systematically assessed the effect of seasonality on EO composition, and production.
The extraction yield of LD was notably higher during spring (1.1%), and summer (0.89%) compared to winter (0.42%). These results highlight a significant variation in EO yield, suggesting a seasonal influence on composition that can be explained by the increase in solar radiation from the intermediate to the dry season. 34 Water stress conditions can increase the percentage of LD EO yield stimulating the plant to produce secondary metabolites, including EO. 33 Studies conducted in Algeria have found 0.79% in dry season which is lower than the yield in our study. 35
The chromatographic profiles of Lavandula dentata essential oils obtained by GC-MS (Figure 1) show that the LD EO for the 3 periods has 8 chemical compounds in common, which β-pinene, β-eudesmol, and eucalyptol are the majority compounds with a significant concentration, for the April, and July periods (Figure 2) observing 3 compounds in common. The chemical composition of LD EO harvested in December reveals an abundance of monoterpene compounds such as eucalyptol (16.3%), and β-pinene (9.7%), see Table 1. It should be noted that some studies conducted during the winter period in Brazil also found high concentrations of eucalyptol, reaching up to 40%, as well as in Argentina, where the dominance of eucalyptol (34.33%) was also observed during the winter period.15,36 These compounds are of great importance both medicinally, and aromatically. In addition, there is a significant concentration of L-trans-pinocarveol (14.67%), and L-fenchone (12.82%), compounds frequently used in the flavor industry. 37 Camphor, pinocarvone, and p-menth-1-en-8-ol are present in significant concentrations. They are commonly used in the perfume industry.6,38,39

Gas chromatogram of Lavandula dentata L. essential oil (LD EO); (a) EO April; (b) EO July; (c) EO December.

Venn diagram of essential oil composition of Lavandula dentata L. (LD).
Chemical Composition of LD EOs.

Abbreviations: LD EO, Lavandula dentata L. essential oil; RT, retention time.
Comparing the April EO to the December EO, there are notable variations with a higher concentration of compounds such as β-pinene (11.22%), which exceeds that of April. Simultaneously, the concentration of eucalyptol decreased to 6.21%. This decrease can be explained by the conditions associated with the plant's growth, and the harvesting period. 11
Additionally, there was a notable presence of pinocarveol (18.01%), and β-eudesmol (11.96%) in low concentrations in December. These compounds possess anti-inflammatory, analgesic, and antioxidant properties. 40 Some compounds, such as camphor (3.49%), caryophyllene oxide (4.30%), and linalool (4.35%), continue to be present but at low concentrations. In comparison with other studies conducted in April in the eastern region of Morocco, similarities were observed, particularly regarding the high concentration of β-pinene (27%), and pinocarveol (14.77%). However, other studies carried out in Marrakech (South-West Morocco) showed a low concentration of β-pinene (0.5%), and pinocarveol (0.7%), with a predominance of camphor (50%). 12 In Tebessa (North-East Algeria), notable differences were observed in the chemical composition of EO, including the predominance of α-terpineolene (51%), and camphor (13.43%). Our LD EO shows different concentrations (3.49%) for camphor. 40
The July profile exhibits the dominance of β-pinene (16.47%) compared to April, and December, along with the notable presence of myrtenol (11.89%) with a high concentration that is absent in the 2 previous profiles. Eucalyptol, linalool, and borneol concentrations remain nearly identical to those in April. A comparison of these concentrations with other studies conducted in eastern Morocco during the summer reveals the presence of α-pinene (4.05%), similar to our EO (4.50%). Additionally, low concentrations of linalool (2.76%), and camphor (1.48%) were found, whereas our profile displays higher concentrations. 41 Studies carried out in Algeria also revealed a low camphor concentration (1.6%), and a predominance of eucalyptol (38.4%). 35
The results of principle components analysis (PCA) show that PC1, and PC2 explain 74.1% of the total variance. PC1 explains 46.6% of the total variance, which means that it is the principal component that explains most of the variance. PC2 explains 27.5% of the total variance, the second principal component that presents the most.
Figure 3 reveals that the chemical compounds, α-pinene, β-pinene, and eucalyptol contribute significantly to both PCs. However, the contribution of eucalyptol to PC1 is smaller than that of the chemical compounds, α-pinene, and β-pinene. This means that eucalyptol is less strongly associated with variation in PC1 than the α-pinene, and β-pinene chemicals. On the other hand, PC2 is mainly influenced by the concentration of eucalyptol, a volatile chemical compound, at low temperatures (December). This observation is consistent with Table 1, which shows a higher December concentration than April, and July. This increase in concentration is probably due to a combination of condensation, and the ripening of LD condensation may favor the accumulation of eucalyptol in the leaves. 40 At the same time, plant maturity may also contribute to the increase in eucalyptol concentration. In Figure 3, α-pinene, and β-pinene are mainly represented on the PC1 axis, which reflects the variation in the concentration of volatile chemical compounds at high temperatures. Points in the upper part of the PC1 axis show a higher concentration of these chemical compounds, while points in the lower part of the PC1 axis show a lower concentration. The interpretation of the increase in the concentration of α-pinene, and β-pinene in April, and July is consistent with the results of the previous analysis. This increase is probably due to the higher temperatures in these months.

Biplot of PCA identified in the LD EOs.
Anti-Inflammatory Activity Using BSA
Figure 4 shows that December, and April have a higher anti-inflammatory activity than July. This difference may be due to the relatively high concentration of eucalyptol (16.33%), L-trans-pinocarveol (14.67%), and β-eudesmol (11.96%) in these profiles, these compounds being known for their anti-inflammatory properties. The low anti-inflammatory activity in July (IC50 = 2.01 ± 0.04 µg/mL) compared to Diclofenac, could be due to the decrease in the concentration of eucalyptol, from 16.33% to 6%. 42

Effect of Lavandula dentata L. essential oils (LD EOs) on albumin denaturation. Values significantly different compared to diclofenac (*P < .05 and **P < .01).
Antioxidant Capacity
DPPH Assay
Figure 5 reveals that the July profile shows high antioxidant activity (IC50 = 0.76 ± 0.2 mg/mL), followed by April (IC50 = 0.84 ± 0.04 mg/mL). In contrast, December exhibits have lower antioxidant activity with an IC50 of 1.16 ± 0.02 mg/mL. The high antioxidant activity observed in the July profile can be attributed to the relatively high presence of compounds with antioxidant properties, such as β-pinene (16.47%), pinocarveol (17.70%), and β-eudesmol (8.74%). Interactions between these chemical compounds can enhance overall antioxidant activity, as combining several compounds can synergistically affect antioxidant activity. 43

Antioxidant activity of LD EO by DPPH assay, (a) IC50 values and (b) % of inhibition.
Additionally, when comparing these results to findings from other regions, a publication in Tunisia reported a similarly low concentration of antioxidant activity during winter (IC50 = 113.29 ± 0.012 mg/mL). 14 In contrast, research conducted in Brazil revealed that a significantly higher concentration, ∼ 20 µg/mL, induced an inhibition percentage equal to 5.7 ± 1.4% of activity during the same season. 5
These results suggest that the chemical composition of the EO profiles varies according to the harvesting period, which may significantly impact their antioxidant activity. Certain compounds, such as β-pinene, pinocarveol, 44 and β-eudesmol, contribute to this increased antioxidant activity in April, and July compared to December. 45
ADMET Analysis
Our investigation emphasized assessing the ADMET properties of compounds found in significant concentrations in the EO. These compounds (02, 03, 07, 11, 12, 17, 26, and 29) demonstrated considerable lipid solubility with XLOGP3 as high as 5.44 and very low polarity as indicated by a topological polar surface area (TPSA) of < 20 Å2 in most instances. Additionally, they exhibited low flexibility, characterized by having zero rotatable bonds depicting molecular structure's rigidity. The favorable “drug-likeness” adhered to Lipinski, Ghose, Veber, and Egan rules with zero violation. The compounds under consideration exhibited a high likelihood of passive absorption through the gastrointestinal tract with 0.55 bioavailability, and penetration of the blood–brain barrier. Moreover, they are not the substrates for P-glycoprotein, a key factor in drug efflux. These compounds showed no inhibition of CYP450, except for ligand 17, which inhibits CYP2C19, exhibiting minimum interaction with drug-metabolizing enzymes, and related toxicity (Table 2).
The ADMET Properties Including Absorption, Bioavailability, Metabolism, Lipinski Violations, and Synthetic Accessibility of Leading Ligands.

Abbreviations: TPSA, topological polar surface area; BBB, blood–brain barrier; BA, bioavailability; SA, synthetic accessibility; ADMET, absorption, distribution, metabolism, excretion, and toxicity; GI, gastrointestinal tract.
Molecular Docking
Molecular docking is a noteworthy tool for investigating the binding affinity, and interactions of identified ligands in EO. This approach provides valuable insights into the potential binding capabilities of the ligands with the specified target protein. The ligand-

2D and 3D representation of co-crystallized ligand with target protein (PDB ID: 3LN1).
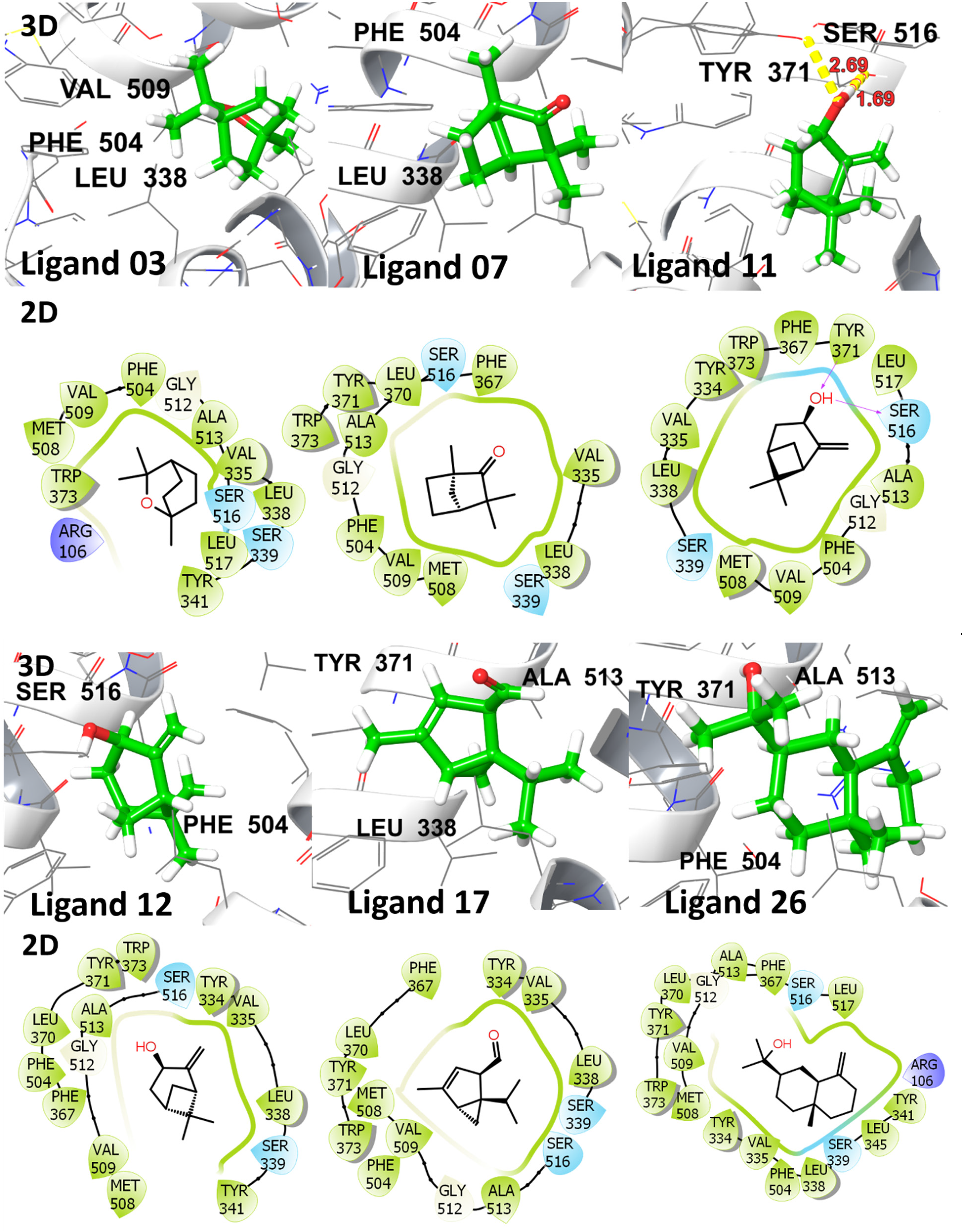
2D and 3D representation of investigated compounds with cyclo-oxygenase 2 (PDB ID: 3LN1).
Molecular Glide Score, H-Bonding Interactions With Distances in Å, Hydrophobic, and Other Interacting Residues for Investigated Ligands With COX-2 Protein (PDB ID: 3LN1).

Abbreviation: HBI, hydrogen bonding interactions.
The antioxidant activity of investigated ligands was evaluated by considering the binding free energy values, and their interactions with 2CAG, as outlined in Table 4. The crystallized ligand exhibited H-bonding interactions with amino acid residues TYR337 responsible for the biochemical environment within the binding pocket. Ligands

2D and 3D representation of investigated ligands with catalase (PDB ID: 2CAG).
Molecular Glide Score, H-Bonding Interactions With Distances (Å), Hydrophobic, and Other Interacting Residues for Investigated Ligands With Catalase Protein (PDB ID: 2CAG).

Abbreviation: HBI, hydrogen bonding interactions.
Despite the decreased concentration of other identified ligands in the EO, they still demonstrate noteworthy binding interactions with anti-inflammatory, and antioxidant proteins. Detailed information on these interactions, and ADMET properties is provided in Tables S1 and S2, and Figures S1 to S10 (Supplemental Material).
Although this study provides valuable information on the chemical composition, and biological properties of LD EO, certain limitations must be taken into account when interpreting the results. The relatively limited harvesting periods (December, April, and July) may not reflect the full range of seasonal variations in LD EO. The volatile compounds identified during each period offer a snapshot, but more extensive sampling throughout the year could provide a more complete understanding of the plant's chemical composition.
Conclusion
In conclusion, this study aims to highlight the importance of the harvesting period on the chemical composition, and biological activities of LD EOs.
The results of this study show significant variations in the chemical composition of LD EOs harvested at 3 times of the year (April, July, and December), and the associated biological activities. These chemical composition variations impact the antioxidant, and anti-inflammatory properties of the essential oil profiles. More specifically, the July, and April profile stands out for its high antioxidant activity compared to the December profile, which can be attributed to the presence of compounds such as β-pinene, and pinocarveol due to their antioxidant properties.
Regarding anti-inflammatory activity, April, and December showed higher activity compared to July. This difference could be due to the relatively high concentration of eucalyptol, L-trans-pinocarveol, and β-eudesmol in these compounds being known for their anti-inflammatory properties.
Molecular docking studies also revealed the strong binding interactions of pinocarveol with antioxidant protein, and L-trans-pinocarveol, β-eudesmol with anti-inflammatory protein, suggesting a promising correlation between the computational predictions, and experimental observations for EO's antioxidant, and anti-inflammatory potential.
These results have significant implications for the potential use of these essential oils in various fields, such as medicine, perfumery, and the flavor industry. Further research is necessary to understand better the mechanisms underlying these variations, and to make the most of the beneficial properties of these EOs from LD.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241230822 - Supplemental material for Seasonal Variations in the Essential Oil Composition and Biological Activities of Wild Lavandula dentata L.
Supplemental material, sj-docx-1-npx-10.1177_1934578X241230822 for Seasonal Variations in the Essential Oil Composition and Biological Activities of Wild Lavandula dentata L. by Amina Labhar, Yahya El-Mernissi, Naoufal Ahidar, Aziz Zouhri, Omar Benamari, Farhan Siddique, Maryam Bashir, Amin Salhi, Mhamed Ahari, Samir Ibenmoussa, Mohammed Bourhia, Ahmad Mohammad Salamatullah, Abdellah ELyoussfi and Hassan Amhamdi in Natural Product Communications
Footnotes
Authors’ Contributions
Amina Labhar, Yahya El-Mernissi, Naoufal Ahidar, Aziz Zouhri, Omar Benamari, and Farhan Siddique: conceptualization, original draft writing, reviewing, and editing. Maryam Bashir, Amin Salhi, Mhamed Ahari, Samir Ibenmoussa, Mohammed Bourhia, and Ahmad: formal analysis, investigations, funding acquisition, reviewing, and editing. Mohammad Salamatullah, Abdellah ELyoussfi, and Hassan Amhamdi: resources, data validation, data curation, and supervision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work is financially supported by the Researchers Supporting Project number (RSP-2024R437), King Saud University, Riyadh, Saudi Arabia.
Ethical Approval
Not applicable.
Statement of Human and Animal Rights
Not applicable.
Informed Consent
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


