Abstract
Background
Periplocae cortex is a well-known Traditional Chinese Medicine with abundant effects, including the treatment of chronic congestive heart failure (CHF). However, the principal components and mechanisms have not been fully elucidated. This study aims to analyze the active ingredients and potential mechanisms of the periplocae cortex in CHF treatment.
Methods
TCMSP, GeneCards, and Comparative Toxicogenomics databases were used to mine the active ingredients and potential targets of the periplocae cortex. Cytoscape 3.9.1 software was employed to construct the “periplocae cortex-active ingredients-potential targets” and protein–protein interaction network. The DAVID 6.8 database was used for Gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis of the predicted targets. The protective effects of β-sitosterol (BSS) were detected in doxorubicin (Dox)-induced H9c2 cells using MTT assay, while the regulatory pathways of BSS were examined using various assays, including real-time quantitative PCR and Western blot. The cardiac troponin I (cTn-I) concentration was also measured.
Results
A total of 13 active ingredients were identified, and 33 targets of CHF were predicted. Network analysis highlighted BSS as the key active ingredient, interacting with 29 targets. GO and KEGG analyses indicated significant involvement of these targets in calcium and apoptosis signaling pathways. Experimental findings demonstrated that BSS enhanced cell viability, reduced intracellular calcium ion concentration, and inhibited caspase 3 activity in Dox-induced H9c2 cells. BSS significantly altered the expression of CHRM1, CASP3, and CASP9 genes (P < .05), and increased CHRM2 expression. Additionally, BSS significantly increased cTn-I concentration. Notably, suppression of calcium and apoptosis signaling pathways partially mitigated BSS's effects on cell viability and cTn-I concentration.
Conclusion
Through network pharmacology and experimental verification, establishes BSS as a key active ingredient in the periplocae cortex for CHF treatment. BSS appears to modulate calcium and apoptosis signaling pathways, thereby exerting therapeutic effects in the treatment of CHF.
Keywords
Introduction
Congestive heart failure (CHF) refers to a pathological condition characterized by the congestion of either the pulmonary or systemic circulation. CHF has a high mortality rate due to inadequate cardiac output to maintain tissue metabolism. The weakening of myocardial contractility is primarily due to coronary heart disease, valvular disease, cardiomyopathy, myocarditis, myocardial metabolic disorders, hypertension, or cardiac overload.1,2 Currently, treatments for CHF include diuretics, angiotensin-converting enzyme inhibitors (ACEI), angiotensin II receptor antagonists (ARB), cardiotonics, β-blockers, and aldosterone receptor antagonists, aiming to lessen disease severity and alleviate symptoms. Nonetheless, these treatments can sometimes lead to adverse reactions like electrolyte disturbances, dry cough, angioedema, arrhythmia, and renal injury.3,4 Additionally, achieving complete recovery of cardiac contractility in CHF patients remains elusive. Recent advancements in CHF therapeutics, such as angiotensin receptor-enkephalinase inhibitors (ARNI), sodium-glucose cotransporter 2 (SGLT-2) inhibitors, and soluble guanylate cyclase (sGC) agonists, have emerged, yet significant uncertainties persist in heart failure (HF) treatment. 5 Concerns include potential rebound effects following abrupt medication discontinuation and the uncertain efficacy of treatments in patients with left ventricular ejection fraction (LVEF) ≥ 60%. Furthermore, there has been limited progress in slowing the progression of HF in patients.
Traditional Chinese Medicine (TCM) is characterized by its holistic approaches and tailored treatment strategies, especially effective in managing chronic and complex illnesses due to its extensive clinical experience. However, its modernization faces challenges due to its intrinsic complexity involving multiple components, targets, and synergistic effects. 6 Recently, network pharmacology has emerged as a globally recognized field, integrating systems biology, multidirectional pharmacology, computational biology, and network analysis. This method constructs intricate networks linking diseases, phenotypes, genes, and drugs, offering a comprehensive view of the drug–disease relationship and aiding in new drug development. Network pharmacology is characterized by its integrity and systematicness, which aligns with the principle of holistic view and dialectical treatment of TCM.7,8 Additionally, molecular docking has become an invaluable technique, revealing intricate details of ligand-receptor interactions at the molecular level. 9 Together, these approaches offer groundbreaking perspectives in identifying active compounds and understanding their mechanisms.
Periplocae cortex commonly known as “Xiangjiapi” or northern “Wujiapi” in China is the dried root bark derived from the Periploca sepium Bge. It is interesting to note that the common name of “Wujiapi” is southern “Wujiapi” which is the root bark from the Acanthopanax gracilistylus W. W. Sminth. According to the Chinese Pharmacopoeia 2020 edition, “Wujiapi” and “Xiangjiapi” are 2 distinct medicinal materials. Although they have similar appearances, their sources, chemical components, pharmacological effects, and toxicity are different. Therefore, they cannot be used interchangeably. The use of the periplocae cortex dates back over 2000 years and was first recorded in Shen Nong Ben Cao Jing. To date, more than 100 compounds have been isolated and identified from the periplocae cortex, including C21 steroidal glycosides, cardiac glycosides, terpenoids, polysaccharides, volatile oils, and fatty acids.10,11 Besides its characteristic antirheumatic effect, the periplocae cortex also exhibits remarkable efficacy in the management of CHF, heart failure edema, and dysuria. 10 Periplocymarin, an effective component extracted from the periplocae cortex, belongs to cardiac glycosides and has been studied for its cardiovascular effects. Research has shown that the extracts of the periplocae cortex exhibited cardiotonic effects on the isolated heart of rats and could strengthen cardiotonic function. 11 As it is well known, periplocin is a poisonous component in the periplocin cortex, and it shows a special cardiotoxic effect. The content of periplocin should be limited in order to reduce the toxicity response when it is used in the clinic. In clinical application, the dosage of periplocae cortex ranges from 3 to 6 g/60 kg/day, which has a therapeutic effect on chronic CHF. 10 Qiliqiangxin capsule containing component periplocae cortex is the first Chinese patent drug with evidence-based medical evidence to positively affect CHF in China. 12 In a multicenter, randomized, double-blind, parallel-group, placebo-controlled study enrolled 512 CHF patients, Qiliqiangxin capsules for 12 weeks reduced the NT-proBNP level. 13 Overall, the periplocae cortex has shown potential in the treatment of cardiovascular diseases, and further research is needed to fully understand its mechanisms of action and clinical applications. Hence, it is imperative to elucidate the action mechanism and active ingredients of the periplocae cortex to ensure its effectiveness and safety. Additionally, pinpointing the key active ingredients, developing monomer preparations, and performing chemical structure modifications as needed could enhance the use of the periplocae cortex in CHF treatment, minimizing potential toxicity. Such efforts are critical in advancing our understanding of the periplocae cortex and its potential therapeutic benefits.
In our study, we utilized network pharmacology, molecular docking, and in vitro validation to investigate the active ingredients, key targets, and potential pharmacological mechanisms of the periplocae cortex against CHF. Our findings have identified that β-sitosterol (BSS), as a crucial active ingredient, interacting with 29 targets and playing a role in both calcium and apoptosis signaling pathways. This finding underscores the significant potential of BSS in treating CHF and other cardiovascular diseases. It provides an important reference point for future drug development and research in this field. In summary, our extensive study presents encouraging prospects for the treatment of CHF, highlighting the therapeutic promise of BSS in cardiovascular medicine.
Materials and Methods
Potential Active Ingredients and Targets Analysis of Periplocae Cortex
The potential active ingredients and the corresponding targets were obtained from the TCMSP database (https://old.tcmsp-e.com/tcmsp.php). Enter “Cortex Periplocae Radicis” in the search box, and filters for retrieving pharmacokinetic data were set to oral bioavailability (OB) ≥ 30% and drug-similarity (DL) ≥ 0.18. Subsequently, the targets of active ingredients retained from the TCMSP database were mapped into standard gene names through the UniProt database (https://www.uniprot.org).
Analysis of the Targets of Periplocae Cortex in the Treatment of CHF
The keyword “convergent heart failure” was searched in the GeneCards database (https://www.genecards.org) to obtain the related genes involved in regulating CHF. Then, genes verified by experiments were screened as the relevant targets of CHF by using the Comparative Toxicology Database (CTD) (http://ctdbase.org). Common CHF-related target genes were obtained from 2 databases. Then, the target genes of CHF and the target gene of active ingredients of periplocae cortex were mapped and screened, and the potential targets of periplocae cortex in the treatment of CHF were obtained by using the online Venn mapping software Bioinformatics (http://bioinformatics.psb.ugent.be/webtools/Venn/).
Potential Target Network Construction
Construction of Active Ingredients-Target Genes Network of Periplocae Cortex
The active ingredients and target genes of the periplocae cortex were integrated into Cytoscape 3.9.1 software to construct a “Periplocae Cortex-Active Components-Potential Targets” network. Within this network, each ingredient or target acting on CHF was represented by a node, while the relationship between the ingredient and the target was represented by a connecting line. By utilizing the “Analyze Network” function in Cytoscape 3.9.1 software, the topological parameters of the network were calculated. The greater the degree of connection between nodes in the network diagram, the more critical the role of the node in the network, and then the important active ingredients and regulatory targets of the periplocae cortex in the treatment of CHF were obtained.
Construction and Analysis of Protein–Protein Interaction (PPI) Network
The potential targets of the periplocae cortex were entered into the String database (https: string-db.org/, Version: 11.5), and defined the species as Homo Sapiens. The “medium confidence (0.400)” option was selected to the minimum required interaction score and selected “hidden disconnected nodes in the network” display options. Finally, the PPI relationship was obtained, downloaded as a TSV format file, and imported into the Cytosape 3.9.1 software. The PPI network was constructed by combining the node degree value and the edge combined score value.
GO and KEGG Pathway Enrichment
The potential targets of the periplocae cortex were entered into DAVID 6.8 database (https://david.ncifcrf.gov/tools.jsp), with the species defined as Homo sapiens. The official gene symbol was selected and the threshold value was set as P < 0.05. Gene ontology (GO) enrichment analysis of potential targets, including biological process (BP), molecular function (MF), and cell composition (CC) BPs were performed. KEGG was used to analyze the signaling pathways of target genes, and the bubble diagram of the enriched signaling pathways was drawn using data analysis software (http://www.bioinformatics.com.cn/).
Molecular Docking
The 3D structure of the target proteins (ADRA1A: 4IYE, CHRM1: 6ZFZ, CHRM2: 5ZKC, CHRNA7: 5AFN, DRD1: 7JVP, HTR2A: 7WC8, CASP9: 2AR9, CASP8: 4JJ7, and CASP3: 5IBP) were retrieved from the protein database (PDB) (https://www.rcsb.org/). The obtained files underwent processing using Discovery Studio 2021 software, which included the removal of water molecules, the addition of hydrogen atoms, and the elimination of any unnecessary ligands. Subsequently, we utilized AutoDockTools-1.5.7 software to analyze the molecule with the highest node count in the target network construction. The selected molecule was exported as a PDBQT format file. The PDBQT format file of receptor protein and ligand small molecule was opened in AutoDockTools-1.5.7 software and generated a docking box while adjusting the parameters. The exhaustiveness parameter was set to 9. Finally, we employed Autodock Vina to perform docking between the receptor protein and ligand small molecule. We analyzed the obtained docking results based on the binding energy (kcal/mol) and the number of hydrogen bonds formed, which provided an evaluation of the binding strength and activity. The results of molecular docking are visualized using Discovery Studio 2021 software.
Dox-Induced H9c2 Cells Injury Model Verification
Experimental Materials and Reagents
BSS was purchased from Glpbio Technology Inc (Montclair). Doxorubicin hydrochloride (DOX ≥ 99.58%), Q-VD-OPh (purity: 99.78%), and amlodipine (purity: 99.76%) were purchased from MedChemExpress Co., Ltd. The Cell Counting Kit-8 (CCK-8 kit) was obtained from Beyotime Biotechnology Co., Ltd. Rat cardiac troponin I (cTn-I) ELISA kit was purchased from Meimian Biotechnology Co., Ltd. Caspase 3 activity assay kit was purchased from Procell Life Science &Technology Co., Ltd. Dulbecco's Minimum Essential Medium (DMEM), penicillin/streptomycin and 0.25% trypsin-EDTA, fetal bovine serum (FBS), and phosphate buffer (PBS) were purchased from Gibco. The Fluo-8® AM fluorescent probe was acquired from Shanghai Maokang Biotechnology Co., Ltd. ReveAid First Strand cDNA Synthesis Kit, SYBR™ Green I nucleic acid gel dye, Nuclease-free H2O and Trizol reagent were purchased from Thermo Fisher Scientific Inc. The dNTP mixed solution, 5×HF Phusion buffers, and DNA polymerase were purchased from New England Biolabs, Inc. The real-time fluorescence quantitative PCR (qRT-PCR) primers were synthesized by Nanjing Kingsley Biotechnology Co., Ltd. Anti-CHRM1 polyclonal antibodies, and anti-CHRM2 polyclonal antibodies were purchased from Solarbio Life Sciences. Caspase-3, Caspase-9, and β-actin antibodies were purchased from Cell Signaling Technology. All other reagents were purchased from other domestic biological companies.
Cell Culture
H9c2 rat cardiomyocytes (CRL-1446) were purchased from the American Type Culture Collection (ATCC), and cultured in DMEM medium containing 10% FBS, 100 U/mL penicillin, and 100 μg/mL streptomycin at 37 oC with 5% CO2. The cells were dissociated with 0.25% trypsin-EDTA solution once the cell density reached ∼ 80% to 90%.
Cell Viability Determination
H9c2 cells were seeded into a 96-well plate at the density of 5 × 103 cells/well for 24 h. Following treatment, the culture medium was discarded, and the cells were incubated with DMEM basic medium supplemented with 0.5 mg/mL CCK-8 at 37 oC for 2 h. Absorbance at 450 nm was then measured using a multi-function microplate reader (Wallace). Cell viability (%) was determined by comparing the results with those of the untreated group.
Determination of Intracellular Ca2+ Concentration
H9c2 cells were seeded into a 96-well plate at a density of 5 × 103 cells/ well for 24 h. The cells were then pre-treated with BSS (5, 10, and 20 μM) for 2 h followed by exposure to 5 μM Dox for 24 h. Subsequently, DMEM basic culture medium supplemented with 4 μM Fluo-8® AM was added, and the cells were incubated at 37 oC for 30 min. The supernatant was discarded, and 100 μL PBS buffer containing 1 mM probenecid was added to reduce the leakage of the de-esterification probe. Fluorescence intensity was measured at an excitation wavelength of 490 nm and an emission wavelength of 515 nm using a multifunctional microplate reader. Following this, 10 μL of CCK-8 solution was added to each well. After a further 2 h incubation, absorbance at 450 nm was recorded. The ratio of fluorescence intensity to absorbance for each group was then calculated.
Determination of Caspase 3 Activity
H9c2 cells were seeded into a 12-well plate at a density of 5 × 104 cells/well for 24 h. The cells were then pre-treated with BSS (5, 10, and 20 μM) for 2 h followed by exposure to 5 μM Dox for 24 h. The Caspase 3 activity was detected according to the manufacturer's instructions. Caspase 3 activity was expressed as units/mg protein.
cTn-I Concentration Detection
The cTn-I concentration was detected according to the manufacturer's instructions. Briefly, the culture supernatant was collected posttreatment and mixed with the appropriate reagents. The addition of the stop solution transforms the color from blue to yellow. The color intensity is quantified at 450 nm using a spectrophotometer. Concurrently, calibration standards are assayed with the samples, enabling the creation of a standard curve that correlates OD with cTn-I concentration. The cTn-I concentration in the samples is subsequently determined by comparing their OD values to this standard curve.
RNA Extraction and RT-qPCR Determination
Total RNA was extracted from cultured H9c2 cells with 500 μL Trizol according to the manufacturer's protocol. The RNA was reverse transcribed into cDNA using the RevertAid First Strand cDNA Synthesis Kit. The related genes were analyzed by RT-qPCR using the ABI StepOnePlus™ sequence detection system (Thermo Fisher Scientific, Inc). SYBR-Green I dye was used to detect the PCR amplification of the target gene. The PCR cycle conditions were as follows: Initial step at 95 oC for 5 min, followed by 38 cycles of 95 oC for 15 s, 60 oC for 30 s, and 72 oC for 15 s, followed by melting curve program (95 oC for 15 s, 60 oC for 60 s, and 95 oC for 15 s). The relative expression level of the target gene was calculated using the 2−ΔΔCt method, and GAPDH was used as an internal control. Each sample was tested at least 3 times. Specific primer sequences are shown in Table 1.
Primer Sequence of Target Gene.

Western Blot Assay
H9c2 cells were lysed with RIPA buffer containing 1 mM PMSF and 1% (v/v) phosphatase inhibitor on ice. The cell lysis supernatant was collected and quantified according to the procedure of the BCA protein assay kit (Thermo Fisher Scientific). SDS-PAGE was used to separate proteins according to molecular weight. The polyvinylidene difluoride (PVDF) membranes were blocked with 5% nonfat dried milk. The blocking solution was removed and specific primary antibodies against CHRM1, CHRM2, Caspase 3, Caspase 9, and β-actin were added and incubated (all used at 1:1000 dilution) overnight at 4 oC. HRP-conjugated antibody (1: 2000) was added to the membranes for 2 h at room temperature. Western blot images and the densities of the bands were performed using the Bio-Rad Gel Imaging Systems (Bio-Rad, Hercules).
Statistical Treatment
The experimental results were statistically analyzed by GraphPad Prism 9.0 software (GraphPad). The data were expressed using mean and standard deviation (mean ± SD). One-way analysis of variance (ANOVA) followed by Dunnett's or Turkey's multiple comparisons test was used to analyze differences. The significant statistical difference was indicated by P < .05.
Results
Analysis of Potential Active Components of Periplocae Cortex
A total of 61 main compounds of periplocae cortex were obtained from the TCMSP database, among which 17 compounds were obtained according to the screening criteria of OB ≥ 30% and DL ≥ 0.18, including poriferast-5-en-3β-ol, BSS, sitosterol, 21-o-methyl-5,14-pregndie-3β,14β,17β, 21-tetrol-20-one, Glycoside K_qt, Glycozolidal, and glycoside E_qt, etc. The targets of these 17 compounds were analyzed, and it was found that BSS and Glycozolidal had more targets. The details are shown in Table 2.
Basic Information on Potential Active Ingredients of Periplocae Cortex.

Analysis of the Targets of Periplocae Cortex in the Treatment of CHF
Among the 17 compounds identified in the periplocae cortex, 4 compounds were found to have no targets. Consequently, the analysis focused on the remaining 13 active compounds, encompassing a total of 43 targets. These were compared with data from the CTD and GeneCards databases. From this comparison, 33 potential targets were identified, which are believed to be relevant for the treatment of CHF using the periplocae cortex. Detailed information on these findings can be found in Figure 1 and Table 3.

Venn diagram of targets of periplocae cortex in the treatment of congestive heart failure (CHF).
Targets for the Treatment of CHF With Periplocae Cortex.

Analysis of “Periplocae Cortex-Active Ingredients-Potential Action Targets” Network
In the study on the periplocae cortex for treating CHF, 33 targets were correlated with 13 active compounds, resulting in a network comprising 48 nodes. This included one node each for CHF and periplocae cortex, 13 nodes for active compounds, and 33 nodes for potential targets, alongside 110 connecting edges. The “Network Analyze” function in Cytoscape 3.9.1 software was used to analyze the topology parameters, such as the degree value and center proximity value of the nodes. Nodes with higher degree and center proximity values were more likely to be significant active compounds or targets. The top 6 active ingredients were BSS, 21-o-methyl-5,14-pregnedine-3β,14β,17β, 21-tetrol-20-one, Glycoside K_qt, glycoside E_qt, delta 5-Pregnenetriol, and xysmalogenin. The top 4 targets were NCOA2, NR3C2, PGR, and NCOA1, which could interact with 9, 9, 8, and 6 active components, respectively. The details are shown in Figure 2 and Table 4.

Topological Characteristics of “Periplocae Cortex-Active Components-Potential Targets” Network.

PPI Network Analysis
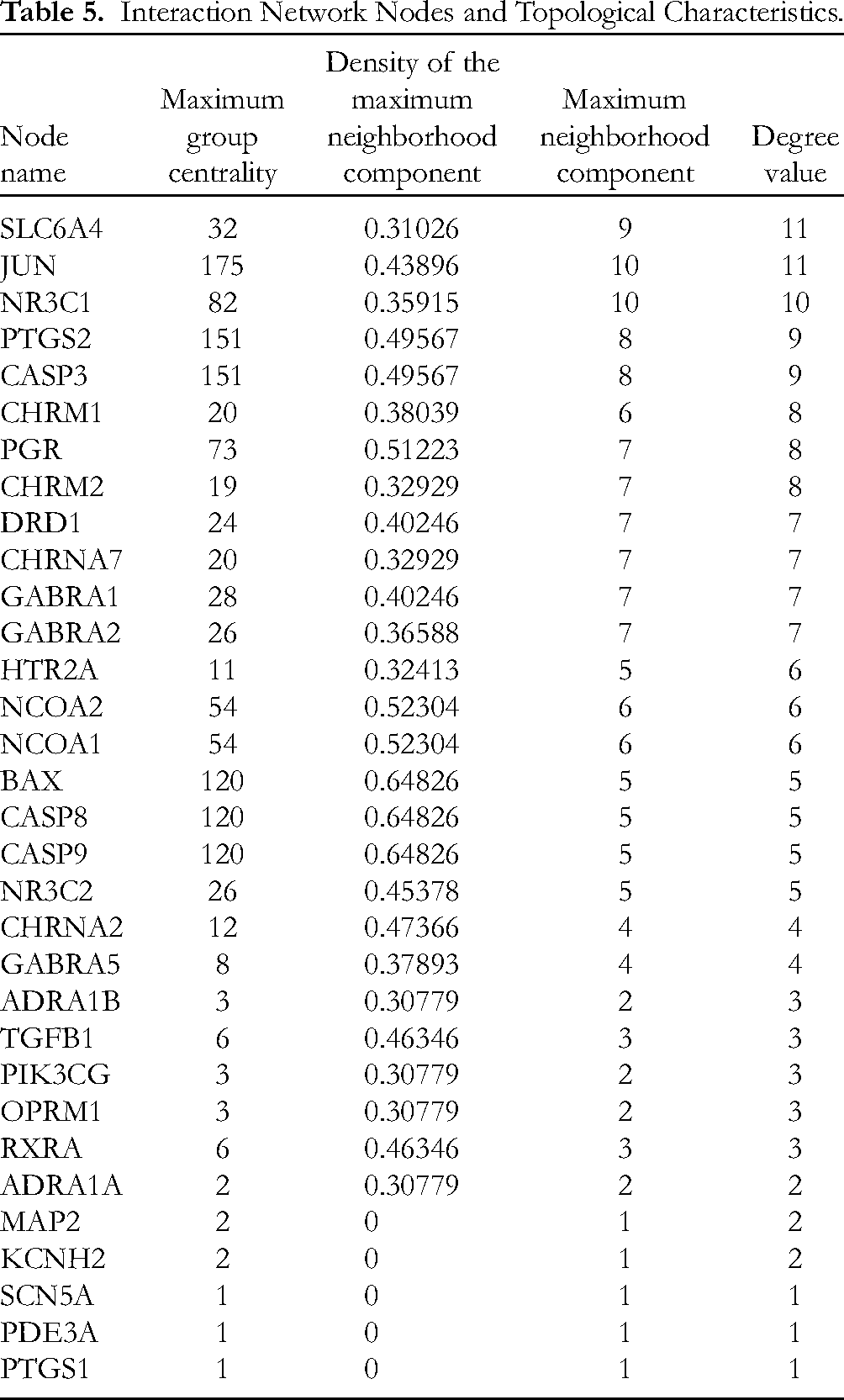
Hub genes, also known as key genes or central genes, play a pivotal role in BPs and regulatory networks. The COUNTIF function in Excel was utilized to analyze 32 interacting proteins, excluding PON2 due to its lack of interacting proteins. The number of interacting proteins for each protein was calculated, and this data was used to plot nodes in a graph. Larger nodes represent a higher count of interacting proteins. The interaction strength, indicated by the “Combined Score” from the String database, was represented by the width of the edges in the network, with wider edges denoting higher scores. These 32 targets were entered into the String database, and the output files were imported into Cytoscape 3.9.1 software to construct a PPI network. This network comprised 86 edges across 32 nodes, with an average node degree value of 5.38. Targets exceeding this average degree value included SLC6A4, JUN, NR3C1, PTGS2, CASP3, CHRM1, PGR, CHRM2, DRD1, CHRNA7, GABRA1, GABRA2, HTR2A, NCOA2, and NCOA1.The average clustering coefficient was 0.596, and it demonstrated a highly significant PPI enrichment, with a P-value of < 1.0 × 10−16. The results are shown in Figure 3 and Table 5.

The protein–protein interaction (PPI) network of targets of the periplocae cortex. Nodes represent the proteins. Edge represents the protein–protein association.
Interaction Network Nodes and Topological Characteristics.
GO Enrichment and KEGG Pathway Analysis
GO Enrichment Analysis
The DAVID software facilitated an analysis of the GO functions for 32 genes, yielding 224 significant GO terms (P < .05). These were categorized into BP, CC, and MF, constituting 62.5%, 16.52%, and 20.98% of the terms, respectively. This breakdown comprised 140 terms in BP, 37 in CC, and 47 in MF. The top 10 terms were analyzed based on the −LogP value in descending order. It was found that the targets in BP were mainly involved in the response to estradiol, response to xenobiotic stimulation, chemical synaptic transmission, response to drugs, regulation of membrane potential, nervous system process, positive regulation of apoptosis, G protein-coupled cyclic nucleotide/receptor signaling pathway, response to lipopolysaccharide, and signal transduction. CC targets mainly involve the presynaptic membrane, postsynaptic module, and plasma membrane. MF targets mainly involved neurotransmitter receptor activity, extracellular ligand-gated ion channel activity, enzyme binding, cysteine endopeptidase activity involved in the execution stage of apoptosis, RNA polymerase II transcription factor activity, ligand-activated sequence-specific DNA binding, cysteine endopeptidase activity involved in the process of apoptosis, inhibitory extracellular ligand-gated ion channel activity, and GABA-gated chloride ion channel activity. The results are shown in Figure 4.

Gene ontology (GO) function enrichment analysis (Top 10).
KEGG Pathway Analysis
Fifty-five signal pathways (P < .05) were obtained by KEGG pathway enrichment analysis. The top 10 pathways were sorted according to −LogP value and a bubble chart was drawn online using Micro-bioinformatics (http://www.bioinformatics.com.cn/login/). It was found that the targets of the periplocae cortex mainly involved the interaction between neuroactive ligands and receptors, morphine addiction, Kaposi's sarcoma-associated herpesvirus infection, calcium signaling pathway, and apoptosis. ClueGO + CluePedia could more vividly show the relationship between pathways and the enrichment of genes in pathways. The participating genes mainly included CHRM2, CHRM1, CHRNA7, DRD1, HTR2A, ADRA1B, ADRA1A, CASP9, CASP8, CASP3, and BAX. The results are shown in Figure 5A and B.

Bubble diagram of Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis (Top 20).
Molecular Docking
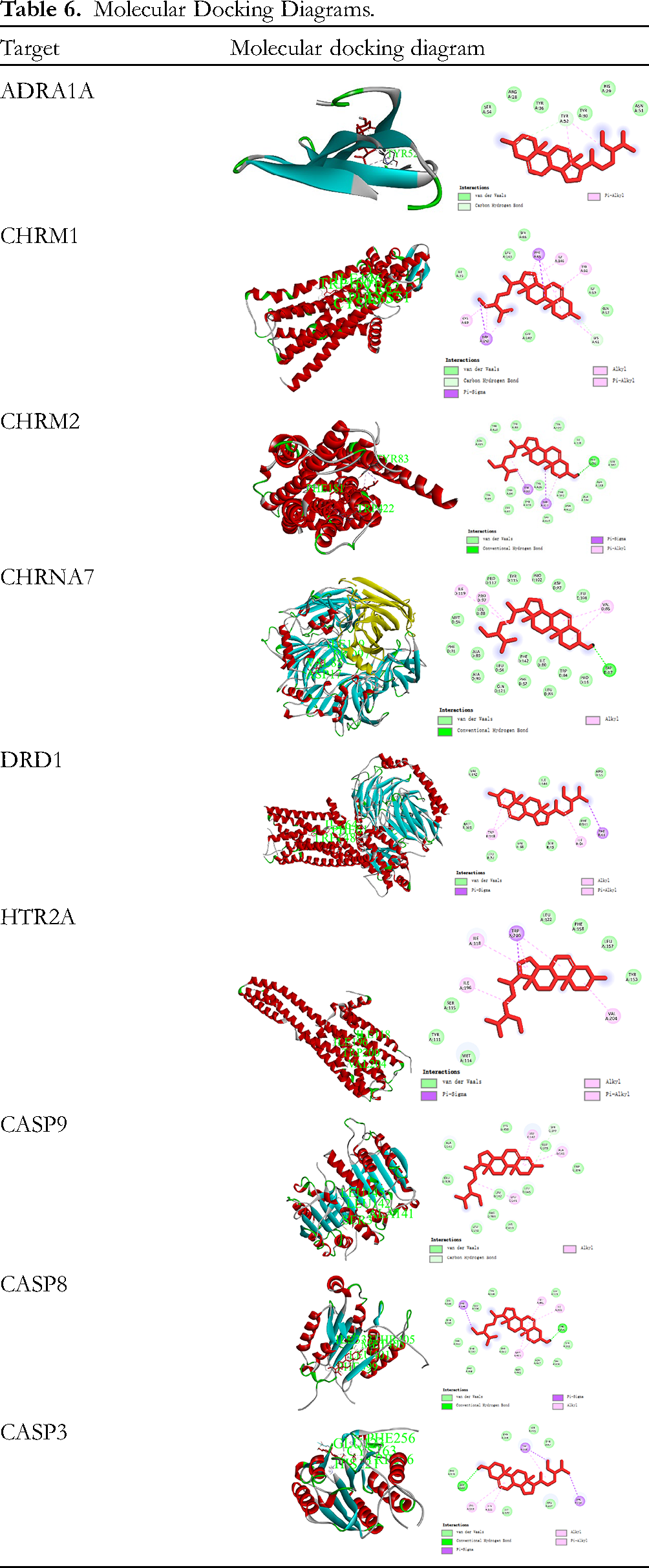
Although 21-o-methyl-5,14-pregnedine-3β,14β,17β, 21-tetrol-20-one, Glycoside K_qt, glycoside E_qt, and delta 5-Pregnenetriol primarily target PGR, NR3C2, NCOA2, and NCOA1, these targets do not rank among the top 10 KEGG signaling pathways. This indicates that calcium signaling pathway and apoptosis genes regulated by BSS, including CHRM2, CHRM1, CHRNA7, DRD1, HTR2A, ADRA1B, ADRA1A, CASP9, CASP3, and BAX, play important roles in CHF treatment. Consequently, the molecular docking analysis focused on targets predominantly regulated by BSS. As shown in Table 6, BSS primarily engages with ADRA1A, CHRM1, DRD1, HTR2A, and CAS9 through weaker interactions like Pi-Alkyl, Pi-Sigma, and Alkyl. However, BSS forms hydrogen bonds with specific amino acid residues: PHE181 in CHRM2, ASP17 in CHRNA7, THR405 in CASP8, and GLU123 in CASP3.
Molecular Docking Diagrams.
BSS Inhibited Dox-Induced Intracellular Accumulation of Ca2+ and Inhibited Caspase 3 Activity in H9c2 Cells
The protective effects of BSS on H9c2 cells subjected to Dox damage were investigated using the CCK-8 assay. As shown in Figure 6A, treatment with 5 μM Dox for 24 h markedly decreased cell viability in H9c2 cells compared to the Ctrl group (P < .0001). However, treatment with BSS protected H9c2 cells against Dox-induced cardiotoxicity in a dose-dependent manner. The cell viability rates after treatment with BSS at concentrations of 10 and 20 μM were 57.9145% and 65.9247%, respectively (P < .05, P < .0001). Dox is a chemotherapy drug that can cause an increase in intracellular Ca2+ levels, which can trigger various cellular responses such as apoptosis and oxidative stress. As shown in Figure 6B, the mean relative fluorescence intensity in the Dox group was significantly higher compared to the Ctrl group (75496.1 vs 162 041, P < .0001). However, BSS treatment at 20 μM significantly inhibited Dox-induced Ca2+ overload in H9c2 cells (127 075 vs 162 041, P < .001). Moreover, BSS could significantly inhibit the increase in caspase3 activity caused by Dox (Figure 6C). In summary, these results collectively indicated that BSS effectively countered Dox-related cardiotoxic effects in H9c2 cells.

Effects of BSS on Ca2+ concentration and caspase-3 activity in H9c2 cells subjected to Dox injury. H9c2 cells were pretreated with BSS at the indicated concentration for 2 h followed by 5 μM Dox for 24 h: (A) cell viability was examined by CCK-8 assay, (B) relative fluorescence intensity, and (C) Caspase 3 activity. Data are expressed as mean ± SD of 3 independent experiments. Data were analyzed using one-way ANOVA and Dunnett's multiple comparisons test. ****P < .0001 versus Ctrl group; #P < .05, ###P < .001, and ####P < .0001 versus Dox group.
BSS Inhibited Calcium and Apoptosis Signaling Pathways in Dox-Induced H9c2 Cells
Bioinformatics analysis has revealed that calcium and apoptosis signaling pathways-related genes were the most important targets in treating CHF with BSS. To further investigate the underlying mechanism, the mRNA levels of related regulators were analyzed in H9c2 cells after 24 h of Dox treatment in the presence or absence of BSS. As shown in Figure 7A to E, compared to the ctrl group, the protein expression levels of CHRM1, CASP3, and CASP9 in the Dox group were significantly increased (P < .01). CHRM2 protein levels were significantly decreased (P < .0001). Compared to the Dox group, BSS significantly decreased the levels of CHRM1, CASP3, and CASP9 (P < .01), while increasing the expression of CHRM2 (P < .05). Similarly, mRNA levels of CHRM1, CASP3, and CASP9 were significantly elevated in the Dox group versus the control group (Figure 8A, E and F, P < .05), with no significant change in DRD1 mRNA levels (Figure 8C), despite an increase in content. Additionally, CHRM2 and ADRA1A mRNA levels were significantly reduced (Figure 8B and D, P < .01). Compared to the Dox group, 20 μM BSS significantly decreased the mRNA levels of CHRM1, CASP3, and CASP9 (P < .05), while increasing the content of CHRM2 mRNA (P < .05). These data indicated that BSS might alleviate the calcium influx and apoptosis caused by Dox-induced myocardial damage.

Effects of BSS on the calcium and apoptosis signaling pathway-related protein expression. (A-E) Representative Western blots (A) and densitometric analysis of the expression of CHRM1 (B), CHRM2 (C), Caspase 3 (D), and Caspase 9 (E). Data are expressed as mean ± SD of 3 independent experiments. Data were analyzed using one-way ANOVA and Dunnett's multiple comparisons test. **P < .01, ***P < .001, and ****P < .0001 versus Ctrl group; #P < .05, ##P < .01, ###P < .001, and ####P < .0001 versus Dox group.

Effects of BSS on the calcium and apoptosis signaling pathway-related genes mRNA contents. (A-F) The mRNA expression levels of CHRM1 (A), CHRM2 (B), DRD1 (C), ADRA1A (D), Caspase 3 (E), and Caspase 9 (F) were detected by qRT-PCR. Data are expressed as mean ± SD of 3 independent experiments. Data were analyzed using one-way ANOVA and Dunnett's multiple comparisons test. *P < .05 and **P < .01 versus Ctrl group; #P < .05 and ##P < .01 versus Dox group.
Inhibition of Calcium and Apoptosis Signaling Pathways Weakened the Protective Effects of BSS in Dox-Induced H9c2 Cells
Calcium channel blocker amlodipine and caspase activity inhibitor Q-VD-OPh were separately added to ascertain if BSS mediates its protective effects through the regulation of calcium and apoptosis signaling pathways. Experimental findings revealed that inhibiting these pathways slightly diminished the protective effects of BSS, yet significant protection was still observed. Similarly, inhibiting these pathways reduced the efficacy of BSS in lowering cTn-I concentration. This suggests that the protective mechanism of BSS relies on both calcium and apoptosis signaling pathways (Figure 9).

Effects of BSS on the survival of H9c2 cells following the inhibition of calcium and apoptosis signaling pathways. H9c2 cells were pretreated with amlodipine (10 μM) or Q-VD-OPh (50 μM) for 2 h, respectively, and then incubated with BSS at the indicated concentration for 2 h followed by 5 μM Dox for 24 h. (A, B) Cell viability was examined by CCK-8 assay. (C) cTn-I concentration (pg/mL) was examined. Data are expressed as mean ± SD of 3 independent experiments. Data were analyzed using one-way ANOVA and Turkey's multiple comparisons test. ****P < .0001 versus Ctrl group; ####P < .0001 versus Dox group; &P < .05, &&&P < .001, and &&&&P < .0001.
Discussion
HF is one of the most concerned clinical syndromes in the cardiovascular field with a high rehospitalization rate, poor prognosis, and high mortality. 14 In recent years, the treatment patterns for HF have changed from “The Golden Triangle” to “New Quadruple Therapy,” and new therapeutic drugs have been launched in succession. 2 As a representative drug of ARNI, sacubitril-valsartan can reduce cardiovascular adverse events in HF patients in the early stages of application, and long-term application can maintain or even reverse ventricular remodeling. However, in clinical practice, HF patients often suffer from progressive deterioration of renal function, and RAAS blockers are often used to protect the kidney. 15 Although it is unclear whether the protective effect of sacubitril-valsartan is better than that of RAAS blockers, patients may use a dosage lower than the tolerance target, resulting in a discount in the curative effect and may lead to a withdrawal rebound in sudden withdrawal of the drug. SGLT-2 inhibitors (empagliflozin or sotagliflozin) may cause adverse reactions, including diarrhea and hypoglycemia. 16 Currently, the drugs available for the treatment of HF are still limited and far from meeting the clinical requirements. Therefore, novel therapeutic drugs are urgently needed.
TCM has been practiced and developed alongside Chinese civilization for thousands of years. TCM is a promising source of potential compounds that are effective and less toxic. 17 Simultaneously, it is of great significance to find symptoms, material basis, or mechanisms consistent with TCM for clinical treatment. However, it is inappropriate to rely on the Western medicine research model (“one disease–one target–one drug” dogma) to do TCM mechanism research. 18 With the rapid development of bioinformatics, system biology, comprehensive pharmacology, and high-throughput histology, network pharmacology has proven to be an effective drug-target analysis approach in the comprehensive analysis of TCM. 19 It describes the complex relationship among diseases, human biological systems, and drug targets from a systematic perspective. Through complementary integration with molecular docking, novel insights into active compounds screening are provided and the mysterious veil of the underlying mechanism of TCM may be uncovered in the future, triggering a revolution in the modernization and internationalization of TCM. 20 However, network pharmacology may miss truly effective compounds due to incomplete data, signal noise in complex networks, limitations of predictive models, and constraints on experimental validation.
CHF belongs to a group of cardiovascular disorders that includes palpitation, asthma, phlegm retention, and edema. The primary pathological basis of CHF is blood stasis and qi stagnation. 21 It was found that periplocin, a component of periplocin cortex, could cause myocardial toxicity in the treatment of CHF. 22 Therefore, it is essential to identify the low toxicity and active molecules of the periplocin cortex. In this study, we demonstrated that BSS was the key active molecule of the periplocin cortex in CHF treatment. Studies have shown that BSS is the most predominant phytosterol and has various biological effects, including anti-anxiety, sedative effects, analgesia, immunomodulation, antibacterial, anticancer, anti-inflammatory, lipid-lowering effects, liver protection, antioxidation, and anti-diabetes.23,24 Previous studies have also shown that BSS is relatively safe and has no genotoxicity or cytotoxicity.25,26 Li et al 27 found that BSS has significant cytoprotective effects, with the highest activity observed at 40 μM in the hypoxia/reoxygenation (H/R) model in the H9c2 cell. Lin et al 28 reported that BSS (10, 20, and 50 μM) could protect H9c2 cells from H/R-induced myocardial injury by regulating the PPARγ/NF-κB signaling pathway. Wong et al 29 also found that BSS (3 and 10 μM) could protect H9c2 cells and rat hearts from oxidative damage by enhancing glutathione redox. However, there was no relevant research on BSS in the Dox-induced H9c2 model. In this study, we investigated the effects of BSS (5, 10, and 20 μM) on the Dox-induced H9c2 model, which is a model of acute ventricular dysfunction and late-onset cardiomyopathy and CHF caused by the anticancer drug Dox. 30 We found that BSS at 10 and 20 μM significantly increased cell viability and decreased cTn-I concentration. These results suggest that BSS may have potential therapeutic applications in the treatment of CHF.
Apoptosis, commonly known as programmed cell death, plays a critical role in myocardial cell death postacute myocardial infarction. This process significantly contributes to the progression of left ventricular remodeling and the onset of symptomatic HF. Consequently, therapies targeting the inhibition of apoptosis could serve as effective measures in preventing or treating HF.31,32 In this study, we found that BSS could significantly reduce the content of Caspase 3 and 9 mRNA, concurrently inhibiting the expression of these proteins in H9c2 cells affected by Dox-induced injury. BSS inhibited Caspase 3 activity in H9c2 cells. More importantly, inhibition of apoptosis signaling pathways weakened the protective effects of BSS. Caspase 3 and 9 are key enzymes involved in the apoptotic process. By curtailing their expression, BSS potentially impedes apoptosis, thereby enhancing the survival of cardiomyocytes in the Dox-challenged H9c2 cell model. Additionally, molecular docking analyses reveal that BSS exhibits a modest interaction with CAS9 and forms hydrogen bonds with the amino acid residues THR405 of CAS8 and GLU123 of CAS3. This implies a potential interaction of BSS with CAS8 and CAS3, both crucial caspases in the apoptotic cascade. The direct interaction between BSS and CAS8 and CAS3 needs to be further verified by additional experiments. These findings suggest that BSS has anti-apoptotic effects and could be a potential therapeutic strategy for HF.
The muscarinic acetylcholine receptor M1 (CHRM1) is a Gq protein-coupled receptor that plays a crucial role in the vascular system by facilitating the conversion of GTP into cGMP, which subsequently leads to the efflux of cytosolic Ca2+. In contrast, the muscarinic acetylcholine receptor M2 (CHRM2) is associated with the Gi protein, inhibiting adenylate cyclase activity and thus diminishing the conversion of ATP to cAMP. This modulation is significant in orchestrating a variety of cellular processes. 33 In this study, we found that BSS markedly attenuated Dox-induced intracellular Ca2+ accumulation, and upregulated CHRM2 mRNA expression while downregulating CHRM1, findings that were corroborated by Western blot analysis. These results imply that BSS may modulate muscarinic acetylcholine receptor activity and cAMP levels, potentially offering a therapeutic strategy for the prevention or treatment of HF. We found that impeding calcium signaling pathways diminished the cardioprotective effects of BSS, as evidenced by the reduced efficacy of BSS in decreasing cardiac troponin I (cTn-I) levels when calcium signaling was inhibited. This suggests that calcium signaling pathways are integral to the protective mechanism of BSS.
Furthermore, the cardiac dopamine D1 receptor (DRD1) has been identified as another potential therapeutic target for the amelioration of ventricular arrhythmias associated with HF. 34 Our study revealed that BSS could downregulate DRD1 mRNA expression, suggesting that BSS may confer protection by modulating DRD1 signaling pathways. Additionally, adrenergic receptors (ADRs) mediate the effects of catecholamines in tissues, and specifically, the expression of ADRA1A (but not ADRA1B) has been linked to reduced myocardial cell death. This suggests that ADRA1A may play a role in myocardial protection. 35 We discovered that BSS could enhance ADRA1A mRNA expression levels in H9c2 cells. Molecular docking results indicated that BSS exhibited weak interactions with ADRA1A, CHRM1, CHRM2, and DRD1. These findings suggest that BSS may exert its cardioprotective effects by modulating the signaling pathways associated with these receptors. Further studies are warranted to elucidate the precise molecular mechanisms by which BSS influences these pathways and to confirm its therapeutic potential in vivo.
This study has certain limitations, one of which is the inability to rule out the potential for BSS to exert cardioprotective effects through pathways not identified in our research. Additionally, our findings are currently confined to in vitro validation. To comprehensively assess the therapeutic efficacy of BSS, future research should focus on evaluating its impact in a DOX-induced HF mice model, thereby providing a more robust and applicable understanding of its potential as a treatment option.
Conclusion
In summary, our research showed that BSS was the key active compound in the periplocae cortex for treating CHF and demonstrated the potential protective effects of BSS against Dox-induced myocardial injury in vitro. The cardioprotective effect of BSS may be related to the calcium and apoptosis signaling pathways. BSS exerts its therapeutic benefits in CHF through a multifaceted approach, targeting multiple pathways, which underscores its significance in the treatment of this condition. These insights are pivotal in guiding the regulation of periplocin content to mitigate myocardial toxicity, an aspect that is vital for the future clinical utility of BSS, particularly in broadening its therapeutic applications for CHF. Further studies are needed to confirm the efficacy and safety of BSS in animal models and clinical trials. Network pharmacology and molecular docking technology have shown great potential in drug discovery and development. However, hurdles such as the precision of drug target predictions, the dependability of compound screening, and the safety of drug candidates remain to be surmounted. It is essential to enhance the accuracy and dependability of network pharmacology approaches and to synergize these with other experimental modalities to expedite the drug discovery process. With collaborative efforts from researchers across various disciplines, we are optimistic that network pharmacology will contribute significantly to the innovation of new therapeutic agents, shaping the future of medicinal development.
Footnotes
Abbreviation
Acknowledgments
The authors thank the Sixth Affiliated Hospital of Guangzhou Medical University, the Science and Technology Bureau of Qingyuan, the Guangdong Bureau of Traditional Chinese Medicine, the State Natural Science Funds Commission, and the Natural Science Foundation of Guangdong Province for their financial support.
Authors’ Contributions
X Z and Z L conceived and designed the research, drafted the original manuscript, and provided financial support. B G, J G, and G X performed the experiments, carried out the data processing, and provided technical support. Q C, Z K, and G X assisted in the manuscript revision. All the authors have read and approved the final manuscript for publication.
Availability of data and materials
The data used to support the findings of this study are available from the first author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Medical Research Fund of Qingyuan People's Hospital (No. 20200101), the Plan on Enhancing Scientific Research in Guangzhou Medical University (No. GZMU-SH-009/016), the open research funds from the Sixth Affiliated Hospital of Guangzhou Medical University, Qingyuan People's Hospital (No. 202201-208), the 2022 Science and Technology Project of Qingyuan (No. 2022KJJH036), the Medical Research Fund of Guangdong Province (No. A2021431), the Research Project of Traditional Chinese Medicine Bureau of Guangdong Province (Nos. 20211459 and 20221470), the Scientific Projects from Guangdong Province (No. 2019A1515110751), and the National Science Foundation of China (No. 82003726).
Ethical Approval
This study does not contain any studies with human or animal subjects. Ethical approval does not apply to this article.
Statement of Human and Animal Rights
This study does not contain any studies with human or animal subjects. The statement of human and animal rights does not apply to this article.
Informed Consent
This study does not contain any studies with human or animal subjects. Informed consent does not apply to this article.
