Abstract
Cancer is one of the leading causes of death worldwide and commonly becomes resistant to chemotherapy, therefore, it is important to search for and evaluate novel sources of nontoxic antitumor agents. The cactus Pachycereus marginatus is native to Mexico and is traditionally recommended to treat gastrointestinal infections. Tumor-bearing mice survival, liver function, and histopathology by P. marginatus extracts and the in vitro effects of isolated compounds lophenol, β-sitosterol, and palmitic acid were investigated, using the L5178Y-R lymphoma murine model. In vivo oral administration of the aqueous partition at 0.5 mg/kg caused 60% survival at 60 days, without altering liver parenchyma and enzymes, as compared with 40% survival induced by vincristine (0.05 mg/kg), and no survival of tumor-bearing mice without treatment. Furthermore, P. marginatus n-hexane extract, lophenol, β-sitosterol, and palmitic acid compounds caused up to 89%, 73%, and 83% in vitro cytotoxicity to L5178Y-R cells, respectively. These results may support the evaluation of P. marginatus extracts and bioactive compounds in clinical studies.
Plants are the most common natural sources of therapeutic agents because of their great biodiversity and proven pharmacological effectiveness; however, only a marginal number of them have been scientifically validated. Plant chemotherapeutic agents represent an alternative to conventional therapy against cancer, the most commonly used are Taxol, vincristine, vinblastine, and irinotecan. 1 Cactaceae family contains about 1500 species, some of which have been reported with anticancer, anti-inflammatory, and hepatoprotective properties. 2-4 Pachycereus marginatus (DC) cactus, also known as Stenocereus marginatus, Cereus marginatus, and organ pipe, has columnar trunks reaching 20 feet in height and is commonly used as fodder, living fences, fuelwood, and as an alternative medicine to treat gastrointestinal infections. 5 It grows wild in dry and hot climate States of Mexico such as Nuevo León, Aguascalientes, Tamaulipas, Morelos, and Veracruz, and in Texas. 6 Cancer is one of the main causes of morbidity and mortality worldwide, with millions of new cases and deaths every year. In men, most cancers affect the prostate, lungs, and gastrointestinal tissues, and in women, cancers in the breast, lung, cervix, and stomach are usually observed; each year 160 000 new cases are reported in Mexico, with 80 000 deaths. 7
Preclinical studies on the antitumor effectiveness and liver function by P. marginatus extracts have not been yet investigated, as well as elucidation of its bioactive compounds. Methanol-aqueous partition caused 60% survival of tumor-bearing mice, which remained alive for 60 days (log-rank test, P < 0.01, as compared with untreated tumor-bearing mice), whereas 100% mortality was observed in extract-untreated tumor-bearing mice (tumor untreated) from 30 days posttumor administration (Figure 1a). Antitumor agent vincristine at 0.05 mg/kg caused 40% survival of tumor-bearing mice from 35 days posttumor administration, which remained alive for 60 days, whereas 100% survival was observed in normal untreated mice not inoculated with tumor cells (Figure 1a). Furthermore, P. marginatus methanol-aqueous partition and vincristine caused significant (P < 0.05) 27% and 70% reduction in tumor volume index 27 days posttumor administration, respectively, compared with untreated tumor-bearing mice control (Figure 1b).
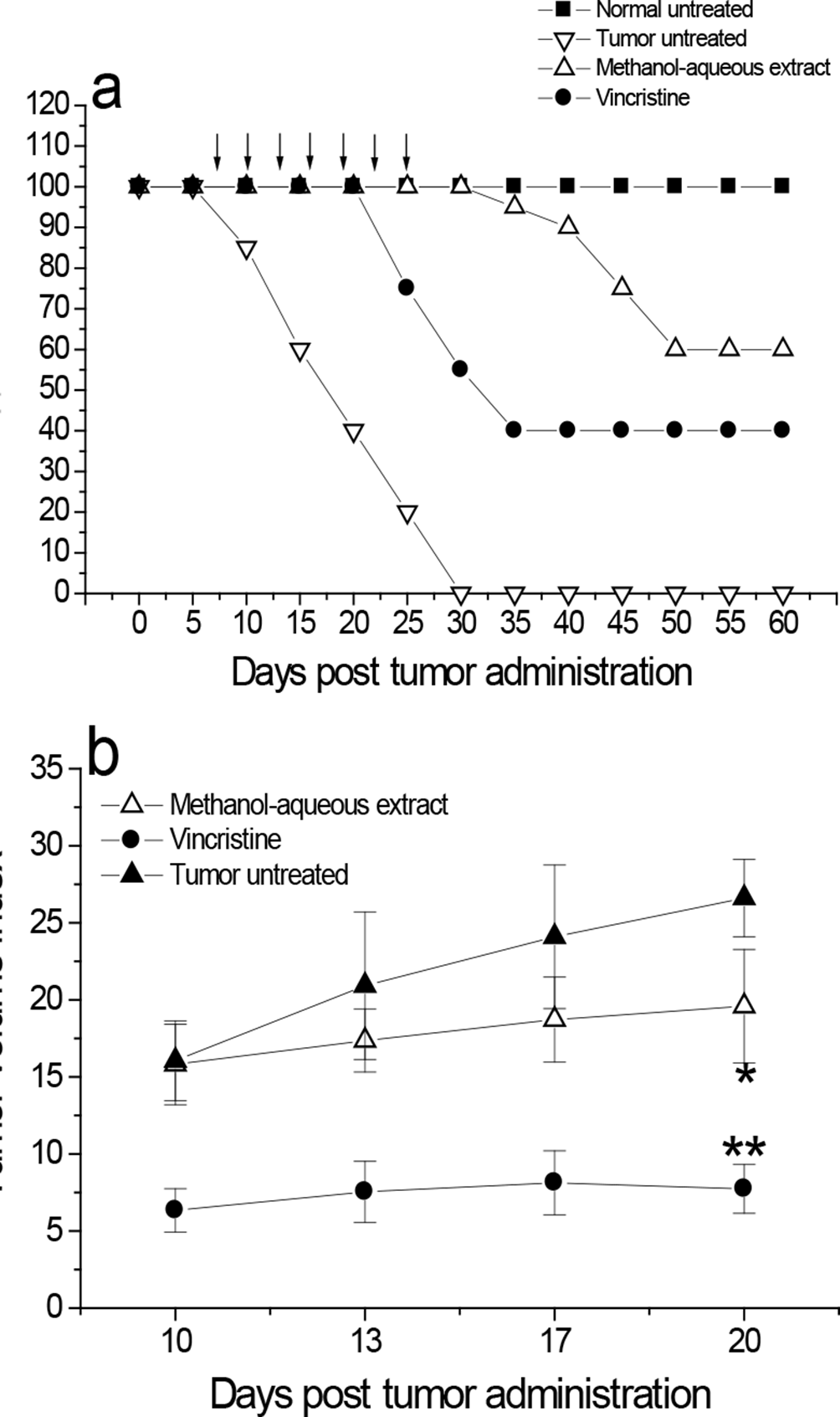
Increased mice survival and reduced tumor volume by P. marginatus methanol-aqueous partition. Female 2-month-old Balb/c mice received a subcutaneous administration (upper right thigh) of 2 × 107 L5178Y-R lymphoma cells/mL. After 7 days of tumor inoculation, L5178Y-R tumor-bearing mice were orally treated with P. marginatus methanol-aqueous partition, as explained in the text; controls included P. marginatus extract-untreated tumor-bearing mice (tumor untreated), normal untreated control without tumor (normal untreated), and vincristine-treated tumor-bearing mice. Mice survival (a) and tumor volume (b) were determined as explained in the text. *, P < 0.05; P < 0.01, as compared with the response of extract-untreated tumor-bearing mice.
In regard to liver function and tissue injury, in the present study, no alterations on these parameters were observed after P. marginatus extract treatments (Figure 2 and Table 1), despite an increase of 2.6 over twice the upper normal limit (2N) following hexane extract treatment (Table 1). Criteria of drug-induced liver injury have been defined by others. 8 Benichou reported that a combined increase in ALP and total bilirubin, provided that one of them is above 2 N the upper normal limit, indicates liver damage, considering that no other biochemical test is specific to liver disorder. Isolated increase even over 2 N in ALP or TB should be considered only a biochemical abnormality and not necessarily a sign of liver injury. As shown in Table 1, the serum level of enzymes and total bilirubin were normal after treatment with P. marginatus extracts, the only increase above 2 N was caused by the hexane extract treatment (Table 1), which might have been a biochemical abnormality, rather than indicating liver injury. 8 Serum levels of total bilirubin, glutamic oxaloacetic transaminase (GOT), glutamic pyruvic transaminase (GPT), and alkaline phosphatase (ALP) were not significantly altered after P. marginatus methanol-aqueous partition treatment (Table 1).

Histological assessment of liver tissue after in vivo P. marginatus extracts treatment. Normal mice were treated by oral administration of 0.2 mL of P. marginatus extracts every 3 days (prepared fresh in PBS), for a total of 7 doses. Controls were normal extract-untreated mice. After killing animals, liver sections were fixed in 10% buffered formalin for histopathological assessment of liver damage. (a) Normal extract-untreated control, (b) P. marginatus hexane extract-treated, (c) P. marginatus chloroform extract-treated, (d) P. marginatus methanol extract-treated, and (e) P. marginatus methanol-aqueous partition-treated mice (100× magnification).
Effect of Pachycereus. marginatus Extracts on Liver Enzymes and Bilirubin Serum Levels.

aValues in the parenthesis represent the fold increase/decrease of the upper normal limit.
Histopathological examination supported results from enzyme analysis by showing absence of liver injury; microscopically, liver slices from control and extract-treated animals showed normal parenchymal architecture, without noticeable alterations (Figure 2). It is known that chemotherapeutic agents can cause side effects such as headache, hypertension, and hepatotoxicity, among others. Paracetamol, for instance, can cause severe liver necrosis by free radicals production. 9 In the present study, P. marginatus extracts did not significantly alter liver parenchyma and serum levels of total bilirubin, GOT, GPT, and ALP enzymes, indicating absence of liver damage (Figure 2 and Table 1).
In search for bioactive antitumor agents, P. marginatus lophenol, β-sitosterol, and palmitic acid isolated compounds caused significant (P < 0.05) 24% to 89%, 17% to 73%, and 33% to 83% toxicity to L5178Y-R cells at concentrations ranging from 15.62 to 500 µg/mL, respectively (Table 2), whereas vehicle control for n-hexane was not cytotoxic, and vincristine caused 75% cytotoxicity at 100 µg/mL (data not shown). Because of the increase of resistance of tumor cells to chemical agents, together with unwanted side effects, it is of critical relevance to discover and develop novel natural alternatives with significant potential against cancer, with little or no adverse effects. Other cactus species with antitumor potential include Lophophora williamsii (Lem. ex Salm-Dyck) Coult. (peyote), which is toxic for murine L5178Y-R and fibroblastoma L929, and human myeloid U937 and mammary gland MCF7 tumor cells, and Lophocereus schottii (Engelm.) Britton & Rose. 2,4 In the present study, evidence has been provided for the isolation of lophenol, β-sitosterol, and palmitic acid from P. marginatus hexane extracts, which were toxic to L5178Y-R cells at concentrations as low as 15.6 µg/mL, in a concentration-dependent manner (Table 2). The triterpene lophenol has not been described in P. marginatus, but it was reported in Lophocereus schottii, Tieghemella heckelii, and Dracaena cochinchinensis and possesses antitumor properties. 10
Toxicity of Lophenol, β-Sitosterol, and Palmitic Acid Against L5178Y-R Lymphoma Cells.
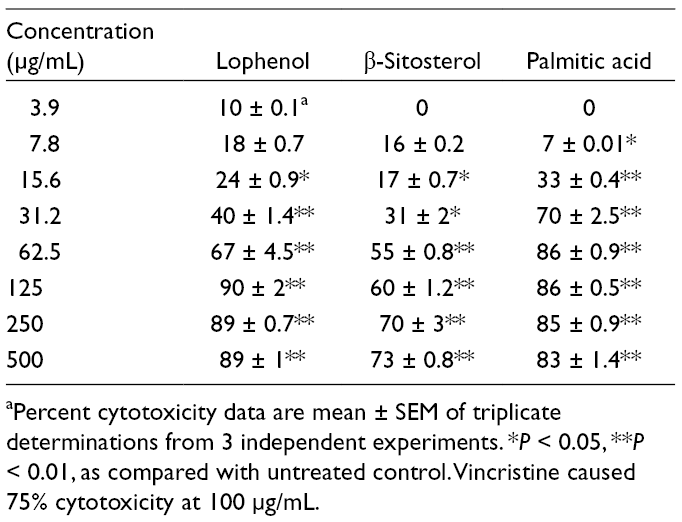
aPercent cytotoxicity data are mean ± SEM of triplicate determinations from 3 independent experiments. *P < 0.05, **P < 0.01, as compared with untreated control. Vincristine caused 75% cytotoxicity at 100 µg/mL.
Similarly, β-sitosterol, the most common plant sterol, has not been yet reported in P. marginatus, but has been shown to inhibit human cancer growth. 11 In addition, palmitic acid has not been reported in P. marginatus, but other species have reported its antitumor properties. 12 In conclusion, the present study demonstrated that P. marginatus extracts inhibited in vivo murine tumor progression, which increased tumor-bearing mice survival, without affecting liver structure and function, and the hexane isolated bioactive compounds lophenol, β-sitosterol, and palmitic acid caused up to 89% tumor cell in vitro toxicity. These results may support the evaluation of P. marginatus extracts and bioactive compounds in clinical studies.
Experimental
Reagents and Culture Media
Penicillin-streptomycin solution, l-glutamine, phosphate buffered saline (PBS), and RPMI 1640 medium were obtained from Life Technologies (Grand Island, NY). Fetal bovine serum (FBS), sodium dodecyl sulfate (SDS), vincristine, N,N-dimethylformamide (DMF), and 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) were purchased from Sigma-Aldrich (St Louis, MO).
The tumor cell line L5178Y-R (mouse DBA/2 lymphoma R, clone CRL-1722) was purchased from The American Type Culture Collection (Rockville, MD), and it was maintained in culture flasks with RPMI 1640 medium, supplemented with 10% FBS, 1% l-glutamine, and 0.5% penicillin-streptomycin solution (referred as complete RPMI 1640 medium) at 37°C, in a humidified atmosphere of 5% CO2 in air; cellular density was kept between 105 and 106 cells/mL. Extraction buffer was prepared by dissolving 20% (wt/vol) SDS at 37°C in a solution of 50% each DMF and demineralized water, and the pH was adjusted to 4.7.
Preparation of Plant Extracts
Pachycereus marginatus was identified by M.Sci. María del Consuelo González de la Rosa, Chief of Facultad de Ciencias Biológicas Herbarium at Universidad Autónoma de Nuevo León (UANL), Mexico, with voucher specimen number 025588. Sixty grams of the powdered stems were sequentially extracted with 600 mL of hexane, chloroform, and methanol by Soxhlet system during 40 hours each. Extracts were concentrated to dryness using a rotary evaporator Büchi (Brinkmann Instruments Inc., Switzerland) to yield 11.6, 11.4, and 23.1 g, respectively, stored at 6°C until use, and dissolved in distilled water for biological activity testing. The methanol-aqueous partition of the extract was prepared by using 11 g of methanol extract and dissolving it in methanol, then in hexane, and shaking for 10 minutes. The resulting solution was separated with a funnel and washed with ethyl acetate, followed by washing with water and then dried.
Animals
Six-week-old female Balb/c mice (22-28 g) were purchased from Harlan Sprague-Dawley Inc. (Indianapolis, IN). They were kept in a pathogen- and stress-free environment at 24°C, under a light-dark cycle (light phase, 06:00-18:00 hours), and given water and food ad libitum. Institutional guide for the care and use of laboratory animals was followed.
In Vivo Tumor Growth Inhibition by Pachycereus marginatus Methanol-Aqueous Partition
L5178Y-R cell suspension was washed twice in PBS by centrifuging at 2000 rpm for 10 minutes and adjusted to 2 × 107 cells/mL in complete RPMI 1640 medium. Next, female 2-month-old Balb/c mice received 100 µL subcutaneous administration (upper right thigh) of this suspension. After 7 days of tumor inoculation, L5178Y-R tumor-bearing mice were treated by oral administration of 0.2 mL methanol-aqueous partition (0.5 mg/kg) every 3 days (prepared fresh in PBS), for a total of 7 doses. Controls were extract-untreated tumor-bearing mice (tumor untreated), normal untreated control without tumor (normal untreated), and vincristine (intravenous, 0.05 mg/kg)-tumor-bearing mice. Mice survival was measured daily for 60 days. To determine tumor volume by external caliper, the longitudinal tumor diameter (length) and the transverse tumor diameter (width) were determined using the following formula: Tumor volume (mm3) = 1/2(length × width2). Tumor volumes were taken on day 0 (7 days after tumor administration and first treatment), and on days 7 (day 14 posttumor injection), 10 (day 18 posttumor injection), 13 (day 21 posttumor injection), 17 (day 24 posttumor injection), and 20 (day 27 posttumor injection). The mean volume index was calculated as follows: mean final tumor volume (mm3) at day 27 posttumor injection (before animal death)/mean initial tumor volume (mm3) at 7 days after tumor administration.
In Vivo Hepatotoxicity Assay and Liver Damage Assessment
Mice were orally administered with 200 µL of the methanol-aqueous partition at 0.5 mg/mL; after 10 days of treatment, blood was drawn by cardiac puncture under ether anesthesia to determine serum total bilirubin, GOT, GPT, and ALP, in the Laboratorio de Análisis Químico Clínicos of Facultad de Ciencias Químicas at UANL. After animals were euthanized, livers were excised and weighed. Liver tissues were embedded in paraffin, sectioned, and stained with hematoxylin and eosin for histological studies. 13
Isolation and Characterization of Compounds with Antitumor Activity From Pachycereus marginatus
The chemical nature of some of the active compounds in the n-hexane P. marginatus extract was elucidated. For this, 1 g of n-hexane extract was chromatographed on a 20 g-silica gel column and eluted with stepwise gradients of n-hexane-chloroform, chloroform-ethyl acetate, and methanol. The eluted material was divided into fractions A (43.8 mg), B (789.5 mg), C (77.5 mg), D (621 mg), E (1.01 g), and F (1.57 g), based on their TLC (CHCl3 100%) profiles. The most L5178Y-R cytotoxic fractions D, E, and F (in vitro assay data not shown) were further fractionated as explained above. Fraction D (500 mg) was chromatographed and eluted as detailed above and the eluted material was divided in fractions DA (18 mg), DB (76 mg), DC (160 mg), DD (17 mg), and DE (22 mg), based on their TLC (CHCl3-EtOAc 9.8:0.8) profiles. DC fraction contained a main compound with Rf = 0.31, which was additionally chromatographed with 8 g silica gel and stepwise gradients of chloroform-ethyl acetate. In turn, eluted fractions were divided into fractions named DCA (8 mg), DCB (48 mg), DCC (31 mg), DCD (65 mg), and DCE (6 mg), based on their TLC profiles.
Fraction DCD contained a main compound with Rf = 0.31, whose structure was established as lophenol (C28H48O) by analyzing 1- and 2-dimensional NMR spectroscopic results (1H-NMR, 13C-NMR, DEPT-135, COSY, HSQC, HMBC) and comparing with literature data. 14 Fraction E was chromatographed and eluted as detailed above and the eluted material was divided into fractions named EA (19.8 mg), EB (168 mg), EC (107 mg), ED (251 mg), and EE (405 mg), based on their TLC (CHCl3-EtOAc 9:1) profiles. Fraction ED contained a main compound according to its TLC profile (Rf = 0.5), which was chromatographed on a 10 g-silica gel column and eluted as mentioned above and the eluted subfractions were divided in fractions EDA (9 mg), EDB (46 mg), EDC (114 mg), and EDD (56 mg), based on their TLC profiles. EDC fraction (Rf = 0.5) was further chromatographed and eluted with stepwise gradients of chloroform-ethyl acetate. Subfractions were divided in the fractions EDCA (13.6 mg), EDCB (51 mg), EDCC (9.5 mg), and EDCD (26 mg), based on their TLC profiles. This later fraction contained a compound with Rf = 0.5, whose structure was defined as β-sitosterol (C29H50O), 15 according to 1- and 2-dimensional NMR spectroscopic results and comparing with literature data. In addition, 600 mg of fraction F were chromatographed on a 20 g-silica gel column and eluted with stepwise gradients of chloroform-ethyl acetate, and methanol. Next, eluted fractions were collected and combined based on their TLC profiles into the fractions FA (17.3 mg), FB (36.7 mg), FC (79 mg), FD (108.3 mg), and FE (340.5 mg). Fraction FC contained a main compound with Rf = 0.66, which was further chromatographed in an 8 g-silica gel column and eluted with stepwise gradients of chloroform-ethyl acetate, and methanol. Eluted subfractions were then collected and combined based on their TLC profiles in the fractions FCA (37.1 mg), FCB (6 mg), FCC (8.7 mg), and FCD (17.3 mg). Fraction FCA contained a compound with Rf = 0.66, with some polar impurities, because of which it was further chromatographed as explained above and eluted subfractions were collected and combined based on their TLC profiles, in the fractions FCAA (15.8 mg), and FCAB (17.8 mg). Fraction FCAA contained a compound with Rf = 0.66, corresponding to palmitic acid, 16 according to 1- and 2-dimensional NMR spectroscopic results and comparing with literature data (C16H32O2).
Pachycereus marginatus Extract Isolated Compounds Effects on L5178Y-R Cells Viability
L5178Y-R cell suspensions were washed 3 times in RPMI 1640 and suspended and adjusted to 5 × 104 cells/mL in complete RPMI medium. One hundred microliters of the cell suspensions were then added to flat-bottomed 96-well plates (Becton Dickinson, Cockeysville, MD), containing 100 µL triplicate cultures of complete RPMI 1640 (unstimulated control), the compounds lophenol, β-sitosterol, and palmitic acid at various concentrations in complete RPMI 1640, and fractions-free vehicle (vehicle was similarly processed as with P. marginatus fractions but without plant material), and vincristine (100 µg/mL) as a positive control. After incubation for 44 hours at 37°C with 5% CO2, MTT (0.5 mg/mL, final concentration) was added to all wells, and cultures were additionally incubated for 4 hours. Next, cell cultures were incubated for 16 hours with extraction buffer (100 µL/well) and optical densities, resulting from dissolved formazan crystals, were then read in a microplate reader (DTX 880 Multimode detector, Becton Dickinson, Austria) at 570 nm. The percentage of cytotoxicity was calculated as follows: % Cytotoxicity = 100-[(A570 in extract treated cells/A570 in untreated cells) (100)].
Statistical Analysis
Results were expressed as mean ± SEM of the response of 3 replicate determinations per treatment (in vitro study) or 5 mice per experimental group from 3 independent experiments. The level of significance was assessed by ANOVA and Dunnet’s t-test. Survival curves were calculated by the Kaplan-Meier method, and the statistical significance of differences in the survival curves between the groups was evaluated by the log-rank test.
Footnotes
Acknowledgments
We would like to thank Laboratorio de Inmunología y Virología from Facultad de Ciencias Biológicas at UANL for supporting the developing of this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Programa de Investigación Científica y Tecnológica (PAICYT) of UANL, Mexico to RGF.
