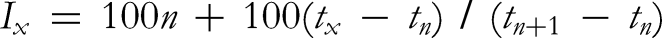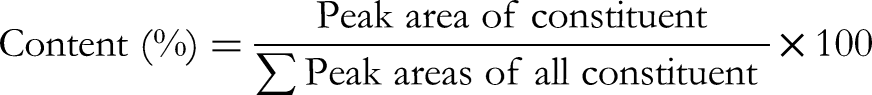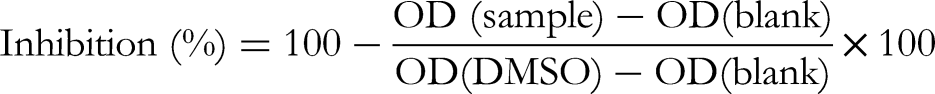Abstract
Purpose of the Research
Peliosanthes micrantha Aver.et N.Tanaka has been found in central highland Dak Nong province, Vietnam, and used locally to treat various diseases. This research studied phytochemicals, spectroscopic analysis, and cytotoxic activities of P. micrantha rhizomes (PM-R).
Methods
In this study, steam-distilled essential oil of P. micrantha rhizome (EO) was analyzed by gas chromatography - mass spectrometry (GC-MS). Phytoconstituents of P. micrantha rhizome ethanolic extract (PM-R-ET) were separated by column chromatography and their structures were determined. Attenuated total reflectance (ATR) – Mid Fourier-transform infrared spectroscopy (FTIR) was used to characterize its dry powder (PM-R-DP).
Major Findings
The results showed that EO contained eleven constituents. Two compounds, pumilaside A (2) and β–sitosterol (4), were successfully isolated from PM-R-ET, and unambiguously elucidated by nuclear magnetic resonance (NMR) and mass spectrometry (HRMS). ATR-FTIR spectrum of PM-R-DP discovered 26 different functional groups of its compounds. PM-R-ET showed IC50 value of 96.14 ± 6.12 µg/mL against HT29 cell lines.
Conclusion
Our findings partly confirm previous Ultra High-Performance Liquid Chromatography-Quadrupole Time-of-Flight Mass Spectrometry (UHPLC-QTOF-MS) study and show two isolated compounds and EO constituents, spectroscopic properties, and cytotoxic activity from the P. micrantha rhizomes.
Introduction
The genus Peliosanthes Andrews currently referred to Asparagaceae, is indigenous to Asia. 1 Many studies on the discovery and ecological characteristics of the Peliosanthes species reported that the Peliosanthes grow mainly in subtropical to tropical regions within Asia. 2 In 2013, the medicinal herb Peliosanthes micrantha Aver.et N.Tanaka belonging to Asparagaceae, was discovered in Vietnam, 3 and the same species (called Sam cau Krông Nô or Hue da la nho in Vietnamese) was found in central highland Krong No district, Dak Nong province, Vietnam. According to the indigenous peoples of Dak Nong province, the rhizomes of P. micrantha were decocted or alcohol-extracted and used for tonic, detox, hepatic, body nourishment, and vitality male enhancement. In Cao Bang province, P. teta was found and used for various ailments. 4 Only a few research of chemistry and biological activities have been published even for the genus Peliosanthes and especially for P. micrantha species. Till today, glycoside J-3, J-4, and three spirostanol glycosides were isolated from P. sinica species. 5 Our previous research presented that P. micrantha rhizome ethanolic extract (PM-R-ET) showed mild DPPH antioxidant activity. 6
High-Performance Liquid Chromatography-Quadrupole Time-of-Flight Mass Spectrometry (HPLC-QTOF-MS) is a powerful tool in both chromatographic separation and mass spectrometric detection of the secondary metabolite compounds in herbal extracts, and establishing the relationship between the profile of compound and the biological activities. 7 After the analysis of chemical components is carried out, it is essential to apply the methods for the guided separation of the metabolite compounds in the herbal extracts. Zhang L. et al 8 have successfully applied the Ultra High-Performance Liquid Chromatography-Quadrupole Time-of-Flight Mass Spectrometry (UHPLC-QTOF-MS)-guidance strategy including finding, tracking, purity determination, and structural elucidation of targeted compounds as well as HPLC chromatographic conditions transplantation to prepare the sulfur-containing ginsenoside Rg1 derivative from sulfur-fumigated ginseng. Xue J. et al 9 have developed one integrated data-mining strategy that combines molecular networking, in-house polygonal mass defect filtering, and diagnostic fragment ion filtering to identify phytochemicals in Stephania tetrandra based on LC-MS data; then applied to isolate two new bioflavonoids. In Vietnam, although the application of HPLC-QTOF-MS/MS to identify the metabolites was used in some research groups, 10 the discovery of new compounds in the medicinal plants was mainly carried out by the traditional column chromatography 11 or other new instrumental separation methods. 12
Essential oils are natural volatile fractions distilled from herbal plants and formed by classes of substances such as esters of fatty acids, aldehyde alcohols, ester, aliphatic hydrocarbons, mono and sesquiterpenes, etc 13 The gas chromatography - mass spectrometry (GC-MS) technique was most frequently used to identify and quantitatively determine the constituents of volatile compounds, hydrocarbons, alcohols, acids, esters, etc, in the given plant. Meher et al 14 have used the GC-MS technique to analyze the essential oil of both the leaf and the rhizome of Zingiber purpureum species.
Fourier-transform infrared spectroscopy (FTIR) between 4000 cm−1 to 400 cm−1 is an established time-saving method to characterize and identify functional groups of metabolites in herbal plants. The absorption band of a specific bond is called the characteristic band of this bond. 15 Canbay et al 16 have used FTIR analysis to identify the functional groups of the bioactive compounds in grapeseed extracts obtained from ethyl acetate, water, and petroleum ether. FTIR analysis can be used with the Attenuated total reflectance (ATR) technique for qualitative functional group analysis of extracts or foods. 17
Plant-based medicines have good potential as a primary source of chemotherapeutic drugs. Cytotoxic compounds such as metabolites including phytosterols are currently used for cancer chemotherapy. 18 A variety of plants have been evaluated to investigate their cytotoxic activities. Many tumor cell lines such as human breast adenocarcinoma (MCF-7), the human promyelocytic leukemia tumor (HL60), human neuroblastoma (SH-SY5Y), African green monkey kidney fibroblast (CV1-P), rat brain glioma (C6), mouse macrophage (RAW264.7), mouse fibroblast (3T6) cells have been tested for in vitro cytotoxic potential. 19
Recently, we reported the use of UHPLC-QTOF-MS to tentatively identify phytochemicals in an ethanol extract of the P. micrantha rhizomes and dereplicated one known compound of glycoside J-3 (
In this paper, we reported the tentative identification of eleven constituents in P. micrantha rhizome essential oil by GC-MS. The isolation and structural identification of two known natural products, pumilaside A (
Material and Methods
Chemicals and Reagents
All reagents, n-hexane, ethyl acetate, dichloromethane, methanol, anhydrous sodium sulfate, NaHCO3, L-glutamine, sodium piruvate, 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT), sulforhodamine B, and ellipticine were purchased from Sigma-Aldrich Chem. Co. (St. Louis, MO., USA). Retention Index Standard Product No. R 8769 was purchased from Sigma-Aldrich Chem. Co. (St. Louis, MO., USA). Silica gel (Qingdao Ocean Chemical Group Co. Ltd, China), C18 columns, and reversed-phase Triart C18 column (YMC Co, Ltd, Japan), silica gel G plates (Qingdao Marine Chemical Inc., China). Deionized water for HPLC; HPLC grade acetonitrile, methanol, reagent grade formic acid (≥98%) were obtained from Fisher (USA). Dulbecco's Modified Eagle's Medium (DMEM), Minimum Essential Medium with Eagle salt (MEME), and fetal bovine serum (FBS) were obtained from Life Technologies, Inc., (Gaithersburg, MD, USA). TCA (Trichloroacetic acid), acetic acid were purchased from Bio Basic Inc. (Canada). Tris-base, SRB Dimethyl sulphoxide (DMSO) were obtained from MP Biomedicals (Strasbourg, France). Sulforhodamine B and Ellipticine were purchased from Sigma Aldrich (St. Louis, MO, USA). Human lung carcinoma A549 cells, human breast carcinoma MCF-7 cells, human hepatocarcinoma HepG2 cells, human cervical carcinoma Hela cells, and human colorectal carcinoma HT-29 cells were obtained from Prof. J. M. Pezzuto, Long-Island University (US) and Prof. Jeanette Maier, University of Milan (Italy).
Plant Material
The rhizomes of P. micrantha were collected in Dak Nong Province, Vietnam in October 2021 and identified by Dr Nguyen Sinh Khang, Institute of Ecology and Biological Resources, Vietnam Academy of Science and Technology (VAST). The voucher specimen (PM-R-2021) has been lodged at the Center for High Technology Research and Development, VAST.
Steam-Distilled Essential oil Preparation and GC-MS Analysis
Powdered P. micrantha rhizomes (300 g) were placed on a stainless steel holed disk over distilled water (3 L) in our 5 L apparatus with a chilled condenser. Essential oil was vaporized by steam distillation for 4 h and separated. The isolated oil was dried over anhydrous sodium sulfate for 12 h to obtain essential oil, P. micrantha rhizomes (PM-R-EO), weighed, and placed in a refrigerator. Experiments were triplicated.
GC–MS analysis of the PM-R-EO was carried out as previously described
11
on a 456-GC (SCION) equipped with a flame ionization detector, SQ Select (SCION), and a Rxi-5ms column (30 m × 0.25 mm (i.d.), film thickness 0.25 µm df, RESTEK). The injector and detector temperatures were set at 250 °C and 250 °C, respectively. The column temperature progress was initiated at 80 °C for 1 min., followed by an increase to 170 °C at 5 °C/min, then to 280 °C at 50 °C/min, and finally kept isothermally at 280 °C for 3 min. The carrier gas was helium at a flow rate of 1 mL/min. The sample was injected by splitting. The split ratio injected was 30 µL of essential oil. The mass selective detector conditions were as follows: ionization voltage 70 eV, emission current 40 mA, and acquisitions scan mass range 50-500 amu under full scan, scan velocity 1 s/scan. Retention Index was calculated for GC non-isothermal ramp using Van den Dool and Kratz equation (Van den Dool and Kratz RI) areas by the following formula:
Where: Ix = retention index of component x
n = carbon number of n-alkane peak eluting immediately before sample peak
tx = retention time of sample peak
tn = retention time of n-alkane peak eluting immediately before sample peak
tn + 1 = retention time of n-alkane peak eluting immediately after sample peak.
Identification of peaks and constituents was done with the NIST 17 library (resemblance percentage > 90%). Constituent content (%) was calculated as relative peak area to total peak areas by the following formula:
Ethanol Extraction
The fresh rhizomes of P. micrantha (8.700 kg) are washed, sliced, and dried for 5 days in an oven at 80 °C and ground into powder (PM-R-DP). Dried powder of the rhizomes of P. micrantha (2.800 kg) was extracted with 96% ethanol (17.5 L × 3 times) at room temperature in an ultrasonic 20 kHz and 1 kW extractor as described previously. 6 The combined extract was concentrated to dryness under reduced pressure, yielding a residue of PM-R-ET (123.30 g, brown solid). PM-R-ET was flushed with nitrogen gas and stored at −20 °C for future use.
Isolation and Purification
PM-R-ET was reconstituted in ethanol 96% and sequentially partitioned with n-hexane, ethyl acetate (EtOAc) to give the n-hexane portion H0 (23.1 g), EtOAc portion EA0 (39.1 g) and ethanolic portion E0 (42.6 g). The EtOAc-soluble EA0 (39.1 g) portion was further fractionated on silica gel chromatography column (CC 63-200 μm) eluting with gradient elution of increasing polarity solvent mixture of n-hexane–EtOAc (ie, 100:1→ 90:10 → 80:20 → 60:40 → 40:60 → 20:80 → 0:100, 2 L each) to obtain 7 fractions (EA1 → EA7). The EA1 fraction was separated on CC (40-63 μm) and then eluted by n-hexane–EtOAc (20: 1, v/v) to give 3 subfractions (EA1.1 → EA1.3). The EA1.2 subfraction was again separated on CC (40-63 μm) and then eluted by n-hexane–EtOAc (15: 1, v/v) for 2 subfractions (EA1.2.1, HĐ-1). A white powder (
Structural Characterization
Nuclear magnetic resonance (NMR) spectra for structural elucidation of isolated compounds were measured on a Bruker Avance-500 spectrometer (Bruker, Karlsruhe, Germany) in DMSO-D6 operating at 500 MHz for 1H NMR, HMBC, and HMQC, 150 MHz for 13C NMR, respectively. Tetramethylsilane (TMS) was used as an internal standard. EI mode MS spectra of isolated compounds were measured on an X500R QTOF mass spectrometer (AB SCIEX, USA).
ATR-FTIR Analysis
ATR-FTIR was one of the technologies used to investigate the chemistry of the P. micrantha rhizomes. Before ATR-FTIR analysis, dry P. micrantha rhizomes were ground to a fine powder using an agate mortar, then sifted using sieves with mesh sizes ranging between no.100 and no. 120, aimed at isolating particles with dimensions falling within the 0.125-0.150 mm range. The collected sample was PM-R-DP. This sieving process minimized the potential impact of radiation scattering attributable to variable particle sizes, thus ensuring analytical consistency. The ATR-FTIR measurements were conducted using an FTIR Spectrometer (Nicolet Apex, Thermo Scientific), covering a wavelength spectrum between 4000 cm−1 and 400 cm−1 as previously described. 20 Each sample was subjected to 128 scans with a spectral resolution of 4 cm−1, and measurements were replicated six times to affirm the reliability of the data. As a first step, the dry ground rhizome powders were analyzed for spectral band positions to identify the signatures of the major functional groups. Assignments of the main bands were carried out by analyzing the acquired spectra and comparing them with the literature.
Cell Culture and Cytotoxicity Assay
MTT was dissolved in PBS (pH 7.4). A549, MCF-7, HepG2, Hela, and HT-29 cells were were kept in DMEM or MEME supplemented with L-glutamine, sodium piruvate, NaHCO3, penicillin/streptomycin, 10% FBS, Trypsin-EDTA (0.05%). They were maintained in 25 cm2 flasks at 37 °C in a humidified incubator with 5% CO2. The cytotoxic effects of PM-R-ET on cancer cells were evaluated by measuring the metabolic activity using the MTT assay.
MTT assay, as previously described by Skehan et al,
21
was performed to assess the cytotoxicity of the extract of P. micrantha. The test extract, PM-ET, was dissolved in DMSO. The stock samples were dissolved in DMSO 100% at a concentration of 20 mg/mL. 10 µL of six serial dilutions of test extract (ie, 0.8, 4.0, 20.0, 100, 200, and 500 µg/mL) were brought in 96-well plates in the culture mediums (without FBS). The cells were grown and regulated to appropriate concentrations, and then 190 µL of the cell medium was brought into the 96-well plates containing 10 µL of test extracts. Ellipticine as a reference was used at different concentrations of 0.08, 0.4, 2.0, and 10.0 μg/mL. DMSO 10% mL was used as the negative control. The blank negative control without cell and sample was arranged and measured in the well plates of the same test serial. Following incubation for 72 h, the medium was removed, 10 μL MTT (5 mg/mL in PBS) was added to each well, and the cells were incubated for an additional 4 h at 37 °C at 5% CO2. After 4 h, the supernatant was removed, and 150 μL DMSO was added to each well. The absorbance at 540 nm was recorded using a BIO-TEK spectrophotometer. The formula used to calculate the inhibition percent (%) was:
IC50 was calculated by TableCurve 2Dv4 software. Experiments were performed in triplicate.
Statistical Analysis
Packages of Excel (Microsoft Co, Redmond, WA, USA) and SPSS (version 19.0 statistical software, SPSS Inc, Chicago, USA) were used for statistical analysis. Results represented means ± sd of replicated determinations. Comparison and separation of the mean values were performed using Tukey's multiple comparison test.
Results
GC–MS Analysis
The essential oil of PM-R-EO was obtained at a yield of 0.01% based on the fresh material weight. Eleven components of the essential oil were found using GC–MS (Figure 1).

GC-MS chromatogram of PM-R-EO sample.
The major constituents present in the PM-R-EO were tentatively identified as a high alkane, 1-docosene (
Chemical Constituents of PM-R-EO Sample.
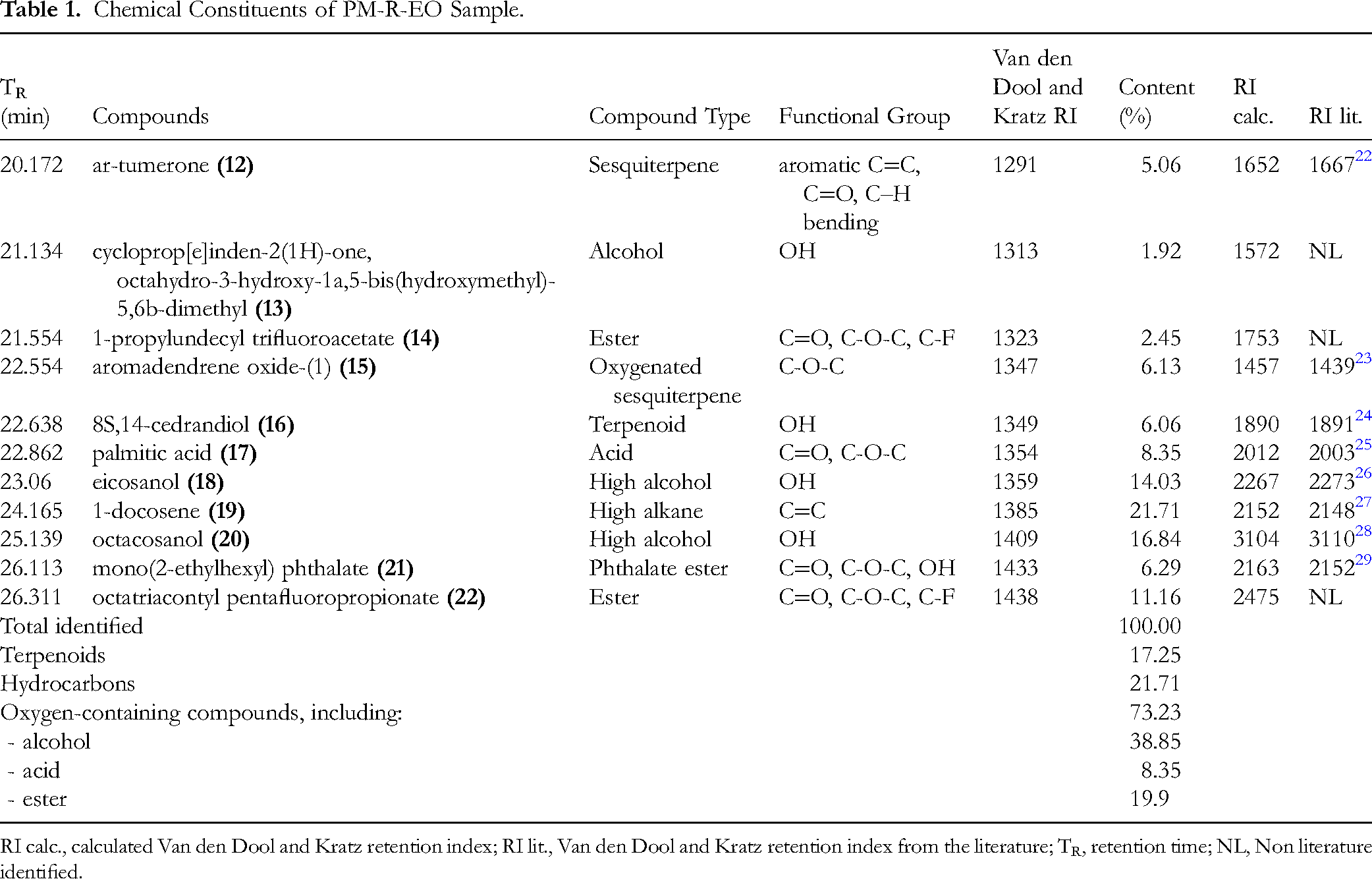
RI calc., calculated Van den Dool and Kratz retention index; RI lit., Van den Dool and Kratz retention index from the literature; TR, retention time; NL, Non literature identified.
Isolation and NMR Characterization
Our isolation protocol for PM-R-ET has given two compounds. Compound (
Compound (
ATR-FTIR Spectroscopy
ATR-FTI spectrum of PM-R-D was shown in Figure 2 and presented 28 vibrations.

ATR-FTIR spectrum of PM-R-D sample.
The vibration assignments are in Table 2. The FTIR absorptions at 603 cm−1, 652 cm−1, and 677 cm−1 in the region below 800 cm−1 were known as the skeletal bands relating to the carbohydrate skeletal and/or the benzene ring stretching vibrations.19,33 An absorption peak at 926 cm−1 in the region between 900-1200 cm−1 defined the coupled C-C and C-O stretching and C-OH bending vibrations. 34 Two peaks at 1314.00 cm−1 and 1027 cm−1 were assigned to C=O vibration and C–O absorptions respectively. 35 The absorption peaks at at around 1013 cm−1 in the region of 1010-1060 cm−1 and 1615 cm−1, corresponding to double bonds, C=O and S=O of polysaccharides.36,37 Two absorption peaks at 1641 cm−1 and 1635 cm−1 were appeared in the 1623-1640 cm−1 region, characteristic of double bonds stretching. Three absorption peaks at 1013 cm−1, 1027 cm−1, and 1030 cm−1 in the region between 1000-1200 cm−1 might indicate the pyranose forms of the polysaccharides.38,39 IR bands at 814 cm−1, 828 cm−1, and 922 cm−1 could relate to the α-anomeric carbon configurations of the polysaccharide.37,39,40 FTIR spectrum presented the peaks at 1029 cm−1, 1248 cm−1, and 1732 cm−1 which were assigned as polysaccharide characteristic bands due to different acetyl stretching vibrations.41,42 A small peak at 1414 cm−1 in the 1200-1500 cm−1 region was assigned to O–H/C–H vibrations. 43
Assignments of ATR-FTIR Functional Groups and Constituents of P. micrantha Rhizomes.

In high wavenumber region of double bonds stretching between 2300-2940 cm−1, the vibrations at 2366 cm−1, 2832 cm−1, and 2936 cm−1 were also assigned to C=C stretching. 44 An absorption peak at 2855 cm−1 was assigned to CH3 cyclic hydrocarbon stretching or to C-H stretching. 45 In high wavenumber region of O-H vibrations, there are small O-H stretching vibrations at 3697 cm−1, 3841 cm−1, and 3437 cm−1.45,46 The FTIR spectrum showed a broad intensity peak at 3332 cm−1, and a relatively weak band at 3743 cm−1, both are the characteristic N-H stretching absorptions of secondary amine and amides, respectively. 33
Peak at round 2918 cm−1 was assigned to C-H and alkyl bonds of the polysaccharides.37,47 A small peak at 3282 cm−1 was assigned to O-H vibration of the polysaccharides. 42
Cytotoxic Activity
The cytotoxic activity of ethanolic extract from P. micrantha rhizomes, PM-ET, was presented in Table 3. The sample inhibition increased while HT29 cell concentrations increased from 0.8 to 500 µg/mL. Its IC50 cytotoxic value against HT29 cells was 96.14 ± 6.12 µg/mL. It did not show significant cytotoxicity against HepG2, A549, Hela, and MCF-7 cells (IC50 > 100 µM).
Cell Cytotoxicity of Alcohol Extract from P. micrantha Rhizomes.

*Values are the mean of three replicates ± standard deviation.
Discussion
GC–MS Analysis of Essential Oil
Among eleven constituents tentatively identified in PM-R-EO, the presence of oxygen-containing compounds was highest (total 73.23%), followed by one hydrocarbon (21.71%), and finally three terpenoids (total 17.25%). Three terpenoids were of the same concentration of around 6.00%. One aromatic sesquiterpene, ar-tumerone (5.06%) (
Some compounds identified in the PM-R-EO were previously extracted. From the extract of one edible mushroom mutant of Schizophyllum commune strains,
52
1-docosene and some triacontyl pentafluoropropionate derivatives such as triacontyl pentafluoropropionate and dotriacontyl pentafluoropropionate could be identified by GC-MS. However, we could not find any documentation in the literature of the natural source of octatriacontyl pentafluoropropionate (22) accounting for up to 11.16% of all identified EO constituents. Eicosanol (
Isolation and NMR Characterization
Only two phytoconstituents, pumilaside A (
ATR-FTIR Spectroscopy of P. micrantha Rhizome Powder
Infrared spectroscopy was used to investigate the chemical composition of the P. micrantha rhizomes powder. At first, the samples were analyzed concerning the spectral band positions to identify the signatures of the major functional groups. Then, the assignments of the main bands were carried out by analyzing the acquired spectra and comparing them with the literature. We first assigned IR vibrations to the metabolites in the rhizomes. ATR-FTIR vibrations showed a small peak at 1641 cm−1 that could be of aliphatic C=C stretching in 1-Docosene, the main component of the EO (21.71%), 35 while one other small peak at 1635 cm−1 that could be of C=C cyclic alkene stretching which might come from β-sitosterol, 45 and ar-tumerone. 48 In the higher wavenumber double bonds region, the vibrations at 2366 cm−1, 2832 cm−1, and 2936 cm−1 were also assigned to C=C stretching of β-sitosterol. 46 The presence of a benzene ring stretching at 652 cm−1 might come from pumilaside A and pumilaside C. 32 The FTIR peaks at 1314 cm−1 and 1027 cm−1 could present C=O vibration and C–O absorption of alcohols, carboxylic acids, esters, and ethers specific functional groups in the rhizomes of P. micrantha, such as 1-propylundecyl trifluoroacetate, palmitic acid, mono(2-ethylhexyl) phthalate, octatriacontyl pentafluoropropionate, and/ or ar-tumerone in the essential oil, respectively. 35 An absorption peak at 2855 cm−1 was assigned to CH3 cyclic hydrocarbon stretching or to C-H stretching of β-sitosterol. 45 β-sitosterol isolated from Momordica charantia and Aristolochia indica showed a small peak at 3437 cm−1 assigned as OH group stretching.45,46
The results of FT-IR spectroscopy could be used to elucidate the presence of the related pyranose molecular structures and polysaccharides. The FTIR absorptions at 603 cm−1 and 677 cm−1 in the region below 800 cm−1 were known as the skeletal bands relating to the carbohydrate skeletal vibrations. A relatively extensive absorption peak at 926 cm−1 defined the coupled C-C and C-O stretching and C-OH bending vibrations of characteristic absorptions of polysaccharide, 33 or glycosidic linkages of different carbohydrate configurations. 34 The absorption peak at 1615 cm−1 was caused by the C=O polysaccharide symmetrical deformation. 36 The FTIR broad intensity peak at around 1013 cm−1, corresponding to asymmetric stretching of S=O to confirm the attachment of sulfonyl groups possibly to sulfated steroidal glycoside J-3, glucopyranoside pumilaside A, and polysaccharides.5,31,37 Such information indicated the presence of polysaccharides in the water extract of P. micrantha rhizomes. As O-linked acetyl groups commonly in plant-derived polysaccharides show O-linked C=O stretching vibration at around 1740 cm−1, and O-linked C–O stretching vibration at around 1245 cm−1, 58 the FTIR spectrum presented a typical acetyl C=O stretching vibration at 1732 cm−1 and a typical acetyl C–O stretching vibration at 1248 cm−1. A peak at 1029 cm−1 was assigned as a polysaccharide-characteristic band due to asymmetric stretching of C-O-C tensile vibrations and C–O–H linkages.41,42 The monosaccharide adopts four different configurations in solution including two types of sugar ring (furan and pyran). Once a glycosidic bond is formed, the configuration of anomeric carbon is fixed and two different anomeric carbon configurations (α or β) are formed in a monosaccharide solution. 38 As pyranose rings were represented by three absorption peaks in the region of 1000-1200 cm−1, three typical vibrations at 1013 cm−1, 1027 cm−1, and 1030 cm−1 indicated the pyranose forms of the polysaccharides in the P. micrantha rhizomes.38,39 In addition, IR bands at 814 cm−1, 827 cm−1, and 922 cm−1 confirmed the existence of α-anomeric carbon configurations of the polysaccharide.37,39,40 Spectra absorbance at 2918 cm−1 was assigned to the stretching vibration of aliphatic C-H absorption and asymmetric stretching vibration of the C–H bond in the alkyl groups of the polysaccharides.37,47 A small peak occurred at 1414 cm−1 which is assigned to O–H/C–H deformation and C–H/C–H2 stretching vibrations in the 1200-1500 cm−1 region. 43 In addition, a small peak at 3282 cm−1, assigned to O-H symmetric and asymmetric stretching vibration of the polysaccharides. 42
Cytotoxic Activity of P. micrantha Rhizome Ethanolic Extract
The cytotoxic activity of alcohol extract from P. micrantha rhizomes was evaluated against A549, Hela, HepG2, HT-29, and MCF-7 cells using MTT assay. At the same extract dilution concentration of 200 µg/mL, the inhibition rates decreased from 78.70, 71.03, 40.32, 31.49, and 30.45 against HT29, Hela, HepG2, A549, and MCF7, respectively. This sequence was also observed for the extract concentration of 100 µg/mL. Surprisingly, the extract did not show the highest cytotoxicity against HT29 cells compared to other cancer cells in other tested concentrations. It showed the highest cytotoxicity against HepG2 cells in the low range between 0.8-20 µg/mL. Meanwhile, it always presented the lowest cytotoxicity against A549 cells in the tested range. The IC50 value of PM-R-ET against HT29 cells was 96.14 ± 6.12 µg/mL. The study established the IC50 against the rested cancer cell lines higher than 100 µg/mL but did not yet determine their values. Ellipticine was used as reference, yielding IC50 values of 0.40 ± 0.03, 0.34 ± 0.03, 0.32 ± 0.02, 0.36 ± 0.03, and 0.39 ± 0.02 µg/mL for A549, Hela, HepG2, HT-29, and MCF-7, respectively. The results showed that the PM-R-ET was much less cytotoxic against all the tested cancer cells compared to the reference. In this work, we have isolated pumilaside A (
Limitations of the Study
The results of this research provided information about the first chemical investigation of the Vietnamese plant P. micrantha rhizome ethanolic extract. Additionally, the study dealt with its ATR-FTIR spectroscopy characteristics and cytotoxic activities. The significance of this study is the first presentation of the chemical compositions of isolated compounds, volatile compounds, and IR assignments of the functional groups of the constituents in its rhizomes, and anticancer activity. However, the chemical, spectroscopic, and cytotoxic investigations were carried out only for the alcoholic extract. Therefore, the main limitations can be considered that: 1) only a few constituents could be isolated, not yet quantified and their roles in the anticancer activity would not be verified, and 2) there might be different phytochemical profiles of P. micrantha rhizome alcoholic, hydroalcoholic, and aqueous extracts which could present different biological activities. It is necessary to expand research using different extraction solvents, and then to further isolate the phytoconstituents from both P. micrantha rhizomes and leaves to reduce limitations and provide a more complete contribution to the chemical literature of Vietnamese natural flora. Further studies are needed for in-depth biological activities of P. micrantha rhizomes.
Conclusion
This research studied the presence of chemical compounds in P. micrantha rhizomes including eleven constituents in the essential oil, and two isolated constituents in the ethanolic extract. ATR-FTIR spectra of P. micrantha rhizome powder presented several functional groups that could come from the presently and previously identified chemical compounds. Its ethanolic extract showed IC50 cytotoxicity of 96.14 ± 6.12 µg/mL against HT29 cells. The findings would encourage further in vitro and in vivo tests to verify the pharmaceutical activities of P. micrantha rhizomes.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261418394 - Supplemental material for Chemico-Physical Characterization and Cytotoxicity of the Peliosanthes micrantha Rhizomes Collected in Highland Province Dak Nong of Vietnam
Supplemental material, sj-docx-1-npx-10.1177_1934578X261418394 for Chemico-Physical Characterization and Cytotoxicity of the Peliosanthes micrantha Rhizomes Collected in Highland Province Dak Nong of Vietnam by Le Ngoc Hung, Do Ngoc Thuy, Nguyen Thi Huong, Pham Thi Nhung, Phung Van Trung and Do Thi Nguyet Que in Natural Product Communications
Footnotes
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Vietnam Academy of Science and Technology [grant number UDNDP.04/2022-2023].
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.