Abstract
Keywords
Introduction
In the wake of the pivotal revelation of penicillin in 1928, researchers initiated a promising endeavor to investigate and formulate antibiotics for addressing illnesses induced by detrimental bacteria. This era, commonly denoted as the “golden age of antibiotics.”1,2 witnessed the effective application of medicinal chemistry principles to synthesize antibiotics with hybridized, semisynthetic attributes, thus enhancing their physicochemical properties.3-5 Unfortunately, indiscriminate antibiotic use, improper prescription practices, their excessive deployment in agriculture, a dearth of novel antibiotic discoveries, and the formidable challenges associated with regulatory approval have collectively led to the emergence and proliferation of antibiotic-resistant bacterial strains.6-8 This proliferation of antibiotic-resistant bacteria stems from improper and excessive antibiotic usage, unsuitable prescription practices, the widespread application of antibiotics in agriculture, a lack of innovative solutions, and regulatory hurdles. In 2017, the World Health Organization (WHO) identified a group of critical disease-causing microbes, collectively known as ESKAPE bacteria, including Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter spp. 1 Urgent research is imperative to discover novel treatments for these bacteria, as they have developed the ability to evade various therapies and transfer their resistance to other organisms.9,10
Furthermore, antibiotic-resistance genes can be transmitted between bacterial strains, facilitating their dissemination. Simultaneously, the development of new antibiotics has become more challenging and costly, limiting treatment options in the face of escalating antibiotic resistance.11,12 Consequently, bacterial infections are becoming increasingly challenging to treat, leading to treatment failures, severe complications, and, in some cases, fatalities. Medical procedures are also growing riskier due to the heightened threat of antibiotic-resistant infections.
The exploration of essential oils as a substitute for conventional antibiotics has emerged as a prominent topic in scientific literature. Essential oils, extracted from aromatic plant species, have garnered attention for their potential in combatting bacterial infections, particularly those caused by bacteria resistant to traditional antibiotics.13-19 These essential oils encompass a variety of bioactive compounds with established antimicrobial properties. Their composition varies across plant species, offering a diverse array of potential therapeutic options. Researchers have been investigating the effectiveness of essential oils in inhibiting the growth of pathogenic bacteria, with some studies reporting promising outcomes.14,16,20 One of the key advantages of essential oils is their versatility, as they can be obtained from numerous plant sources, each characterized by a unique chemical composition. This versatility enables the targeting of a wide spectrum of bacterial strains. 21 Such diversity is particularly advantageous in the context of antibiotic resistance, where a single drug may prove ineffective against multiple resistant strains. This attribute is especially crucial in clinical and medical applications, as it minimizes the risk of adverse reactions in patients.
Combination therapy, involving the concurrent use of multiple antibacterial agents, whether of synthetic or natural origin, including essential oils, has emerged as a potent and prudent strategy for combating antibiotic-resistant bacterial pathogens. 22 This therapeutic approach offers multifarious advantages, encompassing the circumvention of antibiotic resistance mechanisms, enhanced bactericidal efficacy through synergistic or additive interactions, reduced dosage requirements, and an expanded range of targeted microbial species.22,23 However, it is imperative to recognize the diverse potential interactions inherent in these combination regimens, encompassing synergistic, additive, partial, and antagonistic effects. This underscores the necessity for an insightful understanding of such interplay.22-26 Expanding our comprehension of these complex interactions will contribute to the development of more effective treatment strategies against antibiotic-resistant bacteria, addressing a critical healthcare challenge of our time.
Ptychotis verticillata Duby., belonging to the Apiaceae family, is an annual plant that thrives at a height of 15 to 35 cm, adorned with a glaucous hue and featuring a slender, pivoting root. 27 Its stem, erect and striated, displays a delicate structure with numerous spreading branches. The umbels, small and drooping before flowering, consist of 6 to 12 capillary rays, markedly uneven, with very short inner ones. This plant is an endemic plant to Morocco, and is locally referred to as Nûnkha and under various other names, is a medicinal and fragrant plant found extensively in North African regions. It is widely employed in traditional medicine for its roles as an antispasmodic, antidiabetic, and antipyretic agent.28,29 Additionally, it is valued for its antiseptic properties, along with its notable antifungal, antibacterial, and anti-cancer attributes.19,30
The principal objective of this investigative endeavor pertains to a comprehensive evaluation of both the phytochemical composition of Ptychotis verticillata essential oil (PVEO) and its attendant antibacterial activity against a diversified array of bacterial strains. These strains encompass a tandem ensemble of Gram-negative organisms, notably Escherichia coli, P aeruginosa, and K pneumoniae, as well as three Gram-positive counterparts: S aureus, Micrococcus luteus, and Bacillus subtilis. In tandem with these evaluations, we are meticulously scrutinizing potential interactions between PVEO and three widely employed antibiotics, specifically Amoxicillin, Ampicillin, and Erythromycin. This methodological approach, ensconced in the overarching imperative of combating antibiotic resistance, aspires to proffer novel antimicrobial modalities, while simultaneously shedding light upon the mechanisms by which PVEO may potentiate or act synergistically alongside these antibiotic agents.
Results and Discussion
Chemical Composition
The chemical composition of PVEO was determined using gas chromatography-mass spectrometry (GC-MS) technique. This analysis revealed that PVEO consists mainly of 21 volatile compounds (Table 1; Figure 1, 2; MS spectra were given in Supplemental file). GC-MS analysis identified the presence of bioactive volatile compounds in PVEO, which are responsible for its antioxidant, anticancer, antifungal, and anti-inflammatory activities.31,32 The major compounds in this essential oil include γ-terpinene, which accounts for 38.96%, β-cymene with 19.38%, thymol with 18.07%, and carvacrol with 6.99%. Several studies have highlighted the biological properties of these major compounds, either individually or in synergy, particularly the synergy of thymol with carvacrol, as demonstrated in scientific works carried out by Bouhtit et al, 33 Campos-Requena et al, 34 and Masoumi et al. 35
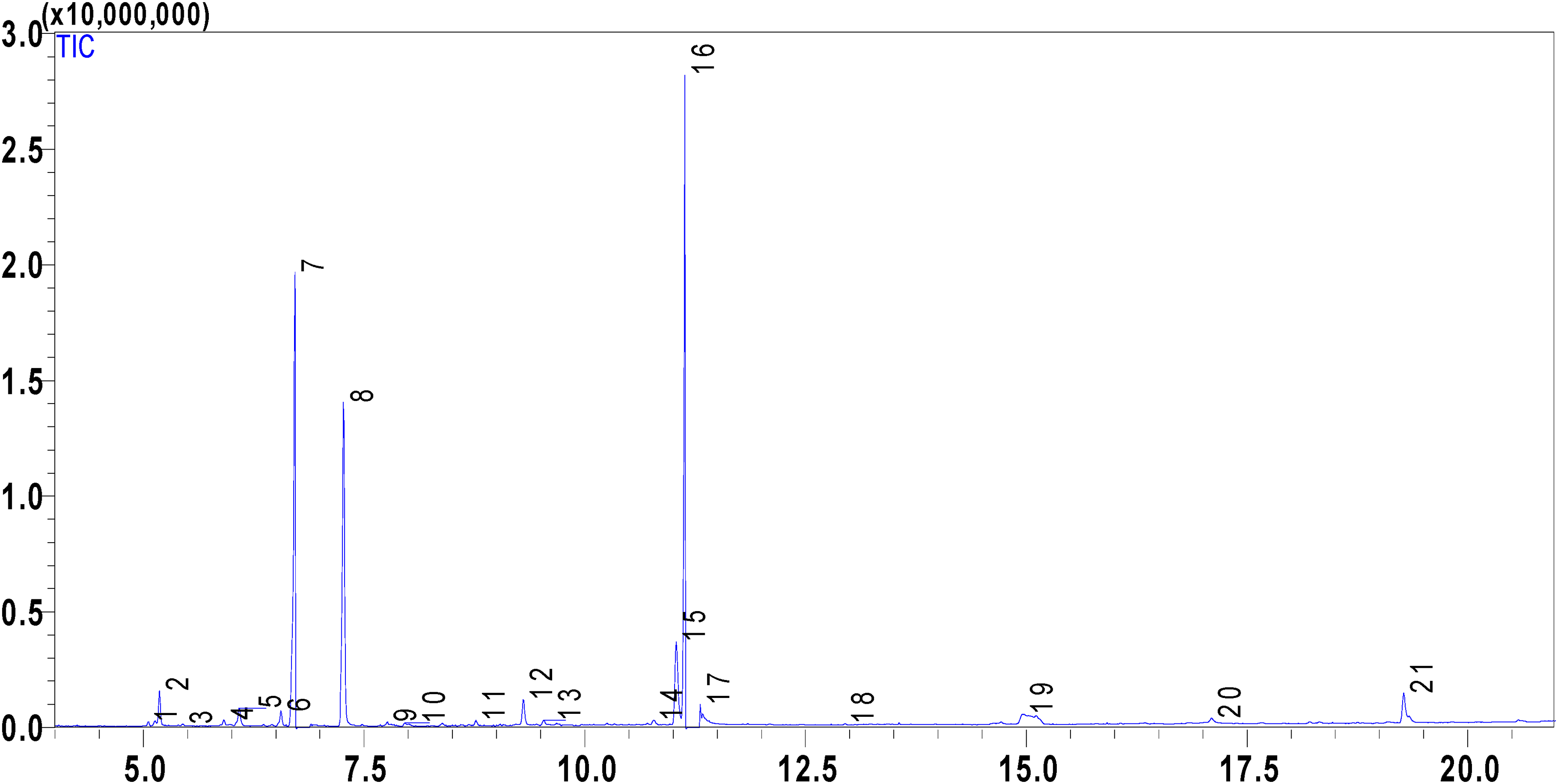
Chromatogram of the chemical composition of PVEO using GC-MS. Abbreviations: PVEO, Ptychotis verticillata essential oil; GC-MS, gas chromatography-mass spectrometry
Chemical structures of PVEO's volatile compounds using GC-MS. CID were retrieved from PubChem NCBI (https://pubchem.ncbi.nlm.nih.gov/). Abbreviations: PVEO, Ptychotis verticillata essential oil; GC-MS, gas chromatography-mass spectrometry; CID, compound identifiers.
Determination of Phytochemical Composition of PVEO, Using GC-MS.
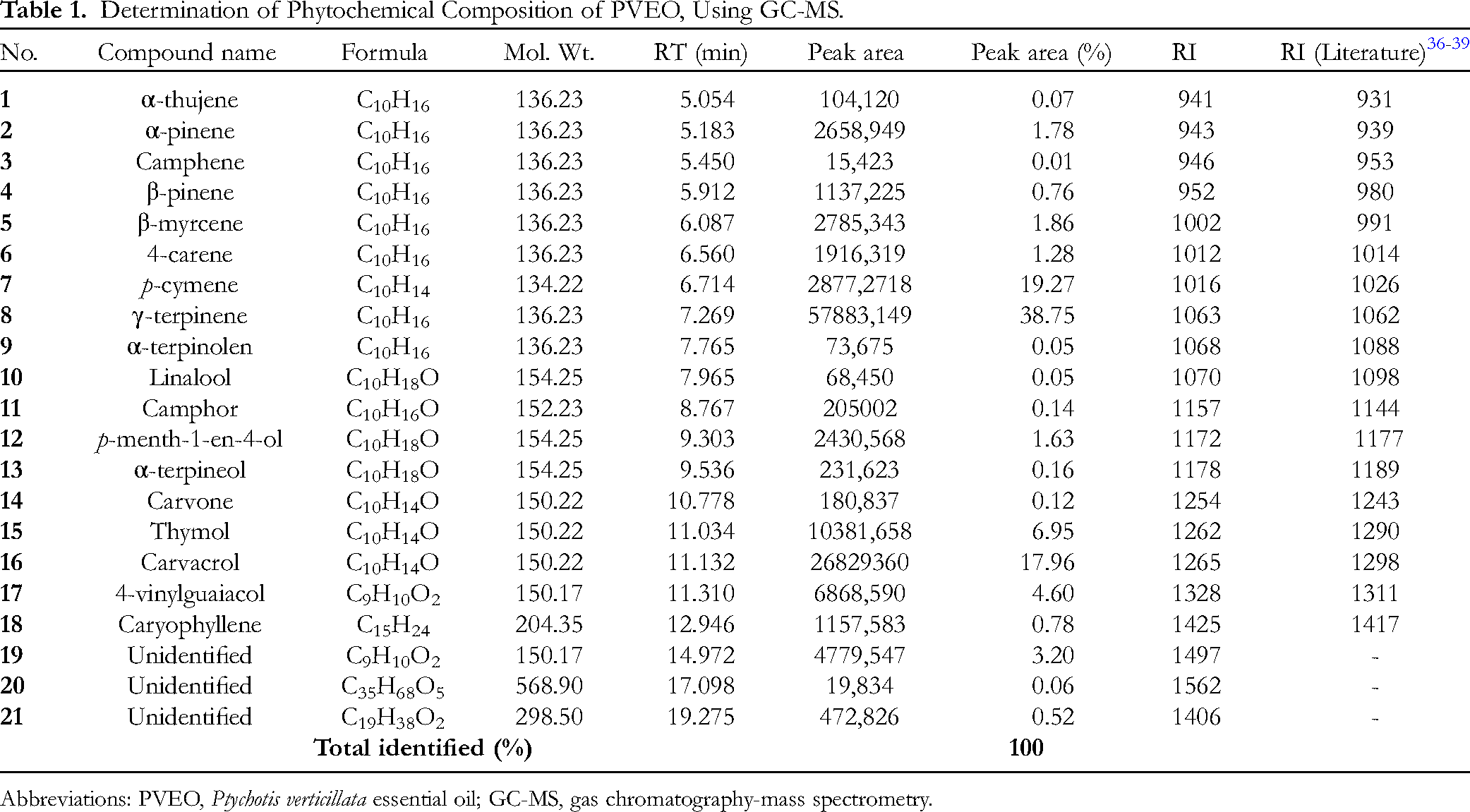
Abbreviations: PVEO, Ptychotis verticillata essential oil; GC-MS, gas chromatography-mass spectrometry.
It is noteworthy that there were additional compounds present in lower quantities in the essential oil, including α-thujene, α-pinene, camphene, β-pinene, β-myrcene, (+)-4-carene, α-terpinolene, Linalool, camphor, p-menth-1-en-4-ol, α-terpineol, phenol, 4-ethenyl-2-methoxy-, caryophyllene, and carvone. These compounds may contribute to the biological and pharmaceutical activities of the essential oil through potential synergistic interactions. It's noteworthy that these results differ from those reported by Taibi et al 19 for the essential oil of the same plant originating from the same region. These discrepancies can be attributed to various factors, including environmental conditions, plant age, vegetative growth stage, genetic factors, and variations in abiotic factors such as temperature, altitude, humidity, and air quality in different harvesting locations.40,41 As a result, significant variations are observed in both the quantity and proportion of bioactive compounds. For instance, in the essential oil from the Touissit region, only 12 constituents are identified, with thymol (37.05%), D-limonene (22.97%), γ-terpinene (15.97%), m-cymene (12.14%), and carvacrol (8.49%) predominating. 30 It is crucial to underscore that the biological activities linked to essential oils predominantly result from complex and synergistic interactions among numerous chemical constituents present within the oil, rather than depending on the actions of isolated individual compounds.42,43
Antibacterial Activity (Minimum Inhibitory Concentration)
PVEO exhibited substantial antibacterial efficacy against all tested bacterial strains, with minimum inhibitory concentration (MIC) values ranging from 200 µg/mL for E coli to 2000 µg/mL for M luteus. Ampicillin displayed a range of MIC values, from 0.4 µg/mL against B subtilis to 1 µg/mL against S aureus, and K pneumoniae. Similarly, Amoxicillin demonstrated MIC values, spanning from 0.75 µg/mL for M luteus to 1.5 µg/mL for S aureus, and K pneumoniae. On the other hand, erythromycin's MIC values ranged from 0.4 µg/mL for M luteus to 1.25 µg/mL for B subtilis, and P aeruginosa (as per Table 2).
The Minimum Inhibitory Concentration of PVEO and Three Conventional Antibiotics (Ampicillin, Amoxicillin, and Erythromycin) Against Different Bacterial Strains.

Abbreviations: PVEO, Ptychotis verticillata essential oil; MIC, minimum inhibitory concentration.
The chemical composition of PVEO is primarily characterized by the presence of γ-terpinene, β-cymene, thymol, and carvacrol. Studies have elucidated the interactive nature of thymol and carvacrol, contributing significantly to their remarkable antibacterial potency. Lambert et al. 44 demonstrated that while carvacrol and thymol exhibited some individual antibacterial activity, they synergistically enhanced their efficacy when used in combination. 44 This suggests that the concurrent application of these two compounds yielded more substantial inhibition of bacterial growth than when used individually. 44 Guarda et al 45 also observed this synergistic antibacterial action between these two bioactive molecules. Many studies have demonstrated that carvacrol and thymol are potent antibacterial agents against both Gram-positive and Gram-negative bacteria. The most frequently reported mechanism of antibacterial action of both isomers involves the disruption of bacterial membrane leading to bacterial lysis and leakage of intracellular contents resulting in death. 46 Additionally, the notable presence of p-cymene in significant quantities may contribute to their remarkable antibacterial effectiveness, as p-cymene has been recognized for its diverse antimicrobial properties in previous studies.47,48 These results confirm those obtained by Taibi et al, 19 who also demonstrated that PVEO from the eastern region of Morocco (Touissit), has significant antibacterial activity against a wide range of bacteria. The efficacy of this essential oil was evaluated against Listeria innocua, E coli, S aureus, and P aeruginosa. MIC values obtained in this study were in the order of 500 µg/mL against L innocua and P aeruginosa, 250 µg/mL against E coli and S aureus respectively. 19
Synergistic Studies of PVEO with Synthetic Antibiotics
Utilizing a combinatorial therapy approach holds great potential in addressing the formidable challenge posed by antibiotic resistance. This innovative method involves the strategic integration of synthetic antibiotics with natural substances, such as essential oils, as part of the therapeutic arsenal.49,50 These novel combinations of antimicrobial agents can yield a spectrum of interactions, encompassing synergy, addition, antagonism, or even an absence of a discernible effect. Of particular significance are the synergistic interactions that manifest in these combinatorial regimens, where the therapeutic whole becomes greater than the sum of its individual parts. These synergistic interactions offer a multifaceted approach to combating bacterial infections, exhibiting enhanced efficacy and reduced toxicity, owing to their concerted action on multiple fronts. 51
The potential of these synergistic combinations is twofold: Not only do they provide a promising solution to the growing menace of antibiotic resistance, but they also present an effective strategy against bacterial strains that have developed resistance to multiple drugs. By targeting bacteria through multiple mechanisms simultaneously, these combinations can deter the emergence of antibiotic resistance, making it considerably more challenging for bacteria to evolve mechanisms of survival in the face of such multifaceted attacks. 52
The study findings, as elucidated in the subsequent tables (Tables 3, 4, and 5), offer valuable insights into the impact of combining PVEO with the antibiotics (ampicillin, amoxicillin, and erythromycin) on the MIC for various bacterial strains. The MIC values, quantified in µg/mL, represent the minimum concentrations required to inhibit the growth of these bacterial strains. These results illuminate the potential of combining PVEO with conventional antibiotics to achieve lower MIC values, indicating the enhanced inhibitory effect of these combinatorial therapies on bacterial growth. Such information not only expands our understanding of the utility of these synergistic combinations but also underscores the need for further exploration and optimization of such approaches in the ongoing battle against antibiotic-resistant bacteria.
Results of the Combinations Between PVEO, and Ampicillin

MICa: MIC when the individual component is tested independently; MICc: MIC of each component within the combination that achieves the most significant growth inhibition; FIC: calculated as the ratio MICc/MICa; FICI: FIC of Ampicillin + FIC of PVEO. Abbreviations: PVEO, Ptychotis verticillata essential oil; FICI, Fractional Inhibitory Concentration Index; FIC, fractional inhibitory concentration; MIC, minimum inhibitory concentration.
Results of the Combinations Between PVEO, and Amoxicillin.

MICa: MIC when the individual component is tested independently; MICc: MIC of each component within the combination that achieves the most significant growth inhibition; FIC: calculated as the ratio MICc/MICa; FICI: FIC of Amoxicillin + FIC of PVEO. Abbreviations: PVEO, Ptychotis verticillata essential oil; FICI, Fractional Inhibitory Concentration Index; FIC, fractional inhibitory concentration; MIC, minimum inhibitory concentration.
Results of the Combinations Between PVEO, and Erythromycin.

Abbreviations: PVEO, Ptychotis verticillata essential oil; FICI, Fractional Inhibitory Concentration Index; FIC, fractional inhibitory concentration; MIC, minimum inhibitory concentration.
Synergistic Potential of PVEO with Ampicillin
The results of the combined action of PVEO with ampicillin are outlined in Table 3. Ampicillin, a member of the penicillin class of antibiotics, is frequently utilized for the treatment of diverse bacterial infections. It operates by disrupting the synthesis of the bacterial cell wall, gradually weakening and eventually leading to the breakdown of the bacterial cell. 53 Ampicillin exhibits efficacy against a broad spectrum of bacterial infections, including respiratory tract infections, urinary tract infections, skin and soft tissue infections, as well as specific gastrointestinal infections.53,54 The results were interpreted as follows: A Fractional Inhibitory Concentration Index (FICI) value of ≤ 0.5 indicated synergy, 0.5 < FICI ≤ 0.75 suggested partial synergy, 0.75 < FICI ≤ 2 indicated no effect, and FICI > 2 indicated antagonism. 55 Synergy was evident against E coli with a FICI of 0.45. Furthermore, partial synergy was observed against S aureus with an FICI of 0.60, B subtilis with an FICI of 0.66, K pneumoniae with an FICI of 0.68, and M luteus with an FICI of 0.55. Notably, this combination exhibited no discernible effect against P aeruginosa. Overall, there were reductions in the MIC values of PVEO and ampicillin in synergistic and partially synergistic interactions, ranging from two to fourfold, respectively.
Synergistic Potential of PVEO with Amoxicillin
The results of the combined action of PVEO and amoxicillin are outlined in Table 4. Amoxicillin, a widely prescribed antibiotic, belongs to the penicillin family of antibiotics and is employed to treat a diverse range of bacterial infections. Amoxicillin operates by inhibiting bacterial growth and is effective against various types of infections, including respiratory tract infections, ear infections, urinary tract infections, skin infections, and certain dental infections. 56 Synergy was evident against M luteus with a FICI of 0.43. Furthermore, partial synergy was observed against S aureus with an FICI of 0.63, B subtilis with an FICI of 0.56, and K pneumoniae with an FICI of 0.55. Notably, this combination exhibited no discernible effect against E coli, and P aeruginosa. Overall, there were reductions in the MIC values of PVEO and amoxicillin in synergistic and partially synergistic interactions, ranging from two- to seven-fold, respectively.
Synergistic Potential of PVEO with Erythromycin
Erythromycin is an antibiotic medication belonging to the macrolide class of drugs.57,58 It is used for a variety of bacterial infections. 58 The mechanism of action of erythromycin involves inhibiting bacterial growth and interfering with their capacity to synthesize essential proteins, ultimately resulting in the suppression of bacterial proliferation. 59 Erythromycin exhibits efficacy against a broad spectrum of bacteria, encompassing both gram-positive and gram-negative species.60,61 It is frequently prescribed for the management of respiratory tract infections, skin and soft tissue infections, ear and sinus infections, and certain sexually transmitted diseases such as syphilis and chlamydia. 62 It is also used as a preventive treatment for rheumatic fever in individuals who are allergic to penicillin. 63
For S aureus, the MIC of PVEO alone was 500 µg/mL, while erythromycin had a MIC value of 0.75 µg/mL. However, when combined, the MIC values decreased significantly to 75 µg/mL for PVEO and 0.2 µg/mL for erythromycin, resulting in an fractional inhibitory concentration (FIC) of 0.27 and an FICI of 0.42, indicating a synergy interaction. E coli showed a similar trend with a reduction in MIC values when PVEO and erythromycin are combined. The MIC of PVEO dropped from 200 to 50 µg/mL, and it decreased for erythromycin from 1 to 0.2 µg/mL, leading to respective FIC values of 0.25 and 0.20, again indicating synergy. B subtilis displayed a substantial reduction in MIC values as well. The MIC for PVEO decreased from 700 to 150 µg/mL, and erythromycin's MIC decreased from 1.25 to 0.1 µg/mL. The resulting FIC was 0.08, and the FICI was 0.29, demonstrating a synergy interaction. In contrast, P aeruginosa did not exhibit a significant change in MIC values when PVEO and erythromycin were combined, indicating no combinatorial effect. K pneumoniae displayed a partial synergy interaction, with reduced MIC values for both components and a FICI of 0.52. For M luteus, a synergy interaction between PVEO and erythromycin was found, with a corresponding FICI of 0.35. These findings shed light on the efficacy of the combinatorial therapy. Overall, three- to ten-fold reductions were observed in the MIC of PVEO and erythromycin, for S aureus, and M luteus, in synergistic and partially synergistic interactions. These findings underscore the potential of combining PVEO with erythromycin to enhance the antibiotic's effectiveness against specific bacterial strains, providing valuable insights into the fight against antibiotic resistance.
MICa: MIC when the individual component is tested independently; MICc: MIC of each component within the combination that achieves the most significant growth inhibition; FIC: calculated as the ratio MICc/MICa; FICI: FIC of Erythromycin + FIC of PVEO.
These findings suggest that PVEO has the potential to augment the efficacy of the three antibiotics tested, suggesting that PVEO could be used to enhance therapeutic efficacy in treating infections with ampicillin, amoxicillin, and erythromycin. Secondary metabolites present in essential oils exert various mechanisms of action against bacteria. Some metabolites, such as phenols, terpenes, and ketones, inhibit bacterial cell growth by disrupting essential metabolic processes.64,65 In contrast, others inflict damage upon the bacterial cell membrane, resulting in the leakage of cellular constituents and eventual cell death. Some interfere with bacterial DNA and RNA synthesis, while others inhibit enzymes crucial to bacterial survival. Some metabolites induce oxidative stress by producing free radicals, while others disrupt cellular energy production. Essential oils often combine these metabolites, enhancing their efficacy by targeting various aspects of bacterial metabolism. However, the specific mechanisms of action may vary according to the chemical composition of the oil and the sensitivity of the bacteria targeted. Antibiotics also exert various mechanisms of action against bacteria. The main mechanism of action of ampicillin and amoxicillin is to inhibit bacterial cell wall synthesis, thereby weakening the bacteria and rendering them vulnerable. On the other hand, Erythromycin acts by inhibiting bacterial protein synthesis by binding to ribosomes, thus disrupting the formation of new proteins essential to bacterial growth and survival. 66
The synergistic interactions observed between PVEO and antibiotics in this study could result from a common action between the different mechanisms of action of antibiotics and bioactive molecules present in the essential oil. This joint action could result in an improvement in cell permeability, which in turn would promote increased uptake of antibiotics by bacterial cells. This effect is likely linked to the mechanism by which essential oil components interact with cellular membrane channels, unlocking these channels and facilitating the transit of antimicrobial agents to their intracellular targets. The synergy arising from the combined activity of secondary metabolites present in essential oils and antibiotics holds promise as a strategy to augment the effectiveness of antibacterial treatments. This merits further investigation for potential clinical applications. 67
These results confirm previous studies that have already reported several essential oils extracted from different medicinal plants.51,68-72 Further research is essential to deepen understanding of this essential oil's mechanism of action and identify the most effective combinations of essential oils and antibiotics in a medical context. In-depth studies are crucial to elucidate the specific interactions between essential oil components and the antibiotics studied and to determine how these interactions influence the bacterial response.
Materials and Methods
Plant Material
In the spring of 2022, samples of Ptychotis verticillata Duby. were procured from a local market in the eastern region of Morocco. The plant material was grown in its native environment within Ahfir city, located in the Eastern region of Morocco, and harvested during the spring of 2022. Subsequently, the plant material underwent a drying process at ambient room temperature, in shade. The samples underwent meticulous taxonomic identification procedures, culminating in the deposition of a designated reference specimen labeled as HUMPOM17* at the Faculty of Sciences, University Mohammed Premier, Oujda, Morocco.
Essential Oil Extraction
PVEO was obtained from the aerial sections of the plant using the hydrodistillation method, and a modified distillation equipment was employed for this extraction process. A quantity of 100 grams of the dried aerial parts of the plant was used each time for the extraction of the essential oil, using a modified Clevenger apparatus (the modifications consist of a Graham condenser to improve the cooling system), as previously documented in studies conducted by Sadgrove et al. 73 The yield of the extraction was noted as 4.6 ± 0.3%.
Chemical Composition
The qualitative and semiquantitative analysis of PVEO was conducted using a gas chromatograph with a mass spectrometer detector, as previously described in the work of Kandsi et al. 74 A Shimadzu GC system from Kyoto, Japan, was utilized in combination with an MS QP2010 (Shimadzu Scientific Instruments) to identify and separate compounds. Separation was achieved using a BPX5 capillary column with 95% dimethylpolysiloxane + 5% phenyl. Pure helium (99.99%) was used as the carrier gas at a constant flow rate of 1.69 mL/min, and the injection, ion source, and interface temperatures were fixed at 250 °C. The column furnace was programed to increase from 50 °C (for 1 min) to 250 °C at a rate of 10 °C/min, maintaining the temperature for 1 min. Sample components were ionized in EI mode at 70 eV, and the mass range studied was 40 to 300 m/z. Subsequently, 1 μL of each prepared oil was injected in fractionation mode (fractionation ratio 90:1). 38 Three evaluations were conducted per sample, and compounds were identified by comparing the retention times to verified standards and mass spectrum fragmentation models found in databases or on NIST compounds. Laboratory Solutions (v2.5) was used to collect and process data. 38 MS spectra are shown in the supplementary file.
Determination of MIC
The assessment of MIC for both PVEO and antibiotics employed a modified broth dilution method as previously described in references.18,19,75 Initially, a freshly cultured microbial inoculum, of the studied bacterial strains of (E coli, P aeruginosa, K pneumoniae, S aureus, M luteus, and B subtilis), was prepared using Muller Hinton broth (MHB) and standardized to conform to the 0.5 McFarland standard. Subsequently, the essential oil was dissolved in dimethyl sulfoxide to create a stock solution at a 1:1 volume ratio, with a final volume adjusted to 1 ml. Similarly, PVEO was diluted in MHB in a series of dilutions ranging from 3000 to 25 µg/ml, while antibiotics were subjected to dilution procedures with a series ranging from 5 to 0.0025 µg/ml. Subsequently, 100 µL of the appropriately diluted essential oil/antibiotic solution was introduced into sterile tubes, each containing 90 μL of MHB and 10 μL of a microbial suspension with a concentration of 106 CFU/mL, resulting in a final volume of 200 μL. For the assessment of bacterial growth, both positive controls (comprising MHB and microbial suspension) and negative controls (devoid of inoculum) were integrated into the experimental setup. These sterilized tubes were then incubated at 37 °C for a duration of 24 h. Following incubation, 40 µL of a p-iodonitrotetrazolium violet solution was introduced to gauge bacterial growth over an additional 20 min. The appearance of a pink coloration indicated bacterial growth, whereas tubes that did not exhibit a change in coloration were regarded as having reached the MIC for the particular combination of essential oil and antibiotic under examination. 17 The experiments were carried out in a triplicate.
Synergistic Studies of PVEO with Synthetic Antibiotics
The assessment of synergistic interactions between PVEO and synthetic antibiotics (amoxicillin, ampicillin, and erythromycin) was conducted employing the broth dilution method. Within sterile tubes, a mixture was prepared, comprising 50 µL of each antibiotic dilution, 90 µL of Muller-Hinton Broth, and 10 µL of microbial suspension. Subsequently, this mixture was incubated at 37 °C for a duration of 24 h. Upon completion of the incubation period, an additional 40 µL of a 0.4 mg/mL p-iodonitrotetrazolium violet solution was introduced to monitor bacterial growth over an additional 20 min. The presence of a pink coloration signified bacterial growth. To assess the interaction between the two agents and calculate the FICI, the FIC index was employed. The FICI was determined using the following formula: FICI = (Agent I MIC + Agent II MIC), where Agent I MIC represents the MIC of Agent I when used in combination with Agent II divided by the MIC of Agent I when used alone, and Agent II MIC represents the MIC of Agent II when used in combination with Agent I divided by the MIC of Agent II when used alone. In this context, Agent I and Agent II denote distinct components, namely PVEO and antibiotics. The interpretation of results was as follows: FICI ≤ 0.5 indicated synergy, 0.5 < FICI ≤ 0.75 suggested partial synergy, 0.75 < FICI ≤ 2 indicated no effect, and FICI > 2 indicated antagonism. 55 The experiments were carried out in a triplicate.
Conclusion, Limitations, and Future Considerations
In the current study, the evaluation of PVEO revealed significant antimicrobial activity, demonstrating heightened efficacy against some Gram-positive and Gram-negative bacterial strains. When utilized in conjunction with conventional antibiotics (Ampicillin, Amoxicillin, and Erythromycin), PVEO demonstrated synergistic interactions with specific bacterial strains, as indicated by FIC values falling within the range of 0.40 to 0.50. This synergy resulted in a noteworthy reduction of MIC values, ranging from two- to ten-fold, when compared with the individual MICs of all tested antibiotics. The combined use of essential oils and antibiotics not only reduced the minimum effective dosage required for bacterial inhibition but also mitigated the potential for the emergence of antibiotic-resistant strains. Furthermore, the incorporation of essential oils as adjuncts exhibited the potential to ameliorate adverse effects and curtail treatment costs. These findings underscore the potential for the development of a novel class of antimicrobial agents, leveraging the collective impact of diverse antimicrobial compounds. This approach holds great promise in addressing the growing challenge of antimicrobial resistance. It is crucial to acknowledge certain limitations within the current study. Notably, the observed synergistic effects of PVEO with antibiotics were found to be strain-specific, and the investigation focused on a selected subset of Gram-positive and Gram-negative bacterial strains. The broader applicability of these findings to a diverse range of bacterial species remains an area for future exploration. Given the intricate chemical composition of essential oils, comprising numerous compounds contributing to their overall activity, the study identified major bioactive components, but the potential synergistic effects of minor components were not extensively examined. The clinical applicability of these findings also necessitates further scrutiny, considering factors such as bioavailability, toxicity, and pharmacokinetics in living organisms. Moreover, while the study demonstrated the efficacy of combined essential oils and antibiotics in vitro, the translation of these findings to clinical settings warrants additional investigation. Future studies should consider aspects like bioavailability, toxicity, and pharmacokinetics in living organisms. Additionally, the research indicates the need for further exploration into the mechanisms underlying these synergistic interactions at the molecular and cellular levels. In conclusion, while this study provides valuable insights, there is a clear call for more comprehensive investigations to fully understand the potential and limitations of combining essential oils with antibiotics in combating microbial infections.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231222785 - Supplemental material for Chemical Profiling, Antibacterial Efficacy, and Synergistic Actions of Ptychotis verticillata Duby Essential Oil in Combination with Conventional Antibiotics
Supplemental material, sj-docx-1-npx-10.1177_1934578X231222785 for Chemical Profiling, Antibacterial Efficacy, and Synergistic Actions of Ptychotis verticillata Duby Essential Oil in Combination with Conventional Antibiotics by Mohamed Taibi, Amine Elbouzidi, Mounir Haddou, El Hassania Loukili, Reda Bellaouchi, Abdeslam Asehraou, Youssef Douzi, Mohamed Addi, Ahmad M Salamatullah, Hiba-Allah Nafidi, Mohammed Bourhia, Musaab Dauelbait, Bouchra El Guerrouj and Khalid Chaabane in Natural Product Communications
Footnotes
Acknowledgment
The authors are grateful to the Ministry of Higher Education, Scientific Research and Innovation of Morocco for supporting this work under the PRIMA project “OLI4FOOD”. The authors also would like to extend their sincere appreciation to the Researchers Supporting Project, King Saud University, Riyadh, Saudi Arabia for supporting this work through the project number (RSP-2024R437).
Author Contributions
Conceptualization was done by MT, AE, EHL, and MA Data curation was done by MT and AE. Formal analysis was carried out by MT, AE, MH, EHL, and MA. Funding acquisition was done by AMS, HAN, and MB. Investigation was done by MT and AE. Methodology was handled by MT, AE, MH, RB, and CK. Project administration was done by AA, BE, and CK. Resources were handled by AMS, HAN, MB, and BE. Software was handled by MT, AE, EHL, and YD. Supervision was done by AA, BE, and CK. Validation was done by RB, AA, YD, MA, MB, BE, and CK. Visualization was done by MA. Writing—original draft was done by MT, AE, and MA. Writing—review & editing was done by M.T., A.E., AMS., H.A.N., M.B., M.D., and C.K.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is financially supported by the Ministry of Higher Education, Scientific Research and Innovation of Morocco for supporting this work under the PRIMA project “OLI4FOOD“. This work is also financially supported by the Researchers Supporting Project number (RSP-2024R437), King Saud University, Riyadh, Saudi Arabia.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
