Abstract
Natural products are an inexhaustive reservoir of bioactive compounds with diverse effects protecting human health as components of nutrition and as active substances of drugs. The objective of the present study was to evaluate the bactericidal and carbohydrate digestive enzyme inhibitory activity of twelve native plants from Cyprus, that are consumed for medicinal and/ or culinary purposes. Each plant was sequentially extracted with solvents of increasing polarity, from non-polar to polar solvents. At first, the total phenolic (TPC) and flavonoid (TFC) content of the extracts were determined. The bactericidal potential of plant extracts was tested against six bacteria using the broth microdilution method. Furthermore, their inhibitory effects on digestive enzymes, namely α-glucosidase and α-amylase, were also determined. Results demonstrated a substantial diversity in TPC (2.3-483.5 mg gallic acid equivalent g−1) and TFC (4.1-394.6 mg catechin equivalent g−1) of the plant extracts; a great impact of solvent was found. Furthermore, potent antibacterial activity (minimum bactericidal concentration ≤ 500 µg ml−1) of capper, mountain oregano, rosemary, silver thistle, and vine leaf extracts against Gram-positive bacteria was determined. Regarding carbohydrate digestive enzyme inhibitory effects, the inhibition of α-glucosidase enzyme is higher than 80%, when rosemary, silver thistle, and vine leaf extracts are used at a concentration of 500 µg g−1. Overall, the present study describes the antibacterial and inhibitory effect against carbohydrate digestive enzymes of unstudied plant species or known plants from the unexplored island of Cyprus. It provides valuable data for the nutraceutical value of native edible plants as well as assesses these plants as potential sources of antibacterial and carbohydrate digestive enzyme inhibitory agents for drug discovery.
Keywords
Introduction
Natural products extracted from plants, either as pure compounds or fractions, provide countless health effects owing to their chemical diversity. Plant extracts have shown considerable potential in a range of applications in the food industry, pharmaceutics, and cosmetics. 1 Among biological effects, the antimicrobial and antidiabetic potency of natural products have attracted scientific interest due to their significance for the pharmaceutical and food industry.2,3
The discovery of new antibacterial compounds is of great importance because of the continuing global concerns about antibiotic resistance. There is a need to develop new antimicrobials and alternatives that could reduce the development and spread of antibiotic resistance among bacterial pathogens. 4 Thus, numerous pure phytochemicals and/or fractions have been assessed and recommended as antimicrobial agents for diverse purposes. 5 It is noteworthy that several patents have been registered for the isolation and commercialization of natural products as antimicrobial compounds. 6 Phenolic compounds, alkaloids, saponins, terpenoids, iridoids, and glucosinolates are some of the phytochemical groups that are linked with antibacterial activity.
Diabetes mellitus is the most common endocrine disorder resulting from a defect in insulin secretion, insulin resistance, or both. It is the third leading cause of morbidity and mortality, after heart attack and cancer. 7 Although synthetic drugs are mainly used for the amelioration of the altered glycaemic status in diabetic subjects, the utilization of natural products for the treatment of diabetes and associated conditions has been adopted in various healthcare systems around the globe. Besides in vivo studies, many clinical trials also support the antidiabetic effects of natural products and/or pure compounds. 8 The reduction of the post-prandial glucose levels through inhibition of the degradation of the oligo and disaccharides is one of the therapeutic strategies. The inhibition of carbohydrate-hydrolyzing enzymes such as α-amylase and α-glucosidase is essential to control post-prandial glucose levels as they are responsible for the digestion of starch and glycogen. 9 Flavonoids, alkaloids, terpenoids, anthocyanins, glycosides, and phenolic compounds are found to act as enzyme inhibitors with an inhibitory effect comparable to acarbose, a well-known anti-diabetic drug. 9
Polyphenols or phenolic compounds constitute one of the most numerous and widely distributed groups of substances in the plant kingdom and are closely related to the antibacterial and antidiabetic activity of several cultivated and wild plants.10,11 They are an inexhaustible reservoir of successful drug leads derived from Earth's biodiverse flora. As it is well known that more than 95% of the world's biodiversity has not been evaluated for any biological activity, screening wild and native plants for new bioactive compounds is a challenging perspective. 6
Cyprus is the third largest island of the Mediterranean basin and is very privileged from the floristic point of view as it exhibits great biodiversity in its flora compared to other European countries, as it is located at the crossroads of three continents. Furthermore, there is accumulated knowledge on the use of plant materials to treat or prevent several diseases, as several ethnic groups left their cultural traces on the island. Thus, the objective of the present work was to evaluate the antibacterial and carbohydrate digestive enzyme inhibitory effects of native edible plants from Cyprus, which are consumed for medicinal and culinary purposes. The study includes unexplored plant materials as well as plants that have been studied in other geographical regions.
Results and Discussion
Twelve edible native plants grown in Cyprus were harvested and studied in the present work. As Table 1 shows caper, silver thistle, crown daisy, and tuberous hawkbit are utilized for culinary purposes, whereas field marigold, wild clary, Greek mountain tea, and white mustard are usually consumed as decoction/infusion for health benefits. In addition, common mallow, oregano, rosemary, and vine leaves are consumed for both health effects and culinary purposes. For all selected plants, there are previous studies supporting their antimicrobial and/or antidiabetic effects. Apart from silver thistle, all plants have antibacterial and/or antifungal activities. Regarding their antidiabetic effects, there are mainly in vitro and ethnopharmacological studies supporting the potency of plants to treat diabetes and related conditions. Although the biological activities of some of the plants have been well-studied, the antibacterial and carbohydrate digestive enzyme inhibitory effects of plants grown in Cyprus were investigated for the first time. Cyprus is an isolated ecosystem that has an intense Mediterranean climate with prolonged hot summer.
12
It is expected that the combined drought and high temperatures induce secondary metabolism in plants to biosynthesize higher amounts of bioactive phytochemicals.
13
On the other hand, there are plant species such as
Information about Names, Edible Parts, Consumption Mode, Antimicrobial and Antidiabetic Effects of Twelve Studied Plants.

TPC and TFC of Plant Extracts
At first, the TPC and TFC of the extracts were determined, as phenolic compounds are widely distributed in plants and are correlated with their antibacterial and antidiabetic effects.10,11 The results demonstrated that polar solvents, namely water and methanol, recovered the highest amounts of phenolics compared to acetone and hexane (Figure 1). More specifically, TPC ranged from 41.48 ± 0.68 mg gallic acid equivalent (GAE) g−1 to 483.54 ± 11.82 mg GAE g−1 for aqueous extracts, from 35.40 ± 2.81 mg GAE g−1 to 259.05 ± 4.06 mg GAE g−1 for methanolic extracts, from 9.42 ± 0.24 mg GAE g−1 to 82.02 ± 1.35 mg GAE g−1 for acetonic extracts, and from 2.26 ± 0.34 mg GAE g−1 to 56.06 ± 0.36 mg GAE g−1 for hexanic extracts. The superiority of polar solvents to extract phenolic compounds from plant tissues has been thoroughly reported in the literature. 48 Among the plants examined, rosemary, wild sage, and capper had the highest phenolic contents in medium to high-polarity solvents, whereas the richest medium to low-polarity extracts came from oregano, rosemary, and capper. On the other hand, field marigold, silver thistle, and tuberous hawkbit had the lowest phenolic contents. Finally, results demonstrated the impact of the solvent on the extraction efficacy; Figure 1 reveals the appropriate solvent for each plant for extracting phenolic compounds.
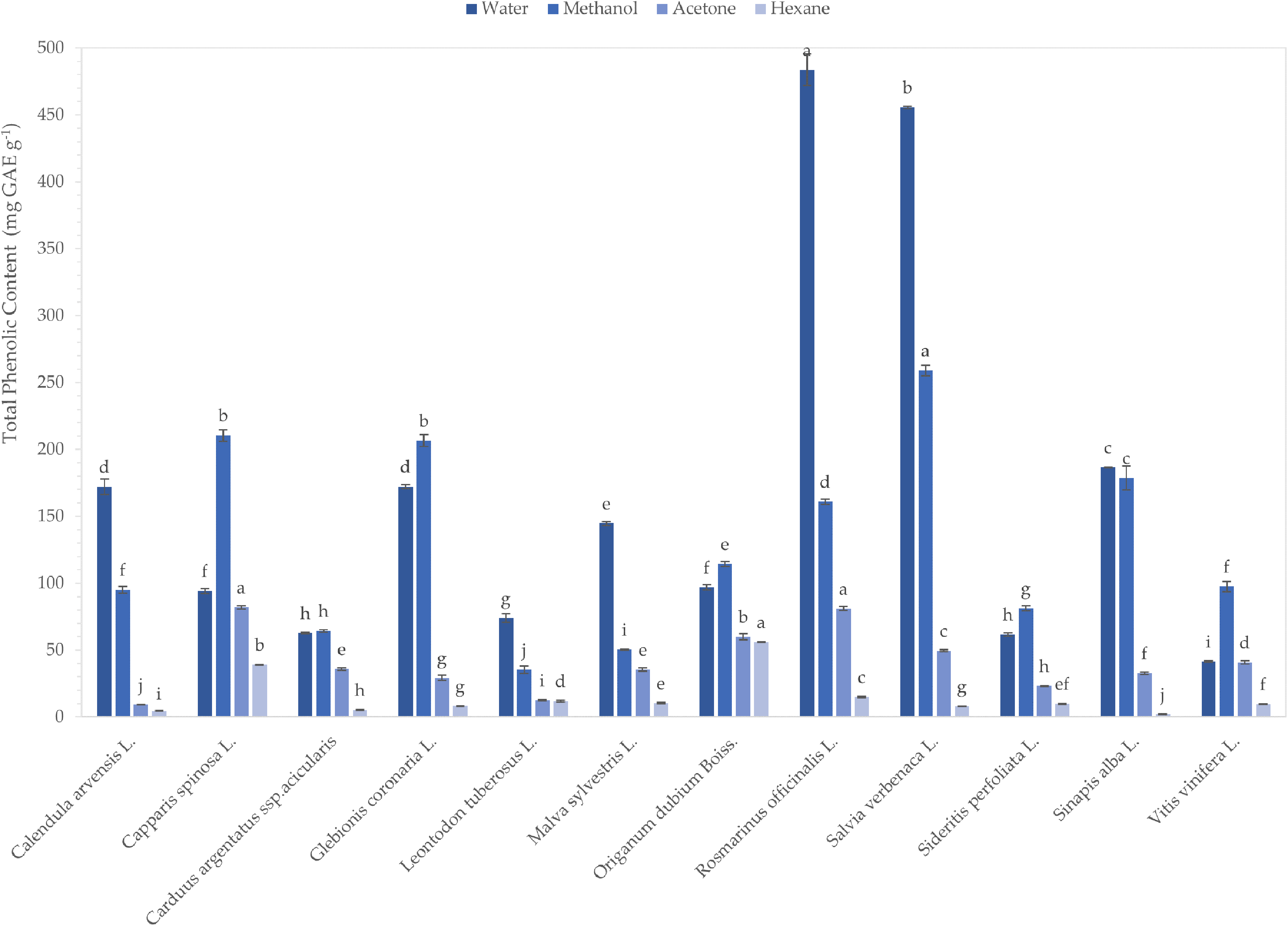
Total phenolic contents of extracts derived from twelve native plants grown in Cyprus. Results were expressed as mg gallic acid equivalent (GAE) g−1 material. The data is indicated as the mean ± SD. Different letters indicate statistically significant differences in contents (
Flavonoids are the main subgroup of polyphenols associated with the antidiabetic potency of plants. 49 Figure 2 demonstrates a great diversity in TFC values for the plants examined; TFCs ranged between 4.11 ± 0.10 mg catechin equivalent (CE) g−1 and 394.64 ± 6.92 mg CE g−1. The results showed that the lowest recovery of flavonoids was achieved when hexane was used for the ultrasonic extraction of plant materials. Similar to TPCs, oregano, rosemary, and capper extracts contain the highest concentration of flavonoids. Our findings provide useful information for the selection of appropriate solvents for recovery of flavonoids and a knowledge base of TPCs and TFCs for wild edible plants grown in Cyprus.

Total flavonoid contents of extracts derived from twelve native plants grown in Cyprus. Results were expressed as mg catechin equivalent (CE) g−1 material. The data is indicated as the mean ± SD. Different letters indicate statistically significant differences in contents (
Antibacterial Potency of Plant Extracts
The antibacterial potential of twelve edible plants grown in Cyprus was determined and presented in Table 2. In general, plant extracts showed higher bactericidal activity against Gram-positive bacteria rather than Gram-negative ones. It is well documented that plant-derived compounds have little or no inhibitory activity against Gram-negative bacteria, whereas they have significant activity against Gram-positive bacteria.
50
However, noteworthy susceptibility of tested bacteria was found among Gram-positive bacteria. The growth of
Minimum Bactericidal Concentration (MBC, mg mL−1) of Hexanic, Acetonic, Methanolic, and Aqueous Extracts of Twelve Edible, Native Plants Grown in Cyprus Against Six Bacteria.
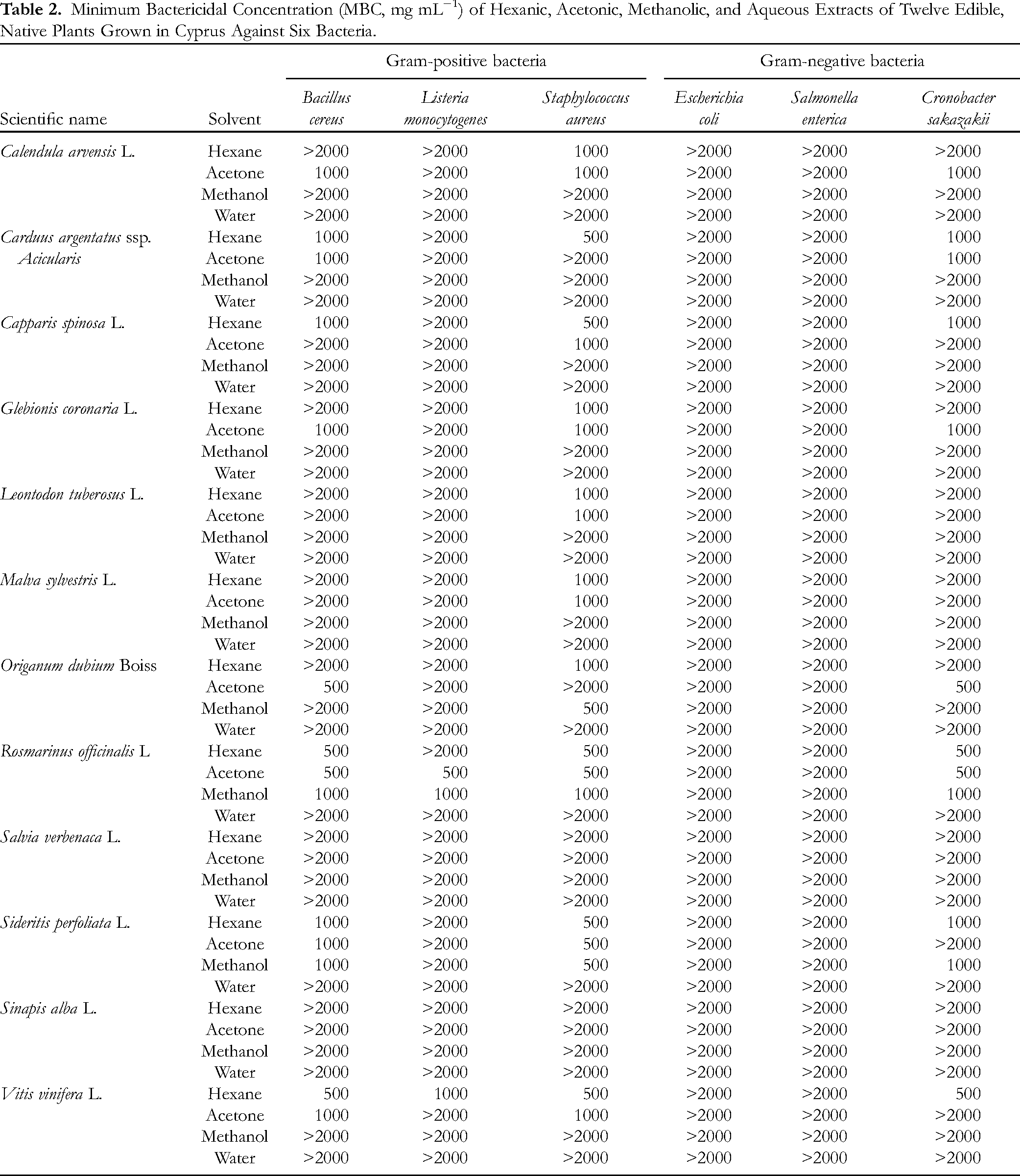
The plant extracts were inefficient to inhibit the growth of Gram-negative bacteria. Unfortunately, the tested plants had no bactericidal activity against
MBC values provide a valuable classification of studied edible plants. The most active plants were rosemary and grapevine leaves. Although the antibacterial potency of rosemary has been well studied, the corresponding potency of grapevine leaves from a native grape variety called “Mavro” was estimated for the first time. A group of six plants, namely field marigold, capper, silver thistle, crown daisy, oregano, and Greek mountain tea had a bactericidal activity against three tested bacteria (
Inhibitory Effect of Plant Extracts on Carbohydrate Digestive Enzymes
The inhibitory activity of all plant extracts on diabetes-related enzymes was previously assessed by in vitro and in vivo studies (Table 1). The serial exhaustive extraction method using a solvent of increasing polarity, from non-polar (hexane) to polar (water), was used to prepare crude extracts to focalize to the active components. Thus, the inhibition of the extracts on the enzymes was studied for each solvent separately. Results show that all plants had higher inhibitory effect against α-glucosidase compared to the α-amylase enzyme (Figure 3a). The % inhibitory activity of the hexanic extracts fluctuated from 20.29 ± 0.76% to 87.82 ± 0.54% for α-glucosidase, while a weak inhibition was found against α-amylase enzyme (1.96 ± 0.66-13.44 ± 2.04%). It is obvious that hexanic extracts cannot be considered as potential α-amylase inhibitors. On the other side, the hexanic extracts of rosemary and vine leaves had a potent inhibitory effect against the α-glucosidase enzyme. Recently, carnosol, a diterpene abundant in the hexanic extract of rosemary, was identified as a potent α-glucosidase inhibitor. 55 Regarding vine leaves, information is available only for polar extracts of grape skins and seeds.56,57 Thus, the assessment of α-glucosidase inhibitory activity of hexanic extract components has to be further studied in order to identify the active constituents.
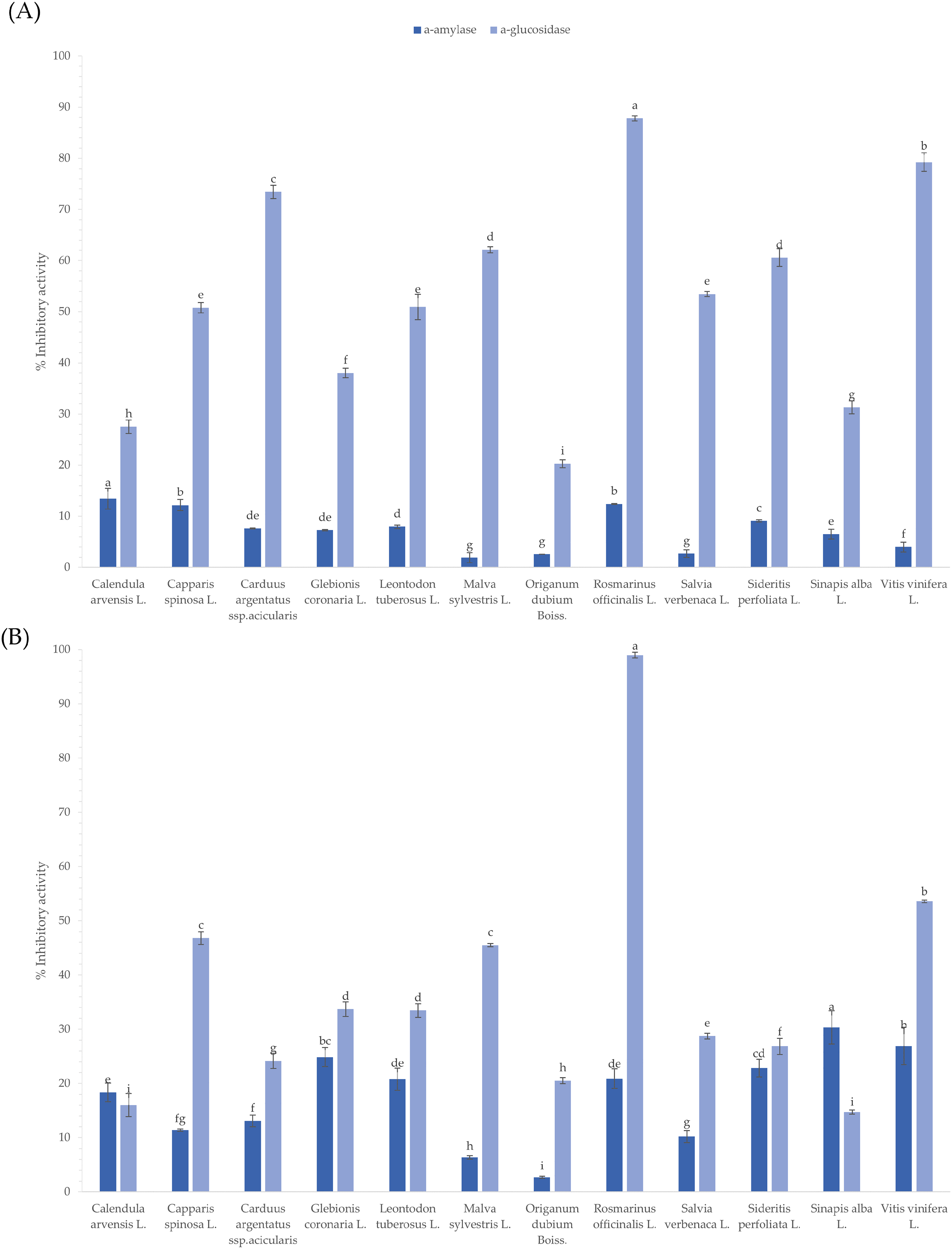
The percent α-glucosidase and α-amylase inhibition (%) of hexanic (A) and acetonic (B) extracts derived from twelve native plants. The data is indicated as the mean ± SD. Different letters indicate statistically significant differences in contents (
Subsequently, the inhibition activity of acetonic extracts was determined. Acetone is more polar than hexane, so the extracts are expected to contain medium-polar phytochemicals. The results demonstrated a similar trend for the acetonic and hexanic extracts (Figure 3(a) and (b)). The α-amylase inhibition activity of all acetonic extracts was less than 33.02%. Their α-glucosidase inhibitory activity values ranged from 14.78 ± 0.37% to 98.96 ± 0.52%. Indisputably, the acetonic extract of rosemary was an effective α-glucosidase inhibitor as its inhibition activity reached nearly 99%. This activity is possibly correlated with the presence of carnosol, rosmanol, and carnosic acid, as acetone is considered a suitable solvent for their recovery. 58 Results also showed that vine leaves, common mallow, and caper presented a medium inhibitory activity against the α-glucosidase enzyme. It is rather difficult to isolate an active constituent of pharmacological importance, but our findings support an additional nutraceutical value for three edible plants.
The utilization of methanol produces the most common plant extracts in the field of natural products research as it can simultaneously recover different classes of phytochemicals. According to our results, the most active extracts for inhibiting α-glucosidase were produced from vine leaves, caper, and wild clary (Figure 4(a)). The potent activities of caper and wild sage are probably related to their high content of phenolic compounds and flavonoids (Figures 1 and 2). Regarding the α-amylase assay, the highest activity was found for crown daisy, followed by field marigold and vine leaves. It is interesting that capper and wild clary had the weakest inhibition activity against α-amylase enzyme, while both plants exerted remarkable anti-α-glucosidase activity.

The percent α-glucosidase and α-amylase inhibition (%) of methanolic (A) and aqueous (B) extracts derived from twelve native plants grown in Cyprus. The data is indicated as the mean ± SD. Different letters indicate statistically significant differences in contents (
The inhibitory effect of aqueous extracts varied greatly as their % inhibition value ranged from 0.51% to 40.06% for α-amylase and from 0.70% to 83.12% for α-glucosidase, respectively. In contrast to other solvents, a close correlation was found between the two enzyme assays (r = 0.718). Unambiguously, the potential of vine leaf extract was great as it possessed significant inhibition on both digestive enzymes. Its activity is likely related to the presence of flavonoid glucosides as glycosylated derivatives of quercetin and kaempferol. 58 Previous work also highlighted that glycosylated flavonoids are more active than aglycone ones. 56 In addition to grapevine leaves, the aqueous extract of rosemary had noteworthy inhibitory activity against α-glucosidase and α-amylase. Greek mountain tea and tuberous hawkbit also acted reasonably as inhibitors of both digestive enzymes.
Results demonstrated the usefulness of the serial extraction method using solvents of increasing polarity to evaluate the inhibitory effects of edible plants against digestive enzymes. The use of a single extraction solvent may give an incorrect estimation of the plant's potential. Although the inhibitory effects of extracts against carbohydrate digestive enzymes were determined at specific concentration level due to the plethora of extracts, the present study delivers interesting findings. In particular, it demonstrates for the first time the inhibition activity of
Conclusions
This is the first report showing the antibacterial and carbohydrate digestive enzyme inhibitory effects of edible native plants from Cyprus. The present study provides insight into the antibacterial and in vitro antidiabetic activity of a variety of extracts from different plants utilized for medicinal and/or culinary purposes in Cyprus. Results contribute to the ongoing scientific investigation on the application of plant extracts as anti-bacterial and antidiabetic agents for the food and pharmaceutical industry. Furthermore, it delivers useful data for the nutraceutical potential of commonly consumed plants. Results also classify the plants based on their ability to inhibit the activity of two carbohydrate digestive enzymes related to diabetes. More specific, capper, rosemary, silver thistle, and vine leaf can be considered as valuable sources of α-glucosidase inhibitors; whereas their inhibitory effect against α-amylase is not promising. Mountain tea, oregano, rosemary, and vine leaf extracts had significant bactericidal potential against Gram-positive bacteria at a decent concentration. Based on these findings, further purification and testing of the effective plants are recommended to identify the major active ingredients responsible for the antibacterial and antidiabetic activity of these extracts.
Materials and Methods
Chemicals and Reagents
All chemicals were of analytical reagent grade. Hexane, dimethyl sulfoxide (DMSO), gallic acid, sodium nitrite (NaNO2), aluminum chloride (AlCl3), sodium dihydrogen phosphate anhydrous (NaH2PO4), disodium hydrogen phosphate dodecahydrate (Na2HPO4×12H2O), and soluble starch were purchased from Scharlau Chemie (Barcelona, Spain). Acetone and methanol were acquired from Honeywell (Charlotte, North Carolina, USA). Folin–Ciocalteu reagent, sodium carbonate (Na2CO3), catechin, p-nitrophenyl-α-D-glucopyranoside (PNG), and 3,5-dinitrosalicylic acid (DNS) were obtained from Sigma-Aldrich (Steinheim, Germany). Sodium hydroxide (NaOH) and sodium chloride (NaCl) were obtained from Merck (Darmstadt, Germany). The enzymes α-glucosidase from
Plant Materials
Twelve native and edible plants, namely
Preparation of Plant Extracts
Plant extracts were prepared by mixing approximately 5 g of dry powdered material with 30 mL of each solvent in the following order: hexane, acetone, methanol, and water. The obtained mixture was then placed in an ultrasonic bath (UCI-50, 35 KHz, Raypa-R. Espinar, S.L., Terrassa, Spain) and sonicated for 60 min. The temperature was set at 60°C when hexane, methanol, and water were used as the extraction solvents, while in the case of acetone, the temperature was reduced to 40°C. After the ultrasound treatment of the sample, it was allowed to cool at room temperature and then centrifuged for 10 min at 2500 rpm. The supernatant was collected and the remaining solid was extracted again with the next solvent, according to the solvent sequence described above. In this way, fractions of different polarities were collected. The obtained extracts were dried and stored at −20°C, until further analysis. Five replicates were performed for each extraction.
Determination of TPC and TFC
TPC of the plant extracts was determined using a 96-well microplate Folin–Ciocalteu method. 64 Prior to spectrophotometric analysis, the extracts were redissolved in 20% (v/v) DMSO and filtered through a 0.45-μm membrane filter to remove any insoluble particles. Then, 20 μL of extract solution was mixed with 100 μL of Folin–Ciocalteu reagent (1:4 v/v diluted with water), and the mixture was shaken for 1 min in a flat-bottom 96-well microplate. The mixture was allowed to stand for 4 min and then 75 μL of a saturated solution of sodium carbonate (100 g L−1) was added. The obtained mixture was shaken for 1 min and then allowed to stand in the dark at room temperature for 2 h. The absorbance of the reaction mixture was then measured at 750 nm using a Thermo Scientific Multiskan GO spectrophotometer (ThermoFisher Scientific, MA, USΑ). Gallic acid was used as a standard for calibration, and total phenolics were expressed as mg GAE per g of dry extract.
TFC of the extracts was investigated using the aluminum chloride colorimetry method. 65 Briefly, 25 μL of each extract was mixed with 100 μL of distilled water and 10 μL of a 50 g L−1 sodium nitrite solution in a flat-bottom 96-well microplate. After waiting for 5 min, 15 μL of AlCl3 solution (100 g L−1) was added to the reaction mixture. Then after waiting for another 6 min, aliquots of 50 μL of NaOH solution (1 mol L−1) and 50 μL of distilled water were added and the reaction mixture was shaken for 30 s. The absorbance of the mixture was measured at 510 nm using a Thermo Scientific Multiskan GO spectrophotometer. Catechin was used as the reference standard, and TFC values were expressed as mg CE per g of dry extract.
Assessment of Antibacterial Potency of Plant Extracts
The bacteria used for this study were three Gram-positive bacteria
Assessment of the Inhibitory Effect of Plant Extracts on Carbohydrate Digestive Enzymes
In brief, a mixture containing the extract solution (100 μL, 0.5 mg mL−1) and 50 μL of 0.1 mM phosphate buffer (pH = 6.8) containing a-glucosidase (1.0 U mL−1) was prepared and incubated at 37°C. After 10 min of incubation, 50 μL of PNG (5 mM in 0.1 mM phosphate buffer, pH = 6.8) was added and the reaction mixture was allowed to stand for 5 min. Finally, the absorbance of the mixture was measured at 405 nm against a blank solution where PNG was replaced with buffer. Control which represents 100% enzyme activity was prepared by replacing the extract solution with 20% (v/v) DMSO. Results were expressed as % inhibitory activity of the extracts compared to the control. 68
The α-amylase inhibitory activity of the extracts was determined using the DNS colorimetric method. 69 Briefly, 100 μL of the extract solution (10 mg mL−1 in 20% v/v DMSO) was mixed with 100 μL of α-amylase solution (2 U mL−1 in 20 mM sodium phosphate containing 6.7 mM NaCl, pH 6.9) and the resulting mixture was incubated at 35°C for 10 min. Then, 200 μL of the substrate solution (soluble starch, 1% w/v in the same buffer) was added and the reaction mixture was incubated at 35°C for 20 min. Finally, the reaction was terminated by adding 200 µL of DNS reagent. The mixture was then boiled for 10 min, cooled down, and diluted appropriately (1:10 with water) before its analysis at 540 nm. The absorbance of the reaction mixture was measured against a blank sample containing the extract solution, starch solution, and DNS (without enzyme). Results were expressed as % inhibitory activity of the extracts compared to the control.
Statistical Analysis
All experimental assays were performed in triplicate. The results obtained were expressed as mean values ± standard deviation (SD). The means were compared and statistical differences were obtained through one-way ANOVA followed by Duncan's multiple range test at a 95% confidence level. The differences between individual means were considered significant at
Footnotes
Acknowledgments
The authors would like to thank Dr Nikolaos Nikoloudakis (Department of Agricultural Sciences, Biotechnology & Food Science, Cyprus University of Technology for identifying plant material.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was co-funded by the European Regional Development Fund and the Republic of Cyprus through the Research & Innovation Foundation (Project: Natura Platform, EXCELLENCE/0421/0205).
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
