Abstract
A green study was undertaken for the extraction of plumbagin, an important natural naphthoquinone, from the roots of Plumbago pulchella Boiss. In the first stage, four solvents were comparatively evaluated, acetone, n-hexane, ethyl acetate, and methanol; attending Principle #5, only green solvents (ethyl acetate and acetone) were underscored. In a second step, considering Principle #6, various green alternative activating modes were assessed: two electromagnetic methods, microwave (MW) and near-infrared (NIR), in addition to mechanical approaches, ultrasound (US), and tribochemistry-mechanochemistry (TB-MCh). These green modes were studied in comparison to the classical mantle heating (MH) and finally with maceration (M). MW and US-MH ensued the better modes, and EtOAc (green) and n-hexane (non-green) displayed the best yields (0.027% and 0.025%, respectively). Focusing on Principle #11, the monitoring and quantification of the extractions were performed by GC-MS employing 8-hydroxyquinoline as a reference. Some other involved Principles (#1, #3, #7, #12) will be commented on in the discussion. As an important complement, a statistical technique based on the analysis of variance (ANOVA) and response surface methodology was used to establish the degree of dependence of the amount of plumbagin extracted with the parameters time and amount of root, in addition to a GC-MS validation process carried out according to ICH Q2 R2 2022 guidelines. Finally, it is convenient to highlight that the target plant is native to Mexico, and according to a profound literature search, no chemical information for Plumbago pulchella Boiss is accessible.
Introduction
Nowadays the green chemistry paradigm has acquired great importance wishing; to highlight that it is focused on the molecular level, accomplishing sustainability.1,2 In this sense, it is unequivocally positioned as an important scientific tool to prevent pollution (or at least minimize it). Green chemistry is engaged by a protocol, the Twelve Principles, 3 it being important to emphasize that along with it, the green chemical researchers design, develop, and many times innovate processes to attain low or no risk to the environment and consequently to human health. It is also important to highpoint, that no chemical activity is completely risk-free and waste-free. Therefore, the “Twelve Principles of green chemistry” must be assumed and accomplished as a scientific reflection. 4 In addition, it is noteworthy that green chemistry is currently considered an excellent way to contribute to sustainable development.2,5 Consequently, chemists are required to attend both the “UNESCO-Decade Call (2005–2014) for a sustainable education (teaching, investigation, and diffusion),” in addition to Agenda 30-ONU (2015-2030), a protocol related to the 17 sustainable development goals.
Plumbagin (5-hydroxy-2-methyl-1,4-naphtoquinone) (Figure 1), a naturally occurring metabolite of the Plumbago genus, has created great interest. It exhibits many valuable pharmacological activities 6 : anti-cancer,7‐11 leishmanicidal, 12 bactericidal, 13 anti-fungal, 14 antifertility, 15 hypolipidemic, 16 hyperglycemic, 17 and antioxidant, 18 among others. The major commercial species of the Plumbago genus is Plumbago zeylanica, which is highly cultivated in the south of India. However, it is important to mention that the content of plumbagin in this genus is low, Plumbago zeylanica 0.25 to 1.34%, Plumbago rosea 0.57 to 1.0%, and Plumbago capensis 0.15 to 0.43%. 6

Plumbagin structure.
As a consequence of the last comments, some research groups have focused their efforts on improving the extraction of plumbagin, employing several extraction methods: maceration,19‐21 serial maceration, 21 dynamic maceration, 20 mantle heating,20,21 and ultrasound.20,21 However, some of them have shown disadvantages: the use of chloroform 20 or its mixture with dichloromethane, 19 which are considered toxic solvents, 22 and the requirement of long extraction times, even days, employing as solvents, n-hexane, methanol, water, chloroform, or ethyl acetate.19‐21
After an exhaustive search of the literature, and consequently, to our knowledge, there is minimal information related to the chemistry and extraction of Plumbago pulchella Boiss: only three studies were found: cytotoxic activity, 23 skincare formulation, 24 and the hyperaccumulation occurring in ultramafic vegetation. 25 It is important to highlight that this species is native to Mexico (Figure S1; Supporting Information). 26
As a part of our research program, green chemistry, attending both “UNESCO-Decade Call (2005-2014) for a Sustainable Education (teaching, investigation, and diffusion)” and the Agenda 30-ONU (2015-2030), we have performed interesting comparative natural product isolation studies.27,28 This is in addition to accomplishing various green synthetic strategies to achieve the pharmacological activity of many important molecules.29‐33
Related to the above comments, this work is offered as a green approach for the extraction of plumbagin from the roots of Plumbago pulchella Boiss: evaluating mainly green solvents (Principle #5), ethyl acetate, and acetone, estimating green alternative activating modes (Principle #6), microwave (MW) and near-infrared (NIR) electromagnetic irradiations, also mechanical approaches such as ultrasound (US), and tribochemistry-mechanochemistry (TB-MCh), in comparison with classical mantle heating (MH) and maceration (M). It is significant to mention that the quantification and monitoring (Principle #11) of the extractions were determined by GC-MS. Moreover, a statistical technique (ANOVA) was used to establish the degree of dependence of the amount of plumbagin extracted with the parameters time and amount of root. In addition, a GC-MS validation study was performed working with the ICH Q2 R2 2022 guidelines.
Results and Discussion
Plumbagin/5-hydroxy-2-methyl-1,4-naphtoquinone was identified from its physical, spectroscopic, and mass spectrometric data and comparison with literature34‐36: identification complemented by new information: HRMS-DART+ (19.8 eV) m/z (% ra) [assignment]: 189(100%) [M + 1]+, accurate mass 189.05562, exact value 189.05517, error: 0.45 ppm, unsaturations 7.5, for C11H9O3, protonated molecule [M + H]+.
To establish appropriate primary conditions, the results of previously reported works19‐21 were considered: the solvents used were chloroform, 20 dichloromethane, 19 n-hexane, EtOAc, MeOH, and water, 21 in addition to chloroform–MeOH and chloroform–dichloromethane 19 mixtures. The employed modes used for extraction were Soxhlet (MH), 20 and serial maceration extraction (M), 21 the times were 2.5, 5, and 7.5 h.19‐21
In the first stage, the MH mode was assessed considering two quantities of plant roots, 50 and 1 g, employing four solvents: n-hexane, EtOAc, MeOH, and acetone (chloroform and dichloromethane are unquestionable non-green), for three extraction times: 2.5, 5, and 7.5 h, screening the extracts by GC-MS (Table 1). As can be seen, the solvents n-hexane and EtOAc provided better extraction yields (2.26 and 2.25 mg/g at 7.5 h). However, using acetone, considered a green solvent, this level was not reached. It is also noteworthy that both n-hexane and MeOH have been recently considered highly toxic according to the TRI-list 22 ; disrupting the green chemistry protocol. 37 They were, therefore, discarded from the green study.
Quantification of Plumbagin by GC-MS (1 g of Root).

The provided data are the mean of three replications.
Consequently, according to the previously obtained results, the green study was implemented using only EtOAc (in this work, the best green solvent) for the extraction of plumbagin, with 10 or 120 mL for 1 or 50 g of roots, respectively, at low extraction times of 7.5, 5, and 2.5 h, using for comparison, the following extraction modes: MH, US, TB-MCh, MW, US-MH, NIR, and M, monitoring the extraction by quantification of plumbagin by GC-MS.
The obtained data, in general, the mean of three replicates, of all the extractions are summarized in Table S1 (Supporting Information), and the corresponding graphical representation (providing enhanced objectivity) is shown in Figure 2.
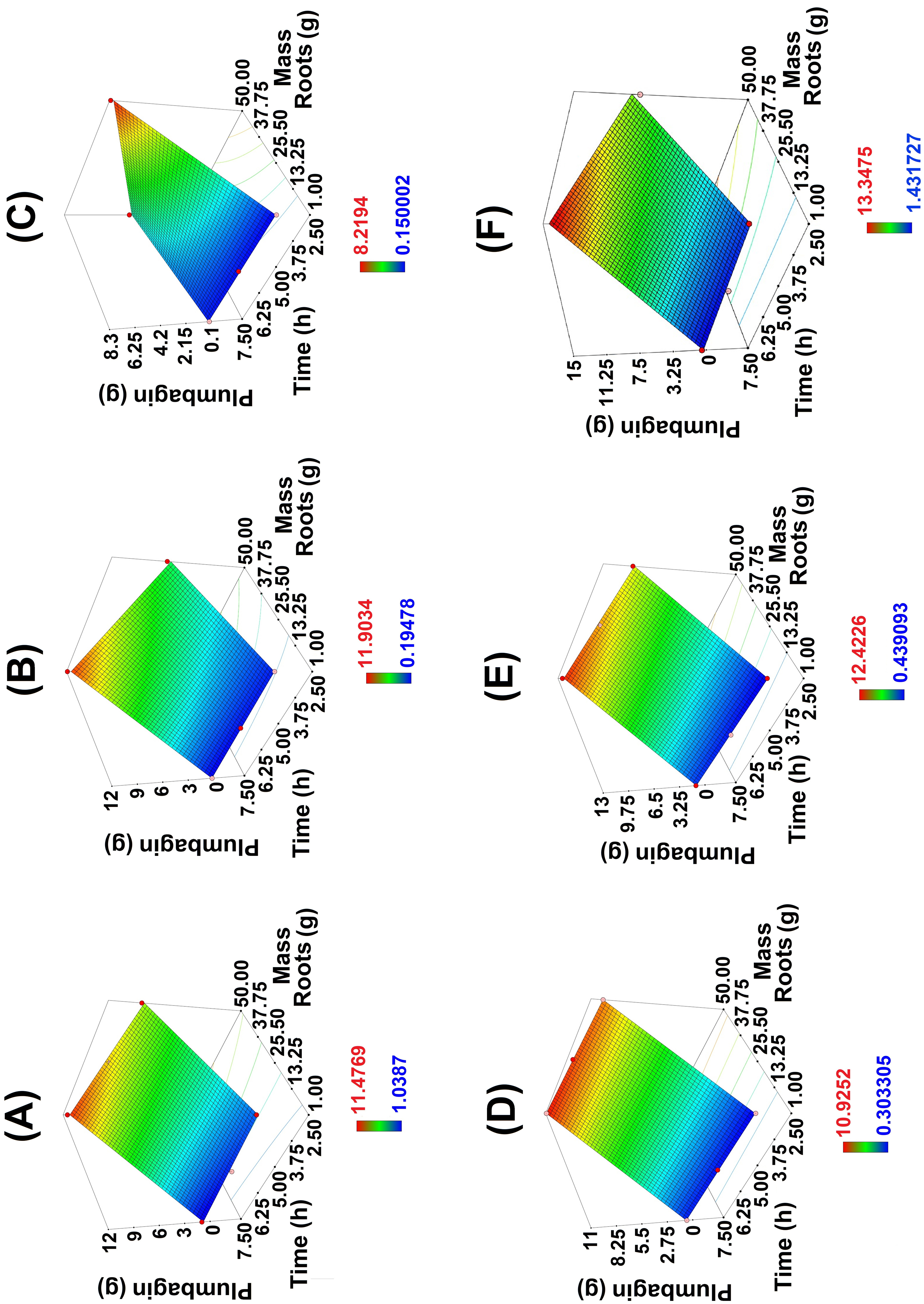
Quantification of plumbagin employing GC-MS for 50 g of roots, (A) MH, (B) NIR, (C) TB-MCh, (D) US, (E) US-MH, and (F) MW.
Statistical ANOVA Analysis
Through the analysis of variance and response surface methodology, a random experimental factorial design was generated, using the data from Table 2.
Levels of Variables Considered for Experimental Design.
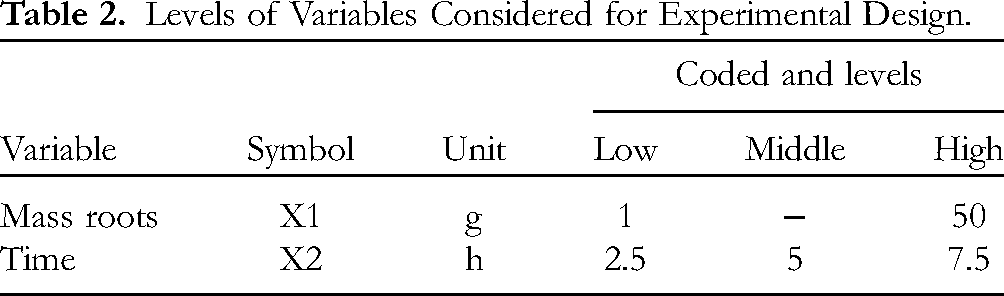
The results of the analysis were evaluated based on statistical values of the sum of squares (SS), degrees of freedom, the mean SS (MSS), Fisher variation ratio (F-value), and probability (P-value). Results of the ANOVA analysis are shown in Table 3, and the interaction between the process variables is illustrated by the 3D response surface plots (Figure 2). The optimum process parameters are calculated using the fitted model 2FI and validated by the experiments.
ANOVA for the Response Surface Model 2FI Suggested Extraction.
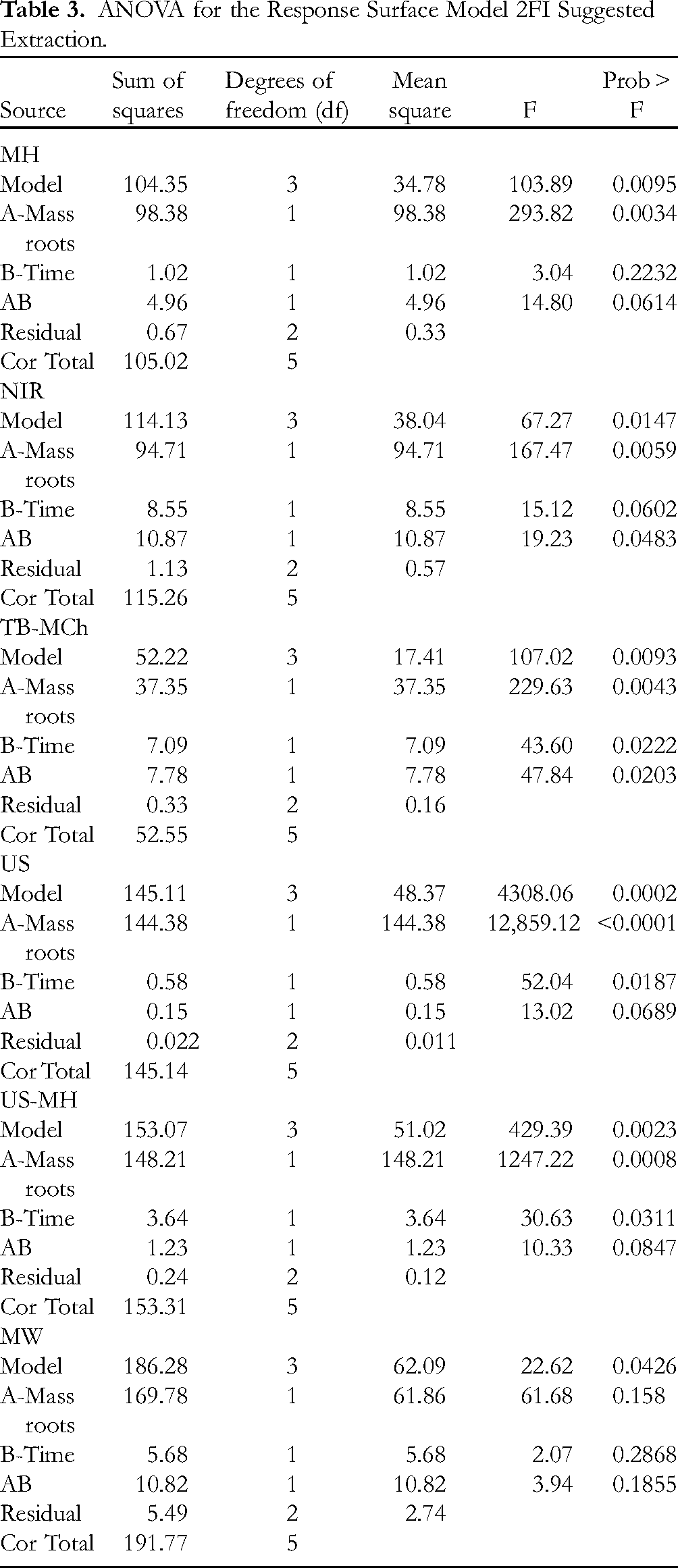
The Prob > F values were 0.0059, <0.0001, 0.0034, 0.0008, 0.0043, and 0.158 for the extractions using the NIR, US, MH, US-MH, TB-MCh, and MW methods, respectively. These results were less than 0.050, indicating that this model term is significant. 38 Regarding time, was found that the Prob > F values for the activation modes of US, US-MH, and TB-MCh were 0.0187, 0.0311, and 0.0222, respectively, indicating that time is also significant for these extraction methods. For the MW, NIR, and MH activation methods, the Prob > F values were 0.2868, 0.0602, and 0.2232, respectively. These results were greater than 0.05, and, therefore, not significant for the MW, NIR, and MH activation modes. However, if the values of Prob > F are not greater than 0.100, they can be considered significant. Thus, in all those cases, only NIR is significant.
The analysis of the standard deviation, R-squared (R2), adjusted R squared (Adj R squared), predicted R squared (pred R squared), and the adequate precision (Adeq precision) of the models are in reasonable agreement (Table 4). Adequate precision (Adeq precision) measures the signal-to-noise ratio. A ratio greater than 4 is desirable. Ratios of 18.31, 122.23, 21.85, 42.94, 23.63, and 10.30 for NIR, US, MH, US-MH, TB-MCh, and MW modes, respectively, showed adequate signals. These results indicate that this model can be used to navigate in the design space (Figure 2).
Descriptive Statistics Data Analysis for the 2FI Model.

Adj, adjusted; Pred, predicted; Adeq, adequate.
Abbreviations: MH, mantle heating; NIR, near-infrared; TB-MCh, tribochemistry-mechanochemistry; US, ultrasound; MW, microwave.
Based on the ANOVA analysis, the final equations that represent the behavior of the model for the extraction of plumbagin based on the activation modes NIR, US, MH, US-MH, TB-MCh, and MW are represented by equations S1-S6 (Supplementary Information).
Considering the comments, which agree with the data shown in Figure 2, Table 5 displays the best yields, both in weight and in percent, of plumbagin, for the different employed methods for the extraction of the target molecule. It is convenient to note that to obtain better objectivity the same results are displayed in Figure 3.

Summarized yields in percent of plumbagin, using 50 g of root: (A) plumbagin yield (mg), and (B) yield (% plumbagin).
Summarized Yields of Plumbagin.

Abbreviations: MH, mantle heating; NIR, near-infrared; TB-MCh, tribochemistry-mechanochemistry; US, ultrasound; MW, microwave.
Prime Results
The response surface data of the 2FI model (Figure 2A–E) are conveniently summarized in Table 5 and Figure 3A–B. The optimal conditions for the extraction of plumbagin, employing EtOAc, the better green solvent, and 50 g roots were: MH time 7.5 h (11.48 mg/0.023%), NIR time 7.5 h (11.90 mg/0.024%), TB-MCh time 2.5 h (8.22 mg/0.016%), US time 7.5 h (10.93 mg/0.022%), US-MH time 7.5 h (12.42 mg/0.025%), and MW time 7.5 h (13.35 mg/0.027%).
In this regard, the most convenient green approaches correspond to US-MH time 7.5 h (12.42 mg/0.025%), employing EtOAc as the solvent. Nevertheless, the use of MW (1300 Watts, 65 °C, 0.7799 bar) as activating mode for the extraction of the target compound must be highlighted since at the first assessment of 50 g roots, time 5 h, and EtOAc-greener solvent, the higher yields were achieved (13.34 mg corresponding to 0.027%).
Complementarily, it is convenient to comment that the maceration mode (M), with excellent incidence in Principle #6 of green chemistry, was discarded since even after more than 12 h (50 g/EtOAc), only traces of plumbagin were detected, as evidenced by monitoring by GC-MS, see Supplementary Information (Figures S3–S8).
Conventional Mantle Heating-Thermal Conditions for (Reflux) Extraction
It is well known that MH processes are governed by temperature and solvent recycling 28 ; in this work, plumbagin was obtained with good yield (mg and %) (Figures S3–S8). In this case, the solvent, EtOAc, has a convenient dielectric constant of 6.0 D 39 to improve the solvent/plumbagin interactions, favoring the solubility-selectivity of the target compound. According to the response surface of the 2FI model shown in Figure 2(A), the optimal conditions for the extraction of plumbagin with MH were 7.5 h and 50 g roots.
NIR Promoted Extraction
The plumbagin yield obtained by NIR was very close to that of the MH process, according to the TIC chromatogram (Figures S6–S8). In addition, infrared irradiation can induce different vibration modes in the molecules providing high energy efficiency.40,41 Additionally the irradiation can be directly absorbed by the root tissues and solvent. Consequently, this irradiation can directly heat the solvent-Plumbago roots mixture, facilitating the extraction of the metabolite from the sample matrix.27,28 According to the response surface of the 2FI model shown in Figure 2(B), the optimal conditions for the extraction of plumbagin with NIR were 7.5 h and 50 g roots.
TB-MCh-Assisted Extraction
An appropriate explanation related to the low plumbagin yields for this procedure (Figures S3–S8) is the poor production of tribomicroplasma in the vicinity of the sliding contact area between the Plumbago roots and the mortar-pestle, 42 in addition to lower friction conditions provoking lesser intensity toward the extract mixture. 43 According to the response surface of the 2FI model shown in Figure 2(C), the optimal conditions for the extraction of plumbagin with TB-MCh were 2.5 h and 50 g roots.
US-Assisted Extraction
Extraction assisted only by ultrasound displayed a yield slightly lower than that of MH and NIR (Figures S3–S5). In other words, the cavitation effects produced good interaction with the cellular structure in the tissues and disruption of the cell walls, releasing the metabolite of interest by a convenient solvent penetration.27,28 According to the response surface of the 2FI model shown in Figure 2(D), the optimal conditions for the extraction of plumbagin with US were 7.5 h and 50 g roots.
Ultrasound-Reflux-Assisted Extraction
This procedure offered better plumbagin extraction, in comparison to the previously reported methods19‐21 (Figures 3, and S3–S8); however, it was not for this study. At this point, the synergy between the cavitation effect and the direct heating by a typical reflux process increased the yield by a higher disruption of the root cell walls and solvent/solute interactions, facilitating the release of the target molecule due to better mass transfer of the metabolite from the sample matrix.27,28 According to the response surface of the 2FI model shown in Figure 2(E), the optimal conditions for the extraction of plumbagin with US-MH were 7.5 h and 50 g roots.
Microwave-Assisted Extraction
It is convenient to highlight that the MW mode afforded the best extraction yield of 13.35 mg/50 g roots (Figures 3 and S6–S8) and the best yield percent (0.27%) of plumbagin (Table 5). Appropriate facts to explain these results are in agreement between the solvent and the plumbagin polarity (convenient dielectric constant) 39 required by the MW procedure to achieve suitable interactions. In addition, MW engenders high pressures, supporting a rupture of the cell wall of the roots, favoring the interaction of solvent-plumbagin.28,44‐46 According to the response surface of the 2FI model shown in Figure 2(F), the optimal conditions for the extraction of plumbagin with MW were 5 h and 50 g roots.
Conventional Extraction by Maceration
This procedure provided the lowest extraction value and longest extraction time (days) in comparison to MH, US, US-MH, NIR, TB-MCh, and MW. This was probably due to lower mass transfer of the metabolite from the sample matrix and poor solvent/solute interactions 28 ; heat is necessary to obtain a good extraction.
Green Chemical Discussion
The green chemistry protocol is offered as an eco-study for the extraction of plumbagin from the roots of Plumbago pulchella Boiss. In this sense, green chemistry is not different from traditional chemistry but only has special considerations for the implementation of new designs to prevent pollution. 3 According to Anastas and Warner, 3 the green chemistry protocol is based on Twelve Principles. However, in this study, the only principles discussed are those with evident concern toward the green chemistry protocol. (a) Related to Principle #1 (it is better to prevent waste than to treat or clean up waste after it is formed). 3 This is shown by the TIC chromatogram in which peaks other than that of plumbagin must be due to other metabolites (not of interest in this study). Hence the employment of methanol was avoided, and the selectivity was improved by ethyl acetate diminishing the waste. (b) Ethyl acetate, a green solvent,22,47 was the best option, provoking the incidence of Principle #5 (the use of auxiliary substances [eg, solvents, separation agents, etc] should be made unnecessary wherever possible and innocuous when used). 3 (c) A high prevalence of Principle #6 (energy requirements should be recognized for their environmental and economic impacts and should be minimized. Synthetic methods should be conducted at ambient and pressure), 3 diminishing the energetic consumption and the economic impacts; this fact is unequivocally involved since several green alternative activating modes, microwave and NIR electromagnetic irradiations, also mechanical approaches such as ultrasound, and tribochemistry-mechanochemistry, in comparison with the classical mantle heating (MH) and maceration (M). (d) Principle #7 (raw material of feedstock should be renewable rather than depleting wherever technically and economically practicable) 3 was involved. The roots of Plumbago pulchella Boiss are renewable, and the species is perennial. (e) Significant incidence of Principle #11 (analytical methodologies need to be further developed to allow for real-time, in process monitoring and control prior to the formation of hazardous substances) 3 is highlighted and displayed since the monitoring of the extractions was by GC-MS. 48 (f) Finally, Principles #3 (wherever practicable, synthetic methodologies should be designed to use and generate substances that possess little or no toxicity to human health and the environment) 3 and #12 (substances and the form of a substance used in a chemical process should be chosen so as to minimize the potential for chemical accidents, including releases, explosions, and fires) 3 were implicated, since EtOAc has been categorized as a green solvent due to its low toxicity, explosiveness, flammability, and good degradability. 47
Conclusions
A green study for the extraction of plumbagin, an important natural naphthoquinone, from the roots of Plumbago pulchella Boiss was accomplished. According to a profound literature search, it seems that it is the first chemical contribution for the target plant; in this sense, it is convenient to highlight that the evaluated specimen is a native of Mexico.
Ethyl acetate, in general, was the best solvent, giving the best yields (weight and %) with MW irradiation; the US-MH procedure also gave good results.
Evaluation of the extraction of plumbagin was validated by the application of profound statistical analysis.
Finally, unequivocally a green approach is offered since a metrical evaluation has been performed. In this sense, the obtained results are in excellent agreement with the Green Chemistry Protocol, which is currently considered an excellent way to contribute to Sustainable Development; in addition, the results complied with both the “UNESCO-Decade Call (2005-2014) for a Sustainable Education (teaching, investigation, and diffusion)” and the Agenda 30-ONU (2015-2030).
Ethical Approval
Ethical approval is not applicable to this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informal consent is not applicable.
Material and Methods
General
Plumbagin, 8-hydroxyquinoline (≥99%, 8-HQ) and HPLC grade n-hexane were purchased from Sigma-Aldrich Chemistry (St. Louis, MO, USA). n-hexane, ethyl acetate, acetone, and methanol were technical grade and were used as received from Materiales y Abastos Especializados S.A. de C.V. (Zapopan, Jalisco, Mexico). The plumbagin extractions were accomplished from the roots of Plumbago pulchella Boiss. The identification and presence of the target molecule were monitored by GC-MS (Figures S2), in addition to comparative thin-layer chromatography (TLC), using n-hexane/ethyl acetate 85:15 as eluent, employing silica gel 60-F254 coated aluminum sheets and visualization with a 254 nm UV lamp.
Microwave-assisted extraction of 50 g of roots was achieved using a Multiwave Pro Solv 60 Hz reactor (Anton Paar, México). However, for 1 g of material, the microwave-assisted extraction was performed using a CEM Focused Microwave™ Synthesis System (CEM Corporation, Matthews, NC, USA). For the NIR irradiation extractions, a commercially available Flavor Wave device was used (Model AX-767MH, 1350 W/110 V/120 V-60 Hz–220 V/240 V-60 Hz) (Made in China). US-assisted extraction was performed using a Branson 1510 ultrasonic bath (80 W, 40 kHz) (Gaithersburg, MD, USA). Mecanochemical-assisted extraction experiments were performed using an IntelliMixer device model RM-2M (ELMI Ltd, Latvia) for samples of 1 g, and a mortar and pestle (volcano rocks) was used for samples of 50 g. In the NIR and US experiments, the temperatures were determined using an infrared thermometer (Infrared + Type K Thermomether, Extech Instruments, Sigma-Aldrich 2509388-1, St. Louis, MO, USA), with the laser point directed toward the center of the extraction mixture. GC-MS was carried out and validated (see Supplementary Information) using a 6850 G2630A/G3170A gas chromatogram in line with a JMS 5975C spectrometer (Agilent Technologies, CA, USA). GC was performed using a 5% phenylmethylsilicon column (30 m × 0.25 μm, 0.25 μm) Agilent 19091s-433e. The linear velocity of the helium carrier gas was 0.9 mL/min. The injector and detector temperatures were 250 and 300 °C, respectively, and they were held for 6 min with a split mode of 1:50. 8-Hydroxyquinoline (8-HQ) was employed as an internal reference to quantify the plumbagin. Related to the physical and spectroscopic characterization of plumbagin, the melting point was determined using a Fisher Johns apparatus and is uncorrected. The EIMS (70 eV) was determined using a JEOL JMS-700 MStation mass spectrometer (JEOL, Peabody, MA, USA). The HRMS-DART+ data were obtained using a JEOL AccuTOF (Direct Analysis in Real Time) mass spectrometer (JEOL, Peabody, MA, USA). The measurements were performed using a DART experiment with polyethylene glycol 400 as an internal reference at 6000 resolutions and triplet helium as carrier gas at 350 °C.
Plant Material
The roots of Plumbago pulchella Boiss were collected in January 2022 in the northeast of State Park Sierra de Guadalupe, Ecatepec de Morelos, Mexico, at the coordinates: west hill 19.5917678, north hill −99.0597134, height 7767.98 feet. The specimen was identified by authorities of the Ecological Park Ehécatl, zone leader Luis Hernández Ramírez, and biologist Montserrat Ramírez Hernández
Typical Extraction by Maceration
Independent experiments were conducted using 0.5, 1, 5, 10, 30, or 50 g of dried and milled roots of Plumbago pulchella Boiss. These samples were mixed with 10, 15, 30, 60, 90, or 120 mL of n-hexane, ethyl acetate, acetone, or methanol and macerating for 2.5, 5, 7.5, or 12 h at room temperature. 21 Each experiment was systematically monitored by TLC (n-hexane/ethyl acetate 85:15; in silica) employing an authentic commercial sample of plumbagin; after this time, the extracts were filtered and dried under vacuum.
Extraction Using Typical Mantle Heating-Thermal Conditions (Reflux)
Independent experiments were conducted with either 1 g or 50 g of dried and milled roots of Plumbago pulchella Boiss with either 10 or 120 mL of ethyl acetate, n-hexane, MeOH, or acetone, respectively, and refluxed for 2.5, 5, or 7.5 h at 63–65 °C. 21 Each extract was systematically monitored by TLC (n-hexane/ethyl acetate 85:15; on silica gel) employing an authentic commercial sample of plumbagin; after this time, the extracts were filtered and dried under vacuum.
Typical Extraction Using Nonconventional Activating Sources
NIR irradiation: independent experiments were conducted using either 1 or 50 g of Plumbago pulchella Boiss roots, with either 10 or 120 mL of EtOAc, respectively, for 2.5, 5, or 7.5 h between 63 °C and 65 °C. In this extraction, several irradiations of 10 min each were carried out, with a pause of 5 min between each irradiation, to avoid the solvent boiling. Each extract was systematically monitored by TLC (n-hexane/ethyl acetate 85:15; on silica gel) employing an authentic commercial sample of plumbagin; after this time, the extracts were filtered and dried under vacuum.
Microwave irradiation: independent experiments were conducted. Either 1 or 50 g of the roots of Plumbago pulchella Boiss, along with either 10 or 120 mL of EtOAc, respectively, was irradiated with 300 W of power for 2.5, 5, or 7.5 h, or 1300 W of power at 60 °C for 2.5, 5, or 7.5 h. Each extract, was monitored by TLC (n-hexane/ethyl acetate 85:15; on silica gel) employing an authentic sample of plumbagin; after this time, the extracts were filtered and dried under vacuum.
Tribochemistry-mechanochemical: either 1 or 50 g of Plumbago pulchella Boiss roots, along with either 10 or 120 mL of EtOAc, respectively, were ground either in an intelli mixer with the shake and rocking UU mode with 80 r/min, using five carbon steel balls of 3/16 inches diameter, or mechanical milled with a mortar and pestle, respectively, for 2.5, 5, or 7.5 h at room temperature. Hence, it is important to note that due to solvent evaporation in the second method, it was necessary to add an extra 100 mL of solvent. The extract of each experiment was systematically monitored by TLC (n-hexane/ethyl acetate 85:15; in silica) employing an authentic commercial sample of plumbagin; after this time, the extracts were filtered and dried under vacuum.
Ultrasound: independent experiments were conducted using either 1 or 50 g of Plumbago pulchella Boiss roots with either 10 or 120 mL of EtOAc, respectively. These were sonicated for 2.5, 5, or 7.5 h at room temperature. All the extracts were systematically monitored by TLC (n-hexane/ethyl acetate 85:15; on silica gel) employing an authentic sample of plumbagin; after this time, the extracts were filtered and dried under vacuum.
Ultrasound extraction under reflux: independent experiments were conducted using either 1 or 50 g of Plumbago pulchella Boiss roots with either 10 or 120 mL of EtOAc, respectively. These were sonicated for 2.5, 5, or 7.5 h between 63 °C and 65 °C. The extract of each experiment was systematically monitored by TLC (n-hexane/ethyl acetate 85:15; on silica gel) employing an authentic sample of plumbagin; after this time, the extracts were filtered and dried under vacuum.
Plumbagin Quantification
GC-MS Determinations
Plumbagin quantification in the extract was performed by GC-MS using 8-HQ as an internal standard. The 8-HQ solution was prepared by dissolving 2 mg of 8-HQ in 0.5 mL HPLC grade n-hexane. Then, 2.8 mL of HPLC grade n-hexane was added to each extract. The analysis of the sample was prepared by mixing 20 μL of the extract with 20 μL of 8-HQ, and dissolving the mixture in HPLC-grade n-hexane. Then, 2 μL of the mixture was injected into the GC-MS equipment. The quantification was determined as follows:
GC-MS Validation Process
The GC-MS validation process was carried out according to ICH Q2 R2 2022 guidelines. The suitability of the system was established by verifying the operation of the internal calibration system and using authentic plumbagin standards with known concentration. Linearity, specificity, accuracy, precision, quantification, and detection of limit parameters were determined for validation of the process. The obtained results gave R2 > 0.9919 linearity acceptance, an accuracy of 98.84%, and, related to precision criteria, a variance coefficient of 1.34%. In addition, the detection limit was 20.66 ng/mL, and the quantification limit was 68.87 ng/mL. The specificity of the GC-MS method was validated by injecting 1 μL of pure plumbagin and methanol (100%) as control, individually. Five different concentrations (200–1000 ng) of plumbagin were analyzed in triplicate, and the respective calibration curve was generated. The linearity between peak areas and concentration of plumbagin was calculated using linear regression analysis (Graphic S1–S3). The detection limit and the quantification limit were calculated based on the standard deviation of the intercept (y) and slope of the calibration curve obtained from linear regression (Graphic S4) (see Supplementary Information).
Statistical Process Control
Experimental design and statistical analysis of the results were performed using the software Design Expert 7.0.0 (version 2005, Stat-Ease, In. 2021, MN, USA). The amount of plumbagin obtained in milligram per gram of initial root was selected as the dependent response variable. The mathematical relationship between the response function (Y) and the independent variables (X) can be approximated by a 2FI model equation.
Supporting Information
The following supporting information can be seen (Table S1). Quantification of plumbagin in EtOAc extracts was done by GC-MS. Figure S1. Endemic and native distribution of Plumbago pulchella Boiss in Mexico. Endemic: Baja California, Baja California Sur, Campeche, Chiapas, Chihuahua, Coahuila, Quintana Roo, Sonora, Sinaloa, Tabasco, Yucatán. Native: Aguascalientes, Colima, Ciudad de México, Durango, Guanajuato, Guerrero, Hidalgo, Jalisco, Michoacán, Morelos, México, Nayarit, Nuevo León, Oaxaca, Puebla, Querétaro, San Luis Potosí, Tamaulipas, Tlaxcala, Veracruz, Zacatecas. Figure S2. (a) TIC chromatogram, (b) mass spectrum of plumbagin, and (c) TIC chromatogram of 8-hydroxyquinone (8-HQ). Figure S3. TIC chromatogram of plumbagin extract with (a) mantle heating, (b) near-infrared irradiation, (c) tribochemistry-mechanochemistry, (d) ultrasound, (e) ultrasound-mantle heating, and (f) microwave, using 1 g of root, with 2.5 h extraction. Figure S4. TIC chromatogram of plumbagin extract with (a) mantle heating, (b) near-infrared irradiation, (c) tribochemistry-mechanochemistry, (d) ultrasound, (e) ultrasound-mantle heating, and (f) microwave, using 1 g of root with 5 h extraction. Figure S5. TIC chromatogram of plumbagin extract with (a) mantle heating, (b) near-infrared irradiation, (c) tribochemistry-mechanochemistry, (d) ultrasound, (e) ultrasound-mantle heating, and (f) microwave, using 1 g of root and 7.5 h extraction. Figure S6. TIC chromatogram of plumbagin extract with (a) mantle heating, (b) near-infrared irradiation, (c) tribochemistry-mechanochemistry, (d) ultrasound, (e) ultrasound-mantle heating, and (f) microwave, using 50 g of root, and 2.5 h extraction. Figure S7. TIC chromatogram of plumbagin extract with (a) mantle heating, (b) near-infrared irradiation, (c) tribochemistry-mechanochemistry, (d) ultrasound, (e) ultrasound-mantle heating, and (f) microwave, using 50 g of root, and 5 h extraction. Figure S8. TIC chromatogram of plumbagin extract with (a) mantle heating, (b) near-infrared irradiation, (c) tribochemistry-mechanochemistry, (d) ultrasound, (e) ultrasound-mantle heating, and (f) microwave, using 50 g of root, and 7.5 h extraction. Equation S1. Surface model of the behavior of extraction by MH. Equation S2. Surface model of the behavior of extraction by NIR. Equation S3. Surface model of the behavior of extraction by TB-MCh. Equation S4. Surface model of the behavior of extraction by US. Equation S5. Surface model of the behavior of extraction by US-MH. Equation S6. Surface model of the behavior of extraction by MW. Validation of Gas Chromatography studies by ICH Q2 R2 2022 guidelines.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231191345 - Supplemental material for Plumbagin Extraction: A Green Study
Supplemental material, sj-docx-1-npx-10.1177_1934578X231191345 for Plumbagin Extraction: A Green Study by Fernanda I. Calzada-González, Joel Martínez and Yolanda M. Vargas-Rodríguez, Luis B. Hernández-Portilla, M. Inés Nicolás-Vázquez, René Miranda in Natural Product Communications
Footnotes
Authors’ Note
This article is dedicated to the memory of Professor Pedro Joseph-Nathan.
Acknowledgments
The authors acknowledge Secretaría del Medio Ambiente del Estado de México for authorization to collect the plant specimen. Also, to Draucin Jimenéz Celi (FES-Cuautitlán UNAM) and Anabel Ruiz Flores (UBIPRO, FES-Iztacala UNAM) for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by Dirección General de Asuntos del Personal Académico DGAPA-PAPIIT (grant IN211023) and by Programa Interno de Cátedras de Investigación Convocatoria 2022, FESC-UNAM (grant CI2218).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
