Abstract
The onset of drug resistance represents the leading cause of chemotherapy failure in clinics. In the last decades, natural compounds have emerged as possible anticancer strategies used either alone or in combination with chemotherapeutic drugs, in order to overcome drug resistance. In fact, plant-derived therapies present biological activity and minimal side effects showing promising roles in the resensitization of resistant cancer cells. This work was aimed at investigating the anticancer potential of the natural naphthoquinone plumbagin in a cisplatin-resistant cancer cell line. The results indicated cytotoxic and pro-oxidant activity of plumbagin in both sensitive (A431wt) and cisplatin-resistant (A431/Pt) human cervix squamous carcinoma cell lines. Moreover, plumbagin treatment induced cell cycle arrest and apoptosis in A431/Pt cells and the inhibition of retinoblastoma complex, suggesting a stronger activity on the cisplatin-resistant cell line. Taken together, the data indicate appreciable in vitro anticancer activity of plumbagin, suggesting that this natural compound could become a tool to overcome cisplatin resistance. Although further studies are necessary, this work underlines a promising role of plumbagin in the resensitization of cisplatin-resistant cancer cells.
In the last decades, researchers have identified several natural compounds with high biological activity as a good strategy to counteract drug resistance occurring in antitumor treatments. In fact, the favorable pharmacological and toxicological profiles of these natural substances and their ability to modulate different targets and signaling pathways involved in cancer allow their clinical use either alone or in combined treatment with chemotherapeutic agents. 1
Among these natural compounds, plumbagin (5-hydroxy-2-methyl-1,4-naphthoquinone) is a naphthoquinone extracted from the roots of Plumbago zeylanica L., a medicinal plant also known as “Chitrak” in Ayurveda medicine. 2 -4 Plumbago zeylanica is a large perennial shrub belonging to the Plumbaginaceae family that can be found in a wild state throughout India. 4 Several studies demonstrated the anticancer role of plumbagin in different types of tumors, such as prostate, breast, gastric, ovarian, colon, esophageal, lung, and pancreatic cancers. 2,5 -11
Cisplatin is a chemotherapeutic agent used as first-line treatment for different types of human solid tumors, such as ovarian, cervical, head and neck, and testicular cancers. 12 -14 In addition to the occurrence of severe toxicity, the main issue related to the clinical failure of chemotherapy is the onset of drug resistance. Therefore, new pharmacological and safer strategies have to be pinpointed to counteract the loss of clinical efficacy of cisplatin after the onset of resistance.
The unchecked regulation of the cell cycle is one of the hallmarks of cancer cells. 15 Thus, targeting the cell cycle could become a strategy that leads to the success of anticancer therapies.
The aim of the present study was to investigate the effect of plumbagin in the regulation of the cell cycle in both sensitive and cisplatin-resistant human cervix squamous cell carcinoma cell lines (A431wt and A431/Pt, respectively), evaluating also the effect on cell viability and reactive oxygen species (ROS) production in order to consider the potential anticancer activity and the possible role of plumbagin in overcoming cisplatin resistance.
Results and Discussion
Cytotoxicity
The effect of 24 hours and 48 hours of plumbagin treatment (0.5-100 μM) was evaluated in human cervix squamous cell carcinoma cell lines (A431wt [sensitive clone] and A431/Pt [cisplatin-resistant clone]). 3-(4,5-Dimethyl-thiazol-2-yl)−2,5-diphenyltetrazolium bromide (MTT) assay was performed, and the results showed a significantly decreased cell viability of both A431 and A431/Pt cell lines, in particular after plumbagin 10 µM treatment, as shown in Figure 1a and b.

Effect of plumbagin (0.5-100 µM) on wild-type A431 (black) and cisplatin-resistant A431/Pt (red) cell growth after (a) 24 hours of drug treatment and (b) 48 hours of drug treatment. Differences between various drug concentrations were evaluated by using 2-way analysis of variance followed by Bonferroni's multiple comparison test. * P < 0.05, *** P < 0.001, between cell lines, wild type vs cisplatin-resistant counterpart; ### P < 0.001, treated cells vs control. Data are the mean ± standard deviation of at least 3 determinations.
The half-maximal inhibitory concentration (IC50) for the cytotoxic effect of plumbagin was calculated from MTT assay data by logistic function regression analysis for both cell lines. Estimated values of IC50 at both 24 hours and 48 hours of treatment are reported in Table 1. The IC50 value was significantly lower for the A431wt cell line in comparison with its A431/Pt counterpart, at both considered times.
Cytotoxic Effect of Plumbagin (Calculated as IC50) After 24 Hours and 48 Hours of Treatment in A431wt and A431/Pt Cell Lines.

IC50, half-maximal inhibitory concentration.
aValues were determined by regression analysis and are expressed as mean ± standard deviation of at least 3 replicates. ***P < 0.001; **P < 0.01, analysis of variance followed by Bonferroni’s test, comparison between cell lines at each time.
From these data, it is possible to conclude that plumbagin exerts a cytotoxic effect, concentration dependent, on both sensitive and resistant clones. A431wt cells appeared to be more sensitive to plumbagin treatment compared with the A431/Pt line.
ROS Assay
Literature studies demonstrated that plumbagin presents pro-oxidant activity 16,17 and that high ROS levels cause deoxyribonucleic acid (DNA) damage and cellular death. In the present work, ROS levels were evaluated by flow cytometry after 2 hours of plumbagin treatment. As reported in Figure 2, plumbagin treatment (1 and 5 µM) was able to significantly increase ROS levels, in comparison with the respective control, in both sensitive and resistant clones.

Reactive oxygen species production in A431wt and A431/Pt cell lines in the presence of 2 concentrations (1 and 5 µM) of plumbagin after a 2-hour treatment. Differences between various drug concentrations and control were analyzed by using one-way analysis of variance followed by Bonferroni's multiple comparison test. ** P<0.01, *** P<0.001, treated vs respective control. Data are the mean ± standard deviation of 3 different experiments.
Cell Cycle and Apoptosis
As already known from literature studies, plumbagin is able to arrest the cell cycle. 3,7,18 The effect of plumbagin treatment (1 and 10 µM) on the cell cycle was tested in A431wt and A431/Pt cells, performing the analysis through flow cytometry. Treatment for 48 hours with the substance caused variation in the cell cycle. In particular, in the A431wt cell line, plumbagin 10 µM decreased significantly the G0/G1 and S phases, while it caused a significant increase in the G2/M phase (Figure 3(a)). In the A431/Pt cell line, treatment with plumbagin 10 µM caused a significant increase in the S and G2/M phases while it decreased the G0/G1 phase, determining a consequent arrest of the cell cycle with a block between the G2/M and G0/G1 phases (Figure 3(b)). Moreover, plumbagin treatment increased apoptosis cell death in both sensitive and resistant clones, as observed by the increased percentage of cells in the sub G0 phase with respect to the untreated ones (Figure 4).
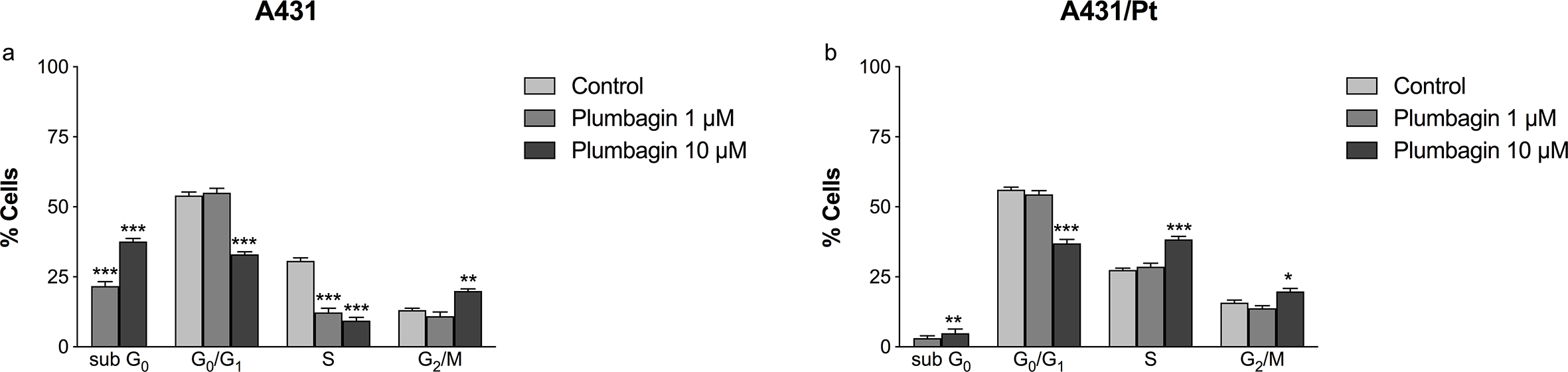
Deoxyribonucleic acid cell-cycle analysis of (a) wild-type A431 and (b) cisplatin-resistant A431/Pt cells. Cells were treated with either medium (control) or plumbagin (1 and 10 µM) for 48 hours and analyzed by flow cytometry. Percentage of cells in G0/G1, S and G2-M phase was calculated using Multicycle software. Differences between various drug concentrations were analyzed by using two-way analysis of variance followed by Tukey’s multiple comparison test. * P<0.05, ** P<0.01*** P<0.001, treated vs control. The cytograms are representative of 3 independent experiments (mean data ± standard deviation are indicated).

Cytograms of 48 hours of (a) A431wt untreated (control) cells; (b) A431wt cells treated with plumbagin 1 µM; (c) A431wt cells treated with plumbagin 10 µM; (d) A431/Pt untreated (control) cells; (e) A431/Pt cells treated with plumbagin 1 µM; (f) A431/Pt cells treated with plumbagin 10 µM. Plots of deoxyribonucleic acid content (propidium iodide fluorescence intensity) vs number of cell events. Percentages of cells in sub G0, G0/G1, S and G2/M phase were calculated using Multicycle software and are represented in the right side of the histograms. All pictures are typical of three independent experiments each performed under identical conditions.
Western Blotting
The protein expression of retinoblastoma was evaluated using Western blotting. The 2 different forms, the hypophosphorylated, inactive form, and the hyperphosphorylated, active one, were compared. As shown in Figure 5a and b, the hyperphosphorylated form decreased compared with the hypophosphorylated, confirming the inhibition of the cell cycle. In fact, the inhibition of the retinoblastoma complex indicates the lack of continuation of the cell cycle.

Relative levels of p-Rb (a) and pp-Rb (b) analyzed by Western blotting in both cell lines upon incubation with plumbagin (1 and 10 µM) for 24 hours. Values are the mean ± standard deviation of 3-4 determinations and are expressed as optical density (OD) in arbitrary units (AU). * P < 0.05; **P < 0.01 vs control (Šídák's multiple comparison test).
The acquired cisplatin resistance of cancer cells is the commonest cause that leads to clinical chemotherapeutic treatment failure. In the last decades, plant-based molecules have gained attention in the field of cancer management thanks to their pharmacological activity, minimally acceptable side effects, and easy availability. Accumulating evidence showed that many natural compounds from numerous biological sources are able to modulate different pathways involved in the survival and growth progression of several types of human cancer cells.
Currently, many phytochemicals have been tested to sensitize tumor cells to chemotherapeutic drugs by inhibiting cell proliferation and pathways related to cell survival. 19 These compounds are mainly flavonoids, polyphenols, terpenes, glycosides, and carotenoids, with particular examples being polyphenols such as genistein, curcumin, silymarin, and ursolic acids. 19 Combinatorial approaches with the application of chemotherapeutic drugs with a natural compound can achieve a synergistic/additive effect that can give advantages in cancer treatment. Our laboratory has previously demonstrated how flavonoids and some polyphenols have appreciable cytotoxic activity and a possible role in resensitizing cisplatin-resistant cell lines, highlighting a selectivity of action and suggesting the possible use of these compounds as coadjuvants in cancer treatment. 20,21 Different natural phenols, such as quercetin, rhein, and capsaicin have been tested, showing cytotoxicity on human ovarian and cervix cancer cell lines. 21 Moreover, it has highlighted the role of quercetin on cell cycle arrest in human ovarian cisplatin-resistant cancer cells 20 ; the combined treatment of curcumin with cisplatin has been demonstrated to be able to arrest the cell cycle and induce cell apoptosis more than cisplatin alone. 22 In the last years, our laboratory has also identified the synergistic effect of silybin and doxorubicin in human colorectal adenocarcinoma cells (LoVo wt and LoVo DOX cell lines). 23
Following the above-mentioned research front and taking advantage of the availability of a validated model of chemotherapeutic drug resistance, this work was aimed at investigating the anticancer role of the natural naphthoquinone plumbagin in human cervix squamous carcinoma cell lines sensitive and resistant to cisplatin treatment. As previously reported, plumbagin has shown cytotoxic anticancer activities in several tumor cells, such as lung, breast, cervical, colorectal, esophageal, and gastric cancer cells. 24 The inhibitory effect of plumbagin treatment on cell viability was demonstrated in our cell model in both sensitive and resistant cell lines.
It is known and well established that the oxidative balance in cancer cells is essential for their survival and that the generation of cytotoxic oxidative stress in cancer cells has emerged as an exploitable strategy for cancer-specific treatment. 25,26 Natural products are known to affect the redox state of cells but are mainly studied for their antioxidant properties instead of their pro-oxidant activities.
Several studies reported that the pro-oxidant activity of some natural compounds may become a strategy to kill cancer cells because of cellular DNA damage. 27 Also for plumbagin, it has been demonstrated that the ability to increase ROS production in human cervical cancer and that high ROS production causes DNA damage, leading to cellular death. 16,17 In line with these observations, this work confirms the pro-oxidant activity of plumbagin in both sensitive and resistant clones, underlying involvement of ROS generation in cell death.
It has been demonstrated that cancer cells take advantage of the unchecked cell cycle in order to increase their survival and proliferation. As the deregulation of the cell cycle is considered a hallmark of cancer, targeting it could become another promising anticancer tool. Also under this aspect, several natural compounds have exhibited inhibitory activity on tumor cell proliferation and cell cycle, offering new hints for potential anticancer treatment. 28 Different studies reported that plumbagin is able to induce cell cycle arrest in different human tumors. In human breast cancer MCF-7 cell lines, plumbagin induced G2/M phase cell cycle arrest 3 ; in PANC-1 and BxPC-3 human pancreatic cell lines, plumbagin induced cell cycle arrest, regulating different key factors, such as cyclin D1, CDK1/CDC2, and cyclin B1. 7 Moreover, a cell cycle arrest has been evidenced in human melanoma A375.S2 cells where plumbagin caused the induction of S-G2/M phase arrest and apoptotic cell death. 18 Interestingly, in the present work, it was observed that in the A431/Pt resistant line plumbagin induces a G2/M phase cell cycle arrest and an increase in the apoptotic rate with respect to the A431wt sensitive cells. It is also known that retinoblastoma protein (Rb) is a key factor in the negative regulation of cell cycle, operating as a major G1 checkpoint, blocking S-phase entry and cell proliferation. During the G0 phase, p-Rb is hypophosphorylated, while until the late mitosis phase it is hyperphosphorylated. In the G1 phase, p-Rb increases the phosphorylation. 29 -31 Data obtained showed that plumbagin is able to decrease the hyperphosphorylated form of the retinoblastoma protein compared with the hypophosphorylated form, causing the inhibition of the retinoblastoma complex. The block of retinoblastoma complex confirms the cell cycle arrest of the resistant cancer cells.
Conclusions
In conclusion, this work shows that plumbagin is able to inhibit the cell viability in our model of both sensitive and cisplatin-resistant cancer cell lines and to induce an increase in ROS production; in addition, the present results demonstrate plumbagin’s ability to arrest the cell cycle in cisplatin-resistant cancer cells and highlight the occurrence of inhibition in the hyperphosphorylated retinoblastoma protein expression.
Since cancer chemoresistance is one of the main factors that hamper the effectiveness of the therapy, increasing the knowledge about possible strategies to overcome this issue becomes a pivotal point in cancer research. Natural products offer a wide variety of phytochemicals with interesting pharmacological properties and limited toxic side effects, suggesting their possible use in combination with traditional chemotherapeutic drugs to overcome drug resistance and, in the final analysis, to improve the efficacy of the therapy. Although deeper studies to elucidate the specific molecular mechanisms linked to the observed effects are necessary, from this work plumbagin appears to be a promising candidate for further investigation as a cisplatin-sensitizing agent.
Experimental
Cells
The wild-type A431 cell line (A431wt) derived from human cervix squamous carcinoma and the cisplatin-resistant variant (A431/Pt) were selected by exposure of A431 cells to increasing cisplatin concentrations for a period of 9 months. 32 Cells were routinely grown in humidified conditions in 5% carbon dioxide at 37°C, incubated with Roswell Park Memorial Institute medium (RPMI 1640), 10% fetal bovine serum (FBS), 4 mM glutamine, 100 U/mL penicillin and 100 µg/mL streptomycin. Cells were collected every 2 days with a minimum amount of 0.05% trypsin–0.02% ethylenediaminetetraacetic acid (EDTA). All reagents were from Cambrex, Lonza (NY, USA) and FBS from Gibco, Invitrogen (Carlsbad, CA, USA).
Chemicals
Plumbagin, with purity >95% (by high-performance liquid chromatography analysis), was purchased from Sigma-Aldrich (St Louis, MO, USA). A 1 mg/mL stock solution of Plumbagin was prepared in dimethyl sulfoxide, stored as small aliquots at −20°C, and then diluted as needed into the cell culture medium.
MTT Assay
The MTT assay was used to determine cell viability. 20 -22 MTT assay consisted of plating cells on 96-multiwell plates, and after overnight incubation, exposing cells to a different concentration range of plumbagin according to the experimental protocol. After 24 hours and 48 hours, 20 µL/well of a 5 mg/mL MTT solution (Sigma-Aldrich, St Louis, MO, USA) was added and incubated for 4 hours at 37°C. The formazan crystals formed following this procedure were dissolved by adding 200 µL of acidic isopropanol, and the absorbance (Abs) was measured at 570 nm using a Victor3X multilabel plate counter (Wallac Instruments, Turku, Finland).
ROS Production
2′,7′-Dichlorofluorescein-diacetate (H2DCF-DA, Molecular Probes, Invitrogen Corp., Carlsbad, CA, USA) was used to detect directly the intracellular steady-state levels of ROS on living cells. Untreated or plumbagin-treated cells were incubated for 2 hours, then 5 µM H2-DCF-DA was added and incubated for 30 minutes at 37°C. After that, a phosphate-buffered saline solution was used to wash the cells; then cells were detached from duplicate wells by 0.05% trypsin and 0.02% EDTA, centrifuged for 5 minutes at 800 g and finally suspended in the medium. Epics XL Coulter Systems (Beckman Coulter, USA) equipped with a 488 Argon laser was the instrument used to analyze directly the cells. Electronically gating data on the basis of forward vs side scatter profiles were used to exclude dead cells; a minimum of 104 cells of interest was further analyzed. Logarithmic detectors were used for the FL1 fluorescence channel necessary for DCF detection. The values of percentages of fluorescent cells were obtained by analysis through EXPO 32 software (Beckman Coulter, Brea, CA, USA).
Cell Cycle and Apoptosis Analysis by Flow Cytometry
Cells were seeded at a constant number on 6-well plates and, after overnight incubation, were exposed to plumbagin (1-10 μM) for 24 hours and 48 hours. At the end of the treatments, cells were washed, detached with 0.25% trypsin–0.2% EDTA and fixed with cold ethanol at 70% for 30 minutes on ice. Following washing, cells were resuspended in a PBS solution to which were added 0.03 mg/mL propidium iodide (Molecular Probes, Invitrogen, UK) and 0.03 mg/mL RNAase-DNAase free (Sigma, St. Louis, MO, USA) and incubated for about 30 minutes at room temperature in the dark. About 3 × 104 cells/sample were analyzed on an Epics XL-flow cytometer (Beckman Coulter, USA), with an Argon Laser λexc = 488 nm and a λem= 575 ± 20 nm. The percentages of cells in the different phases of the cell cycle were calculated with the Multicycle software provided by the manufacturer, considering the diploid cycle, and correcting for cell clusters.
Western Blotting
One hundred millimeter cell culture dishes were seeded with a constant number of cells and, following overnight incubation, were treated with plumbagin (1-10 µM) for 48 hours. Cells were then lysed with ice-cold lysis buffer supplemented with protease inhibitor cocktails (Roche Molecular Biochemicals, Mannheim, Germany), and the protein content was determined by the Lowry procedure (Bio-rad DC Protein Assay, MA, USA). Forty micrograms of protein of each sample was loaded onto a polyacrylamide gel and electrophoretically separated in running buffer. After electrophoresis, the proteins were blotted onto an Hybond-P polyvinylidene difluoride membrane (Amersham Biosciences, Buckinghamshire, UK). Following blocking with a 10% skim milk solution, the membrane was exposed to anti-p-Rb (Ser 807/811) antibody (1:200; Santa Cruz Biotechnology Inc., Heidelberg, Germany) and after washing, incubated with HRP-conjugated antirabbit secondary antibody (1:3500; PerkinElmer, MA, USA). The signal was visualized with an enhanced chemoluminescent kit (Amersham Biosciences) according to the manufacturer’s instructions and analyzed by a Molecular Imager VersaDoc MP 4000 (Bio-rad). Proteins were normalized to β-actin (1:7000 (Mouse, AbCam, Cambridge, UK).
Statistical Analysis
GraphPad Prism version 3.03 (GraphPad, San Diego, CA, USA) software was used for statistical evaluation of experimental results. Data were analyzed using one-way or two-way analysis of variance, followed by an appropriate post-hoc test (Bonferroni or Tukey-Kramer, or Šídák multiple comparison tests). The IC50 values were determined by logistic function regression analysis with a nonlinear curve fitting process using cytotoxicity curves performed in triplicate. A P value of less than 0.05 was considered for determining statistical significance.
Footnotes
Acknowledgments
MM is supported by the University of Padova (CPDA 159453). ER is supported by the University of Padova (DOR, UA.A.D10.020_UA_Ricerca). IG and VC are supported by the University of Padova.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
