Abstract
Objectives
The current work aimed to isolate and identify new interesting small-molecule natural products from the leaves of a well-known medicinal plant Morus alba.
Methods
The ethanol extract of M. alba leaves was partitioned between EtOAc and water, and the EtOAc soluble part was fractionated by macroporous resin column chromatograph (CC) eluted with gradient EtOH. The 80% EtOH eluate was subject to silica gel, reversed phase C18, MCI gel and Sephadex LH-20 CC and finally purified by HPLC to afford four minor compounds
Results
A pair of new quinone enantiomers (
Conclusions
The M. alba leaves represent a rich source of new natural products, and the currently obtained constituents did not exert obvious growth inhibitory activity against the tested tumor cell lines. The bioactivities of the new isolates deserve further exploration in the future.
Introduction
The genus Morus is the most famous plant tribe of family Moraceae in that most Morus species possess significant economic values because of their nature as both food and natural medication in human's long history. 1 M. alba (mulberry) as the type species of genus Morus is an indigenous plant of China but now has spread out to most Asian and European countries. 1 In East Asian countries especially China, M. alba has been used for over a thousand years in different aspects of daily life. 1 Of particular note, all parts of mulberry tree are with various usages/values: the fruit is a popular food in early summer, and the leaf (Mori folium), tender twig (Mori ramulus) and root bark (Mori cortex) are catalogued in Chinse National Pharmacopeia as herbal medicines. 2 Modern researches including natural product investigations have demonstrated that the great values of mulberry tree are closely related to its bioactive chemical constituents, which has been well summarized in several comprehensive reviews.3,4 Among the different plant parts of M. alba, the leaf represents an interesting and specialized one owing to the fact that it is the only part that has been recognized by Chinese National Health Commission to be a homology of both food and medicine. Previous phytochemical studies and biological evaluations into mulberry leaf revealed that it is rich in small-molecule natural compounds including alkaloids, benzofurans, catechins, coumarins, flavonoids, etc, with flavonoids being the predominant components, and more interestingly, these metabolites and the leaf extract were reported to show a variety of pharmacological properties, making mulberry leaf an attractive research topic for years till now.5,6
Recently, several reports on mulberry root barks collected in the authors’ hometown have drawn our attention due to the large number of new diversified structures and promising therapeutical potential for diabetes and renal fibrosis.7–9 As is known to all, natural products vary according to the body part, habitat, collecting time and even local seasonal climate of the source species. Therefore, we wonder if mulberry leaf from the same province also contains such rich new chemical constituents. An intensive phytochemical investigation was thus conducted on mulberry leaf collected in Xiajin County of Shandong province, a place famous for its ancient mulberry trees. As a result, new quinone (

Chemical Structures of Compounds
Materials and Methods
General
Optical rotations: Rudolph VI polarimeter (Rudolph Research Analytical, Hackettstown, USA); UV and ECD data: Chirascan circular dichroism spectrometer (Applied Photophysics Ltd, Surrey, UK); NMR experiments: in CD3OD (δH 3.30, δC 49.15), Bruker Avance DRX600 spectrometer (Bruker BioSpin AG, Fallanden, Switzerland); HR-ESIMS spectra: Agilent 6545 Q-TOF mass spectrometer (Agilent Technologies Inc., Waldbronn, Germany); HPLC analyses and separations: SilGreen-C18 column (5 μm, 10 × 250 mm, Greenherbs Science and Technology, Beijing, China), Daicel CHIRALPAK AD-H and IC columns (both 5 μm, 4.6 × 250 mm, Daicel Chiral Technologies Co. Ltd, Shanghai, China) on Shimadzu LC instrument (Shimadzu, Kyoto, Japan). Conventional column chromatography (CC): D101-macroporous absorption resin (Sinopharm Chemical Reagent Co., Ltd, Shanghai, China), reversed-phase (RP) C18 silica gel (Merck KGaA, Darmstadt, Germany), MCI gel (CHP20/P120, Mitsubishi Chemical Co. Ltd, Tokyo, Japan), Sephadex LH-20 (GE Healthcare Bio-Sciences AB, Uppsala, Sweden) and silica gel (300-400 mesh or H; Qingdao Marine Chemical Co. Ltd, Qingdao, China).
Plant Materials
The leaves of Morus alba L. (family Moraceae) were collected at the end of October 2024 in Xiajin County, Shandong Province (China), and the species authentication was accomplished by Prof. Xiushan Yin from Shandong First Medical University (Accession No.: TMUCIH-2024-006).
Extraction and Isolation
The air-dried leaves (5.0 kg) of M. alba were milled and the powder was percolated with 15 L EtOH at room temperature for four times (once a week). The EtOH extraction was solvent-removed under reduced pressure and the obtained residue (600 g) was partitioned between water (1.0 L) and EtOAc (0.5 L × 4). The organic phases were combined and condensed in vacuum to afford the EtOAc soluble part (256 g), which was then fractionated on D101 macroporous resin CC with 30%, 50%, 80% and 95% (all 5.0 L) EtOH in H2O successively. Each eluate was concentrated to remove the solvent to yield the respective fractions.
The 80% EtOH-H2O eluate (89 g) was first separated by silica gel CC (petroleum ether-EtOAc, 30:1 to 1:1) to give 12 fractions (Fr-A to Fr-L). Fr-J (702 mg) was then fractionated by a RP-C18 column (50%−100% MeOH-H2O) to obtain six further subfractions (Fr-J1 to Fr-J6), among which Fr-J2 was further processed with Sephadex LH-20 CC (CH2Cl2-MeOH 1:1) to yield subfractions Fr-J2-1 to Fr-J2-4. Fr-J2-3 was finally purified by HPLC (50% MeCN-H2O) to give compound
Compound
Compounds
1H (600 MHz) and 13C (150 MHz) NMR Data for Compounds

Assignments with the same superscript might be interchangeable.
Compounds
ECD Calculations
Compounds
Cytotoxic Assay
Tumor cell lines were obtained from the cell bank of Chinese Academy of Sciences, Beijing Branch (Beijing, China) and cultured according to the supplier's instructions. Briefly, MDA-MB231, A549 and Hela cells were maintained in Dulbecco's Modified Eagle Medium supplemented with 10% (v/v) fetal bovine serum (FBS), and 4T1 cells were maintained in Roswell Park Memorial Institute 1640 medium supplemented with 10% (v/v) FBS. All cell lines were incubated at 37 °C in a humidified atmosphere containing 5% CO2.
In brief, tumor cells were seeded in 96-well plates at the density of 6 × 103 cells/well. After 24 h, different concentrations of tested compounds were added and co-incubated for another 24 h. Then, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide solution (MTT, 5 mg/mL, 10 μL) was added to each well and the plate was cultured for another 2 h. Following removal of the culture suspension, 100 μL dimethyl sulfoxide was added to each well. Finally, the optical density values at 490 nm were measured using a microplate reader (Tecan, Austria). Adriamycin (Aladdin, Beijing, China) was used as a reference compound.
Statistical Analysis
Statistical analysis (if applicable) was performed using GraphPad Prism 8.02 (GraphPad Software, San Diego, CA, USA) and a one-way analysis of variance (ANOVA) followed by Tukey's multiple comparison tests.
Results
Compound
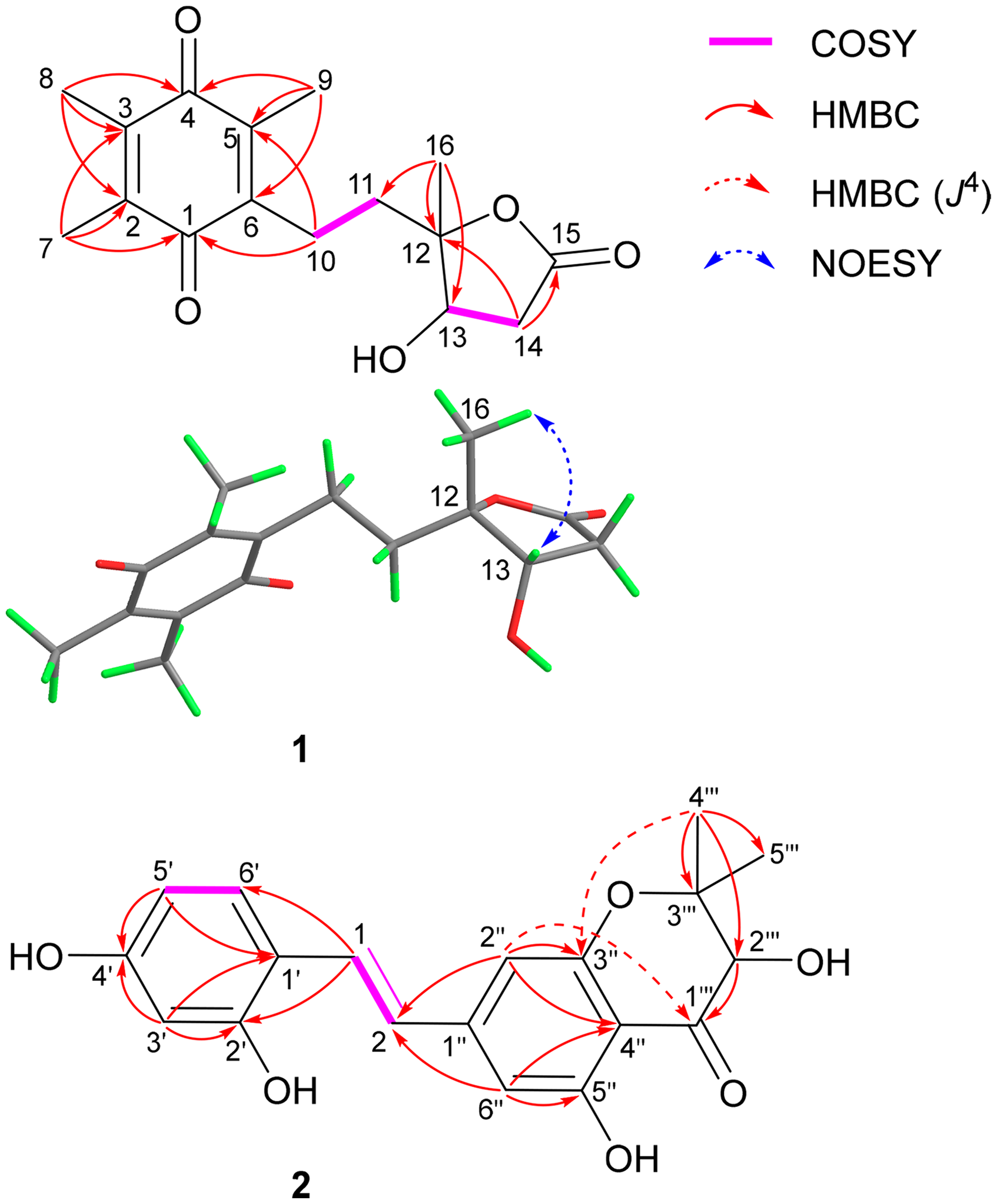
Illustration of key 2D NMR Correlations of Compounds
It was interesting to note that the specific optical rotation of

Experimental and Calculated ECD spectra for Compounds
Compound
Two known minor constituents, namely 4-prenyloxyresveratrol (
As the novel quinone (
Discussion
In the current work, two pairs of new minor enantiomers (
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251385042 - Supplemental material for Minor Non-Cytotoxic Enantiomeric Constituents from the Leaves of Morus alba
Supplemental material, sj-docx-1-npx-10.1177_1934578X251385042 for Minor Non-Cytotoxic Enantiomeric Constituents from the Leaves of Morus alba by Jie Xu, Yanfang Yang, Qianqian Li, Ziyi Dong, Yaoyang Guo and Zhansheng Jiang in Natural Product Communications
Footnotes
Acknowledgements
This project was financially supported by the Tianjin Municipal Health Commission (No. 2024013), and we thank Prof. Xiushan Yin for the authentication of plant materials.
Ethical Considerations
The present study does not involve use of any experimental material/sample derived from animals or human and thus does not require an ethical approval.
Author Contributions
Conceptualization, Jie Xu; methodology, Yanfang Yang; formal analysis, Qianqian Li; investigation, Jie Xu; data curation, Ziyi Dong; writing—original draft preparation, Yaoyang Guo; writing—review and editing, Jie Xu.; supervision, Zhansheng Jiang; project administration, Jie Xu; funding acquisition, Zhansheng Jiang. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Tianjin Municipal Health Commission (grant number 2024013).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
