Abstract
Keywords
Introduction
Essential oils (EOs) are complex mixture of natural volatile organic compounds, which are highly odorous and hydrophobic in nature. They can be obtained from the various parts of plants following different extraction methods. Monoterpenoids and sesquiterpenoids are the dominant categories of chemical constituent possessing the isoprene as the building unit. Due to strong fragrance and excellent medicinal properties, they have been widely used in food manufacturing as fragrances, flavors, and preservatives; in the cosmetic industry, such as skin products and perfumes; as additives to feedstuffs, eg, growth promoters and antioxidants, and as medicines in the pharmaceutical industry, eg, medicines.1,2 A plethora of research has explained about strong antimicrobial, antiseptic, antiviral, antioxidant, anti-parasitic, anti-protozoal, antifungal, anti-inflammatory and insecticidal activities of such essential oils. 3 But, in the recent years, the utilization of synthetic fragrance and flavoring agents, preservatives, and antimicrobial agents has rapidly expanded with little appreciation of their possible health risks to consumers. These synthetic substances, however, are extremely dangerous and harmful to health when consumed in excess of the permitted level. Therefore, essential oils may serve as rational substitutes for ensuring food safety, while maintaining both nutritional content and food quality, in addition to removing human health risks. Since many EOs have a predominance of oxygenated monoterpenoids, which have demonstrated potential antimicrobial properties, they are currently used as flavoring agents. 4
Elsholtzia is a genus including about 42 species under the family Lamiaceae in the world. 5 They are widely distributed in East Asia, Africa, North America, and Europe especially, in China, Korea, Japan, India and Nepal. It has been reported that at least 33 plant species of this genus are found in China. 6 Similarly, at least 12 plant species of this genus are reported in Nepal in accordance to Annotated Checklist of the Flowering plants of Nepal. 7 Among them, Elsholtzia strobilifera (Benth.) Benth and Elsholtzia blanda (Benth.) Benth are taken under the consideration in this research, because they are growing in their natural habitats in Nepal. Most of the Elsholtzia species have a distribution at altitudes between 1000 m and 3000 m above sea level, and grow in hilly grassland, waste areas, forests, thickets or valleys in the warm areas. The plants belonging to the Elsholtzia genus are usually rich in essential oil-bearing species, which are commonly used as domestic folk medicine, and show the remarkable antimicrobial, anti-inflammation, fever relieving, analgesic, antioxidant potential, antitumor and anticancer properties. 8
Elsholtzia strobilifera, also known as Ban Bawari (in Nepali), is an annual aromatic herb distributed at altitudes up to 3700 m. It is commonly found in hills, grasslands, open areas in forests, thicket margins, and valleys of Sichuan, Taiwan, Xizang, Yunnan, India, and Nepal. It is one of the common essential oil-bearing plants growing in the wild stage. 9 This plant species has been reported to have several medicinal values. Earlier studies showed that the EO of E. strobilifera exhibits effective antibacterial and antifungal activity. Elsholtzia blanda, also known as Ban Silam (in Nepali), is an aromatic shrub or subshrub distributed at an altitude up to 1500 m. It is commonly found in East Asia, Africa, North America and European countries, and is more prevalent in China, Korea, Japan, Nepal, and India. 8 Traditionally, the aerial parts of this species are used to treat fever, inflammation, skin diseases, and cholera. Additionally, the plant is important in traditional Chinese medicine (TMC) for treatment of dysentery, acute enteritis, nephritis, and hepatitis.
A survey of the literature has revealed that, this is the first extensive work on the chemical analyses, along with enantiomeric composition, other biological activities and cytotoxicity of EOs of either plant species locally available in Nepal or elsewhere to the best of our knowledge. The aim of the present study, therefore was to identify the chemical components of E. strobilifera and E. blanda EOs from Nepal, to determine the distribution of enantiomers of chiral terpenoids, to evaluate their in-vitro antibacterial activity, antifungal efficacy, antioxidant activity, and cytotoxicity. This study has generated some insightful preliminary data for further study and uses of two Elsholtzia species.
Results and Discussion
Isolation and Yields of Essential Oils
E. strobilifera and E. blanda plant samples collected from two geographical locations of Nepal were hydrodistilled in the 1:5 ratio of plant materials (aerial parts of herbs) to water using Clevenger apparatus. The yields of E. strobilifera and E. blanda essential oils were 0.9 ± 0.1% (v/w) and 0.9 ± 0.2% (v/w), respectively. The EO yields were found to be relatively comparable as reported previously. 10 The differences in EO yields may be due to various reasons, like geographical origin, seasonal variation, as well as differences in extraction methods (eg, temperature, and extraction time). 11 The Elsholtzia EOs were characterized by pleasant odor, transparent to slightly viscous liquids and pale yellow in color.
Chemical Composition of Essential Oils
The hydro-distillation of the aerial parts of E. strobilifera and E. blanda resulted a pale-yellow oils with characteristic pleasant aromas. The GC–MS analysis of EOs has resulted in the identification of 55 compounds representing 98.2% of total E. strobilifera essential oil and 41 compounds representing 94.3% of total E. blanda essential oil, respectively (Table 1).
Chemical Composition of the Essential oil from the Aerial Parts of Elsholtzia strobilifera and Elsholtzia blanda.

Note: RI = retention index values calculated with respect to a series of n-alkanes (C8–C40) on a ZB-5 ms column; components are listed in order increasing RI values.
Major compounds are indicated in bold.
In the essential oil from E. strobilifera, pinocarvone (40.7%) and β-pinene (10.0%) were the most prominent compounds, followed by cis-pinocamphone (7.9%), α-humulene (7.3%), sabinene (4.7%), humulene epoxide II (2.3%), and verbenone (2.2%) as other important identified components. Among all the major classes of terpenoids (monoterpene hydrocarbons, oxygenated monoterpenoids, sesquiterpene hydrocarbons, and oxygenated sesquiterpenoids) in this EO, oxygenated monoterpenoids were the most dominant class with nearly 57.5% of the total EOs. A study carried by Murari and Mathela identified the presence of β-pinene (12.1%), diterpene (10.4%), and carvone (10.5%) as important components in E. strobilifera essential oil. 12 Whereas, in another study carried out earlier showed that E. strobilifera growing in India contained pinocarvone (51.9%) and β-pinene (9.7%) as the main compounds along with yields of 0.3%. 13 In contrast to the above results, a subsequent report of E. strobilifera EO identified the presence of pinocarvone (7.4%), geranial (citral b) (29.9%), neral (citral a) (18.3%), humulene epoxide II (6.3%), and trans-calamenene (3.9%) along with other minor constituents. 8 However, the present study has shown the dominance of pinocarvone and β-pinene along with the identification of 55 compounds, which is highly prominent result as compared to the previous study9,12 and these results are in close agreement with respect to the major components reported by Bisht. 13
The essential oil from E. blanda was found to contain dihydrotagetone (49.1%) and (Z)-tagetone (15.6%) as the most dominant compounds, followed by (E)-tagetone (8%), (Z)-β-ocimene (3.7%), benzaldehyde (2.9%), and β-caryophyllene (2.8%) as other detected compounds in smaller amounts. Regarding the major classes of terpenes in the EOs, the oxygenated monoterpenoids were the most dominant class with nearly 74.2% of the total EOs. The EO of E. blanda from Vietnam was composed predominantly of monoterpenoids, with camphor (25.1%), camphene (22.6%), α-pinene (11.5%), and 1,8-cineole (9.9%) as the four most abundant constituents. 14 The E. blanda EO from India was characterized with geranyl acetate (71.4%), linalool (5.3%), geraniol (3.8%), (E)-β-ocimene (2.9%), β-caryophyllene (1.8%), α-bergamotene (1.7%), (Z)-β-ocimene (1.4%), acetophenone (1.1%), and linalyl acetate (1.1%) as the most abundant compounds. 15 Similarly, two types of E. blanda essential oil from India were reported with linalool as the major component in both the inflorescence parts (77.3%-80.2%) and vegetative aerial parts (57.9%-62.9%). 16 Similarly, the E. blanda EO from Vietnam was found to contain linalool as the dominant component. 17 Some of these previous studies on the composition of E. blanda EOs revealed that E. blanda presents great variability in the concentrations of the major chemical compounds depending on their availability in different countries. Moreover, E. blanda EOs from Nepal exhibited dihydrotagetone and (Z)-tagetone as major constituents which were not reported in the previous studies. The reason behind this might be attributed to the environmental, edaphic, and climatic conditions, because the plant species may alter the biosynthetic pathways of secondary metabolites productions. The most abundant components were oxygenated monoterpenoids and monoterpene hydrocarbon followed by sesquiterpene hydrocarbon, which were more similar patterns those of other species of Elsholtzia genus as reported previously. 18
Enantiomeric Distribution Analysis
The chiral GC–MS is one of the most powerful tools for the authentication and standardization of essential oils. The chiral GC–MS analysis allowed for the identification and percentage composition of various chiral components in both E. strobilifera and E. blanda EOs (Table 2). The EOs of E. strobilifera and E. blanda were found to have 12 and 7 chiral terpenoid components in total for their enantiomeric distributions, respectively. The determination of enantiomeric composition of chiral terpenoids is an effective tool for identification and authentication of EOs. The EOs obtained from different plants may be adulterated, due to the addition of several carrier oils and other foreign components.19,20 However, geographical origin and distillation methods generally do not affect the enantiomeric ratios of chiral components present in the EOs. This study is the first to present the Elsholtzia species of Nepalese origin. The (+)- and (–)-enantiomers have distinctly different biological and organoleptic properties although they have the same physicochemical property. In this study, all the 12 chiral terpenoids detected in E. strobilifera essential oil were levorotatory, whereas, α-pinene detected in E. blanda essential oil was dextrorotatory and other 6 chiral terpenoids were levorotatory. Similarly, (–)-α-thujene was the predominant enantiomer (97.1%) in E. strobilifera essential oil and, (–)-sabinene was another dominant enantiomer (76.6%) in E. blanda essential oil. Furthermore, the levorotatory (–)-β-caryophyllene and (–)-germacrene D were detected as enantiomerically pure form (100%) in both essential oils.
Enantiomeric Distributions of Chiral Compounds in EOs of E. strobilifera and E. blanda.
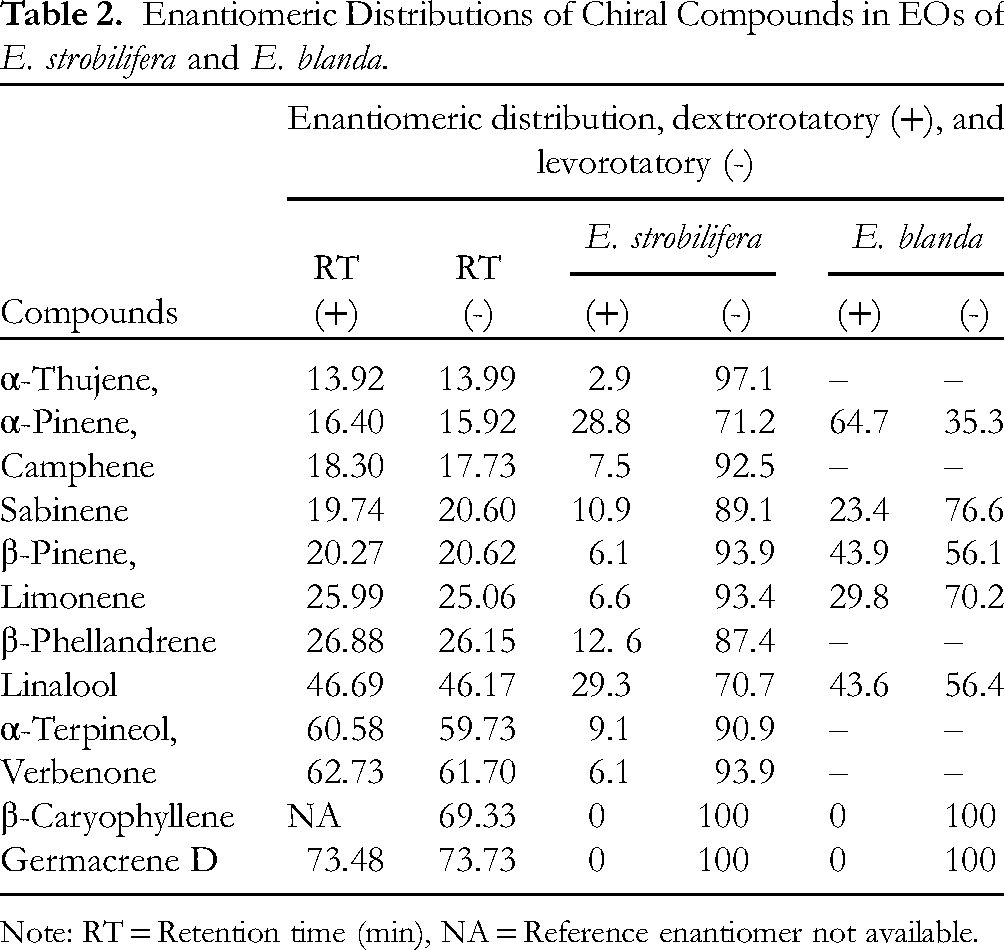
Note: RT = Retention time (min), NA = Reference enantiomer not available.
Through the extensive literature review, there are apparently no elaborative previous studies regarding enantiomeric distribution of chiral compounds for Elsholtzia species. As far as we are aware, this study covers new information for enantiomeric composition of these essential oils. Therefore, this study highlights on the volatile terpenoids as the chiral components of these oils for the first time.
In-Vitro Antimicrobial Activity
The antimicrobial activities of Elsholtzia EOs were evaluated against six microorganisms. Based on the results, E. strobilifera and E. blanda were found to have moderate activity against all microbial stains as compared to positive control in terms of minimum inhibitory concentration (MICs) but their efficacies showed some degree of variation (Table 3). The E. strobilifera EO showed very good antibacterial activity against S. epidermidis with MIC value of 156.3 µg/mL. While, E. blanda exhibited better activity against S. aureus with the MIC value of 156.3 µg/mL. However, both EOs demonstrated weaker antibacterial activities than that of the positive control, gentamicin (MIC < 19.5 µg/mL). On the other hand, both essential oils displayed active antifungal activity against A. niger with MIC value of 156.3 µg/mL, while, these EOs showed reduced antifungal activity against C. albicans and A. fumigatus with MIC value of 312.5 µg/mL. However, the MIC values of two EOs against fungal strains are greater than that of the positive control, amphotericin B (MIC < 19.5 µg/mL) indicating their weaker activity. Nevertheless, it has been suggested that essential oils showing MIC values < 500 μg should be considered to exhibit “strong activity.” 21
Minimum Inhibitory Concentrations (MICs) of E. strobilifera and E. blanda Essential Oils Against Tested Bacterial and Fungal Strains.
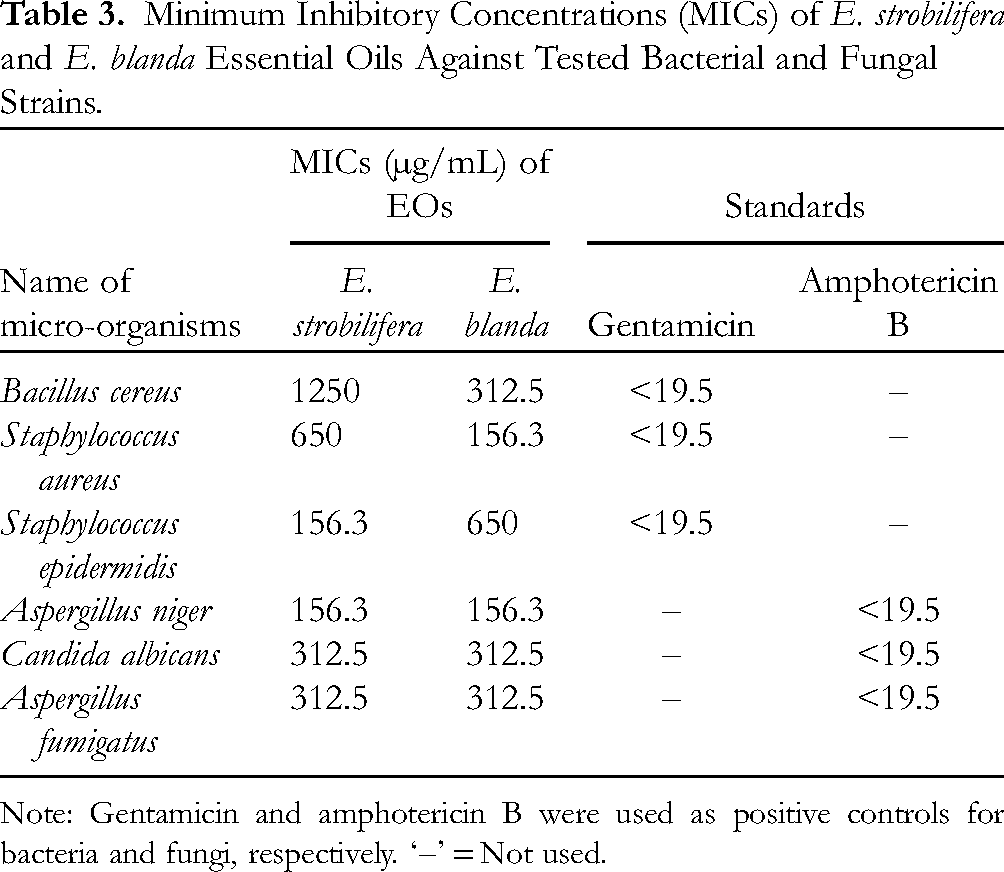
Note: Gentamicin and amphotericin B were used as positive controls for bacteria and fungi, respectively. ‘–’ = Not used.
A previous study had reported that E. blanda EO exhibited good antibacterial efficacy against S. aureus with inhibition zone of 20.6 ± 1.0 mm as compared to standard (21.0 ± 0.1 mm for chloramphenicol). 14 The levels of dihydrotagetones and tagetones found in E. blanda EO likely account for the observed antimicrobial activity. 22 A very limited previous study showed that the EOs from some Elsholtzia species have shown prominent inhibitory activities against several bacteria. 23 The antimicrobial activities of both E. strobilifera and E. blanda EOs could also be related to their relatively high concentrations of oxygenated monoterpenoids, which constitute about 57.5% and 74.1% of the EOs, respectively. However, it is very important to know that both synergistic and antagonistic effect of the constituents resulting in the whole EO extracts a different activity from the sum of the individual components. Moreover, the antimicrobial activity of both EOs could also be related to their major content in oxygenated monoterpenoids, which constitute about 57.5% and 74.1% of the EOs of E. strobilifera and E. blanda. These results showed that antifungal activity of EOs were more prominent than that of antibacterial activity, which were also reported by some previous studies for many other essential oils.24,25 These results would indicate that the EOs from Elsholtzia spp. can also be used as natural preservatives in food against foodborne pathogens and food spoilage organisms.
In-Vitro Cytotoxicity Evaluation
Cell counting kit-8 (WST-8/CCK8) is a convenient and robust way of performing a cell viability assay for screening the cytotoxic activity. 26 The viabilities of NIH-3T3 and MCF-7 cell lines, after the incubation with different concentrations of the two Eos, were found to have significant effects. The results showed that the incubation with various concentrations of EOs affected the viability of NIH-3T3 mouse embryonic fibroblasts and MCF-7 human breast cancer cell lines as displayed by a plot of cell viability (%) versus logarithm concentrations (µg/mL) of the EO samples (Figures 1 and 2). The samples showed the cytotoxic effects on NIH-3T3 and MCF-7 cell lines in a dose-dependent patterns and cytotoxicity was represented as the IC50 values, the concentrations at which 50% of cells were killed in DMEM. This is the first report for these EOs and their cytotoxicity in terms of IC50 values and are presented in Table 4. However, EOs may also enhance cell proliferation at lower concentration. This tendency can be expressed in terms of cell index (%) and some extracts can kill the cells at very high concentration (100 µg/mL), but activate cell growth at low concentration. So, their application as anticancer agents would be insignificant. The cell index for two EOs was represented from a graph of cell index (%) versus the concentration (µM) for cell proliferation. Both EOs have weak proliferation effect at low concentration against the MCF-7 cell line (Figure 3).

Graph representing % cell survival versus logarithm of the concentration (µg/mL) for cytotoxicity of (a) E. strobilifera, (b) E. blanda and (c) standard gemcitabine (Gem-1 and Gem-2) against NIH-3T3 cell line.

Graph representing % cell survival versus logarithm of the concentration (µg/mL) for cytotoxicity of (a) E. strobilifera, (b) E. blandaand (c) standard gemcitabine against MCF-7 cell line
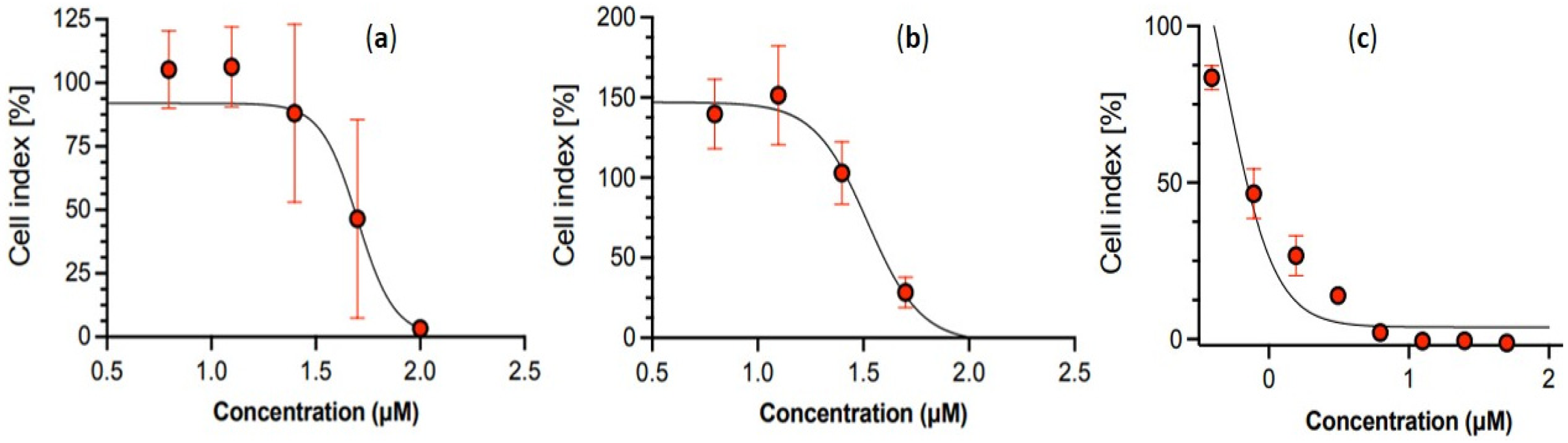
Graph representing cell index (%) versus the concentration (µM) for cell proliferation of (a) E. strobilifera EO, (b) E. blanda EO and (c) standard gemcitabine against human breast cancer cell line (MCF-7).
Cytotoxic Activity of E. strobilifera, E. blanda and Standard Against NIH-3T3 and MCF-7 Cell Lines.

Note: IC50 is median inhibitory concentration.
Both E. strobilifera and E. blanda EO exhibited strong cytotoxic activities against the 3T3 cell lines with IC50 values of 20.2 µg/mL and 35.3 µg/mL, respectively. On the other hand, E. strobilifera and E. blanda EO also displayed relatively strong cytotoxic activities against the MCF-7 cancer cell line with IC50 values of 33.1 µg/mL and 50.1 µg/mL. However, both EO samples demonstrated weaker anticancer activity than that of positive control gemcitabine, with IC50 values of 0.4977 µg/mL and 0.5175 µg/mL for MCF-7 and 3T3 cell lines, respectively. Actually, cytotoxic activities of these two Elsholtzia essential oils are stronger as compared to those of other Lamiaceae species. 27 According to the published guidelines, 28 IC50 value 10 to 100 µg/mL represents promising inhibitory potential against cancer cells, indicating the potent cytotoxic of activities of Elsholtzia EOs. The cytotoxicity of the EOs may be attributed due to some type of synergism of minor components with the other active/major compounds. There are very few reports on the cytotoxicity of Lamiaceae EOs and there are apparently no previous reports on the cytotoxicities of Elsholtzia essential oils.
Antioxidant Activity
DPPH Activity
The antioxidant activity of E. strobilifera and E. blanda EOs was determined by using the DPPH (2,2-diphenyl-1-picrylhydrazyl) radical-scavenging assay using both ascorbic acid and butylated hydroxyltoluene (BHT) as positive controls (Table 5). A standard criterion for measuring the radical-scavenging activity of EO samples is the IC50 value, which represents the concentration of antioxidant needed to reduce the initial DPPH concentration by 50%. This study revealed that E. strobilifera and E. blanda EOs exhibited moderate DPPH free radical-scavenging activities, with IC50 values of 188.8 ± 0.7 μg/mL and 452.8 ± 0.9 μg/mL, respectively. Here, E. strobilifera EO showed slightly stronger antioxidant activity than that of E. blanda EO. However, the antioxidant activities of both EO samples were lower than both ascorbic acid (6.4 ± 0.3 μg/mL) and BHT (12.5 ± 0.1 μg/mL). Many studies have demonstrated the antioxidant properties of EOs from Lamiaceae species. But no previous studies are reported in the literature regarding DPPH radical-scavenging capacity of E. strobilifera and E. blanda essential oils. Since both EOs are dominated by oxygenated monoterpenoids (ketones class), like, pinocarvone in E. strobilifera and dihydrotagetone in E. blanda, the antioxidant activity may be due to mono and sesquiterpenoids of different major classes of compounds in EOs, such as alcohols, ethers, aldehydes, ketones, epoxides or esters. 29 So, observed moderate radical-scavenging activity may be due to the higher activity of the minor components or synergism of the components. However, several studies have shown that the antioxidant activity of the essential oils themselves show the better radical-scavenging capacity than the individual components, which suggests the possible synergistic interactions between different EO components. 30
Antioxidant Activity of E. strobilifera EO, E. blanda EOs, BHT and Ascorbic Acid (Standard).

Note: Values are mean ± standard deviations from three experiments (n = 3).
Ferric-Reducing Antioxidant Power (FRAP)
In order to further assess of the antioxidant activity of the EOs, the total reducing power was investigated using the FRAP assay. Reducing agents transform the Fe3+/ferricyanide complex to the ferrous (Fe2+) form, resulting in an increase in absorbance at 700 nm. The results showed that there were increasing Fe2+ concentration with increase in the concentration of EO samples. The results are expressed in terms of effective concentration (EC50), which are listed in Table 5. The lower the EC50 values, the greater the antioxidant capacity of the EO to convert ferric to the ferrous ion form.
From the analysis, the EO of E. strobilifera demonstrated higher antioxidant ability, with an EC50 value of 348.0 ± 0.1 µg/mL, than that of E. blanda EO, with an EC50 value of 504.7 ± 0.1 µg/mL. On the other hand, both samples showed lower EC50 values as compared to the standard positive control, ascorbic acid, at 177.4 ± 0.1 µg/mL and BHT, at 304.1 ± 0.1 µg/mL. However, E. strobilifera EO showed relatively better reducing property. This reducing property could presumably be due to either the hydrogen atom donating ability or single electron transfer from EO constituents. 31 No previous studies have been reported regarding antioxidant potential of these essential oils in terms of effective concentration (EC50).
Conclusions
In summary, the volatile compounds, chiral terpenoids, antimicrobial potential, cytotoxicity, and antioxidant power of two Elsholtzia essential oils from Nepal have been thoroughly investigated for the first time. The findings showed that the main volatile organic components in both EOs were oxygenated monoterpenoids. Because of these bioactive constituents, EOs were therefore credited with some notable antibacterial and cytotoxic effects, while they only had mild antioxidant activity. The chiral GC–MS analysis indicated that there are some variations in the enantiomeric composition of chiral terpenoids in Elsholtzia essential oils, which could be an important marker for the identification and authentication of these essential oils. Furthermore, because of their noteworthy biological activity and pleasing smells, Elsholtzia EOs may be a viable natural complementary element in foods, perfumes, medicines, and formulations as well. However, the toxicological effects of these EOs must be studied to guarantee their safety and beneficial outcomes for further utilization. Although E. strobilifera and E. blanda, two essential oils of Nepalese origin, have been tested for their ability to inhibit cell proliferation in NIH-3T3 and MCF-7 cell lines, the results of these tests stimulate more research on bioactive chemicals.
Experimental
Collection of Plant Materials
The plant samples of E. strobilifera and E. blanda were collected in October 2021, from Barpak, Gorkha at an elevation of 1808 m with latitude 28°12′44.4″N and longitude 84°44′36.3″E, and in November 2021, from Machchhegaun, Kathmandu at an elevation of 1662 m with latitude 27°39′16.1″N and longitude 85°14′53.2″E during flowering stage. Both plants were collected from wild habitat, air dried at room temperature, and stored. Plants were identified out by taxonomist Ms. Rita Chhetri (Senior Research officer), National Herbarium and Plant Laboratories (KATH), Lalitpur, Government of Nepal and the plant specimens were also deposited at KATH. The herbarium voucher specimen numbers are GKH-02 for E. strobilifera and KTM-10 for E. blanda, respectively.
Extraction and Isolation of Essential Oils
The EOs were obtained by hydro-distillation using a Clevenger-type apparatus 32 for 3 h from 100 g (n = 3) following the standard protocol.33,34 The aerial parts of plant material and water in 1:5 ratios (w/v) were taken during essential oil extraction process. The essential oil thus obtained was dried with anhydrous sodium sulfate and was stored in vials at 4 °C until for further analysis and screening. The yield was calculated based on the volume to weight ratio.
Gas Chromatography–Mass Spectrometry Analysis
The GC–MS analysis of both essential oil samples was as previously described. 35 Identification of the EOs components was carried out by comparison of the mass spectral fragmentation patterns (over 80% similarity match) as well as the comparison of the retention indices, calculated using a homologous series of n-alkanes (C8–C40), with those reported in the literature, 36 and the Aromatic Plant Research Center's own library using the LabSolutions GC–MS solution software version 4.45 (Shimadzu Scientific Instruments). 37 Chiral GC–MS was carried out as previously described. 38 The enantiomers were identified by comparing retention times and mass spectral fragmentation patterns with authentic samples acquired from Sigma-Aldrich, and the enantiomeric ratios were calculated from the peak areas.
Antimicrobial Activity
The in-vitro antimicrobial activities of two EO samples were evaluated in terms of minimum inhibitory concentration (MIC) using micro-broth dilution technique on the bacteria Bacillus cereus (ATCC 14579), Staphylococcus aureus (ATCC 29213), and Staphylococcus epidermidis (ATCC 14990), and fungal strains Aspergillus niger (ATCC 16888), Candida albicans (ATCC 18804) and Aspergillus fumigatus (ATCC 96918) as previously reported.39,40
Cytotoxicity Assay of Essential Oils against Cancer Cell Lines
The cytotoxicity assay against 3T3 (mouse embryonic fibroblast) and MCF-7 (human breast ductal carcinoma) cell lines was carried out. The cell viability in the presence or absence of essential oils was determined using a Cell Counting Kit-8 kit (Dojindo Molecular Technologies, Inc.). The 3T3 and MCF-7cell line (https://www.atcc.org/products/crl-1658) were a kind gift from Prof. Shiro Watanabe and was maintained in standard Dulbecco's modified Eagle's medium (DMEM) with 10% fetal bovine serum (FBS) supplemented with 0.1% NaHCO3% and 1% antibiotic–antimycotic solution. For the cytotoxicity experiments, exponentially growing cells were harvested and plated in 96-well plates (1 × 104/well) in DMEM at 37 °C under humidified 5% CO2% and 95% air for 24 h. After the cells were washed with phosphate-buffered saline (PBS), the medium was changed to serially diluted test samples in DMEM, with the control and blank in each plate. After 72 h of incubation, cells were washed twice with PBS, and 100 μL of DMEM containing 10% WST-8 cell counting kit solution was added to each well. After a 3-h incubation, the absorbance at 450 nm was measured using a MultiscanSkyHigh plate reader (Thermo Fisher Scientific). Cell viability was calculated from the mean values from three wells using the following equation:
Here, only active samples were generally taken just before adding CCK-8 during experiment. Morphological assessment was evaluated just before adding CCK-8. Advanced judgment was carried through using different concentrations for samples with percentage inhibition of 50% and above, to calculate the IC50 (median inhibitory concentration). The cell growth suppression rates are depicted as IC50. Cell viability can be calculated using the ratio of total live/total cells (live and dead). Staining also facilitates the visualization of overall cell morphology. Here, EVOS FL (10 × magnification) was used for this study. The term cell index (%) was also used in order to show the capability of EO extracts to kill the cells at very high concentration (100 µg/mL), but activate cancer cell growth at low concentration. Therefore, its application as an anticancer agent would be minimal.
Antioxidant Activity
DPPH Free Radical-Scavenging Activity (DPPH)
The free radical-scavenging activity of the two EO samples was determined by using DPPH as previously reported. 41 The IC50 (µg/mL) value (concentration of sample required to scavenge 50% of free radicals) was calculated by using a non-linear regression model. Ascorbic acid and BHT were used as positive control. The experiments were conducted in triplicate.
Ferric-Reducing Antioxidant Power Assay (FRAP)
The FRAP assay was used to determine the antioxidant capacities of EO samples as reported previously. 42 The assays were carried out in triplicate, and the results expressed as mean ± standard deviations. Finally, the mean of absorbance values was plotted against the concentration values. Increased absorbance of the reaction mixture indicated the increased reducing power. The results were expressed as effective concentrations (EC50 value in μg/mL) when the absorbance was 0.5 at 700 nm and compared with ascorbic acid and BHT as positive controls.
Statistical Analysis
Statistical analysis of the data obtained were processed and analyzed using Microsoft Excel and OriginPro2016 64Bit (Origin version 9.3, OriginLab Corporation). The results of the antioxidant activity and cytotoxicity of EOs were expressed as the mean of three replicates ± standard deviation (SD).
Footnotes
Acknowledgments
The authors are thankful to the Aromatic Plant Research Center, Lehi, UT, USA, for providing GC–MS and chiral GC–MS data and other financial support to publish this article. The authors are also thankful to Division of Natural Drug Discovery, Institute of Natural Medicine, University of Toyama, Toyama, Japan for supporting the analysis. The authors would like to express their gratitude to the Department of Chemical Science and Engineering, and the Department of Pharmacy at Kathmandu University, Nepal, for the technical assistance. We would also like to thank Mr Sagar Sharma and Ms. Aparna Paudel for their constructive suggestions and support.
Author Contributions
Conceptualization, P.N.P, R. and P.S.; methodology, P.N.P., R.G., P.S. and W.N.S.; validation, P.S., R.G., W.N.S.; formal analysis, P.N.P., R.G., P.S.,S.A,.S.W. and J.M.; investigation, P.N.P., R.G., P.S. S.A., A.A., J.M. and M.P.; data curation, R.G. and P.S.; writing—original draft preparation, P.N.P., A.A. and M.P.; writing—review and editing, P.N.P., R.G., W.N.S. and R.S.; supervision, R.G., P.S. and W.N.S; antimicrobial activities, P.N.P. and P.S. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
Data from this investigation are available in the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Funding
This research was funded by University Grants Commission, Nepal, FRG-78/79-S &T-02.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Sample Availability
Samples of the compounds are available from the authors.
