Abstract
Introduction
Humans are affected by various allergic conditions, such as hay fever, bronchial asthma, and atopic dermatitis. The most well-known mechanism for the onset of allergies, such as hay fever, has long been attributed to type I hypersensitivity (immediate-type hypersensitivity), which involves immunoglobulin E (IgE). 1 Anti-allergic drugs suppress symptoms by disrupting a part of their mechanism of action. However, there are concerns regarding side effects from such medications, such as induced drowsiness and reduced quality of life. Therefore, there is a need to develop functional foods that can safely relieve allergic symptoms. Screening 6 edible plant extracts, we found that water-soluble coriander leaf extract showed an anti-allergic effect in mast cell degranulation assays using RBL-2H3 cells. 2
Coriander (Coriandrum sativum L.) is an annual Apiaceae family plant widely cultivated in Africa, Europe, and Asia. It is commonly known as coriander in English and phakchai in Thai; the leaves and seeds are widely used as savory vegetables and seasoning.3,4 In addition to its use as a seasoning ingredient, it is used in folk medicine in diverse applications, including as a digestive stimulant, antibacterial agent, and for treating airway disorders (bronchitis and cough). In recent years, coriander extracts have shown various physiological activities, including antioxidant,5–7 antibacterial,8,9 and anti-inflammatory10,11 activities. In addition, exposure to coriander extract suppresses the expression of inflammatory mediators in RAW 264.7 cells of lipopolysaccharide-treated mice. 12 Phenolics and carotenoids in coriander are reported to have antioxidant activities. 4 Furthermore, the ethanolic extract of coriander, which contains isoquercetin and quercetin, showed hepatoprotective activity. 13 However, it is still unclear which compounds in coriander are responsible for other health functions.
A recent study showed the anti-allergic effects of water-soluble coriander leaf extract in both cultured cells and pollinosis mice. 2 Furthermore, 8 components have been identified in the methanol-eluate fraction obtained from the water-soluble fraction, which showed degranulation inhibitory activity in RBL-2H3 cells. 14 However, existing in coriander leaves in only small amounts, those components are insufficient to explain the anti-allergic effects of coriander leaves. Active ingredients are expected to present dominantly in the aqueous-eluate fraction and have not yet been isolated. Therefore, in the present study, we isolated the anti-allergic substances from the hot water extract of coriander, determined their structures, and evaluated their activity.
Results and Discussion
Inhibitory Effect of the H2O Extract of Coriander on Degranulation in RBL-2H3 Cells
The hot water extract of coriander (leaves, petioles, and stems) was fractionated using a polystyrene column (MCI gel CHP-20P) to obtain 4 fractions: Frs. 1-4. The H2O eluate fraction (Fr. 2), which was a large amount of all fractions, was separated by Sephadex LH-20 to yield 6 fractions, Frs. 2-1 to 2-6. The inhibitory effects on degranulation were evaluated using RBL-2H3 cells (Figure 1A and B). Frs. 2-1, 2-4, 2-5, and 2-6 showed degranulation inhibitory activity without cytotoxicity (Figure 1C and D); Fr. 2-5 showed the highest activity between 0.63 mg/mL and 2.5 mg/mL, and it was further purified. Fr. 2-5 was separated into 6 fractions using a µ-Bonda Pak C18 column (Frs. 2-5-1 to 2-5-6). All fractions showed degranulation inhibitory activity between 0.25 mg/mL and 1.0 mg/mL (Figure 2A and B). In Fr. 2-5-1, no major compound, other than sugars, was detected by thin-layer chromatography (TLC). Frs. 2-5-2 and 2-5-3 showed considerable cytotoxicity (Figure 2C). Conversely, Frs. 2-5-4, 2-5-5, and 2-5-6 showed no cytotoxicity (Figure 2D). Further analysis was performed using these fractions, which were purified via preparative high-performance liquid chromatography (HPLC) to isolate compounds
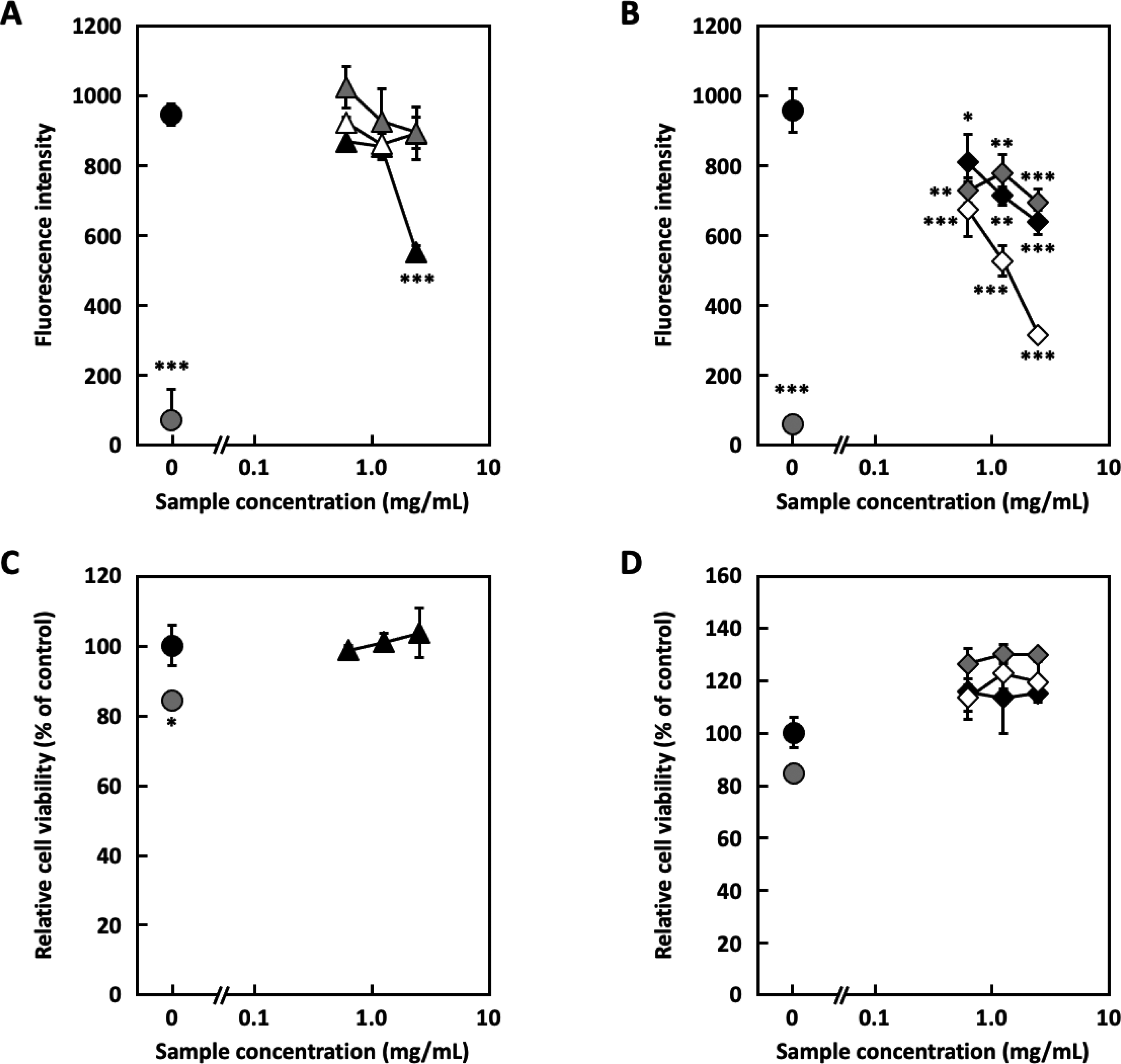
Degranulation inhibitory ability and cytotoxicity of the Sephadex LH-20 fractions in RBL-2H3 cells. (A) Degranulation inhibition of fractions of the Sephadex LH-20 fractions (Frs. 2-1∼2-3) in RBL-2H3 cells. (●: Blank, ●: Control, ▴: Fr. 2-1, △: Fr. 2-2, ▴: Fr. 2-3) (B) Degranulation inhibition activity of the Sephadex LH-20 fractions (Frs. 2-4∼2-6) in RBL-2H3 cells. (●: Blank, ●: Control, ◆: Fr. 2-4, ◇: Fr. 2-5, ◆: Fr. 2-6) (C) Cytotoxicity of the Sephadex LH-20 fractions (Frs. 2-1∼2-3) to RBL-2H3 cells. (●: Blank, ●: Control, ▴: Fr. 2-1) (D) Cytotoxicity of the Sephadex LH-20 fractions (Frs. 2-4∼2-6) to RBL-2H3 cells. (●: Blank, ●: Control, ◆: Fr. 2-4, ◇: Fr. 2-5, ◆: Fr. 2-6) The experiment was performed in triplicate, and data are expressed as mean ± SD (n = 3). Dunnett's test was used to assess the statistical significance of differences (*P < .05, **P < .01, and ***P < .001) compared with the control.
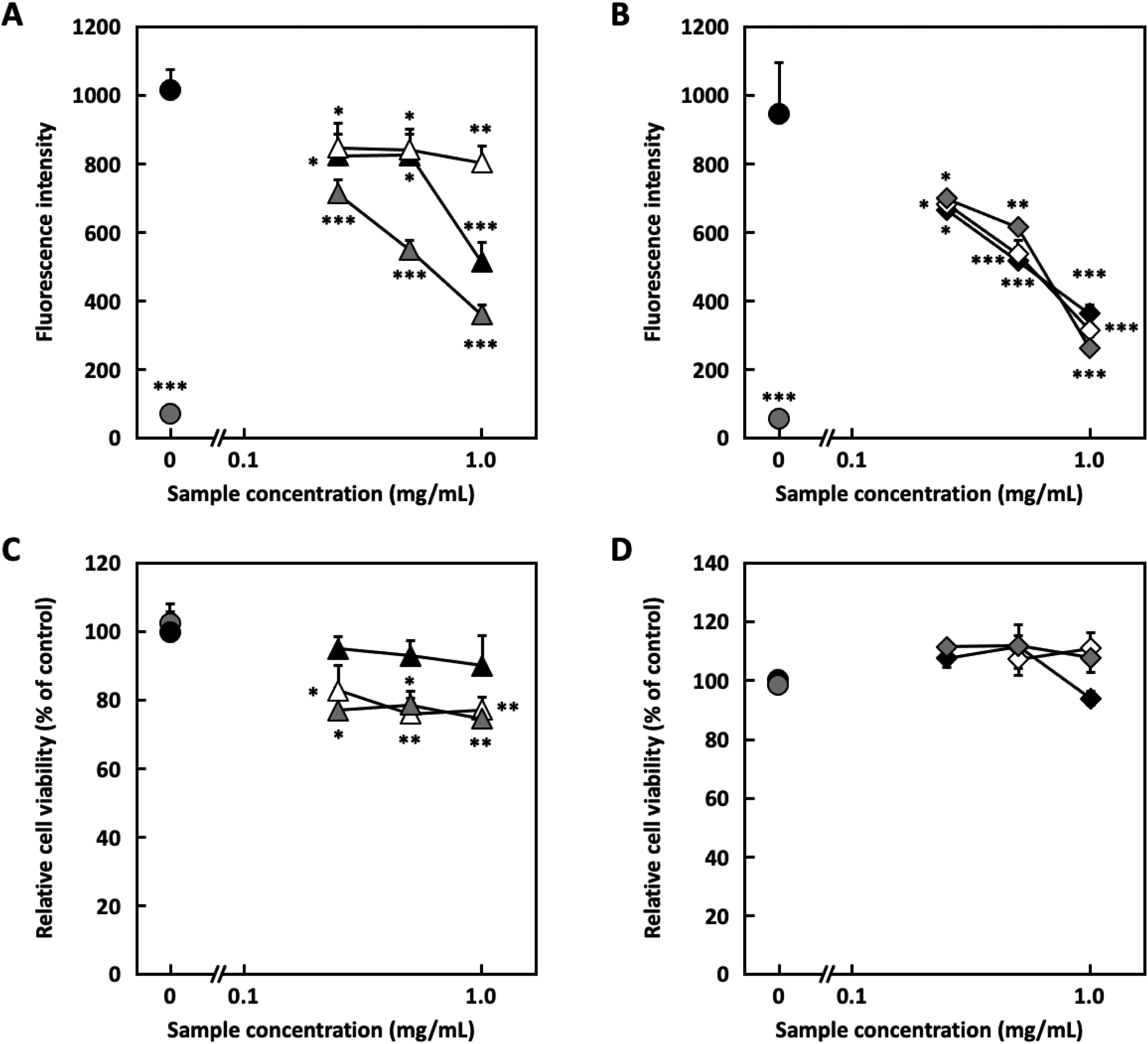
Degranulation inhibitory ability and cytotoxicity of the m-Bonda Pak C18 fractions in RBL-2H3 cells. (A) Degranulation inhibition of fractions of the m-Bonda Pak C18 (Frs. 2-5-1∼2-5-3) in RBL-2H3 cells. (●: Blank, ●: Control, ▴: Fr. 2-5-1, △: Fr. 2-5-2, ▴: Fr. 2-5-3) (B) Degranulation inhibition activity of the m-Bonda Pak C18 fraction (Frs. 2-5-4∼2-5-6) in RBL-2H3 cells. (●: Blank, ●: Control, ▪: Fr. 2-5-4, □: Fr. 2-5-5, ▪: Fr. 2-5-6) (C) Cytotoxicity of the m-Bonda Pak C18 (Frs. 2-5-1∼2-5-3) to RBL-2H3 cells. (●: Blank, ●: Control, ▴: Fr. 2-5-1, △: Fr. 2-5-2, ▴: Fr. 2-5-3) (D) Cytotoxicity of the m-Bonda Pak C18 (Frs. 2-5-4∼2-5-6) to RBL-2H3 cells. (●: Blank, ●: Control, ▪: Fr. 2-5-4, □: Fr. 2-5-5, ▪: 2. (A) Degranulation inhibition of compound 1 Fr. 2-5-6) Experiment was performed in triplicate, and data are expressed as mean ± SD (n = 3). Dunnett's test was used to assess the statistical significance of differences (*P < .05, **P < .01, and ***P < .001) compared with the control.

The structures of compounds
Structural Elucidation of Compounds 1 and 2
Compound

Significant heteronuclear multiple bond correlation (HMBC) correlations of compounds
1H-NMR Spectral Data of Compound

Abbreviation: NMR, nuclear magnetic resonance.
MeOH-d4
Acetone-d6
Acetone-d6, D2O (1:1) m: multiplet.
Compound

3D HPLC profile of the hot water extract of coriander. Column: Cosmosil AR-II (ϕ4.6 × 250 mm); Solvent: CH3CN in 0.1% TFA /H2O in 0.1% TFA, 10/90 (0 min) → 100/0 (30 min); Flow rate: 1.0 mL/min; Column temp.: 40 °C; Detector: PDA (200 nm → 400 nm); Extraction: 6.0 g of dry powders /150 mL hot water, 10 μL injection. Rt
1H-NMR Spectral Data of Compound
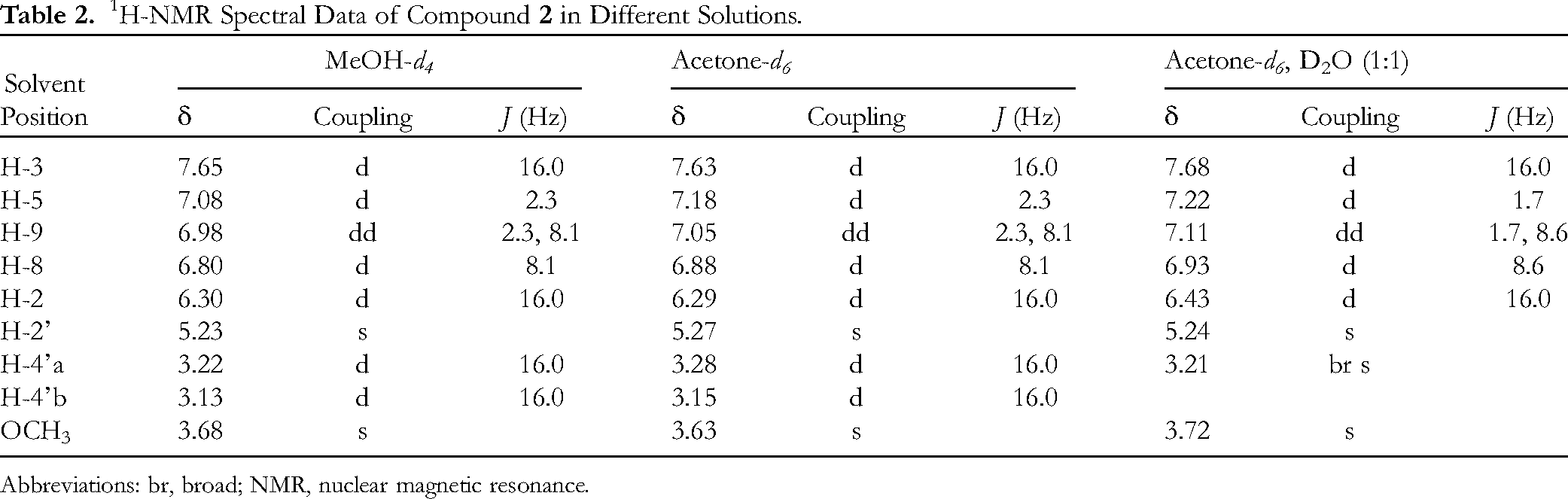
Abbreviations: br, broad; NMR, nuclear magnetic resonance.
Quantification of Compound 1
Since the peak of compound
Inhibitory Effect of Compounds 1 and 2 on Degranulation
β-Hexosaminidase is present in the granules of mast cells and basophils and is released with histamine when mast cells and basophils are activated.22–24 The β-hexosaminidase release assay has been used as an alternative to the histamine release assay in several studies.25,26 β-Hexosaminidase release assays were performed to evaluate the degranulation inhibitory activity of the 2 compounds. Compounds
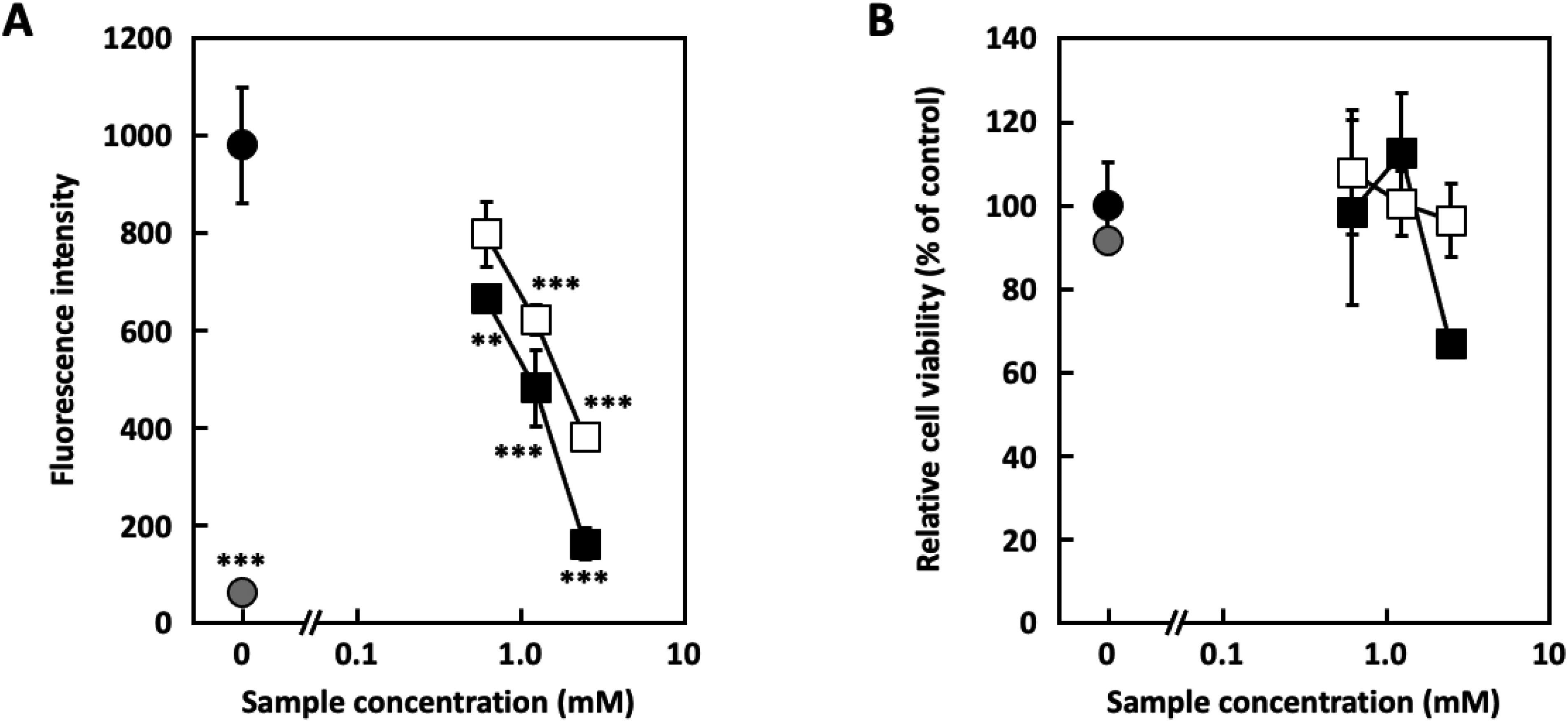
Degranulation inhibition and cytotoxicity of compound
A rudimentary structure-activity relationship study on degranulation activity was conducted using commercially available caffeic and hydroxycitric acid. Hydroxycitric acid was insoluble in DMSO and buffer and could not be examined. Caffeic acid inhibited degranulation (Figure 7), indicating that the caffeic acid structure of compounds

Degranulation inhibition of caffeic acid in RBL-2H3 cells. (●: Blank, ●: Control, 〇: Caffeic acid) The experiment was performed in triplicate. Data are expressed as mean ± SD (n = 3). Dunnett's test was used to assess the statistical significance of differences (**P < .01 and ***P < .001) compared with the control.
Previous studies on the water-soluble fraction of coriander reported that tryptophan, phenylalanine, dihydroxycoumarin glucoside, (3S)-3-methyl-6-hydroxyisocoumarin 8-O-β-D-glucopyranoside, quercetin glucuronide, and rutin contribute to the inhibition of degranulation.14 Although these components showed almost the same activity range compared to compounds
Conclusions
The hot water extract of coriander inhibited degranulation in RBL-2H3 cells. Compounds
Experimental
General Experimental Procedures
Electrospray ionization-mass spectrometric (MS) analysis was performed on a maXis II mass spectrometer (Bruker Co.). MeOH (Fujifilm Wako Pure Chemical Co.) was used as the solvent for LC/MS. MeOH in 5 mM dibutylammonium acetate (Ion-Pair Reagent for LC-MS; Tokyo Chemical Industry Co., Ltd) was added at a 0.1 mL/min flow rate. NMR spectra were recorded using a JEOL ECA 500 NMR spectrometer (JEOL). 1H-NMR spectra were recorded in MeOH-d4, Acetone-d6, and Acetone-d6, D2O (1:1) solutions. The 13C-NMR spectra were recorded in MeOH-d4 solution. The chemical shifts (δ) are reported in parts per million (ppm) and J values in Hz, using MeOH-d4 for 1H-NMR (3.31 ppm) and 13C-NMR (49.0 ppm) as an internal standard, and using Acetone-d6 for tetramethyl silane (0 ppm) as an internal standard. P-1020 (JASCO Co.) was used as the optical rotation measurement device. The UV spectrum was measured using a V-750 spectrophotometer (JASCO Co.). Preparative HPLC was performed using a SHIMADZU LC-20AT pump, SHIMADZU RID-20A detector, Sugai U-620 column heater, and column of X-Select CHS Prep C18 (5 mm, ϕ10.0 × 250 mm; Waters Co.) with a flow rate of 2.0 mL/min and column temperature of 40 °C. Flash chromatography was performed on a µ-Bonda Pak C18 column (Radial-Pak Cartridge ϕ25 × 100 mm × 2; Waters Co.). TLC was performed on pre-coated silica gel 60 F254 (Merck Ltd), and detection was achieved by spraying with 10% H2SO4 followed by heating. Column chromatography was performed on MCI gel CHP20P (Mitsubishi Chemical Co.) and Sephadex LH-20 columns (GE Healthcare Bioscience Co.).
HPLC analysis of the aqueous extract was performed using a photodiode array detector SPD-M10A, interface box SCL-10A, dual HPLC Pump LC-10AD, column oven CTO-10AC, and degasser DGU-14A (SHIMADZU Co.). Cosmosil AR-II (5 μm, 250 × 4.6 mm; Nacalai Tesque) was used as the HPLC column. The solvent comprised acetonitrile with 0.1% trifluoroacetic acid (TFA)/H2O with 0.1% TFA, used as follows: 10/90 (vol/vol) (0 min) → 100/0 (30 min) → 10/90 (31 min) → 10/90 (40 min). The flow rate was set to 1.0 mL/min. The column temperature was 40 °C. The detection wavelength was 200 to 400 nm.
Dulbecco's modified Eagle's medium (DMEM), penicillin, streptomycin, fetal bovine serum (FBS), bovine serum albumin (BSA), mouse anti-dinitrophenyl (DNP) monoclonal IgE, and DNP–human serum albumin (HSA) conjugates were purchased from Sigma-Aldrich (St. Louis). Unless otherwise noted, all other chemicals were purchased from Fujifilm Wako Pure Chemicals or Nacalai Tesque.
Plant Material
Coriandrum sativum L. (taxonomical form: CRUISER) was collected on November 23, 2019, from a farm in Gunma, Japan. Fresh coriander (leaves, petioles, and stems) was blanched in hot water at 80 °C for 20 s, drained, and then frozen at −40 °C. The frozen material was freeze-dried (FDU-1200 in a dry chamber, EYELA Co.) for 48 h. The freeze-dried material was ground in a hammer mill and sieved through a 48-mesh sieve. A voucher specimen (CS2019G) was deposited in the Natural Product Laboratory of the Faculty of Pharmaceutical Sciences, Sojo University.
Extraction and Isolation
To a powder of freeze-dried coriander (leaves, petioles, and stems) (48.59 g), distilled H2O (1.0 L) was added, followed by extraction using sonication (Ultrasonic Cleaner: US-105 (200 W), SND Co.) for 4 h (60 min × 4) at 50 °C. This process was performed 4 times. The mixed extracts were concentrated under reduced pressure to obtain the H2O extract (23.63 g). This was dissolved in H2O (150 mL) and heated at 100 °C for 10 min to evaluate its thermal stability. After cooling the solution to room temperature (20 °C-25 °C), it was subjected to separation using an MCI gel (ϕ50 × 160 mm) and successively eluted with H2O, 50% MeOH, and 100% MeOH (each 1.0 L) to yield 4 fractions. The first fraction (2.19 g) had passed through the MCI gel, the second fraction (18.99 g) was an eluate of H2O, the third fraction (1.04 g) was an eluate of 50% MeOH, and the fourth fraction (0.21 g) was an eluate of MeOH. Part of the second fraction (2.52 g) was loaded onto a Sephadex LH-20 column (ϕ30 × 1000 mm, eluted with 50% MeOH), collected into test tubes (each 20 mL), and divided based on the results of TLC observation to obtain 6 fractions: Fr. 2-1 to 2-6. The fraction relatively rich in aromatic compounds (Fr. 2-5, 367 mg) was applied to a µ-Bonda Pak C18 column [ϕ25 × 100 mm × 2, and eluted with a H2O-MeOH gradient (H2O containing 10%, 20%, and 30% MeOH; 150 mL of each gradient solution)] to yield 6 fractions (Frs. 2-5-1 to 2-5-6). The 30% MeOH eluate fractions, Frs. 2-5-4 to 2-5-6, were combined (75 mg) and purified using preparative HPLC [X-Select CHS Prep C18 (ϕ10 × 250 mm, eluting with a 40% MeOH in 0.1% TFA)] to yield compounds
Compound 1 [2S,3S-2-O-caffeoyl-hydroxycitric acid]
A pale yellow solid; [α]D25 = + 30.1° (c 0.17, H2O); UV λmax log ε 3.99 (331 nm); 1H NMR: see Table 1; 13C NMR δ 174.6 (C-5′), 173.2 (C-6′), 170.0 (C-1′), 167.9 (C-1), 149.8 (C-7), 148.5 (C-3), 146.8 (C-6), 127.6 (C-4), 123.4 (C-9), 116.6 (C-8), 115.3 (C-5), 113.7 (C-2), 77.4 (C-3′), 77.0 (C-2′), 41.0 (C-4′); HR-ESI-MS m/z 369.0460 [M - H]− (calculated for C15H13O11, 369.0463).
Compound 2 [2-O-caffeoyl-hydroxycitric acid 6′-O-methylester]
A pale yellow solid; [α]D25 = + 33.1° (c 0.28, H2O); UV λmax log ε 3.75 (332 nm); 1H NMR: see Table 2; 13C NMR δ 174.5 (C-5′), 171.7 (C-6′), 169.9 (C-1′), 167.8 (C-1), 149.9 (C-7), 148.5 (C-3), 146.8 (C-6), 127.6 (C-4), 123.4 (C-9), 116.6 (C-8), 115.3 (C-5), 113.7 (C-2), 77.4 (C-3′), 77.0 (C-2′), 52.4 (-OCH3), 41.2 (C-4′); HR-ESI-MS m/z 383.0615 [M - H]− (calculated for C16H15O11, 383.0620).
Quantification of Compound 1
Boiling H2O (15 mL) was added to a sample of freeze-dried coriander (1.0 g) following extraction via sonication for 1 h at 50 °C. The extraction was repeated 3 times, and the combined filtrate was mixed to a total volume of 50.0 mL. An aliquot of the sample (10 μL) was injected 5 times into the HPLC system. Compound
Cell Culture
Rat basophilic leukemia RBL-2H3 cells were obtained from the American Type Culture Collection and cultured in DMEM supplemented with 100 U/mL penicillin, 100 μg/mL streptomycin, and 10% FBS at 37 °C in a humidified 5% CO2 to 95% air atmosphere.
β-Hexosaminidase Release Assay
A β-hexosaminidase fluorescence assay kit (Cell Biolabs Inc.) was used in this study. RBL-2H3 cells suspended in DMEM containing 10% FBS and 50 ng/mL of anti-DNP IgE were seeded into a 96-well culture plate (Corning Inc.) at 4 × 104 cells/well and cultured for 24 h. After washing with modified Tyrode's (MT) buffer (135 mM NaCl, 20 mM HEPES, 5.6 mM glucose, 5 mM KCl, 1.8 mM CaCl2, 1 mM MgCl2, and 0.05% BSA, pH 7.4) 2 times, the anti-DNP IgE-sensitised cells were treated with 120 μL of MT buffer containing various concentrations of either the sample or distilled H2O as a blank and control for 10 min. Next, 10 μL of DNP–HSA solution diluted in MT buffer at a concentration of 0.625 μg/mL was added to each well, and the plate was incubated for 30 min. MT buffer (10 µL) was added to blank-treated cells. After incubation, 50 μL of the supernatant was transferred to a 96-well black-bottom microplate. Subsequently, 50 μL of the substrate was added to each well, and the plate was incubated at 37 °C for 15 min. After incubation, 100 μL of neutralization buffer was added to each well, and the absorbance was measured at an excitation wavelength of 340 nm and an emission wavelength of 465 nm using an Infinite 200 PRO plate reader (Tecan Group Ltd).
Measurement of Cell Viability
Cytotoxicity was evaluated using Cell Count Reagent (Nacalai Tesque) based on WST-8, following the manufacturer's instructions. Anti-DNP IgE-sensitized cells were treated with various concentrations of sample and stimulated with DNP-has, as described above. After washing with DMEM, fresh medium containing 5% Cell Count Reagent was added to each well, and the plate was incubated at 37 °C for 20 to 40 min. After incubation, the absorbance was measured at 450 nm using an iMark microplate reader (Bio-Rad Laboratories).
Statistical Analysis
Experiments were performed in triplicate, and the data are expressed as the mean ± standard error of mean. Statistical analyses were performed using GraphPad Prism version 8.43 (GraphPad Software). One-way analysis of variance followed by Dunnett's multiple comparison test was used to assess statistical significance. Values of *P < .05, **P < .01, and ***P < .001 were considered significant.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231183603 - Supplemental material for Isolation and Quantification of 2-O-Caffeoyl-hydroxycitric Acid, an Active Component in Hot Water Extracts of Coriander (Coriandrum sativum L.), Which Inhibits Degranulation of RBL-2H3 Cells
Supplemental material, sj-docx-1-npx-10.1177_1934578X231183603 for Isolation and Quantification of 2-O-Caffeoyl-hydroxycitric Acid, an Active Component in Hot Water Extracts of Coriander (Coriandrum sativum L.), Which Inhibits Degranulation of RBL-2H3 Cells by Tsuyoshi Ikeda, Takuya Sugahara, Honoka Kanamitsu, Keisuke Nakashima, Hiroyuki Onda, Nanami Yoshino, Kosuke Nishi and Momoko Ishida in Natural Product Communications
Footnotes
Acknowledgments
The authors express our appreciation to Dr Daisuke Nakano, Prof. Junei Kinjo, and Mr Hiroshi Hanazono of Fukuoka University for assistance with ESI TOF MS. The authors also sincerely thank Ms. Kasumi Gondo, Ms. Nanako Ishizaka, and Ms. Kaho Sakai of Sojo University for their collaboration in the extraction and isolation of the compounds.
Authors Contribution
Supervision, T. S.; conceptualization, T. I., T. S., H. O., and M. I.; data curation, H. K., K. N.; Investigation, T. I., H. K., K. N., and M. I.; Writing – original draft, T. I. and M. I.; Writing – review & editing, T. S., H. O., N. Y., and K. N.; Project administration, H. O. and N. Y. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
