Abstract
The current study was undertaken to isolate and identify anti-inflammatory and antiallergic active compounds in the ethyl acetate and butanol fractions of an ethanolic extract of the aerial parts of Stellera chamaejasme (SCAE). A new maltol glucoside, maltol 3-(6″-(2-(E)-butenoyl)-glucoside) and benzoic acid, daphnoretin (a coumarin), 6 flavonoids (apigenin, genkwanin, taxifolin, apigenin 7-O-glucoside, luteolin 7-O-sambubioside, genkwanin 5-O-glucoside), and 2 lignans (matairesinoside and lariciresinol) were isolated from SCAE by chromatographic separation. Their chemical structures were elucidated by analyzing spectroscopic data, including 1-dimensional and 2-dimensional nuclear magnetic resonance (Fig1). Maltol 3-(6″-(2-(E)-butenoyl)-glucoside) significantly inhibited β-hexosaminidase release by 85.5% in immunoglobulin E and dinitrophenyl/bovine serum albumin (DNP/BSA)-induced RBL-2H3 cells. Benzoic acid from S. chamaejasme inhibited β-hexosaminidase release (by 80.2%) in IgE and DNP/BSA-induced RBL-2H3 cells and interleukin-4 messenger ribonucleic acid expression (by 21.9% inhibition) in propidium iodide-induced RBL-2H3 cells.
Allergies are caused by hypersensitivity of the immune system due to allergen-induced activation of immunoglobulin E (IgE). 1 IgE-mediated allergies are classified as Type I hypersensitivities and include allergic conjunctivitis, allergic asthma, allergic rhinitis, and atopic dermatitis. 2,3 In particular, IgE is activated by binding to high-affinity IgE receptor (FcεRI) present on either mast cells or basophils. 4 Activated mast cells induce degranulation and secrete various types of autacoids (eg, β-hexosaminidase and histamine) and inflammatory cytokines (eg, interleukin [IL]-4 and IL-13), which are responsible for the Th2 allergic response. 5,6 Thus, inhibiting mast cell activation is viewed as an effective means of treating and preventing allergic diseases. Steroids and antihistamines are used to treat allergy symptoms, but their usages are accompanied by various side effects. 7 -9 Therefore, researchers are actively searching for plant-derived natural materials that are safe, have no side effects, and that inhibit allergic reactions.
Stellera chamaejasme L. (SCAE) is known as “Langdu” in Chinese Pharmacology and belongs to the family Thymelaeaceae. It is a traditional Chinese herb that is widely distributed in northern and southwestern China. 10 The roots of S. chamaejasme have been used from ancient times to treat scabies, tinea, stubborn skin ulcers, chronic tracheitis, and edema. 11 -13 However, recent pharmacological studies have shown that it possesses antiviral, antitumor, anti-inflammatory, anticancer, and immunomodulatory activities. 14 -17 Previous phytochemical studies have isolated and identified various compounds in S. chamaejasme, such as lignans, coumarins, diterpenes, and flavonoids,. 18,19 In the present study, we isolated a new maltol derivative, maltol 3-[6″-(2-(E)-butenoyl)-glucoside] and 10 other compounds from a 95% ethanol extract of the aerial parts of S. chamaejasme and investigated their anti-inflammatory and antiallergic effects by assessing their abilities to inhibit IL-4 expression and β-hexosaminidase release in RBL-2H3 mast cells (Figure 1).

Structures of 11 phenolic compounds isolated from Stellera chamaejasme.
Results and Discussion
Compound
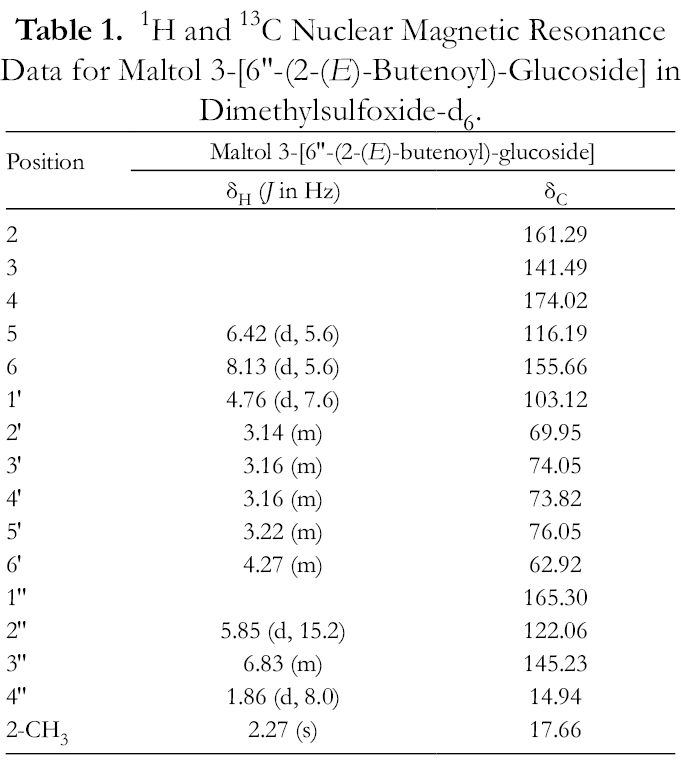
1H and 13C Nuclear Magnetic Resonance Data for Maltol 3-[6″-(2-(E)-Butenoyl)-Glucoside] in Dimethylsulfoxide-d6.

Selected 1H-1H correlated spectroscopy (COSY) and heteronuclear multiple bond correlations of maltol 3-[6″-(2-(E)-butenoyl)-glucoside] from Stellera chamaejasme.
A β-hexosaminidase assay was used to assess the antiallergic effects of the 11 compounds
isolated from S. chamaejasme (Figure 3). β-Hexosaminidase levels (a marker of RBL-2H3
cell degranulation) in cells were increased by 3.05-fold after treatment with IgE and
dinitrophenyl/bovine serum albumin (DNP/BSA). Treatment with ketotifen (the positive
control) at 35 µM inhibited β-hexosaminidase release from RBL-2H3 cells by 58.9%. Of the 11
compounds isolated, genkwanin most potently inhibited β-hexosaminidase release.
Interestingly, the histamine inhibitory effects of flavonoids were dependent on their
glycoside-binding activities. In a previous study, flavonoid aglycones were reported to
inhibit histamine release more than flavonoid glycosides.
31
Similarly, we observed that flavonoid aglycones—apigenin (

Anti-inflammatory effects of 11 compounds from Stellera chamaejasme on interleukin-4 messenger ribonucleic acid expression in PI-induced RBL-2H3 cells. Results are expressed as the means ± SDs of 2 independent experiments. # P < 0.05 versus vehicle control; * P < 0.05 versus PI. DNP-BSA, dinitrophenyl-bovine serum albumin; keto, 35 µM ketotifene; IgE, immunoglobulin E; PI, propidium iodide.
To determine whether maltol 3-[6″-(2-(E)-butenoyl)-glucoside]
(

Antiallergic effects of 11compounds from Stellera chamaejasme on β-hexosaminidase release from immunoglobulin E/DNP-BSA-induced RBL-2H3 cells. Results are expressed as the means ± SDs of 2 independent experiments. # P < 0.05 versus vehicle control; * P < 0.05 versus IgE/DNP-BSA. CsA, 1 µM cyclosporin; DNP-BSA, dinitrophenyl-bovine serum albumin; IgE, immunoglobulin E; IL-4, interleukin-4; PI, propidium iodide.
This is the first report describing the antiallergic effect of a new maltol derivative, maltol 3-[6″-(2-(E)-butenoyl)-glucoside], from S. chamaejasme. Experiments with this compound revealed that it significantly reduced β-hexosaminidase release in IgE/DNP-BSA-stimulated RBL-2H3 cells. It has already been reported that S. chamaejasme extract has a preventive effect on the development of allergic contact dermatitis in oxazolone-induced and 2,4-dinitrochlorobenzene-induced mice. 32 Flavonoid glycosides, luteolin 7-O-glucoside and stechamone, and maltol isolated from S. chamaejasme also showed strong antiallergic effects in hapten-induced dermatitis-like skin lesions in murine models as main components. 32 -34 The present study extends these findings by showing antiallergic and anti-inflammatory activities of various compounds contained in S. chamaejasme in vitro RBL-2H3 cells. Further studies using appropriate animal models are warranted in order to clarify the antiallergic effect of maltol 3-[6″-(2-(E)-butenoyl)-glucoside].
Experimental
General Experimental
HR-ESI-MS were performed on an Agilent 6530 Accurate-Mass Q-TOF LC/MS spectrometer system (Agilent Technologies, CA, USA). One-dimensional (1H and 13C) NMR spectra were recorded using a 400 MHz Varian Unity INOVA NMR spectrometer (Varian, Palo Alto, CA, USA), and two-dimensional (COSY, HMQC, HMBC, and NOESY) NMR spectra were recorded on a Bruker Avance NEO 500 MHz spectrometer system (Bruker, Billerica, MA, USA). High-performance liquid chromatography (HPLC) analyses were carried out on a Shimadzu system (Shimadzu Corporation, Kyoto, Japan), comprising a LC-20AT pump, SPD-20A UV detector set at 254 nm, and a CBM-20A system controller. Column chromatography was performed on silica gel (230-400 mesh, Merck, Germany) and Sephadex LH-20 gel (25-100 mM mesh, Pharmacia, Sweden). Preparative thin-layer chromatography (TLC) (1.05715.0001, Merck, Germany) and TLC (1.05554.0001, Merck, Germany) used precoated silica gel 60 F254, and compounds were visualized by spraying with p-anisaldehyde solution.
Plant Material
Stellera chamaejasme L. aerial parts were collected from Shangri-La (Diqing Prefecture, Yunnan Province, China). The plant was identified by Dr Sang Woo Lee (Korea Research Institute of Bioscience and Biotechnology), and voucher specimens were deposited at the Medicinal Herb Garden, Pusan National University (PNU-0023).
Stellera chamaejasme Extraction and Isolation
The air-dried aerial parts of S. chamaejasme (6.11 kg) were sonicated with 95% ethanol (EtOH, 12 L × 6) for 90 minutes and extracted overnight at room temperature. After evaporation under reduced pressure, the EtOH extract (SCAE, 569.8 g) was suspended in water (H2O,5 L), and partitioned consecutively with n-Hex (12 L, 37.0 g), ethyl acetate (12 L, 20.8 g), and n-butanol (BuOH, 12 L, 34.4 g).
The EtOAc fraction (20.8 g) was subjected to silica gel column chromatography
(dichloromethane [CH2Cl2]-MeOH = 20:1 → 0:1) to yield 13 fractions
(SCE1 to SCE13). Crystallization of fraction SCE2 (314.7 mg) from MeOH gave compound
The n-BuOH fraction (34.4 g) was subjected to silica gel column
chromatography (CH2Cl2-MeOH = 10:1 → 0:1) to yield 8 fractions (SCB1
to SCB8). Silica gel column chromatography (CH2Cl2-MeOH = 20:1 →
0:1) of fraction SCB2 (300.2 mg) yielded 4 subfractions (SCE2-1 to SCE2-4). Subfraction
SCB2-4 (38.6 mg) was separated by preparative TLC
(CH2Cl2-MeOH-H2O = 10:1:0.5) to give compound
Cell Culture of RBL-2H3
Rat basophilic leukemia (RBL-2H3) cells were obtained from American Type Culture
Collection (ATCC, #CRL-2256, Bethesda, MD). Cells were grown in Eagle’s minimum essential
medium supplemented with 10% heat-inactivated fetal bovine serum, 2 mM
Analysis of IL-4 mRNA Expression
RBL-2H3 was sensitized with either dimethylsulfoxide (DMSO) or compounds (10 µM) for 30 minutes and then restimulated with phorbol 12-myristate 13-acetate and ionomycin for 16 hours. To measure IL-4 mRNA expression, total RNA was extracted using RNAiso Reagent (TaKaRa, Shiga, Japan), according to the manufacturer’s guidelines, and subjected to a quantitative real-time polymerase chain reaction (PCR). Primer sequences were used as follows: IL-4, F, 5′-ACC TTG CTG TCA CCC TGT TC-3′ and R, 5′-TTG TGA GCG TGG ACTCAT TC-3′; β-actin, F, 5′-TCA TCA CCA TCG GCA ACG-3′, and R, 5′-TTC CT GAT GTC CAC GTC GC-3′. Real-time PCR data were analyzed with the 7500 System SDS software version v1.3.1 (Applied Biosystems).
Release Activity of β-Hexosaminidase in RBL-2H3 Cells
RBL-2H3 cells (1 × 105 cells per well) in 24-well plates were sensitized with
anti-DNP IgE for 24 hours. The cells were washed with Siraganian buffer and then
preincubated in the buffer for 10 minutes. After that, either DMSO or compound
Statistics
The values are expressed as means ± SDs. The statistical significance was carried out by one-way analysis of variance. P values <0.05 were considered significant.
Supplemental Material
Supplementary Material 1 - Supplemental material for Antiallergic Effects of Phenolic Compounds Isolated From Stellera chamaejasme on RBL-2H3 Cells
Supplemental material, Supplementary Material 1, for Antiallergic Effects of Phenolic Compounds Isolated From Stellera chamaejasme on RBL-2H3 Cells by Beom-Geun Jo, Sim-Kyu Bong, Jonghwan Jegal, Su-Nam Kim and Min Hye Yang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this research was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Ministry of Science & ICT (NRF-2019M3A9I3080263, NRF-2019M3A9I3080265, and NRF-2019M3A9I3080266).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
