Abstract
Background
Viral infections pose some of the most serious human health concerns worldwide. The infections caused by several viruses, including coronavirus, hepatitis virus, and human immunodeficiency virus, are difficult to treat.
Methods
This review details the findings of a literature search performed on the antiviral properties of luteolin. The keywords engaged in the search are “virus” along with “luteolin.”
Results
Luteolin possesses antiviral properties, which is the basis for the current review. It is an important natural flavonoid with numerous important biological properties, including anti-inflammatory, immune regulatory, and antitumor effects, and is found in vegetables, fruits, and several medicinal plants. Recent studies have revealed that many traditional Chinese medicines that contain luteolin inhibit the replication of coronaviruses.
Conclusion
Luteolin effectively inhibits the replication of coronavirus, influenza virus, enterovirus, rotavirus, herpes virus, and respiratory syncytial virus, among others. In particular, it prevents viral infection by improving the body's nonspecific immunity and antioxidation capacity and inhibiting many pathways related to virus infection and replication, such as MAPK, PI3K-AKT, TLR4/8, NF-κB, Nrf-2/hemeoxygenase-1, and others. It also regulates the expression of some receptors and factors, including hepatocyte nuclear factor 4α, p53, NLRP3, TNF-α, and interleukins, thereby interfering with the replication of viruses in cells. Luteolin also promotes the repair of damaged cells induced by proinflammatory factors by regulating the expression of inflammatory molecules. The overall effect of these processes is the reduction in viral replication and, consequently, the viral load. This review summarizes the antiviral effect of luteolin and the mechanism underlying this property.
Introduction
An outbreak of coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), was reported in China in 2019, which later escalated into a global pandemic in 2020. The disease has caused millions of deaths and huge economic losses globally. Epidemics caused by new coronaviruses are predicted to increase in the coming years. Coronaviruses mutate rapidly, are highly pathogenic, and are challenging and expensive to treat. Accordingly, there is an urgent need to develop effective and cheap solutions to manage infections caused by SARS-causing viruses. Viruses are constantly mutating and becoming resistant to antivirals, and this resistance is promoted by the low bioavailability of these antivirals. As such, the use of these drugs is often restricted.1,2
A virus is an acellular particle comprising nucleic acid (DNA or RNA) coated with a protein shell. Viruses are obligate intracellular parasites that only replicate inside host cells. Most viruses are highly contagious and pathogenic, and as obligate intracellular parasites, viruses must utilize the biosynthetic machinery of host cells to replicate and multiply. Different from the drugs used in treating cardiovascular, cerebrovascular, and metabolic diseases, ideal antiviral drugs should inhibit or kill viruses without causing significant effects on the normal growth and metabolism of host cells. At present, small molecular drugs for treating viral infections mainly include viral DNA or RNA synthetase inhibitors, hormones, chloroquine, and others. However, these drugs cause numerous side effects, including body rashes, osteoporosis, leukopenia, and anemia.
There is a need to develop more effective, safe, and cheap antivirals. Traditional Chinese medicines play a unique role in the fight against viral infections. Luteolin is the main active ingredient in various effective formulas. Luteolin (3′, 4′, 5, 7-tetrahydroxyflavone; Figure 1) is a natural flavonoid found in vegetables, fruits, and medicinal plants such as celery, chrysanthemum, sweet pepper, carrots, onion leaves, broccoli, Polygonum cuspidatum, licorice, and honeysuckle, among others. It is a common dietary additive with no known side effects on the human body,3,4 and it possesses several pharmacological properties, including anti-inflammatory, antiallergic, uric acid-lowering, antitumor, antibacterial, and antiviral properties. The flavonoid is also used for cough and chest congestion relief.
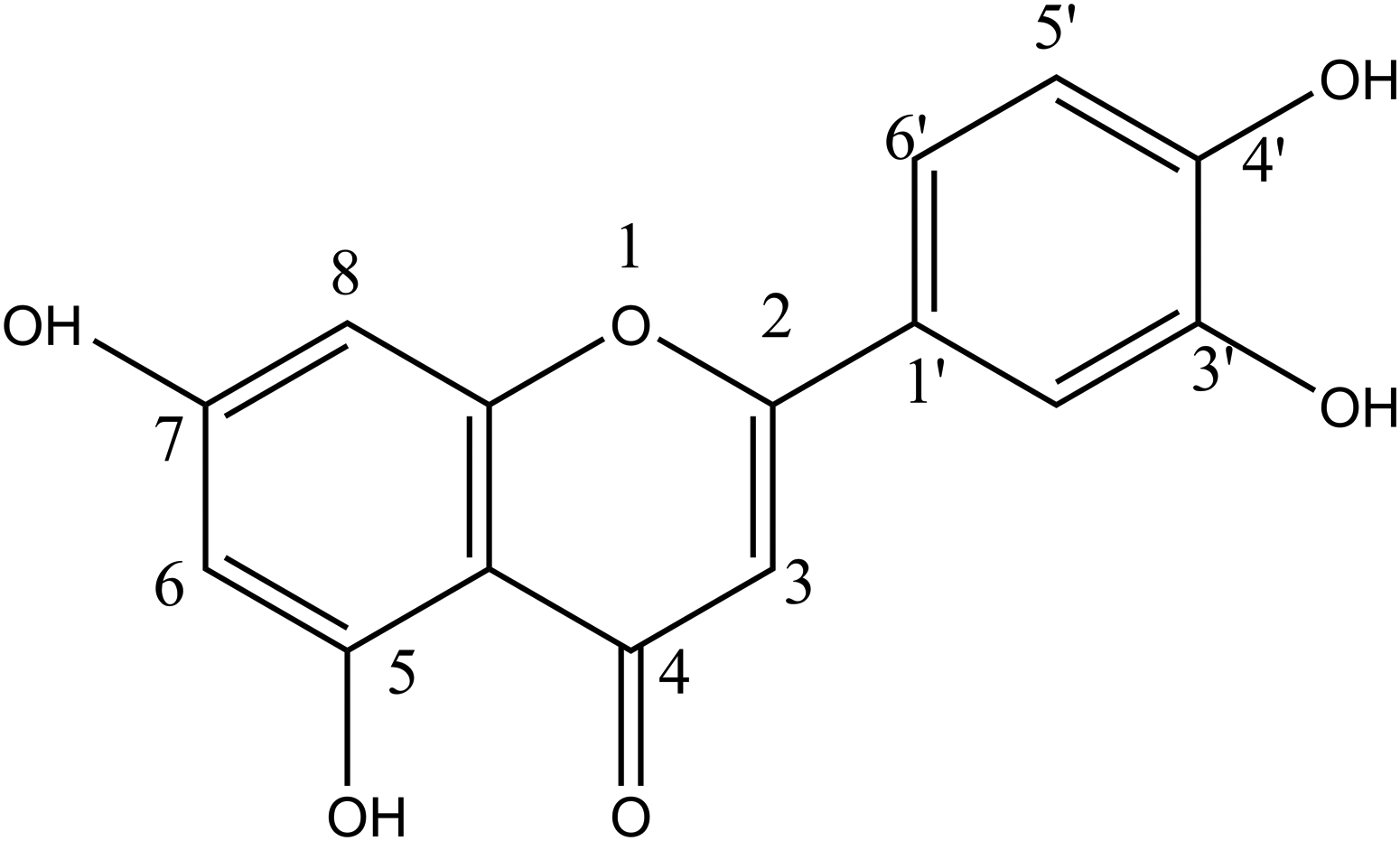
Chemical structure of luteolin.
Luteolin can modulate inflammation, lower uric acid, and treat cardiovascular diseases, amyotrophic lateral sclerosis, and infections caused by SARS and hepatitis viruses.5,6 According to Guo et al, 7 luteolin inhibits cytotoxicity and hemolytic effects by chelating streptolysin O toxin. Prasher et al 8 summarized the multifaceted cancer pathways that luteolin targets. These include through creating redox stress, reactive oxygen species (ROS) generation, inducing autophagy, cell cycle arrest, inhibition of cell proliferation, and the onset of apoptosis, finally leading to cancer cell death. Lei reported that luteolin demonstrated its anti-inflammatory properties by inhibiting the expression of various proinflammatory factors, including IL-4, IL-5, IL-6, IL-8, IL-13, IFN-γ, TNF, IL-1β, and COX-2. 9 The most important effect of luteolin is its potent antioxidative power, which includes excellent radical scavenging and cytoprotective properties. 10 It is also the main active ingredient in some clinical compounds, and ascorbic acid in Shenling Baizhu Powder used for regulating the expression of peroxisome proliferator-activated receptor γ contains luteolin. Luteolin and kaempferol are the key components in the Jiu-Wei-Yong-An formula, which is used for treating atopic dermatitis by regulating the MAPK pathway. Lianhua Qingwen capsule is another luteolin-based drug effective for treating COVID-19.11-13
Luteolin has received widespread attention in recent years as a viable alternative for treating viral infections and related diseases. Structural fitting analysis has shown that luteolin can inhibit the replication of influenza virus, enterovirus, rotavirus, herpes virus, respiratory virus, coronavirus, and other viruses. It primarily prevents viral infection by strengthening the body's nonspecific immunity and inhibiting several pathways, including MAPK, PI3K-AKT, TLR, NF-κB, and Nrf -2/hemeoxygenase-1 (HO-1), which are related to the replication of viruses. It also regulates the expression of many factors, such as hepatocyte nuclear factor 4α (HNF4α), p53, NLRP3, TNFα, and interleukins, which modulate the expression of viral receptors on cells. Luteolin thus regulates the expression of inflammatory factors and promotes the repair of cells damaged by antiviral response molecules. In this review, the antiviral and anti-inflammatory effects of luteolin and the mechanisms underlying these processes have been summarized.
Antiviral Perspectives
RNA Viruses
Influenza Virus
Influenza virus is a member of the Orthomyxoviridae family, which comprises the human and animal influenza viruses. The human influenza viruses include the A, B, and C types. They are a constant and serious threat to humans and many animal species and cause thousands of deaths and huge economic losses yearly. The influenza A virus (IAV) is the most infectious among the 3 human influenza viruses. 14 Between 1918 and 1919, IAVs killed 5% of the world's population.15-17 All influenza viruses contain hemagglutinin (HA) and neuraminidase (NA) surface glycoproteins. NA participates in the replication, transmission, and pathogenesis of the virus. Therefore, blocking the function of NA is a promising treatment option in influenza infection.18-20 At present, the commonly used antiviral drugs (such as zanamivir and oseltamivir) in clinical practice inhibit the release of viral particles from the host cell, thereby stopping the spread of infection. However, they do not ameliorate the long-term autoimmune inflammatory injury caused by the influenza virus. As early as 1940, flavonoids were shown to modulate inflammation and inhibit the growth of bacteria. 21 Luteolin is a flavonoid that can regulate the secretion of antioxidant factors, enhance antioxidation, inhibit viral replication, and reduce the damage caused by oxygen free radical generation during influenza virus infection. Suppressing the TLR3, TLR7, and NF-κB/MLCK pathways and the expression of β-COP and NOX4 proteins inhibit virus replication, modulate inflammation, and promote body repair. In addition, luteolin prevents the replication of viruses by inhibiting the PA-N terminal domain of influenza RNA-dependent RNA polymerase (RdRp).
NA is an enzyme that breaks glycosidic bonds. It also cleaves the sialic acid molecules on the cell surface, thereby freeing the virus to infect cells in the host organism. As such, NA mediates the early stage of viral entry into endothelial cells. 22 Blocking the functions of NA inhibits viral entry into the host cells. 23 Lee et al 24 found that luteolin effectively inhibited the replication of influenza viruses in MDCK cells and the activity of NA in the influenza virus subtypes H1N1, H3N2, and H5N1. As such, NA is potentially an effective target for treating and preventing influenza virus infections.
The TLR family of receptors mediates innate immune system responses. 25 TLR7 and TLR3 recognize viruses and initiate intracellular signal transduction pathways. 26 The TLR signal transduction pathways are divided into the MyD88-dependent and MyD88-independent pathways. 27 MyD88 is a key linker molecule in the TLR signaling pathway and participates in transmitting signals upstream of TLR7 during disease progression. TLR3 specifically recognizes double-stranded RNA (dsRNA). After binding to the viral dsRNA, TLR3 induces the nuclear/cytoplasmic translocation of NF-κB and expression of IFN-α/β through the MyD88-independent signaling pathway, further promoting the release of proinflammatory cytokines that regulate the immune response of cells. 28 TLR7 recognizes single-stranded RNA (ssRNA) and exerts antiviral effects through the MyD88-dependent and MyD88-independent pathways. 29 TRAF3 is a member of the TRAF family of proteins and participates in maintaining the normal functioning of the immune system. It also regulates the activities of the NF-κB classical and NF-κB nonclassical pathways. 30 NF-κB is a critical transcription factor that regulates immune response, growth, and differentiation of cells as well as the expression of inflammatory mediators and immune genes, especially those related to the early immune response. NF-κB is also a critical transcription factor in the TLR signal transduction pathway, where it regulates the expression of anti-inflammatory and other immune response molecules. 31 In H1N1 infection, luteolin can modulate the expression of key target genes and proteins in the TLR signaling pathway by promoting the expression of several nonspecific immune response and antioxidant molecules, including TLR3, TLR7, MyD88, TRAF3, and NF-κB 32 (Figure 2).

Schematic representation of the inhibition of replication and spread of the influenza virus by luteolin.
Luteolin alleviates the destruction of the lung endothelial barrier (LEBD) caused by the IAV by inhibiting the TNF-α-induced apoptosis of HPMVEC cells via the suppression of NOX4 and inhibition of the NF-κB/MLCK pathway. 33 It also inhibits the replication of the influenza virus, relieves inflammation in the lungs, and regulates water metabolism and immune response (Figure 3). Intercellular connections and cytoskeleton are the basic components that maintain the integrity of the endothelial barrier. NF-κB inactivation and MLCK inhibition prevent TNF-α-induced F-actin cytoskeleton injury, thereby disrupting the permeability of the endothelial barrier. 34 The stimulation of TNF-α increases the expression of phosphor-p65 and p-MLC but modulates that of the tight junction proteins occludin and ZO-1. However, luteolin reverses the activation of the NF-κB pathway and loss of tight junction proteins induced by IAV and TNF-α, thereby reducing microvascular endothelial leakage and apoptosis. These findings suggest that luteolin enhances the integrity of the LEBD and, thus, is a potential target for the treatment of severe influenza. NOX4, a member of the NADPH oxidase family, protects the vascular system against inflammatory damage and oxidative stress. 35 Nuclear transcription factors such as activator protein-1 and NF-κB promote the transcription of NADPH oxidase. In addition, NF-κB induces inflammation by first activating NOX4, leading to NF-κB signal transduction. 36
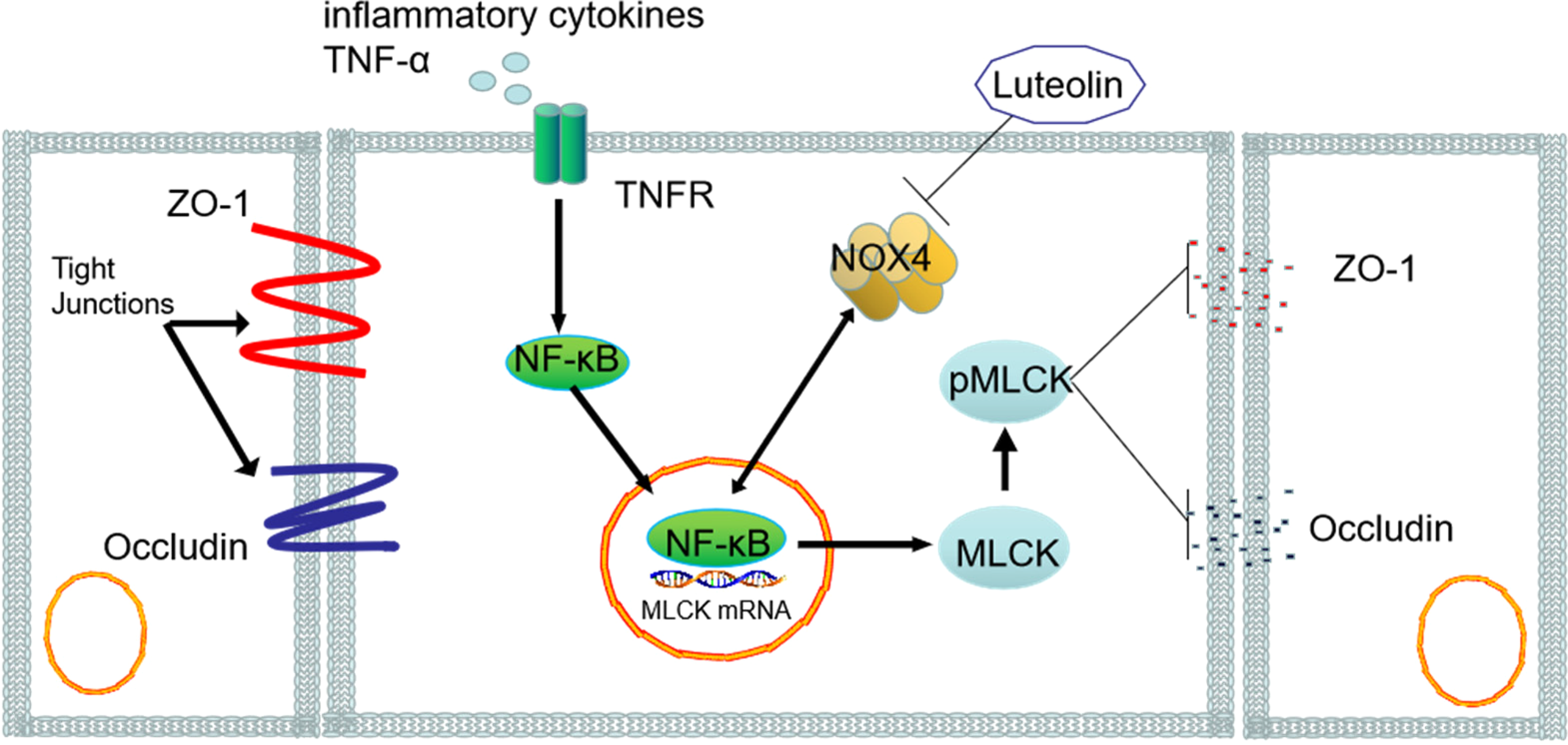
Protective effects of luteolin against influenza A virus (IAV)-induced pulmonary microvascular endothelial damage by regulating the NOX4/NF-κB/MLCK pathway.
The COPI complex comprises 9 different subunits. β-COP, 1 of the 9 subunits, is an essential part of the coatomer complex. The COPI complex participates in the transportation of contents between the Golgi apparatus and the endoplasmic reticulum and mediates the entry and endocytosis of the influenza virus.37,38 Disrupting the activities of this complex can impair endosome sorting, multivesicular body formation, and/or membrane transport.39-41 In addition, δ-COP (ARCN1) knockdown suppresses the nuclear translocation of Vrnp, 42 whereas β-COP knockdown significantly reduces the replication of IAN. 43 COPI knockdown inhibits infection with IAV mainly by blocking the internalization of the virus and early-to-intermediate/late endosomal tracking. 44 In mammalian cells, Golgi BFA resistance factor 1 (GBF1) activates the recruitment of COPI. GCA compounds inhibit the function of GBF1, causing the rapid dissociation of COPI from the Golgi apparatus and eventual decomposition of the Golgi apparatus. Haiyan revealed that luteolin reduces the replication of IAV in vitro by interfering with the expression of the I complex, an essential coat viral protein. Although luteolin does not interfere with the function of COPI, it exerts its antiviral activity by inhibiting the expression of this protein. 45 Further, luteolin inhibits the absorption and internalization of IAV by reducing the expression of the β-COP protein.
The initiation of viral mRNA transcription relies on short oligomers of host pre-mRNA, generated through a unique “catch–hatch” mechanism. The process begins with the binding of the PB2 subunit to the 5′ cap (m7 GTP) of the host precursor mRNA. A PA subunit then cuts the RNA fragment approximately 10-13 nucleotides downstream of the 5′ cap to generate the primers. PB1 uses an RNA fragment as the template for viral mRNA synthesis. RdRp is highly conserved in all influenza strains, and the “catch-hatch” mechanism has been observed in all viruses belonging to the Orthomyxoviridae family. Accordingly, RdRp is a promising target for treating influenza virus infections.46-49 In light of the above findings, Zima et al 50 proposed that luteolin and its homologs effectively inhibit the endonuclease of influenza virus against the PA-N terminal domain of influenza RdRp (PA-Nter). Several experiments, including the AlphaScreen technology, gel-based endonuclease inhibition detection, and structural analysis of PA-Nter and luteolin, have revealed that luteolin can effectively inhibit endonuclease activity.
Flavivirus
Dengue fever virus (DENV), which causes dengue fever, is a member of the Flaviviridae family. All 5 DENV serotypes (DEN 1-5) can infect humans. Even though the threat of DENV is gradually increasing globally, there is currently no effective antiviral against it. About 390 million cases of dengue fever infection occur yearly in >100 countries in the tropical and subtropical regions, with 25% of them progressing to clinical diseases.51,52 Dengue fever has become the main cause of hospitalization and even death for some patients, especially as a secondary infection. Antibody-dependent enhancement may cause a more severe disease. 53 Nonstructural protein 3 (NS3) is a protease of DENV that can potentially be used to treat DENV infection.54-56 NS3 molecules comprise the protease (NS3pro) and helicase domains. The NS3pro domain relies on a cofactor, NS2B, for its catalytic action and serves as a center for the assembly of the DENV replication complex, thereby modulating viral pathogenesis and suppressing the host immune response. These processes are central to the assembly of the DENV virion, as well as to the regulation of viral pathogenesis and the host immune response.57-59 Dwivedi et al 60 revealed that luteolin effectively inhibits the formation of the DENV NS2B/NS3pro complex, and as such, is a potential drug for treating dengue fever.
Luteolin slightly and noncompetitively inhibits the activity of the recombinant dengue virus NS2B/NS3pro, which mediates the replication and assembly of viruses. The prM protein is cleaved by the host preprotein convertase furin, the M stump of the membrane. The “pr” peptide remains attached to the virus particle until the end of the virus particle assembly. Upon release from the host cell, the peptide dissociates and transforms the immature virus particles into infectious particles. Consequently, this disrupts the virus maturation process, producing fewer mature virus particles and effectively reducing or cutting off the viral replication cycle. The in vitro suppression of the dengue virus in various cell lines and PBMC appears to be caused by the accumulation of immature viral particles. Peng et al 61 found that luteolin inhibits the late stage of the dengue virus life cycle in cells, possibly by inhibiting the host proprotein convertase furin in a noncompetitive manner, resulting in the impartial cleavage of the prM protein. This disrupts the virus maturation process, producing immature virus particles and effectively reducing or preventing subsequent virus replication. Luteolin primarily exerts its effect by blocking the furin activity.
Japanese encephalitis virus (JEV) is a member of the Flaviviridae family, one of the most economically important viruses in the world. JEV is an enveloped virus with 11-kb long single-stranded positive-stranded RNA. Its genome comprises a single long open reading frame (ORF) flanked by 5′ and 3′ untranslated regions. The fatality from human viral encephalitis may be as high as 30%. Also, 30% to 50% of patients with viral encephalitis develop permanent neurological or psychiatric sequelae. 62 According to the World Health Organization (WHO), there are 68 000 new cases of Japanese encephalitis (JE) each year, resulting in 15 000 deaths. The majority of the cases mainly occur in children from China, India, and Southeast Asian countries.63,64 Today, JE is considered a global public health problem. The JEV genome codes for a polyprotein, which is cleaved by viral and cellular proteases to produce 3 structural {capsid (C), pre membrane (prM), and envelope (E)} and 7 nonstructural (NS) proteins (NS1, NS2A, NS2B, NS3, NS4A, NS4B, and NS5).65,66 Although effective vaccines are available, no effective antiviral therapy against JE has been developed. The flavivirus E protein is a viral HA that mediates the receptor-specific attachment of the virus to the cell surface and induces the host's protective immunity. 67 The E protein mediates the pathogenicity of viruses by inducing cell tropism and mediating the penetration of the virus to susceptible cells. 68 Fan et al 69 found that luteolin significantly reduces the expression of JEV E protein in a dose-dependent manner. These findings suggest that luteolin prevents the attachment of JEV to its receptors, but it does not show antiviral activity in the viral binding and entry stages.
Respiratory viruses
Human respiratory syncytial virus (RSV) belongs to the Pneumovirus genus in the Paramyxoviridae family, and it is the main cause of respiratory infections such as pneumonia and bronchiolitis in infants and children. Severe RSV infection is characterized by repeated wheezing or asthma. RSV is also an important cause of respiratory infections in the elderly (aged ≥65 years). Despite the continuous and positive progress in developing RSV vaccines, effective RSV treatment is still lacking.70-73 The controversial efficacy, toxicity, and the long and cumbersome inhalation of the traditional nucleoside drug ribavirin have restricted its use for RSV treatment. 74 Wang et al 75 assessed the in vitro antiviral activity of luteolin against RSV based on the cytopathic effect (CPE) reduction test and found that it inhibits the replication of RSV by regulating the MiR-155/SOCS1/STAT1 signaling pathway. 76 Luteolin exerts its antiviral activity by increasing the expression of IFN-stimulated genes (ISG) through the phosphorylation of STAT1. The SHIP1, WEE1, VHL, TP53INP1, PU.1, BCL2, BCL6, SOCS1, and SOX genes are the direct targets of microRNA-155 (miR-155).77-79 MiR-155 induces the phosphorylation of STAT1 and expression of the ISG by directly targeting SOCS1, the negative STAT1 regulator. 80 MiR-155 and its target gene SOCS1 are the key regulators of effector CD8 T cells and perform this function by regulating cytokine signal transduction through the STAT5 pathway. Inhibiting miR-155 abrogates the downregulation of SOCS1 expression caused by luteolin, whereas upregulating the expression of miR-155 enhances the inhibitory effect of luteolin. Studies have shown that luteolin inhibits RSV replication by inducing the expression of miR-155. It also enhances the antiviral effect of IFN-α/β against RSV.
Chikungunya virus
Chikungunya virus (CHIKV) belongs to the Togaviridae family in the Alphavirus genus, and is a single-stranded positive-sense RNA of approximately 11-12 kb with a capsid diameter of about 60 to 70 nm. There is only 1 CHIKV virus serotype, which is divided into 3 genotypes: The West African type, the Central-East-South African type, and the Asian type. 81 CHIKV infection is characterized by atypical clinical manifestations of the nervous, urinary, and cardiovascular systems, as well as those of the skin and eye. Persistent debilitating rheumatism may last for several years in individuals of all age groups and sex.81-83 Arbidol is a broad-spectrum synthetic antiviral drug effective against CHIKV that exerts its antiviral property by blocking either the attachment or entry of the virus into the host cells at the very early stage of infection. However, resistance to this drug limits its clinical application.84,85 Murali et al 86 revealed that natural compounds such as luteolin, containing the 5,7-dihydroxyflavone unit, inhibited CHIKV replication. Flavonoids containing the dihydroxy flavonoid structure inhibited the replication of CHIKV rather than its entry into the cell. 87
Human immunodeficiency virus
Human immunodeficiency virus (HIV), which causes acquired immunodeficiency syndrome (AIDS), is an RNA retrovirus that impairs the function of the immune system. The current use of combination antiretroviral therapy for HIV infection has significantly increased from 43% in the mid-1990s to 78% in 2010. 88 However, this therapy does not target resting CD4+ T cells, which harbor latent HIV. The HIV-1 transcription transactivator (Tat) protein increases RNA pol II activity, which promotes the transcription of viral genome elongation by binding to the positive transcription elongation factor b (pTEFb) complex (cycT1 and CDK9). Host factors such as Tat-related histone acetylase (TAH), p300/CBP, GCN5 and P/CAF, and P300/CBP and GCN5 acetylate enhance the activity of Tat at Lys 50 and 51. P/CAF acetylates Lys 28 on Tat and enhances the recruitment of the pTEFb complex. Therefore, given that Tat can interrupt the viral life cycle, it is an important therapeutic target for managing HIV/AIDS.89-94 Luteolin weakens the function of HIV-1 Tat, further underscoring its potential for HIV/AIDS management. Mehla et al 95 demonstrated that luteolin inhibits the replication of latent HIV-1 and effectively blocks the transactivation of LTR, which is mediated by clade B and C-Tat. However, it has no effect on Tat expression and the subcellular localization of this protein. Thus, luteolin exerts its antiviral effect probably via the Tat-LTR pathway and downstream viral proteases.
Rotavirus
Rotavirus is a dsRNA virus belonging to the Reoviridae family. It infects mature villous epithelial cells of the small testis, and is characterized by fever, vomiting, and diarrhea in children. It is estimated that rotavirus infection causes 453 000 deaths every year among children aged <5 years. 96 The current vaccines against the virus are expensive, and as such, cheap but effective natural antivirals can be a valuable alternative. Although luteolin inhibits the replication of rotavirus, the precise mechanism underlying this process is unclear. 97
Hepatitis virus
Hepatitis C virus (HCV) is a positive-sense ssRNA virus. The RNA genome is 9.6 kb in size. Approximately 170 million people worldwide are infected with HCV, 98 and current treatments for infection by it include a long-term dose of ribavirin and PEGylated IFN-α, which clears the virus in 40%-50% of patients. 99 However, the severe side effects of these drugs, including anemia, neutropenia, and leukopenia, underline the need for alternative treatment. HCV encodes a 3.01-kb polyprotein in a single reading frame. 100 In infected cells, this polyprotein is cleaved at multiple sites by a combination of cellular and viral proteases to generate several structural and NS viral proteins, including C, E1, E2, P7, NS2, NS3, NS4A, NS4B, NS5A, and NS5B. Similar to other positive-strand RNA viruses, HCV replication involves the initial synthesis of a complementary negative RNA strand, which is the template for generating new positive-strand RNAs. HCV-replicase (NS5B) is one of the NS proteins. It is an RdRp that catalyzes the replication of the viral RNA. Therefore, NS5B has become the primary target for the development of HCV antivirals. 101
Eclipta alba extract strongly inhibits the activity of HCV replicase RdRp in vitro. 102 Moreover, the extract effectively inhibits the replication of HCV. Weiderlactone, luteolin, and apigenin are the main anti-HCV compounds in the Eclipta alba extract, and these compounds inhibit HCV replication in vitro in a dose-dependent manner. HCV NS5B has 5 different binding sites for small molecules, which block the activity of the enzyme when bound by the compounds. These compounds synergistically inhibit the activity of RdRp by binding NS5B, further validating the different binding sites of the NS5B molecule. A combination of weiderlactone and luteolin in the ratio of 1:1 synergistically inhibits the activity of HCV RdRp.
Coronavirus
The genetic blueprint of SARS-CoV-2, an RNA virus, is similar to that of SARS-CoV and MERS-CoV. SARS-CoV-2 is a member of the β-Coronavirus genus. As of February 2021, 100 million COVID-19 cases have been confirmed globally, resulting in >2 million deaths. The common symptoms of SARS-CoV-2 infection include fever, dry cough, myalgia, loss of smell, and respiratory obstruction. 103 Acute respiratory distress syndrome, systemic inflammatory response syndrome, and multiple system inflammatory syndrome have also been observed in severe COVID-19 cases.104-106 Asymptomatic cases are common, and carrier patients rapidly spread the disease undetected. 107
Abdel Massih et al 108 demonstrated that luteolin inhibits furin activity, a lytic enzyme implicated in the pathogenesis of metabolic syndrome. The cleavage of furin is essential in activating the enzyme in several viruses, including SARS-CoV-2. Luteolin also inhibits the NF-κB pathway, suppresses the expression of TNF-a, IL-6, and IL-1b, and inhibits MPO activity. Flavonoids potentially inhibit the activity of several critical viral targets, including Mpro, S protein RBD, RdRp, human ACE-2 receptor, and TMPRSS2, which are necessary for the entry and replication of viruses.109,110
Gour et al 111 reported that in LPS-induced acute lung injury, luteolin treatment significantly reduces the expression of IL-6 and TNF-α in mouse models by inhibiting the Smad3 pathway, which is activated by NF-κB and transforming growth factor-β1 (TGF-β1). Also, luteolin inhibits the proteolytic activity of SARS-CoV 3CLpro and suppresses the replication of SARS-CoV in Vero cells. It inhibits the entry of SARS-CoV-2 into host cells by denaturing the spike protein, the main protease, and nucleocapsid protein of the virus.
In their 2019 report, Huang et al 112 proposed that angiotensin-converting enzyme 2 and 3C-like protease (3CL pro) are potentially critical targets for antiviral drugs. Of note, luteolin inhibits inflammation and other immune responses through COX-2, CASP3, COX-2, CASP3, IL-17, arachidonic acid, HIF-1, NF-κB, Ras, and TNF signaling pathways by targeting AEC2 and 3CL proteins. IL-6, MAPK1, MAPK14, MAPK8, and REAL scavenge free radicals. Based on the relaxation complex scheme (RCS), classical molecular dynamics (MD) simulation, and meta-dynamics simulation, Shadrack et al 113 revealed that luteolin blocks the entry of SARS-CoV-2 into the cell. Related research revealed that Akt1 is a potential target for the treatment and prevention of COVID-19. Another study showed that luteolin effectively blocks the entry of SARS-CoV-2 into the host cells. 114
DNA Viruses
Hepatitis B virus
HBV infection is a global public health problem. Chronic hepatitis B (CHB) infection increases the risk of liver cirrhosis and hepatocellular carcinoma (HCC). 115 CHB infection is the dominant global cause of HCC, accounting for 55% of HCC cases worldwide and ≥80% in the eastern Pacific region and sub-Saharan Africa, the regions with the highest incidence of cancer. 116 Although all countries have implemented vaccination against HBV, 350-400 million people are still infected worldwide. 117 Nucleoside (acid) analogs and interferons effectively inhibit HBV replication.
HNF4α is a critical transcription factor that regulates the transcription and replication of HBV. It promotes the HBV promoter activity by directly binding to DNA.118,119 Unlike other transcription factors, HNF4α primarily exists in the nucleus as a homodimer and regulates gene transcription by binding to DNA. 120 Bai et al 121 demonstrated that luteolin inhibits the expression of HNF4α and its binding to the HBV promoters in HepG2.2.15 cells. Although luteolin activates the extracellular signal-regulated kinase (ERK), the suppression of ERK abrogates the inhibition of HNF4α induced by luteolin. Likewise, blocking ERK weakens the anti-HBV activity of luteolin. Luteolin directly inhibits the transcription of HNF4α mRNA in a manner different from the phosphorylation-mediated degradation of HNF4α protein. However, how ERK mediates the inhibition of HNF4α mRNA expression remains unclear.
Existing evidence shows that the MAPK pathway participates in the HNF4α-regulated transcription of HBV genes. In particular, the activated MAPK pathway participates in the IL-6-mediated suppression of HBV replication by regulating the expression of HNF4α and HNF1α and Ras-mediated MAPK activation. The activation of the MAPK pathway by PKC inhibits the expression of HNF4α. Although luteolin activates the MAPK pathway, the resultant role of MAPKs varies significantly in different cell types in pathological and physiological conditions. It is, therefore, essential to clarify the role of the MAPK pathway on the effect of luteolin on HBV replication. Luteolin activates ERK and JNK but inhibits p38. The JNK pathway mainly regulates the apoptosis of cells and inflammation. However, apoptosis may not be the mechanism by which luteolin suppresses HBV replication as luteolin has almost no effect on cell death. Further, although luteolin activates JNK and inhibits the expression of p38, these processes do not participate in the anti-HBV effect of this flavonoid. By contrast, the JNK and p38 pathways may be related to other unidentified pharmacological effects of luteolin.
Blocking the ERK pathway only eliminates the downregulation of HNF4α by luteolin. In addition, the inhibition of ERK abrogates the inhibitory effect of luteolin on HBV replication. These findings show that to some extent, luteolin exerts its effect by inhibiting the expression of HNF4α. However, how luteolin activates ERK in liver cells remains unclear. In summary, luteolin inhibits HBV replication in vitro and in vivo at the transcriptional level by downregulating the expression of HNF4α, which is mediated by ERK (Figure 4).

Mechanism by which luteolin regulates HNF4α, Hepatocyte nuclear factor 4α (HNF4α) secretion via the MAPK pathway.
Luteolin inhibits the activity of protein kinase C (PKC), and PKCβ negatively regulates the IFN-induced JAK/STAT activation.122,123 Tai et al 124 found that luteolin activates the JAK/STAT pathway, opening a new avenue for clinical research in this direction. Luteolin increases the expression of endogenous antiviral genes by activating the JAK/STAT pathway. It inhibits the PKC inhibitor, and PKCβ negatively regulates the IFN-induced JAK/STAT activation. Therefore, whether luteolin activates the JAK/STAT pathway by inhibiting PKCβ needs further study.
The HBV X protein (HBx) in the HBV is a potential target for liver cancer treatment.125,126 This protein does not directly bind to DNA. Yet, it regulates cytoplasmic signal transduction pathways such as Ras, Raf, and c-jun, MAPK, NF-κB, AP-1, JAK-STAT, FAK, and PKC, as well as Src-dependent and phosphatidylinositol 3-kinase signaling cascade by interacting with nuclear transcription factors.127,128 Using molecular docking prediction, Pathak et al 129 found that luteolin disrupts the synthesis of the HBx protein. Cui et al 130 further reported that luteolin-7-O-glucoside restored the normal mitochondrial membrane potential damage induced by ROS, indicating that this compound inhibits HBsAg and HBV replication through a mitochondria-related mechanism. HBV binds to the outer mitochondrial membrane through the HBX. This regulates the mitochondrial calcium uptake, cytosolic calcium levels, and signals that drive HBV replication. Luteolin-7-O-glucoside inhibits the mitochondrial calcium uptake, which may regulate the cytosolic calcium signaling and inhibit HBV replication.
Herpes virus
Herpes virus is a group of enveloped DNA viruses in the Herpesviridae family. To date, >100 herpes viruses have been discovered. Herpes viruses can be divided into 3 broad categories: α, β, and γ. These viruses have a broad host range and primarily infect the skin, mucus membranes, and nerve tissues, thereby seriously affecting the health of humans and other animals. The common herpes viruses include the herpes simplex virus type 1 (HSV-1), HSV-2, and Epstein–Barr virus (EBV). These viruses cause diverse infections and are characterized by the 2 life cycles of latent and lytic phases. HSV affects more than one-third of the world's population and causes diseases such as mild cold sores, keratoconjunctivitis, genital herpes, and life-threatening encephalitis. After penetrating the skin or mucosal barrier, HSV mainly infects epithelial cells, where they are transported to the ganglia. This follows a lifetime latent infection, but the virus is reactivated regularly. 131 Although the reactivated virus can cause several diseases, the mechanism underlying this process is not well understood.
There is no effective prevention and treatment therapy for HSV infections. Acyclovir, ganciclovir, maribavir, retinoic acid, curcumin, and artemisinin inhibit the lytic cycle of EBV. HSV infection is regulated by several cell-signaling pathways and transcription factors. For instance, the MRPK pathway promotes the replication of the virus.132-134 Moreover, HSV activates IκB kinase, which triggers the degradation of IκB in the early stages of infection, thereby inducing the sustained activation of NF-κB in epithelia, neurons, and lymphocytes. Luteolin inhibits the activation of PI3K-AKT, MAPK-ERK, and mTOR pathways, indicating a potential option for treating nasopharyngeal carcinoma caused by EBV. The P38, MAPK, and JNK pathways participate in EBV reactivation by N-methyl-N′-nitro-N-nitrosoguanidine (MNNG). Also, ROS plays a crucial role in EBV reactivation following MNNG induction. Thus, luteolin, a ROS scavenger, can inhibit EBV reactivation by blocking this pathway. 135
Durbadal Ojha et al 136 found that luteolin regulates the NF-κB signaling pathway and thereby inhibits HSV-2 replication by downregulating the expression of TNF-α, IL-1β, IL-6, NO, iNOS, and IFN-γ.
Latent membrane protein 1 (LMP1), encoded by EBV 2, is usually expressed in nasopharyngeal carcinoma and regulates several signaling pathways that promote cell growth, transformation, and metabolic reprogramming. 137 LMP1 promotes adipogenesis by increasing the phosphorylation of 4E-BP1 and p70S6K (validated mTOR targets) in nasopharyngeal epithelial cells. The overexpression of fatty acid synthase (FASN) promotes the replication and pathogenesis of EBV. FASN is induced by the EBV-encoded lytic protein BRLF-1 through the p38-MAPK pathway and is required for lytic viral gene expression. 138 Given that FASN is usually overexpressed in virus-related cancers, adipogenesis appears to be critical for viral infection and cancer progression. Luteolin inhibits lipogenesis, LMP1-induced SREBP1 activity, lipid synthesis, and the proliferation of nasopharyngeal epithelial cells. Also, luteolin inhibits the activation of the PI3K-AKT, MAPK-ERK, and mTOR pathways, which regulate LMP1-induced SREBP-mediated adipogenesis.139,140 Finally, luteolin arrests the cell cycle at the G1 phase and inhibits the reactivation of EBV in NPC cells (Figure 5).

Overview of the MAPK pathway.
Rta is a transcription factor expressed in the early stages of the EBV lytic cycle to activate genes required for this process. Wu et al 141 found that luteolin inhibits the expression of EBV cleavage genes necessary for inducing the EBV lytic cycle. Immunofluorescence analysis further revealed that luteolin modulates the activation of EBV, thereby reducing the production of viral particles. Luteolin suppresses the activity of Zta (Zp) and Rta (Rp) promoter proteins. Additionally, it inhibits the Sp1-luc activity, indicating that it exerts its antiviral effect on EBV by inhibiting the binding of Sp1 on DNA. CHIP analysis revealed that luteolin inhibits the activities of Zp and Rp by deregulating Sp1 binding. In short, luteolin inhibits EBV reactivation by suppressing the promoter activity of Zp and Rp, indicating that luteolin can prevent EBV infection. Sp1 binds to the promoter region of the herpes simplex virus IE gene, which is essential for initiating the lytic cycle. 142 Sp1 is a key factor required for IE62-mediated transactivation, which is important for VZV replication. 143 ROS is also critical to the reactivation of EBV after MNNG induction. 144
Pseudorabies (PR), also known as Aujeszky disease, is a highly contagious disease caused by the PR virus (PRV), a herpes virus. In the absence of a specific host, PRV infects several mammals, including pigs, sheep, cattle, and other animals, and causes severe infection and acute death. 145 Lin et al 146 found that luteolin inhibits the activation of NF-κB by inhibiting the phosphorylation of signal transducers STAT1 and STAT3, which activate transcription. Luteolin significantly upregulates the expression of p-Nrf2 and its downstream target, reducing the expression of proinflammatory mediators and cytokines such as COX-2 and iNOS. HO-1 is an inducible, anti-inflammatory, and immunosuppressive enzyme that inhibits apoptosis, inflammation, and oxidative stress both in vitro and in vivo.147-149 HO-1 exerts antiviral effects against HSV2, bovine viral diarrhea virus, porcine reproductive (virus), and respiratory syndrome virus in vitro.150-152 Men et al 153 proposed that luteolin effectively inhibits PRV replication by inhibiting the expression of viral mRNA and gB protein.
Enterovirus
enterovirus 71 (EV71), a member of the Picornaviridae family, causes hand, foot, and mouth disease in infants and young children. 154 An EV71 outbreak in China in 2012 caused >2 million clinical infections and 567 deaths. 155 At present, there is no specific vaccine or antiviral drugs for EV71 and CA16. 156 Luteolin inhibits the replication of EV71 through 3C protease, and Coxsackievirus B3 (CVB3) by suppressing the NF-κB signaling pathway. It also promotes the repair of damaged tissues caused by inflammation.
EV71 has a 7.5-kb genome 157 that codes for a single polyprotein precursor, which is then cleaved into 4 structural proteins (VP1–VP4) and 7 functional proteins (2A-2C and 3A-3D).158,159 VP1, the key structural protein of EV71, possesses the majority of the viral epitopes. Therefore, VP1 is usually used in the detection, identification, classification, and phylogenetic analysis of various EV71 genotypes.160,161 Meanwhile, 3C protease plays an essential role in the life cycle of EV71. This protease cleaves the polyprotein precursor into constitutive proteins and interacts with several host factors that are essential for protein and nucleic acid synthesis.162-164 In addition, 3C protease induces the apoptosis of host cells. In short, these findings suggest that 3C protease is a potential target of anti-EV71 therapy. Luteolin inhibits the replication of EV71 in vitro and the synthesis of VP1-related mRNA transcripts and proteins by blocking the activity of 3C protease. 165
CVB3 is a single, positive-stranded RNA virus in the Enterovirus genus of the Picornaviridae family, and it is the leading cause of human viral myocarditis. TNF-α inducible protein 3 alleviates CVB3-induced myocarditis by inhibiting the NF-κB signaling pathway, thereby promoting inflammation. 166 Luteolin modulates the production of mature virions and synthesis of CVB3 RNA and protein in a dose-dependent manner. 167 Luteolin inhibits CVB3 synthesis at least in part by inhibiting the phosphorylation of p38, MAPK, and JNK, and nuclear translocation of NF-κB, which suppresses the CVB3-induced oversecretion of proinflammatory cytokines. CVB3 replication is mainly regulated via the NF-κB signaling pathway. CVB3 infection can induce the degradation of the NF-κB-IκB complex. The released NF-κB dimer is phosphorylated and transferred to the nucleus to activate the transcription of various genes involved in inflammation and the immune response.
Human papillomavirus
Human papillomavirus (HPV) is a type of papilloma vacuolar virus in the family Papillomaviridae. HPV is a spherical DNA virus that can induce the proliferation of the squamous epithelium of human skin and mucus membranes. There is currently no treatment for HPV infection, and the existing cervical dysplasia and cancer therapies involve either destroying or removing infected tissues using cytotoxic agents or surgery. The expression and functioning of HPV early protein 6 (E6) are necessary for the replication and virus-related tumorigenesis of cervical cancer. HPV E6 induces the degradation of tumor suppressor protein p53.168-170 HPV E6 is a tumorigenic protein that binds to the ubiquitin ligase E6AP (E6-related protein), altering its target specificity. The loss of p53 function suppresses the host cell defenses, promoting virus-induced cell cycle activation during virus replication. Thus, interrupting the interaction between E6 and E6AP and the stabilization of p53 can reduce the viability and proliferation of HPV-infected cells. Luteolin inhibits HPV replication by interrupting the interaction between E6 and E6AP. 171
Ham also confirmed that luteolin killed HPV-infected cervical cancer cells in a dose-dependent manner. 172 Luteolin suppresses the expression of HPV E6 and E7 oncogenes, restores the normal expression of related factors pRb and p53, and increases the expression of E2F5. In addition, it enhances the expression of death receptors and downstream factors such as Fas/FasL, DR5/TRAIL, and FADD in HeLa cells and activates the caspase cascade. In particular, luteolin enhances the activity of caspase-3 and −8, which induces the apoptosis of cells in a dose-dependent manner. Activated caspase-3 induces the activity of caspase-8 and vice versa.173,174 Luteolin also disrupts the mitochondrial membrane potential as well as the release of cytochrome c and inhibits the expression of Bcl-2 and Bcl-xL. Overall, luteolin exerts its anticancer activity by inhibiting the expression of HPV E6 and E7 and the cross-activation of caspase-3 and −8.
Cotton leaf curl Multan virus
Cotton leaf curl Multan betasatellite (CLCuMuB) is a small, icosahedral, single-stranded DNA (ssDNA) molecule. Cotton leafroll disease is a major threat to cotton production throughout South Asia and Africa. The disease is caused by the ssDNA single-decibel Gomo virus, the cotton leafroll virus, and its related satellite molecules. Cotton leafroll virus is a member of the Geminiviridae family and has 2 satellites: β and α. 175 CLCuMuB has recently been classified and proposed to be a member of the Tolecusatellitidae family in the Betasatellite genus. 176 Homology modeling and docking analysis revealed that luteolin binds to βC1 protein encoded by the cotton curl Multan β satellite. 177 βC1 is involved in the intracellular transport of proteins by corestricting with the endoplasmic reticulum in cells. Moreover, βC1 disrupts an autophagy-related ubiquitin-like protein.178,179 It interacts with various proteins, such as carboxymethyltransferase, retinoblastoma-associated protein, and receptor kinase, salicylic acid, biquitin-conjugating enzyme, and ATP-binding cassette transporter. 180 Therefore, luteolin can inhibit the replication of the cotton leaf roller Multan virus, which is mediated by the βC1 protein.
Anti-inflammatory Perspectives
Among the many inflammatory cell complexes, the spot-like receptor (NLR) family is the most common inflammatory factor, consisting of NLRP3, a dot-like protein containing the CARD domain (ASC) and procaspase-1, related to apoptosis resistance. Luteolin inhibits the activity of various inflammatory factors, including MDA, SOD, GSH, TNF-α, and IL-1β.
Luteolin protects against LPS-induced acute liver injury in mice by inhibiting the TXNIP-NLRP3 axis. 181 Luteolin modulates liver damage by inhibiting the activation of NLRP3 inflammasome through the suppression of TXNIP, ASC, caspase-1, IL-1β, and IL-18. In addition, luteolin inhibits LPS-induced liver inflammation by inhibiting the production of inflammation-related genes, TNF-α, IL-10, and IL-6. In addition, luteolin modulates LPS-induced liver cell damage by inhibiting oxidative stress damage and regulating MDA, SOD, and GSH levels. However, by inhibiting TXNIP in vitro, the protective effect of luteolin on liver injury caused by acute LPS in mice was blocked. Jinhui Dong demonstrated that luteolin ameliorates inflammation and Th1/Th2 imbalance in rats with allergic rhinitis via the TLR4/NF-κB pathway. 182 NLRP3 inflammasome is a protein complex containing caspase-1 that regulates the release of IL-1β and plays a vital role in the innate immune response. Luteolin inhibited caspase-1 activation and IL-1β secretion in J774A.1 macrophages under various stimuli (including ATP, nigericin, and silica crystals). Lee et al 183 also found that luteolin inhibited the activation of NLRP3 by interfering with ASC oligomerization. The activation of NLRP3 inflammasome in macrophages requires 2 signals. The first is mediated by NF-κB activation, which induces pre-IL-1β synthesis, an inflammasome substrate. The second signal is necessary for the inflammation of specific assemblies that activate caspase-1, thereby inducing the release of IL-1β from the cell. Luteolin not only interrupts signal 1 but also prevents ASC oligomerization by targeting signal 2, which inhibits the activation of NLRP3 inflammasome.
Conclusion
Luteolin is a natural flavonoid with no known side effects on the human body. Recent studies have elaborated on the mechanism underlying the antiviral property of luteolin. In particular, it interferes with viral replication and promotes the apoptosis of virally infected cells via the MAPK, NF-κB, TLR, and other related pathways (Table 1). In addition, luteolin protects cells against inflammatory damage and further promotes the repair of cells, tissues, and organs caused by virus infection and inhibits the activation of inflammatory factors and modulates inflammation via the TLR4/NF-κB-related pathways. Overwhelming evidence suggests that luteolin and several related compounds, such as honeysuckle, dandelion, Lianhua Qingwen capsules, and Double Huang lian oral solution, which are commonly used in clinical practice, possess good antiviral activity. It is worth noting that as a multitarget drug, the mechanism of luteolin exerting antiviral effects is complex, and the existing research level is not enough to fully elucidate. Other nonspecific effects of luteolin are also involved in its antiviral effects, such as regulation of oxidative stress, induction of autophagy or apoptosis in infected cells, and regulation of immune response. Unfortunately, existing research is not focused on the overall mechanism of action of luteolin. The limitations of experimental conditions have constrained the study of viruses. Thus, numerous aspects regarding infection and viral control remain unresolved. For instance, the specific mechanism underlying the action of numerous traditional antivirals is not fully understood.
Summary of the Antiviral Mechanism of Luteolin.

Footnotes
Author Contributions
Peng Lu, Daoyin Gong, and Zhixing Cao contributed to the conception of the study and revised it critically for important intellectual content; Tingrui Zhang and Yali Ren contributed significantly to analysis and manuscript preparation; and Huanan Rao, Jieting Lei, Guiying Zhao, and Miao Wang helped perform the analysis with constructive discussions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Science Foundation of China (82174193, 81973189, 81891012, U19A2010, and 81873073), Natural Science Foundation of Sichuan Province (No. 2022 NSFSC1535), and Xinglin Scholar Talent Research Supporting Program (CDUTCM) (QNXZ2018007 and QNXZ2018011).
