Abstract
Introduction
Ganoderma sinense Zhao, Xu et Zhang is a member of the genus Ganoderma (lingzhi) which contains 181 species mainly distributed in the tropics and semitropics.1,2 As 1 of 2 definitive raw drug materials of lingzhi reported in the Chinese Pharmacopoeia 2020, G sinense is well-known in East Asia and has been utilized extensively for health food and as a crude drug with a long history. However, few studies have been reported on the chemical constituents and pharmacological activities of G sinense, especially G sinense spores. Our earlier research reported that lipids obtained from G sinense spores exhibited antitumor and immune-enhancing properties3,4 and described how to extract ergosterol ramification and elucidate its structure with their tumoricidal effect as well as antimetastatic activities.
5
In a continuation of our investigation on the chemical ingredients from G sinense spores, our work reports the isolation and identification of a new compound, (22E,24R)-3β,5β,6α,7α,14β-pentahydroxyergosta-8,22-dien-15-one (

Structures of the compounds isolated from Ganoderma sinense spores.
Results and Discussion
Compound 1 (Figure 1) behaves as a whitish powder. Its molecular formula, C28H44O6, was confirmed by using high-resolution electrospray ionization mass spectrometry (HRESIMS). The 1H nuclear magnetic resonance (NMR) spectrum showed 6 methyl protons at δH 1.06 (d, J = 6.7 Hz), 1.02 (s), 0.88 (d, J = 6.8 Hz), 0.79 (d, J = 6.8 Hz), 0.77 (d, J = 6.8 Hz) and 0.75 (s). The 1H and 13C NMR spectra (Table 1) indicated that it contains groups such as 6 methyls, 6 methylenes, night methines (4 methines, 3 oxygenated methines and 2 sp2 methines), and 7 quaternary carbons (2 oxygenated tertiary carbons, 3 sp2 quaternary carbons, and 1 carbonyl carbon) (Figure S1-S4). Thus, its structure was presumed to be a C28 sterol based on the combined HRESIMS and NMR data (Figure 1).
1H and 13C NMR Data for Compound

Abbreviations: NMR, nuclear magnetic resonance; HMBC, heteronuclear multiple bond correlation
600 MHz, coupling constants (J in Hz) are given in parentheses.
150 MHz.
The coupling constant 14.7 Hz confirmed the presence of an E-configuration for the double bond between C-22 (δC 134.3) and C-23 (δC 132.6). 1H–1H correlation spectroscopy (1H-1H COSY) and heteronuclear single quantum coherence identified 2 structural fragments, 1 from C-16 to C-28, and the other from C-1 to C-4 (Figure 2). In the heteronuclear multiple bond correlation spectrum, key relationships (Figure 2) were observed from methyl protons at δH 0.75 (H-18) to C-12, C-13, C-14, and C-17; H-16 to C-13, C-14, C-15 (δC 215.9), and C-17; hydroxyl protons at δH 5.71 (14-OH) to C-8 (δC 125.0), C-13, and C-14 (δC 83.9); H-11 to C-8 and C-9; H3-19 (δH 1.02) to C-1 (δC 30.7), C-5 (δC 74.6), C-9 (δC 144.4), and C-10; and hydroxyl protons at δH 4.82 (5-OH) to C-4, C-5, C-6 (δC 74.8) and C-10. In addition, key relationships in the 1H-1H COSY spectrum were found between H-3 and hydroxyl protons at δH 5.29 (3-OH); H-6 and hydroxyl protons at δH 4.47 (6-OH); H-7 and hydroxyl protons at δH 5.76 (7-OH); and H-11 and H-12.
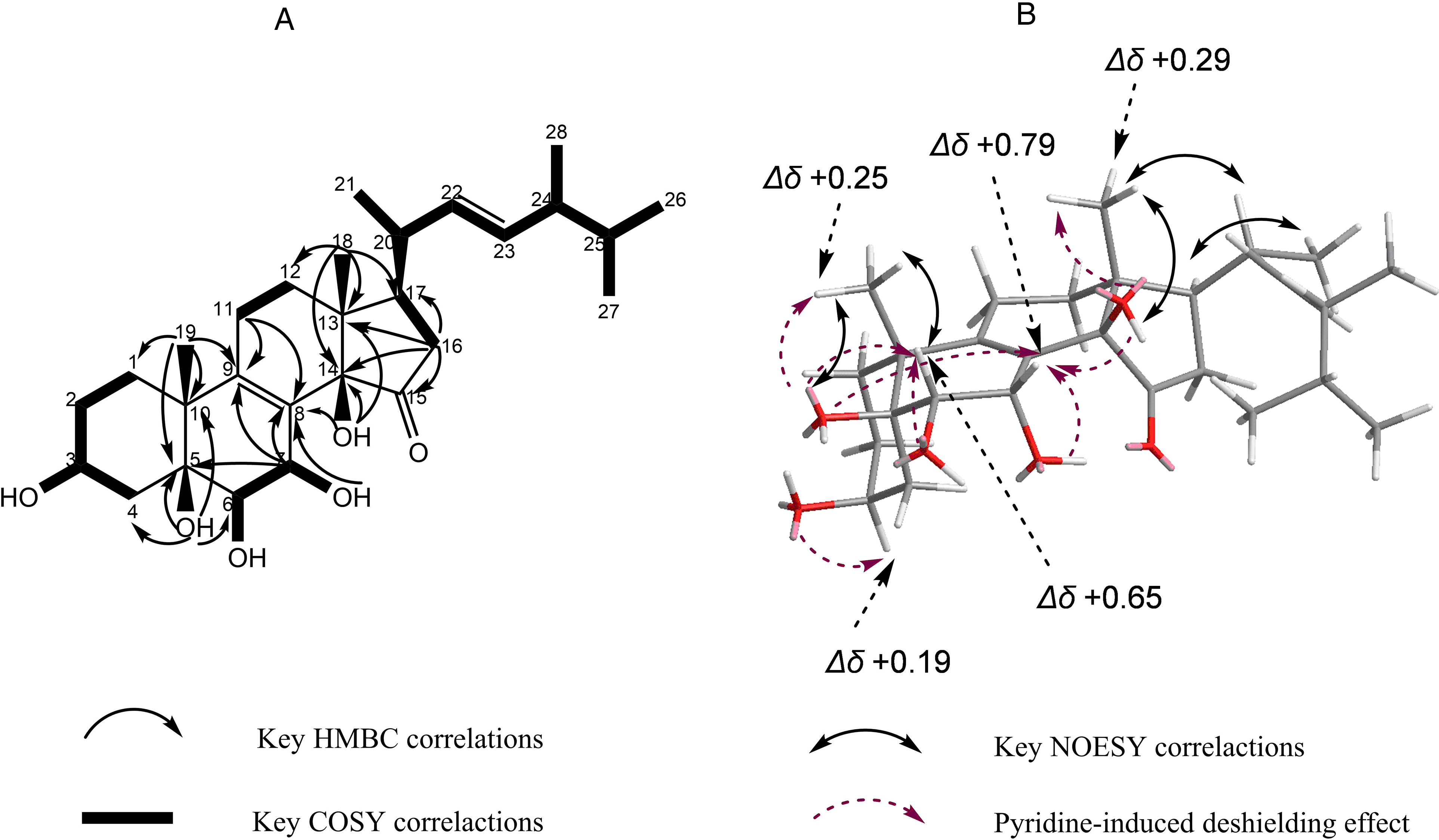
(A) Structure of compound
The relative configuration of compound
The 5 known compounds were identified through comparison of NMR data and ESI data with formerly reported compounds. Their structures were confirmed as (22E,24R)-3β,5α,9α,14β-tetrahydroxyergosta-7,22-dien-6-one (
Compound
In summary, compounds
Experimental Section
General Procedures
Column chromatographies were mainly performed by a Buchi Sepacore® Chromatography system equipped with a C-605 pump controller and C-635 detector. Silica gel (200-300 or 300-400 mesh, Marine Chemical Factory, Qingdao), ODS-A (50 μm, YMC), ODS-AQ (50 μm, YMC), and Sephadex LH-20 (Pharmacia) were utilized for various column chromatographies. Optical rotations were recorded through an MCP 500 Modular Circular Polarimeter. 1H NMR and 13C NMR spectra were obtained through Bruker AVANCE III HD 600 and 500 MHz spectrometers, using tetramethylsilane (TMS) as an internal standard. HRESIMS data were acquired by an LTQ Orbitrap Elite spectrometer.
Fungal Materials
G sinense spores were obtained in August 2020 from a Lingzhi base on a mountain of Fujian Province, cultivated by the Food Engineering Research Centre of the State Ministry of Education, Sun Yat-Sen University. They were authenticated by Dr Wenhua He. A voucher sample (HMAS 77207M) has been stored before. 5
Extraction and Isolation
G sinense spore oil (1.21 kg) was extracted from sporoderm-broken spores (4.0 kg) by supercritical fluid with carbon dioxide, as described previously.
17
The G sinense spore oil (1.21 kg) was further separated by silica gel column chromatography (SGCC) and eluted by ligroine, petroleum ether (60-90 °C)/ethyl acetate (9:1), petroleum ether/ethyl acetate (4:1), petroleum ether (60-90 °C)/ethyl acetate (3:2), and dichloromethane/methanol (1:1) to obtain 5 fractions (ZI-ZVI). Fraction ZVI (7.1 g) was chromatographed by SGCC and eluted with a dichloromethane/methanol gradient (99:1-1:1) to obtain 6 fractions (Z1–Z6). Z1 (3.1 g) was loaded on a reversed-phase SGCC eluted with an acetonitrile/water gradient (2:3-4:1) to afford Z1a (30 mg) and Z1b (35 mg). Z1a was chromatographed by SGCC eluted with a petroleum ether (60-90 °C)/ethanol gradient (19:1-93:7) and purified by recrystallization with acetonitrile to produce compound
Cytotoxicity Assay
The obtained ergosterol derivatives
Characterization of Compound 1
White solid;
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231171513 - Supplemental material for Polyhydroxy Sterols Isolated From Ganoderma sinense Spores and Their Cytotoxic Activities
Supplemental material, sj-docx-1-npx-10.1177_1934578X231171513 for Polyhydroxy Sterols Isolated From Ganoderma sinense Spores and Their Cytotoxic Activities by Danhong Lian, Xin Zhong, Lian Li, Li Gu, Yimei Zheng and Xin Liu in Natural Product Communications
Footnotes
Acknowledgments
The authors would like to thank Instrumental Analysis & Research Center, Sun Yat-Sen University, for NMR and HRESI measurements.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by the National Nature Science Foundation (81503333) of the People's Republic of China.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
