Abstract
A series of novel ergosterol peroxide derivatives with acrylate or propionate side chain were synthesized. All compounds
Keywords
Introduction
Cancer is a persistent and serious disease that affects nearly one-third of humanity worldwide.1 Chemotherapy is still one of the most effective methods of cancer treatment today, but adverse side effects limit the clinical use of many drugs.2,3 Therefore, it is an important task to develop new antitumor drugs with high efficiency and low toxicity. Natural products have a long history of medicinal use and are one of the main sources of anti-cancer drugs.4–8 The discovery and structural modification of natural lead compounds is an important field of natural drug research, and is one of the important directions for the development of new anti-tumor drugs.9–11
Ergosterol peroxide (5α,8α-epidioxiergosta-6,22-dien-3β-ol, EP), a natural steroid that extracted from Ganoderma lucidum in the early stage of our group (
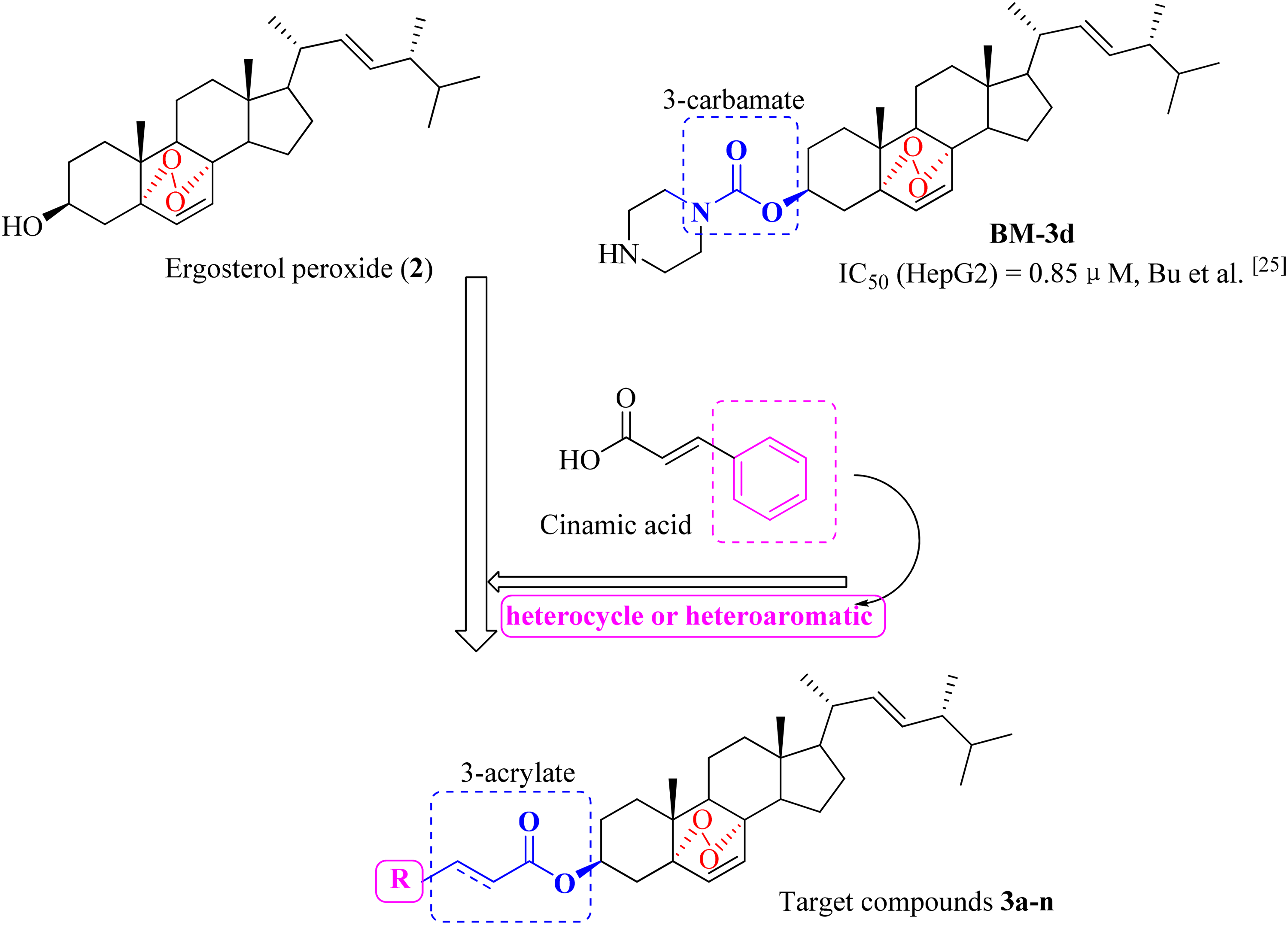
Structure of ergosterol peroxide and its 3-carbamate derivative (
Previously, we reported that the 3-carbamate derivatives of ergosterol peroxide had significant efficacy against several human carcinoma cell lines. Among them, compound
Cinnamic acid is an organic acid isolated from cinnamon or benzoin, contains a ubiquitous α, β-unsaturated acid moiety (acrylic group) presenting potential therapeutic effects in the treatment of cancer (
Results and Discussion
The synthesis process of novel ergosterol peroxide derivatives is shown in Scheme 1. Ergosterol peroxides (

Synthetic pathway to compounds
Compounds
In Vitro Cytotoxicity Activity of Compounds
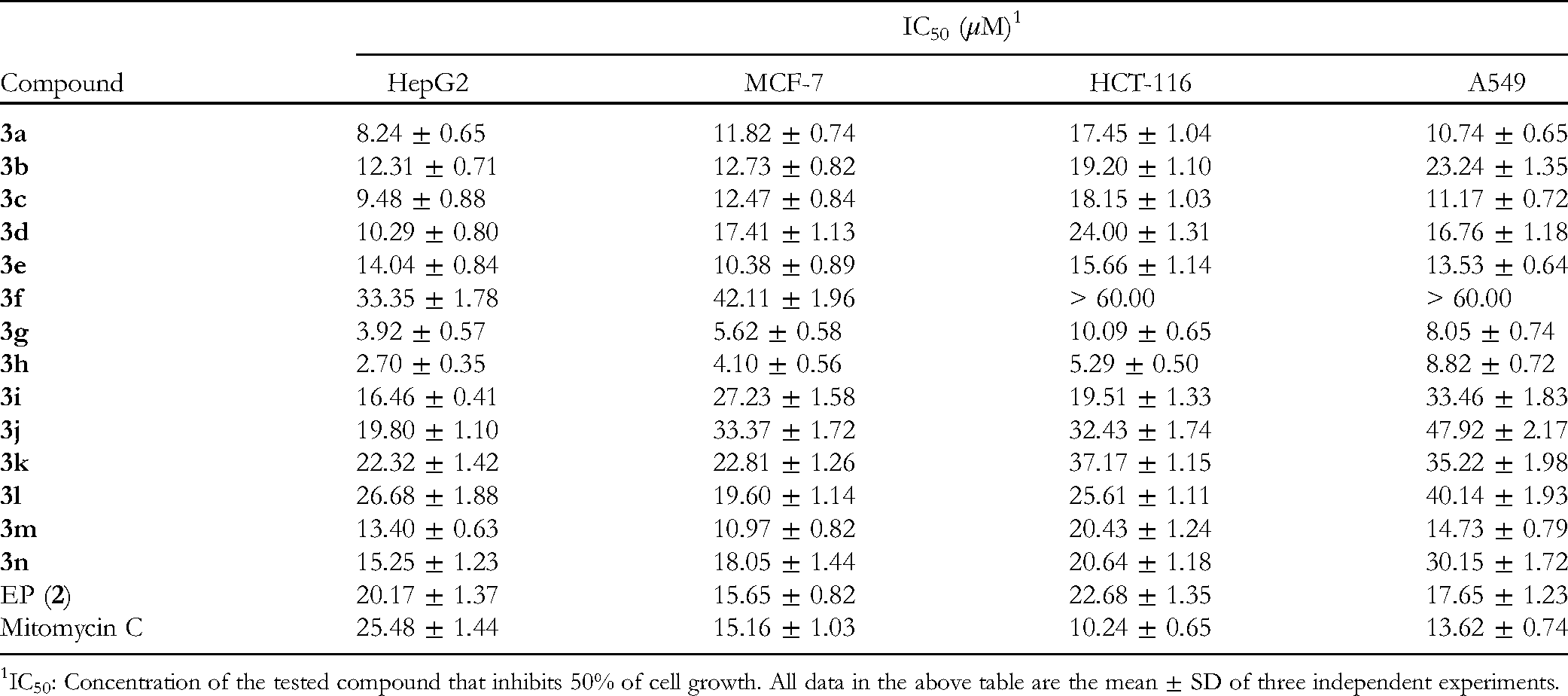
IC50: Concentration of the tested compound that inhibits 50% of cell growth. All data in the above table are the mean ± SD of three independent experiments.
For the HepG2 cell line, most compounds of
The above results suggested some structure-activity relationship (SAR) considerations. First, compounds
Experimental
General
All reagents and solvent were purchased from Energy Chemical (Shanghai, China) and used without further purification. All reactions were monitored by thin-layer chromatography (TLC) silica gel plates (Yinlong, Qingdao, China). The compounds were purified by flash column chromatography (300–400 mesh silica gel, Yinlong, Qingdao, China). HRMS (high-resolution mass spectra) data were carried out by UPLC G2-XS Q-TOF (Waters, USA) and TripleTOF 4600 (AB SCIEX, USA) in a positive ion mode with ESI source. 1H NMR (600 MHz) and 13C NMR (150 MHz) spectra (δ, ppm, J, Hz) were recorded on a AVANCE NEO 600 spectrometers (Bruker, Berlin, Germany). Tetramethylsilane was used as an internal standard.
Synthesis of Ergosterol Peroxide (2 )
Ergosterol (
General Procedure for the Synthesis of Novel Derivatives 3a-n
Ergosterol peroxide (
Ergosterol Peroxide-3-Cinnamic Acetate (3a )
White powder. Yield 85%. 1H NMR (600 MHz, CDCl3) δ 7.67 (1H, s, H-3’), 7.51 (2H, dd, J = 6.7 Hz, H-Ar), 7.39-7.36 (3H, m, H-Ar), 6.52 (1H, d, J = 8.5 Hz, H-7), 6.40 (1H, d, J = 16.0 Hz, H-2’), 6.26 (1H, d, J = 8.5 Hz, H-6), 5.21 (1H, m, H-23), 5.16 (1H, m, H-22), 5.13 (1H, m, H-3), 2.22 (1H, d, J = 13.7 Hz), 2.09 (2H, d, J = 13.6 Hz), 2.04 (2H, d, J = 9.8 Hz), 1.96 (1H, s), 1.85 (1H, d, J = 6.7 Hz), 1.73 (2H, d, J = 14.1 Hz), 1.63 (1H, s), 1.59 (2H, d, J = 10.8 Hz), 1.53 (2H, s), 1.47 (1H, d, J = 6.6 Hz), 1.43 (1H, s), 1.37 (1H, s), 1.25 (s, 2H), 1.24 (s, 1H), 1.00 (3H, d, J = 6.6 Hz, H-18), 0.93 (3H, s, H-28), 0.91 (3H, d, J = 6.8 Hz, H-21), 0.84-0.81 (9H, m, H-26, H-27, H-19); 13C NMR (150 MHz, CDCl3) δ 166.2 (C1’), 144.8 (C3’), 135.4 (C22), 135.3 (C6), 134.6 (Ar-1), 132.5 (C23), 131.1 (C7), 130.4 (Ar-4), 129.0 (Ar-2), 128.2 (Ar-3), 118.6 (C2’), 81.9 (C5), 79.6 (C8), 69.8 (C3), 56.3 (C17), 51.8 (C14), 51.2 (C9), 44.7 (C13), 42.9 (C24), 39.9 (C12), 39.5 (C20), 37.1 (C10), 34.5 (C4), 33.4 (C25), 33.2 (C1), 28.8 (C12), 26.5 (C16), 23.5 (C15), 21.0 (C11), 20.8 (C21), 20.1 (C26), 19.8 (C27), 18.3 (C19), 17.7 (C28), 13.0 (C18); HRMS (ESI) m/z, Calcd. for C37H50O4 [M + H]+: 559.3787, found: 559.3784.
Ergosterol Peroxide-3-(4-Methoxyphenyl)Acrylate (3b )
White powder. Yield 93%. 1H NMR (600 MHz, CDCl3) δ 7.62 (1H, d, J = 16.0 Hz, H-3’), 7.46 (2H, d, J = 8.8 Hz, H-Ar), 6.89 (2H, d, J = 8.8 Hz, H-Ar), 6.52 (1H, d, J = 8.5 Hz, H-7), 6.28 (1H, s, H-2’), 6.25 (1H, d, J = 8.5 Hz, H-6), 5.21 (1H, m, H-23), 5.17 (1H, m, H-22), 5.11 (1H, m, H-3), 3.83 (3H, s, Ar-O-CH3), 2.22 (1H, d, J = 10.2 Hz), 2.12-2.05 (2H, m), 2.03 (2H, d, J = 15.4 Hz), 1.96 (1H, d, J = 6.6 Hz), 1.85 (1H, d, J = 6.7 Hz), 1.72 (2H, d, J = 13.5 Hz), 1.63 (1H, d, J = 9.9 Hz), 1.61-1.56 (2H, m), 1.52 (2H, s), 1.47 (1H, d, J = 6.3 Hz), 1.42 (1H, d, J = 11.6 Hz), 1.35 (1H, d, J = 10.5 Hz), 1.25 (2H, s), 1.23 (1H, s), 1.00 (3H, d, J = 6.6 Hz, H-18), 0.92 (3H, s, H-28), 0.91 (3H, d, J = 6.8 Hz, H-21), 0.82 (9H, dd, J = 9.9, 6.7 Hz, H-26, H-27, H-19); 13C NMR (150 MHz, CDCl3) δ 166.5 (C1’), 161.5 (Ar-4), 144.4 (C3’), 135.3 (C22), 135.3 (C6), 132.4 (C23), 131.0 (C7), 129.8 (Ar-2), 127.4 (Ar-1), 116.1 (C2’), 114.5 (Ar-3), 81.9 (C5), 79.5 (C8), 69.5 (C3), 56.3 (C17), 55.5 (Ar-O-C), 51.8 (C14), 51.2 (C9), 44.7 (C13), 42.9 (C24), 39.9 (C12), 39.5 (C20), 37.1 (C10), 34.5 (C4), 33.5 (C25), 33.2 (C1), 28.8 (C12), 26.6 (C16), 23.5 (C15), 21.0 (C11), 20.8 (C21), 20.1 (C26), 19.8 (C27), 18.3 (C19), 17.7 (C28), 13.0 (C18); HRMS (ESI) m/z, Calcd. for C38H52O5 [M + Na]+: 611.3712, found: 611.3710.
Ergosterol Peroxide-3-(4-Fluorophenyl)Acrylate (3c )
White powder. Yield 84%. 1H NMR (600 MHz, CDCl3) δ 7.62 (1H, d, J = 15.9 Hz, H-3’), 7.52-7.47 (2H, m, H-Ar), 7.06 (2H, t, J = 8.4 Hz, H-Ar), 6.52 (1H, d, J = 8.4 Hz, H-7), 6.31 (1H, d, J = 16.1 Hz, H-2’), 6.25 (1H, d, J = 8.4 Hz, H-6), 5.20 (1H, m, H-22), 5.16 (1H, m, H-23), 5.12 (1H, m, H-3), 2.21 (1H, d, J = 8.5 Hz), 2.08 (2H, d, J = 13.5 Hz), 2.02 (2H, d, J = 16.6 Hz), 1.95 (1H, s), 1.85 (1H, s), 1.74 (1H, s), 1.65 (1H, s), 1.62 (1H, s), 1.59 (1H, s), 1.58 (1H, s), 1.52 (2H, s), 1.46 (1H, d, J = 6.6 Hz), 1.42 (1H, s), 1.36 (1H, s), 1.24 (2H, d, J = 10.4 Hz), 1.22 (s, 1H), 1.00 (3H, d, J = 6.6 Hz, H-18), 0.93 (3H, s, H-28), 0.91 (3H, d, J = 6.6 Hz, H-21), 0.83 (9H, d, J = 10.7 Hz, H-26, H-27, H-19); 13C NMR (150 MHz, CDCl3) δ 166.0 (C1’), 163.1 (Ar-4), 143.4 (C3’), 135.3 (C22), 135.2 (C6), 132.4 (C23), 131.1 (C7), 130.8 (Ar-2), 130.0 (Ar-1), 118.3 (C2’), 116.2 (Ar-3), 81.9 (C5), 79.5 (C8), 69.8 (C3), 56.3 (C17), 51.7 (C14), 51.2 (C9), 44.7 (C13), 42.9 (C24), 39.8 (C12), 39.4 (C20), 37.1 (C10), 34.5 (C4), 33.4 (C25), 33.2 (C1), 28.8 (C12), 26.5 (C16), 23.5 (C15), 21.0 (C11), 20.7 (C21), 20.1 (C26), 19.8 (C27), 18.2 (C19), 17.7 (C28), 13.0 (C18); HRMS (ESI) m/z, Calcd. for C37H49FO4 [M + Na]+: 599.3513, found: 599.3519.
Ergosterol Peroxide-3-(4-Chlorophenyl)Acrylate (3d )
White powder. Yield 80%. 1H NMR (600 MHz, CDCl3) δ 7.61 (1H, d, J = 16.0 Hz, H-3’), 7.44 (2H, d, J = 8.5 Hz, H-Ar), 7.35 (2H, d, J = 8.5 Hz, H-Ar), 6.52 (1H, d, J = 8.5 Hz, H-7), 6.36 (1H, d, J = 15.9 Hz, H-2’), 6.25 (1H, d, J = 8.5 Hz, H-6), 5.21 (1H, m, H-22), 5.17 (1H, m, H-23), 5.13 (1H, m, H-3), 2.22 (1H, d, J = 8.4 Hz), 2.08 (2H, d, J = 14.1 Hz), 2.03 (2H, d, J = 16.0 Hz), 1.96 (1H, s), 1.86 (1H, d, J = 6.7 Hz), 1.73 (2H, d, J = 13.9 Hz), 1.65 (1H, s), 1.61 (2H, d, J = 16.9 Hz), 1.53 (2H, s), 1.47 (1H, d, J = 6.6 Hz), 1.43 (1H, s), 1.36 (1H, d, J = 11.6 Hz), 1.25 (2H, d, J = 9.6 Hz), 1.23 (1H, s), 1.01 (3H, d, J = 6.6 Hz, H-18), 0.93 (3H, s, H-28), 0.91 (3H, d, J = 6.8 Hz, H-21) 0.87-0.79 (9H, m, H-26, H-27, H-19); 13C NMR (150 MHz, CDCl3) δ 165.9 (C1’), 143.3 (Ar-4), 136.3 (C3’), 135.4 (C22), 135.2 (C6), 133.2 (C23), 132.5 (C7), 131.1 (Ar-1), 129.3 (Ar-2, Ar-3), 119.3 (C2’), 81.9 (C5), 79.6 (C8), 69.9 (C3), 56.4 (C17), 51.8 (C14), 51.3 (C9), 44.7 (C13), 42.9 (C24), 39.9 (C12), 39.5 (C20), 37.2 (C10), 34.5 (C4), 33.5 (C25), 33.2 (C1), 28.8 (C12), 26.6 (C16), 23.6 (C15), 21.0 (C11), 20.8 (C21), 20.1 (C26), 19.8 (C27), 18.3 (C19), 17.7 (C28), 13.0 (C18); HRMS (ESI) m/z, Calcd. for C37H49ClO4 [M + H]+: 593.3398, found: 593.3407.
Ergosterol Peroxide-3-(4-Trifluorophenyl)Acrylate (3e )
White powder. Yield 82%. 1H NMR (600 MHz, CDCl3) δ 7.67 (1H, d, J = 16.0 Hz, H-3’), 7.63 (2H, d, J = 8.1 Hz, H-Ar), 7.60 (2H, d, J = 8.3 Hz, H-Ar), 6.52 (1H, d, J = 8.5 Hz, H-7), 6.46 (1H, d, J = 16.0 Hz, H-2’), 6.25 (1H, d, J = 8.5 Hz, H-6), 5.21 (1H, d, m, H-22), 5.16 (1H, m, H-23), 5.14 (1H, m, H-3), 2.22 (1H, s), 2.10 (1H, s), 2.07 (1H, s), 2.04 (2H, d, J = 13.2 Hz), 1.96 (1H, d, J = 6.8 Hz), 1.85 (1H, d, J = 6.8 Hz), 1.74 (2H, d, J = 14.3 Hz), 1.65 (1H, d, J = 15.7 Hz), 1.60 (2H, s), 1.53 (2H, s), 1.47 (1H, d, J = 6.6 Hz), 1.41 (1H, s), 1.37 (1H, s), 1.25 (2H, d, J = 10.1 Hz), 1.22 (1H, s), 1.00 (3H, d, J = 6.5 Hz, H-18), 0.93 (3H, s, H-28), 0.91 (3H, d, J = 6.8 Hz, H-21), 0.85-0.81 (9H, m, H-26, H-27, H-19); 13C NMR (150 MHz, CDCl3) δ 165.5 (C1’), 142.7 (C3’), 137.9 (C22), 135.2 (C6), 135.1 (C23), 132.4 (Ar-1), 131.8 (C7), 131.0 (Ar-4), 128.2 (Ar-2), 125.9 (Ar-3), 124.8 (CF3), 121.1 (C2’), 81.8 (C5), 79.4 (C8), 70.0 (C3), 56.2 (C17), 51.6 (C14), 51.1 (C9), 44.6 (C13), 42.8 (C24), 39.7 (C12), 39.3 (C20), 37.0 (C10), 34.3 (C4), 33.3 (C25), 33.1 (C1), 28.7 (C12), 26.4 (C16), 23.4 (C15), 20.9 (C11), 20.6 (C21), 20.0 (C26), 19.6 (C27), 18.1 (C19), 17.6 (C28), 12.9 (C18); HRMS (ESI) m/z, Calcd. for C38H49F3O4 [M + Na]+: 649.3481, found: 649.3473.
Ergosterol Peroxide-3-(Naphthalen-2-yl)Acrylate (3f )
White powder. Yield 92%. 1H NMR (600 MHz, CDCl3) δ 8.50 (1H, d, J = 15.7 Hz, H-Ar), 8.19 (1H, d, J = 8.4 Hz, H-Ar), 7.88 (2H, t, J = 8.5 Hz, H-Ar), 7.74 (1H, d, J = 7.2 Hz, H-Ar), 7.57 (1H, d, J = 7.8 Hz, H-Ar), 7.53 (1H, d, J = 7.4 Hz, H-Ar), 7.49 (1H, d, J = 7.6 Hz, H-Ar), 6.53 (1H, d, J = 8.5 Hz, H-7), 6.49 (1H, d, J = 15.7 Hz, H-2’), 6.27 (1H, d, J = 8.5 Hz, H-6), 5.22 (1H, m, H-22), 5.18 (1H, m, H-23), 5.17 (1H, m, H-3), 2.26 (1H, s), 2.14 (1H, s), 2.09 (2H, d, J = 10.5 Hz), 1.96 (1H, s), 1.86 (1H, d, J = 6.7 Hz), 1.76 (2H, d, J = 14.0 Hz), 1.70 (1H, s), 1.61 (2H, s), 1.55 (2H, s), 1.47 (1H, s), 1.44 (1H, s), 1.40 (1H, s), 1.26 (2H, s), 1.23 (1H, s), 1.01 (3H, d, J = 6.5 Hz, H-18), 0.95 (3H, s, H-28), 0.92 (3H, d, J = 6.7 Hz, H-21), 0.83 (9H, t, J = 8.2 Hz, H-26, H-27, H-29); 13C NMR (150 MHz, CDCl3) δ 166.1 (C1’), 141.8 (C3’), 135.4 (C22), 135.3 (C6), 133.9 (Ar), 132.5 (Ar), 131.6 (C23), 131.1 (Ar), 130.6 (C7), 128.9 (Ar), 127.0 (Ar), 126.4 (Ar), 125.6 (Ar), 125.2 (Ar), 123.6 (Ar), 123.6 (Ar), 121.4 (C2’), 82.0 (C5), 79.6 (C8), 70.0 (C3), 56.4 (C17), 51.8 (C14), 51.3 (C9), 44.8 (C13), 43.0 (C24), 39.9 (C12), 39.5 (C20), 37.2 (C10), 34.6 (C4), 33.5 (C25), 33.2 (C1), 28.8 (C12), 26.6 (C16), 23.6 (C15), 21.0 (C11), 20.8 (C21), 20.1 (C26), 19.8 (C27), 18.3 (C19), 17.7 (C28), 13.1 (C18); HRMS (ESI) m/z, Calcd. for C41H52O4 [M + H]+: 609.3944, found: 609.3951.
Ergosterol Peroxide-3-(Pyridin-4-yl)Acrylate (3g)
White powder. Yield 81%. 1H NMR (600 MHz, CDCl3) δ 8.77 (1H, s, H-Ar), 8.62 (1H, s, H-Ar), 7.87 (1H, d, J = 7.9 Hz, H-3’), 7.65 (1H, d, J = 16.0 Hz, H-Ar), 7.38 (1H, s, H-Ar), 6.53 (1H, d, J = 8.4 Hz, H-7), 6.48 (1H, d, J = 15.7 Hz, H-2’), 6.26 (1H, d, J = 8.4 Hz, H-6), 5.21 (1H, m, H-22), 5.16 (1H, m, H-23), 5.14 (1H, m, H-3), 2.22 (1H, s), 2.09 (2H, d, J = 24.8 Hz), 2.03 (2H, d, J = 15.8 Hz), 1.96 (1H, s), 1.85 (1H, d, J = 6.7 Hz), 1.74 (2H, d, J = 13.8 Hz), 1.65 (1H, s), 1.60 (2H, s), 1.53 (2H, s), 1.47 (1H, d, J = 6.3 Hz), 1.41 (1H, s), 1.35 (1H, s), 1.25 (2H, d, J = 7.8 Hz), 1.23 (s, 1H), 1.00 (3H, d, J = 6.6 Hz, H-18), 0.93 (3H, s, H-28), 0.91 (3H, d, J = 6.8 Hz, H-21), 0.84-0.81 (9H, m, H-26, H-27, H-19); 13C NMR (150 MHz, CDCl3) δ 165.4 (C1’), 150.3 (Ar), 149.1 (C3’), 140.6 (Ar), 135.3 (C22), 132.5 (C6), 131.1 (C23), 124.2 (Ar), 121.3 (C2’), 81.9 (C5), 79.6 (C8), 70.2 (C3), 56.3 (C17), 51.7 (C14), 51.2 (C9), 44.7 (C13), 42.9 (C24), 39.9 (C12), 39.4 (C20), 37.1 (C10), 34.5 (C4), 33.4 (C25), 33.2 (C1), 28.8 (C12), 26.5 (C16), 23.5 (C15), 21.0 (C11), 20.8 (C21), 20.1 (C26), 19.8 (C27), 18.3 (C19), 17.8 (C28), 13.0 (C18); HRMS (ESI) m/z, Calcd. for C36H49NO4 [M + H]+: 560.3740, found: 560.3738.
Ergosterol Peroxide-3-(Pyridin-3-yl)Acrylate (3h )
White powder. Yield 95%. 1H NMR (600 MHz, CDCl3) δ 7.28 (2H, s, H-3’,H-2’), 7.19 (4H, d, J = 7.7 Hz, H-Ar), 6.50 (1H, d, J = 8.5 Hz, H-7), 6.21 (1H, d, J = 8.5 Hz, H-6), 5.21 (1H, m, H-22), 5.16 (1H, m, H-23), 4.99 (1H, m, H-3), 2.08 (1H, d, J = 7.8 Hz), 2.01 (2H, s), 1.98 (1H, s), 1.95 (2H, s), 1.86 (1H, s), 1.76 (1H, s), 1.67 (2H, d, J = 10.0 Hz), 1.57 (2H, d, J = 4.5 Hz), 1.51 (1H, s), 1.50 (2H, s), 1.38 (1H, s), 1.25 (2H, d, J = 7.8 Hz), 1.23 (s, 1H), 1.00 (3H, d, J = 6.6 Hz, H-18), 0.91 (3H, d, J = 6.8 Hz, H-28), 0.88 (3H, s, H-21), 0.84-0.81 (9H, m, H-26, H-27, H-19); 13C NMR (150 MHz, CDCl3) δ 172.0 (C1’), 140.6 (Ar), 135.2 (C3’), 135.1 (Ar), 132.3 (C22), 130.9 (C6), 128.5 (C23), 128.3 (Ar), 126.2 (C2’), 81.7 (C5), 79.4 (C8), 69.5 (C3), 56.2 (C17), 51.6 (C14), 51.0 (C9), 44.6 (C13), 42.8 (C24), 39.7 (C12), 39.3 (C20), 37.0 (C10), 34.3 (C4), 33.1 (C25), 33.1 (C1), 28.6 (C12), 26.2 (C16), 23.4 (C15), 20.9 (C11), 20.6 (C21), 20.0 (C26), 19.6 (C27), 18.1 (C19), 17.8 (C28), 12.9 (C18); HRMS (ESI) m/z, Calcd. for C36H49NO4 [M + H]+: 560.3740, found: 560.3749.
Ergosterol Peroxide-3-(Furan-2- yl)Acrylate (3i )
White powder. Yield 93%. 1H NMR (600 MHz, CDCl3) δ 8.53 (2H, d, H-Ar), 7.18 (1H, s, H-Ar), 6.51 (1H, d, J = 8.5 Hz, H-7), 6.21 (1H, d, J = 8.5 Hz, H-6), 5.21 (1H, m, H-22), 5.16 (1H, m, H-23), 4.99 (1H, m, H-3), 2.95 (1H, s, H-3’), 2.61 (1H, s, H-2’), 2.07 (1H, d, J = 10.1 Hz), 2.01 (2H, d, J = 14.4 Hz), 1.97 (2H, d, J = 12.2 Hz), 1.84 (1H, s), 1.76 (1H, s), 1.68 (2H, d, J = 13.6 Hz), 1.61 (1H, s), 1.57 (2H, d, J = 11.2 Hz), 1.50 (2H, d, J = 9.0 Hz), 1.47 (1H, d, J = 6.7 Hz), 1.42 (1H, s), 1.36 (1H, s), 1.25 (2H, s), 1.23 (1H, s), 1.00 (3H, d, J = 6.6 Hz, H-18), 0.91 (3H, d, J = 6.8 Hz, H-28), 0.88 (3H, s, H-21), 0.83 (9H, m, H-26, H-27, H-29); 13C NMR (150 MHz, CDCl3) δ 171.2 (C1’), 149.4 (Ar), 135.2 (Ar), 135.0 (C22), 132.3 (C6), 131.0 (C23), 124.1 (C2’), 81.7 (C5), 79.4 (C8), 70.0 (C3), 56.2 (C17), 51.6 (C14), 51.0 (C9), 44.6 (C13), 42.8 (C24), 39.7 (C12), 39.3 (C20), 36.9 (C10), 34.6 (C4), 33.1 (C25), 33.1 (C1), 28.6 (C12), 26.2 (C16), 23.4 (C15), 20.9 (C11), 20.6 (C21), 20.0 (C26), 19.6 (C27), 18.1 (C19), 17.6 (C28), 12.9 (C18); HRMS (ESI) m/z, Calcd. for C35H48O5 [M + H]+: 549.3580, found: 549.3575.
Ergosterol Peroxide-3-(Furan-3- yl)Acrylate (3j )
White powder. Yield 86%. 1H NMR (600 MHz, CDCl3) δ 7.63 (1H, s, H-Ar), 7.55 (1H, d, J = 15.8 Hz, H-3’), 7.42 (1H, s, H-Ar), 6.58 (1H, s, Ar), 6.52 (1H, d, J = 8.5 Hz, H-7), 6.25 (1H, d, J = 8.5 Hz, H-6), 6.12 (1H, d, J = 15.8 Hz, H-2’), 5.22 (1H, m, H-22), 5.15 (1H, m, H-23), 5.11 (1H, m, H-3), 2.21 (1H, d, J = 8.5 Hz), 2.07 (2H, d, J = 13.9 Hz), 2.02 (2H, d, J = 12.3 Hz), 1.96 (1H, d, J = 6.6 Hz), 1.85 (1H, d, J = 6.8 Hz), 1.72 (2H, d, J = 13.8 Hz), 1.63 (1H, s), 1.59 (2H, d, J = 11.0 Hz), 1.52 (2H, s), 1.47 (1H, d, J = 6.5 Hz), 1.42 (1H, s), 1.36 (1H, d, J = 10.6 Hz), 1.25 (2H, s), 1.23 (1H, s) 1.00 (3H, d, J = 6.6 Hz, H-18), 0.92 (3H, s, H-28), 0.91 (3H, d, J = 6.8 Hz, H-21), 0.83 (9H, dd, J = 9.9, 6.7 Hz, H-26, H-27, H-19); 13C NMR (150 MHz, CDCl3) δ 166.0 (Ar), 144.4 (Ar), 135.2 (C3’), 135.1 (C22), 134.6 (C6), 132.3 (C23), 130.9 (C7), 122.6 (Ar), 118.2 (2’), 107.4 (Ar), 81.8 (C5), 79.4 (C8), 69.5 (C3), 56.2 (C17), 51.6 (C14), 51.0 (C9), 44.6 (C13), 42.8 (C24), 39.8 (C12), 39.3 (C20), 37.0 (C10), 34.3 (C4), 33.3 (C25), 33.1 (C1), 28.7 (C12), 26.4 (C16), 23.4 (C15), 20.9 (C11), 20.6 (C21), 20.0 (C26), 19.7 (C27), 18.1 (C19), 17.6 (C28), 12.9 (C18); HRMS (ESI) m/z, Calcd. for C35H48O5 [M + Na]+: 571.3399, found: 571.3409.
Ergosterol Peroxide-3-(Thiophen-2- yl)Acrylate (3k )
White powder. Yield 83%. 1H NMR (600 MHz, CDCl3) δ 7.64 (1H, d, J = 15.9 Hz, H-3’), 7.47 (1H, s, H-Ar), 7.32 (1H, s, H-Ar), 7.28 (1H, s, H-Ar), 6.52 (1H, d, J = 8.5 Hz, H-7), 6.24 (1H, d, J = 8.5 Hz, H-6), 6.22 (1H, d, J = 15.9 Hz, H-2’), 5.21 (1H, m, H-22), 5.17 (1H, m, H-23), 5.13 (1H, m, H-3), 2.23-2.20 (1H, m), 2.07 (1H, d, J = 13.2 Hz), 2.03 (2H, d, J = 15.0 Hz), 1.95 (1H, s), 1.85 (1H, d, J = 6.7 Hz), 1.73 (2H, s), 1.64 (1H, s), 1.59 (2H, d, J = 11.2 Hz), 1.52 (2H, s), 1.48 (1H, s), 1.43 (1H, s), 1.37 (1H, s), 1.26 (2H, s), 1.23 (1H, s) 1.00 (3H, d, J = 6.7 Hz, H-18), 0.92 (3H, s, H-28), 0.89 (3H, d, J = 6.9 Hz, H-21), 0.83 (9H, m, H-26, H-27, H-19); 13C NMR (150 MHz, CDCl3) δ 166.3 (C1’), 138.1 (Ar), 137.7 (C3’), 135.2 (C22), 135.2 (C6), 132.4 (C23), 131.0 (C7), 127.9 (Ar), 126.9 (Ar), 125.2 (Ar), 118.2 (C2’), 81.8 (C5), 79.4 (C8), 69.6 (C3), 56.3 (C17), 51.7 (C14), 51.1 (C9), 44.6 (C13), 42.8 (C24), 39.7 (C12), 39.4 (C20), 37.0 (C10), 34.4 (C4), 33.3 (C25), 33.1 (C1), 28.6 (C12), 26.4 (C16), 23.4 (C15), 20.9 (C11), 20.7 (C21), 20.0 (C26), 19.7 (C27), 18.1 (C19), 17.6 (C28), 12.9 (C18); HRMS (ESI) m/z, Calcd. for C35H48O4S [M + Na]+: 587.3171, found: 587.3179.
Ergosterol Peroxide-3-(Thiophen-3- yl)Acrylate (3l )
White powder. Yield 94%. 1H NMR (600 MHz, CDCl3) δ 7.65 (1H, d, J = 15.9 Hz, H-3’), 7.48 (1H, s, H-Ar), 7.33 (1H, s, H-Ar), 7.28 (1H, s, H-Ar), 6.52 (1H, d, J = 8.5 Hz, H-7), 6.25 (1H, d, J = 8.5 Hz, H-6), 6.22 (1H, d, J = 15.8 Hz, H-2’), 5.22 (1H, m, H-23), 5.17 (1H, m, H-22), 5.11 (1H, m, H-3), 2.21 (1H, d, J = 10.2 Hz), 2.08 (2H, d, J = 13.8 Hz), 2.03 (2H, d, J = 14.4 Hz), 1.96 (1H, d, J = 6.6 Hz), 1.85 (1H, d, J = 6.7 Hz), 1.73 (2H, d, J = 13.6 Hz), 1.63 (1H, s), 1.60-1.56 (2H, m), 1.52 (2H, s), 1.47 (1H, d, J = 6.5 Hz), 1.41 (1H, s), 1.36 (1H, d, J = 10.9 Hz), 1.25 (2H, s), 1.23 (1H, s) 1.00 (3H, d, J = 6.6 Hz, H-18), 0.93 (3H, s, H-28), 0.91 (3H, d, J = 6.8 Hz, H-21), 0.84-0.81 (9H, m, H-26, H-27, H-19); 13C NMR (150 MHz, CDCl3) δ 166.3 (C1’), 138.1 (Ar), 137.6 (C3’), 135.2 (C22), 135.1 (C6), 132.3 (C23), 130.9 (C7), 128.0 (Ar), 126.9 (Ar), 125.1 (Ar), 118.1 (C2’), 81.8 (C5), 79.4 (C8), 69.6 (C3), 56.2 (C17), 51.6 (C14), 51.0 (C9), 44.6 (C13), 42.8 (C24), 39.7 (C12), 39.3 (C20), 37.0 (C10), 34.3 (C4), 33.3 (C25), 33.1 (C1), 28.6 (C12), 26.4 (C16), 23.4 (C15), 20.9 (C11), 20.6 (C21), 20.0 (C26), 19.6 (C27), 18.1 (C19), 17.6 (C28), 12.9 (C18); HRMS (ESI) m/z, Calcd. for C35H48O4S [M + Na]+: 581.3171, found: 581.3179.
Ergosterol Peroxide-3-Phenylpropanoate (3m )
White powder. Yield 85%. 1H NMR (600 MHz, CDCl3) δ 7.63 (1H, s, Ar), 7.55 (1H, d, J = 15.9 Hz, H-Ar), 7.42 (1H, s, H-Ar), 6.57 (1H, s, H-Ar), 6.52 (1H, d, J = 8.5 Hz, H-7), 6.24 (1H, d, J = 8.5 Hz, H-6), 6.12 (1H, d, J = 15.8 Hz, H-Ar), 5.21 (1H, m, H-22), 5.16 (1H, m, H-23), 5.12 (1H, m, H-3), 2.20 (1H, d, J = 10.3 Hz), 2.09 (2H, d, J = 11.8 Hz), 2.02 (2H, d, J = 11.7 Hz), 1.95 (1H, s), 1.85 (1H, d, J = 6.7 Hz), 1.73 (2H, s), 1.63 (2H, s), 1.60 (2H, d, J = 6.4 Hz, H-3’), 1.52 (2H, s), 1.47 (2H, d, J = 6.5 Hz), 1.41 (1H, s), 1.34 (1H, s), 1.25 (2H, s), 1.23 (2H, s, H-2’), 1.21 (1H, s), 1.00 (3H, d, J = 6.6 Hz, H-18), 0.92 (3H, s, H-28), 0.91 (3H, d, J = 6.8 Hz, H-21), 0.84-0.81 (9H, m, H-26, H-27, H-19); 13C NMR (150 MHz, CDCl3) δ 166.0 (C1’), 144.4 (Ar), 135.2 (C22), 134.6 (C6), 132.3 (C23), 130.9 (C7), 122.6 (Ar), 118.2 (Ar), 107.4 (Ar), 81.8 (C5), 79.4 (C8), 69.5 (C3), 56.2 (C17), 51.6 (C14), 51.0 (C9), 44.6 (C13), 42.8 (C24), 39.7 (C12), 39.3 (C20), 37.0 (C10), 34.3 (C4), 33.3 (C25), 33.1 (C1), 29.7 (C3’), 29.3 (C2’), 28.6 (C12), 26.4 (C16), 23.4 (C15), 20.9 (C11), 20.6 (C21), 20.0 (C26), 19.6 (C27), 18.1 (C19), 17.6 (C28), 12.9 (C18); HRMS (ESI) m/z, Calcd. for C37H52O4 [M + H]+: 561.3944, found: 561.3943.
Ergosterol Peroxide-3-(Pyridine-4-yl)Propionate (3n )
White powder. Yield 89%. 1H NMR (600 MHz, CDCl3) δ 8.67 (2H, s, H-Ar), 7.40 (2H, s, H-Ar), 6.57 (1H, d, J = 8.5 Hz, H-7), 6.53 (1H, d, J = 8.5 Hz, H-6), 5.21 (1H, m, H-22), 5.16 (1H, m, H-23), 5.14 (1H, m, H-3), 2.22 (1H, s), 2.11 (2H, d, J = 11.9 Hz), 2.03 (2H, d, J = 16.5 Hz), 1.96 (1H, s), 1.86 (1H, d, J = 6.7 Hz), 1.74 (2H, d, J = 14.1 Hz), 1.68 (2H, d, J = 9.3 Hz, H-3’), 1.60 (1H, s), 1.58 (1H, d, J = 7.3 Hz), 1.53 (2H, s), 1.47 (2H, d, J = 6.6 Hz), 1.40 (1H, s), 1.35 (1H, s), 1.26 (2H, s), 1.24 (2H, d, J = 9.9 Hz, H-2’), 1.23 (1H, s), 1.00 (3H, d, J = 6.6 Hz, H-18), 0.93 (3H, s, H-28), 0.91 (3H, d, J = 6.9 Hz, H-21), 0.84-0.81 (9H, m, H-26, H-27, H-19); 13C NMR (150 MHz, CDCl3) δ 165.0 (C1’), 149.9 (Ar), 141.4 (Ar), 135.2 (C22), 132.4 (C6), 131.1 (C23), 123.7 (C7), 122.1 (Ar), 81.8 (C5), 79.5 (C8), 70.3 (C3), 56.2 (C17), 51.6 (C14), 51.0 (C9), 44.6 (C13), 42.8 (C24), 39.7 (C12), 39.3 (C20), 37.0 (C10), 34.3 (C4), 33.2 (C25), 33.1 (C1), 29.7 (C3’), 29.3 (C2’), 28.6 (C12), 26.3 (C16), 23.4 (C15), 20.9 (C11), 20.6 (C21), 20.0 (C26), 19.6 (C27), 18.1 (C19), 17.6 (C28), 12.9 (C18); HRMS (ESI) m/z, Calcd. for C36H51NO4 [M + H]+: 562.3896, found: 562.3887.
In Vitro Cytotoxic Activity
Cytotoxic activities of all synthesized compounds against tumor cells were tested by MTT assay. All human cancer cell lines were purchased from the Cell Bank of Chinese Academy of Sciences (Shanghai, China). Cells at logarithmic growth phase were transferred to 96-well plates (1 × 104 cells/well) for 12 h. After incubation, cells were treated with a graded concentration of compound from 1 μM to 60 μM, and incubation was continued for 48 h. Afterward, cells were treated with 10 μL of MTT solution (Sigma Chemical Co., Ltd, USA) for 2 h, and then 100 μL of DMSO was added in place of this solution. The absorbance was measured at 490 nm on a microplate reader. The IC50 values are expressed as averaged ± SD of three independent experiments.
Conclusions
A series of novel ergosterol peroxide derivatives with acrylate or propionate side chain were synthesized and characterized. All compounds were evaluated for their in vitro cytotoxic activities against four human carcinoma cell lines (HepG2, MCF-7, A549, and HCT-116). The MTT results showed that compound
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221143634 - Supplemental material for Synthesis and Cytotoxic Activity of Novel Ergosterol Peroxide Derivatives with Acrylate or Propionate Side Chain
Supplemental material, sj-docx-1-npx-10.1177_1934578X221143634 for Synthesis and Cytotoxic Activity of Novel Ergosterol Peroxide Derivatives with Acrylate or Propionate Side Chain by Ming Bu, Zhiguo Zhang, Gang Li, Chunhua Xie, Xiaohui Du, Guofang Ma and Hongling Li in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X221143634 - Supplemental material for Synthesis and Cytotoxic Activity of Novel Ergosterol Peroxide Derivatives with Acrylate or Propionate Side Chain
Supplemental material, sj-docx-2-npx-10.1177_1934578X221143634 for Synthesis and Cytotoxic Activity of Novel Ergosterol Peroxide Derivatives with Acrylate or Propionate Side Chain by Ming Bu, Zhiguo Zhang, Gang Li, Chunhua Xie, Xiaohui Du, Guofang Ma and Hongling Li in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Qiqihar Science and Technology Bureau under Grant (grant number LHYD-202015) and Optional project of Qiqihar Academy of Medical Sciences (grant number QMSI2022Z-01).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
