Abstract
One new steroid
The filamentous fungus Aspergillus flavus is one of the most important species in the Aspergillus genus and is distributed worldwide as a prevalent aflatoxin-producing food and feed contaminant.
1
Meanwhile, it can also produce a wide variety of natural products, such as stilbenoids,
2
diketopiperazine,
3
lactones,
4
and steroids
5
. As a widespread class of natural compounds occurring in animals, plants, and microorganism, steroids comprise a wide repertoire of structurally related natural compounds with important functions such as physiological regulators, hormones, and pro-vitamins.
6-8
In our search for bioactive compounds from soil-derived fungus, the strain of JDW-1 was isolated from the rhizosphere soil of economic plant Cinnamomum bodinieri Levl. Its extract exhibited cytotoxicity against the P388 cells in preactive screening. Investigation of the secondary metabolites led to the isolation of a new steroid

The structures of Compounds 1 to
Compound
1H (600 MHz, CDCl3) and 13C (150 MHz, CDCl3) NMR Data for Compound 1.

NMR, nuclear magnetic resonance.
Spectra were recorded at 600 MHz for 1H NMR using dimethyl sulfoxide as solvent and tetramethylsilane (TMS) as internal standard.
The planar structure of

Selected key 1H-1H COSY and HMBC correlations of 1.
The relative configuration and absolute configuration of

The Oak Ridge Thermal Ellipsoid Plot of single-crystal X-ray data of 1.
The cytotoxicities of Compounds
The Cytotoxicity of Compounds 1 and 2.

aADM = doxorubicin (positive control).
The DPPH Radical Scavenging Activities of Compounds 3 to
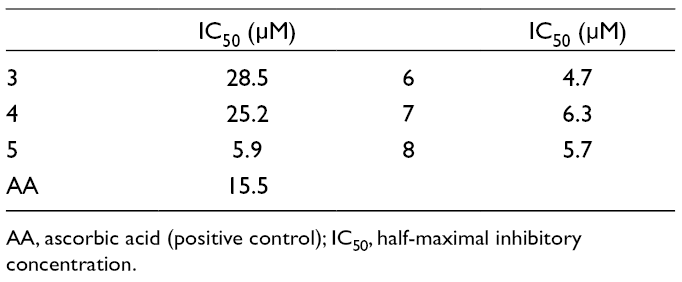
AA, ascorbic acid (positive control); IC50, half-maximal inhibitory concentration.
Steroids, a widespread class of natural compounds occurring in animals, plants, and fungi, had shown great therapeutic value for a broad array of pathologies.
16
Natural steroids had been showing notable cytotoxic activities, and were potential lead compounds and probed to disclose new mechanisms of anticancer.
17,18
In our research, the compound
Experimental
General
Colum chromatography: silica gel and silica gel GF254 (SiO2; 200-300 mesh, 10-40 µm, Qingdao Marine Chemical Inc., China). Sephadex LH-20 (GE Healthcare, Sweden). Semi-prep: high-performance liquid chromatography (HPLC) was performed using an ODS column (YMC-pack ODS-A, 10 × 250 mm, 5 µm). UV spectra were recorded on Waters 2487. IR spectra were taken on a NICOLET NEXUS 470 spectrophotometer in KBr discs. 1H- and 13C-NMR, DEPT, and 2D NMR spectra: JEOL Eclipse-600 spectrometer. High-resolution electrospray ionization mass spectrometry data were obtained using a Thermo Scientific LTQ Orbitrap XL mass spectrometer.
Strain Identification
Fungal identification was performed based on sequencing of the ITS region. The nucleotide BLAST search result showed that the sequence was the most similar to the sequence of Aspergillus flavus.
Extraction and Isolation
The media contained yeast powder, peptone, glucose, and malt extract dissolved in 1 L water with pH 7. The flasks were cultured with standing at 28°C for 30 days. All extracts were evaporated under reduced pressure and concentrated to give a crude extract (20.0 g). The extract was separated by ODS in the gradient elution step of MeOH-H2O. Fraction 7 was further separated on a Sephadex LH-20 column to provide eight fractions. MPLC was applied to Fraction 7 to 7 to exclude the impurities, and isolated by HPLC employing isocratic elution with CH3CN-H2O (65:35; v/v) to obtain Compounds
Biological Assay
The new compound was evaluated for its cytotoxicity against A-549 and Hela cell lines by the MTT method ,
19
and HCT-116, MGC-803, and HO-8910 cell lines by the SRB method.
20
The new compound
X-Ray Crystallographic Analysis of Compound 1
Single-crystal X-ray diffraction data were collected on an Agilent Gemini Ultra diffractometer with CuKα radiation (λ = 1.54184 Å). The structure was solved by direct methods (SHELXS-97) and refined using full-matrix least-squares difference Fourier techniques. Carbon, oxygen, and nitrogen atoms were refined anisotropically. Hydrogen atoms were either refined freely with isotropic displacement parameters or positioned with an idealized geometry and refined riding on their parent C atoms. Crystals suitable for X-ray diffraction were obtained by slow evaporation of a solution in MeOH-H2O. Crystallographic data (excluding structure factors) for
Compound 1
Amorphous powder.
[α]
UV (MeOH) λ max: (log ε) 210 (2.32) nm.
IR (KBr) ν max 3422, 2955, 1674 cm–1.
1H NMR (600 MHz, CDCl3): Table 1.
13C NMR (150 MHz, CDCl3): Table 1.
HRESIMS: m/z [M + H]+ m/z 445.3314 (calcd for C28H44O4 + H, 445.3312).
Crystallographic Data
Monoclinic, C28H44O4, space group P21 with a = 10.8171(4) Å, b = 6.2921(3) Å, c = 15.0930(5) Å, α = 90°, β = 96°, γ = 90°, V = 1021.46(7) Å3, Z = 2, T = 291(2) K, Dc = 1.360 g/cm3, μ = 0.781 mm−1, and F (000) =440. Crystal size: 0.32 × 0.2 × 0.2 mm3. Independent reflections: 3227 with R int = 0.0228. The final agreement factors are R 1 = 0.0476 and w R 2 = 0.1282 [I > 2σ (I)]. The absolute configuration was determined by Flack parameter x = –0.08(19).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the PhD Research Funding of Jinggangshan University (Nos. JZB1801) and National Natural Science Foundation of China (Nos. 81603007).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
