Abstract
Introduction
Lycium barbarum, belonging to the Solanaceae family, is an important economic, ecological and medicinal plants native chiefly to arid and semi-arid areas. As a famous traditional medicine and food in China and other Asian countries, the fruit of L. barbarum is rich in a variety of active ingredients, including polysaccharides, flavonoids, amino acids, betaine, carotenoids, phenolic acids, fatty acids, 1 and are considered to have beneficial effects in strengthening the muscles and bones, nourishing the essence, clearing the eye and calming the nerves, etc. 2 At present, L. barbarum fruit has been widely used in medicine, food, health care, cosmetology, and other fields. From 2008 to 2017 years, the total export volume and amount of L. barbarum fruit were 82 182.08 t and $697 million in China, respectively. 3 Due to the rapidly growing demand for L. barbarum fruit, it has been developed into many cultivated varieties and widely cultivated in different provinces including Ningxia, Qinghai, Inner Mongolia, Hebei, and Xinjiang in China. However, the physicochemical properties and biological activities of the nutritional components are affected by varieties, origins, and harvest time. These factors result in the quality of L. barbarum fruit and its commodities on the market is intermingled. Hence, it is essential to develop strict and effective quality control methods for L. barbarum fruit to accelerate the healthy development of L. barbarum industry.
So far, quality control of L. barbarum fruit is mainly focused on small molecular compounds including carotenes, zeaxanthin dipalmitate, betaine, flavone, and trace elements.4,5 As one of the main active components of L. barbarum fruit, polysaccharides have anti-ageing, immunoregulatory, hypoglycemic, and hypolipidemic properties, as well as providing protection against tumors, radiation, and neurodegenerative diseases, and is an important functional additive in health food. 6 The bioactivity of polysaccharides is closely related to their monosaccharide composition, glycosidic linkage types, molecular weight, and conformation.7,8 In consideration of the important biological activity of polysaccharides in L. barbarum fruit, quality control from this insight should be given attention. Currently, the methods of quality control of polysaccharides in L. barbarum fruit mainly include the total carbohydrate content determination, 9 average molecular weight determination, 10 monosaccharide composition analysis, 7 and saccharide mapping analysis. 11 However, the quality control of polysaccharides is a challenge due to their complex composition and structure.
Notably, the preparation of polysaccharides is the key for its quality control analysis. At present, polysaccharides of the L. barbarum fruit were mainly obtained by water extraction, alcohol precipitation and vacuum drying. These polysaccharide samples contained a large number of small molecular component (glucose, 72.8%), arabinogalactan, pectin, middle molecular weight component LBGP-I-1 (glucose, 51.22%) and LBGP-I-2 (glucose, 32.26%; galactose, 35.48%), which could affect the results of the total carbohydrate content determination, average molecular weight determination, monosaccharide composition analysis, and saccharide mapping analysis, thereby resulting in the quality control analysis for L. barbarum fruit polysaccharides being difficult. 12 A large number of researches showed that it is difficult to distinguish the differences between different batches of L. barbarum fruit according to the monosaccharide composition and molecular weight of L. barbarum crude polysaccharides.6,10,11,13 In addition, the monosaccharide composition of 12 batches Cassiae Semen crude polysaccharides was similar, 14 while the HPLC fingerprints of partially-hydrolyzed Cassiae Semen crude polysaccharides with small molecular weight removed was different and could identify different sources, combining with multiple chemometrics methods. 15 Hence, the preparation of polysaccharides is an important guarantee for effective quality control analysis.
In our previous study reported the biological activity of all L. barbarum polysaccharides (LBP) fractions and found that arabinogalactan was the main active ingredient with the strongest immunoregulatory, inhibition of the growth of cancer cells, anti-ageing activities, and ameliorative chronic colitis.12,16–18 Furthermore, the physicochemical properties characterization revealed that molecular weight of arabinogalactan is 91 kDa with a relative molar ratio 1.08:1 of arabinose to galactan. 12
In this study, our study aimed to set up a fingerprint of L. barbarum fruit polysaccharides depending on the main active ingredient to evaluate their quality from different producing areas, varieties, and harvest time. The main active ingredient of 27 batches L. barbarum fruit polysaccharides (arabinogalactan) were obtained by hot water extraction, fractional precipitation, and dialysis (MWCO 8-14 kDa). Then, gas chromatography (GC) and high-performance gel-permeation chromatography (HPGPC) fingerprints were established. The similarity between multiple fingerprinting profiles was evaluated by chemometrics, using the summer fruit of variety NQ1 from Zhongning (Ningxia) as a reference, which is considered the preeminent source in China. This study is expected to be a reference for quality control of L. barbarum fruit polysaccharides.
Results and Discussion
Polysaccharide Yields of L. barbarum Fruit Arabinogalactan
Twenty-seven batches of L. barbarum fruits from Ningxia, Gansu, Xinjiang, Qinghai, Inner Mongolia, and Hebei were collected, including NQ1, NQ5, NQ7, NQ9, Xiao Jianjiao, Xinjiang Nitraria, MQ1, Hebei wolfberry, and Korean wolfberry. Twenty-seven batches of L. barbarum fruit arabinogalactans were prepared by hot water extraction, fractional precipitation, and dialysis. Polysaccharide yields were sample yields multiplied by the sugar content and were summarized in Table 1.
The Total Carbohydrate Contents and Polysaccharide Yields of the 27 Batches of L. barbarum Fruit Arabinogalactans.

Data are presented as the mean ± SD, n = 3, *P < 0.05, **P < 0.01.
L. barbarum from Ningxia is the only Lycium species listed in the Chinese Pharmacopoeia. 19 S1 (NQ1) is the earliest L. barbarum variety cultivated and recorded in Chinese Pharmacopoeia, while other L. barbarum varieties from Ningxia are its hybrid varieties. Hence, S1 was selected as a reference in the study. As shown in Table 1, the polysaccharide yields of S2, S5, S6, S10, S18, S20, and S25 were significantly (P < 0.05) lower than that of S1. Contrary, the polysaccharide yields of S3, S4, S7, S8, S9, S11, S13, S14, S15, S16, S17, S21, S22, S26, and S27 were significantly (P < 0.05) higher than that of S1. The polysaccharide yields of S12, S19, S23, and S24 were no significantly different from that of S1; thus, in terms of polysaccharide yields, S3, S4, S7, S8, S9, S11, S12, S13, S14, S15, S16, S17, S19, S21, S22, S23, S24, S26, and S27 could be used as substitutes for S1. Of those, S16, S19, S21, and S23 were all autumn fruits, indicating that the autumn fruits could be used as functional food and traditional medicine ingredients as the summer fruits. The bioactivity of polysaccharides is closely related to the monosaccharide composition and molecular weights. Therefore, the results of polysaccharide yield similarity analysis failed to fully reflect differences among samples from different locations, varieties, and harvest seasons. The similarities of monosaccharide composition and molecular weights of L. barbarum arabinogalactan were further analyzed in subsequent experiments.
Fingerprint Analysis of Monosaccharide Composition
The bioactivity of L. barbarum fruit arabinogalactan is related to its monosaccharide composition. 17 Here, fingerprints of 27 batches of L. barbarum fruit arabinogalactans were established, and their similarities were evaluated. As shown in Figure 1A, nine characteristic peaks were obtained for reference GC fingerprint of S1, representing the monosaccharides Rha, Fuc, Ara, Xyl, Man, Glc, Gal, GlcA, and GalA. The GC fingerprinting profiles of 27 samples were established (Figure 1B), and the similarities between 26 samples and S1 were evaluated using the Similarity Evaluation System for Chromatographic Fingerprint of Traditional Chinese Medicine version 2004A (Chinese Pharmacopoeia Committee). As summarized in Table S1 (in Supporting Information), the similarity value for all samples was ≥0.9, indicating that all samples were similar to S1. Therefore, HCA and PCA analysis further evaluated the monosaccharide composition similarity of 27 batches of L. barbarum fruit arabinogalactans.

GC fingerprints of L. barbarum fruit arabinogalactan. (A) GC fingerprint of S1. peak1: Rha, peak2: Fuc, peak3: Ara, peak4: Xyl, peak5: Man, peak6: Glc, peak7: Gal, peak8: GlcA, and peak9: GalA; (B) GC fingerprints of 27 batches of L. barbarum fruit arabinogalactan.
HCA Analysis of GC Fingerprints
HCA analysis of the peak areas of the monosaccharide composition spectra was performed using the intergroup linkage method and the squared Euclidean distance as measure of the sample, and a dendrogram was drawn (Figure 2A). All samples were classified into A, B, C, D, E, and F according to the relative distance of 2.4. Class A was composed of S3, S7, S13, S15, S17, and S10, all dissimilar to S1. S15 and S17 were from the Nomuhong farm (Qinghai Province), and S10 was from Jingyuan (Gansu Province). Previous research found that Zhongning and Yinchuan in Ningxia, the majority area of Xinjiang, and parts of Inner Mongolia were the optimal planting areas of L. barbarum. 20 The result showed that the monosaccharide composition was related to the tree's geographic origin.
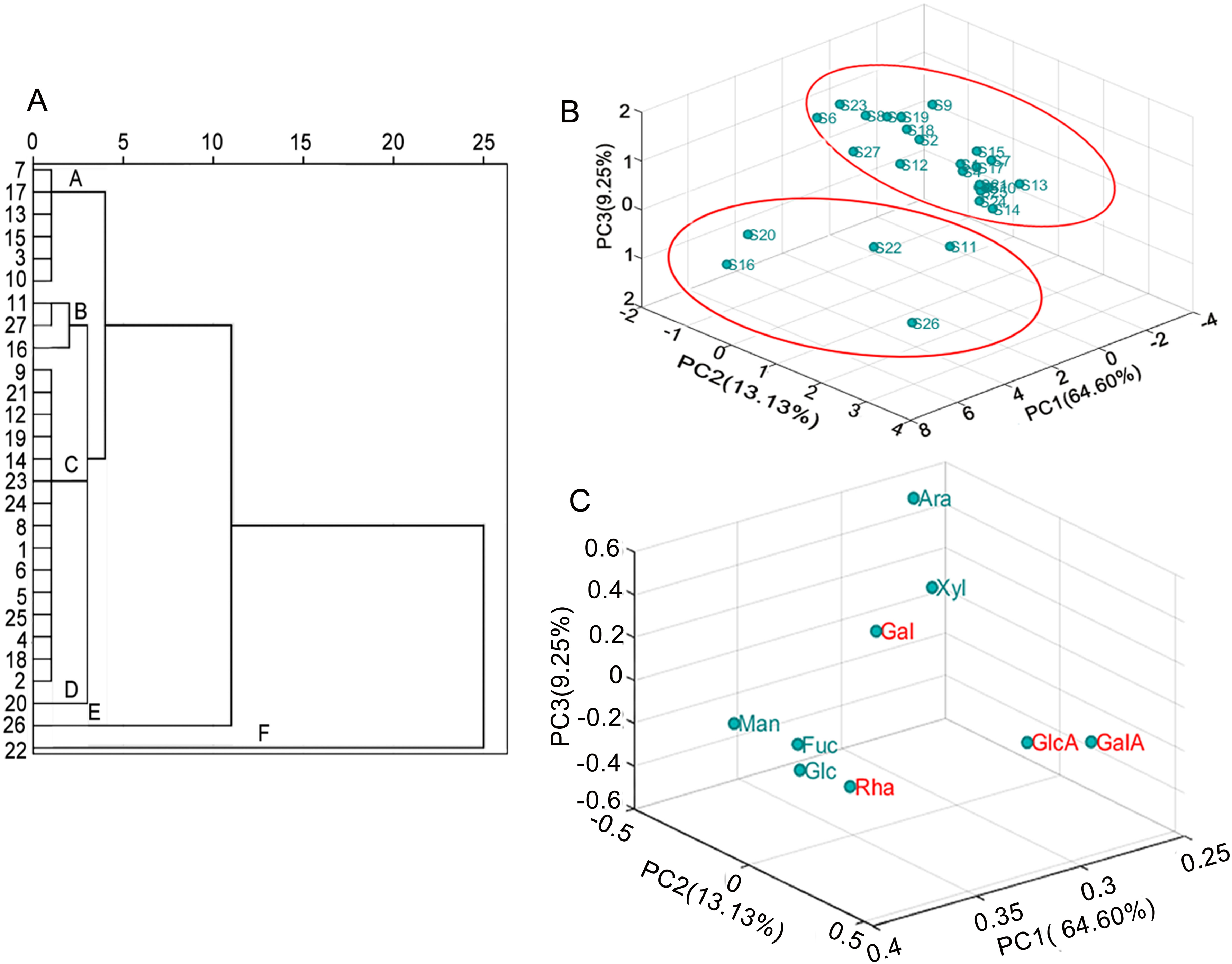
HCA analysis and PCA analysis of GC fingerprints. (A) HCA analysis; (B) The score plot of PCA analysis; (C) The loading plot of PCA analysis.
Class B consisted of samples S11, S27, and S16 from Guazhou (Gansu Province), Qinhuangdao (Hebei Province), and Nomuhong farm (Qinghai Province). Class B samples were dissimilar to S1, indicating that the monosaccharide composition was related to the tree's geographic origin. Class C contained the highest number of samples, all of which were similar to S1, and mainly included the summer fruits of variety NQ1, its cultivars (NQ7 and NQ9), and the autumn fruits of variety Xinjiang Nitraria. In contrast, the summer fruit of the variety Xinjiang Nitraria (S22) was dissimilar to S1, suggesting the monosaccharide composition was related to varieties. Class E and F only contained S26 (Hebei wolfberry, summer fruits) and S22 (Xinjiang Nitraria, summer fruits), respectively, which were not similar to other samples, further indicating that monosaccharide composition was related to varieties. S26 (Class E), the relative molar ratio of Ara to Gal was 0.99, the lowest of all samples except S22. The S22 showed the highest relative percentages of Rha (6.97%) and GalA (17.81%). The relative molar ratio of Ara to Gal of S22 with 0.32 and the Ara relative percentage (12.27%) was the lowest of all samples, while the Glc relative percentage (30.87%) was much higher than that of other samples. Class D only contained S20. The Gal relative percentage (18.77%) was the lowest of all samples, while Glc (15.58%) was the highest except for S16 and S22. Based on these results, Rha, Ara, Glc, Gal, and GalA may be the marker peaks of LBP.
PCA Analysis of GC Fingerprints
To identify the main discriminating factor between L. barbarum fruit arabinogalactan from different regions, varieties, and harvest seasons, the fingerprints of monosaccharide composition were analyzed by PCA. The scree plot in Figure S1 (in Supporting Information) showed that the three principal components (PC1, PC2, and PC3) covered variances of 64.60%, 13.13%, and 9.25%, respectively, thus accounting for 86.98% of the total variance. The PCs could be expressed by the following equations:
PC1 = 0.378X1 + 0.341X2 + 0.271X3 + 0.311X4 + 0.373X5 + 0.351X6 + 0.351X7 + 0.314X8 + 0.295X9
PC2 = 0.242X1 − 0.317X2 − 0.449X3 − 0.007X4 − 0.312X5 − 0.222X6 + 0.113X7 + 0.431X8 + 0.542X9
PC3 = -0.167X1 − 0.368X2 + 0.515X3 + 0.443X4 − 0.180X5 − 0.411X6 + 0.415X7 − 0.042X8 − 0.033X9
In the above equations, X1 to X9 represent Rha, Fuc, Ara, Xyl, Man, Glc, Gal, GlcA, and GalA, respectively. The equation for PC1 points strongly to X1, X7, X8, and X9 as the main factors. The PC2 equation, although weaker than the PC1 equation, it also indicates X3, X8, and X9 as the main factors. Finally, the weakest equation is PC3, which points to X3, X4, X6, and X7 as the main factors. Based on these results, X1, X7, X8, and X9 may be the main factors distinguishing between samples. Principal component analysis (PCA) performed in SIMCA 14.1 further verified the PCA results. The 3D scores plot of PCA showed that most samples clustered into a single group, whereas S11, S16, S20, S22, and S26 were scattered around (Figure. 2B). Therefore, S11, S16, S20, S22, and S26 were different from S1, consistent with the HCA analysis.
The contribution of the nine common characteristic peaks is shown in Figure 2C. The Rha, Gal, GlcA, and GalA values were higher than the remaining peaks. Thus, considering the score plot analysis, Rha, Gal, GlcA, and GalA monosaccharides could be the marker peaks of L. barbarum polysaccharide.
The combined similarity analysis and chemometric analysis showed that S2, S4, S5, S6, S8, S9, S12, S14, S18, S19, S21, S23, S24, and S25 were similar to S1. Of those, S2 was the autumn fruits from Zhongning (Ningxia), and S19, S21, and S23 were all the autumn fruits from Jinghe (Xingjiang), indicating the autumn fruits could be used as functional food and traditional medicine ingredients as the summer fruits. Although the monosaccharide composition analysis reflected some differences among samples, they failed to fully reflect their differences based on locations, varieties, and harvest seasons. Therefore, we further analyzed the similarity of molecular weight in the next section.
The monosaccharide composition fingerprint similarity analysis, and PCA analysis showed that S2, S4, S5, S6, S8, S9, S12, S14, S18, S19, S21, S23, S24, and S25 were similar to S1, consistent with the HCA analysis. The result showed that the difference in monosaccharide composition could be distinguished based on HCA analysis.
Fingerprint Analysis of Molecular Weights
The bioactivity of polysaccharides is closely related to their molecular weight.12,21 Qi et al found that the molecular weight of Corymbia ellipsoidea polysaccharides was an important factor for the activation of RAW264.7 cells to release cytokines as well as for the nuclear factor-kappa B (NF-κB) and mitogen-activated protein kinase (MAPK) signaling pathways. 22 Xu et al found that the antioxidant and hypoglycemic activities of degraded blackcurrant (Ribes nigrum) polysaccharides were stronger than non-degraded components. 23 Zhang et al reported that high-molecular-weight polysaccharides from mulberry (Morus sp.) leaves had stronger bile acid binding ability than those with low molecular weight. 24 Therefore, molecular weight is an important index of polysaccharide quality control. Here, the molecular weights of 27 batches of L. barbarum fruit arabinogalactans were determined following HPGPC fingerprinting. The HPGPC fingerprinting of S1 is shown in Figure 3A. The molecular weights of the four polysaccharide fractions (peaks 1 to 4) were >270, 100.9, <12, and <12 kDa, respectively, based on the retention times (19.34, 7.95, 34.67, and 37.99 min) and the molecular weight standard calibration curve. We only analyzed the similarities of three polysaccharide fractions (from peak 1 to peak 3) because the fourth fraction was buffer salt. In our previous work, L. barbarum fruit arabinogalactan (MW 91 kDa) was the major active constituent.12,15 Here, considering the errors caused by different batches, the second component (100.9 kDa), corresponding to 89.8% of the peak, was the main active ingredient. The HPGPC fingerprints of the 27 samples were established (Figure 3B), and the similarities between 26 samples and S1 were evaluated using the Similarity Evaluation System for Chromatographic Fingerprint of Traditional Chinese Medicine version 2004A (Chinese Pharmacopoeia Committee). Table S1 (in Supporting Information) showed that similarity values (R) were ≥0.9, indicating good similarity among all samples. S2, S3, S5, S7, S9, S10, S11, S15, S16, S17, S18, S20, S21, S22, S26, and S27 were dissimilar to S1 (R < 0.9). Among those, S9, S10, and S11 were from Guazhou (Gansu Province); S15, S16, and S17 were from Nomuhong farm (Qinghai Province); S26 and S27 were from Qinhuangdao (Hebei Province). These results showed that molecular weight was correlated with the producing area. S22, S26, and S27 were from the Xinjiang Nitraria, Hebei wolfberry, and Korean wolfberry varieties indicating that molecular weight was associated with the varieties. Based on these results, the molecular weight was related to the producing area and varieties.
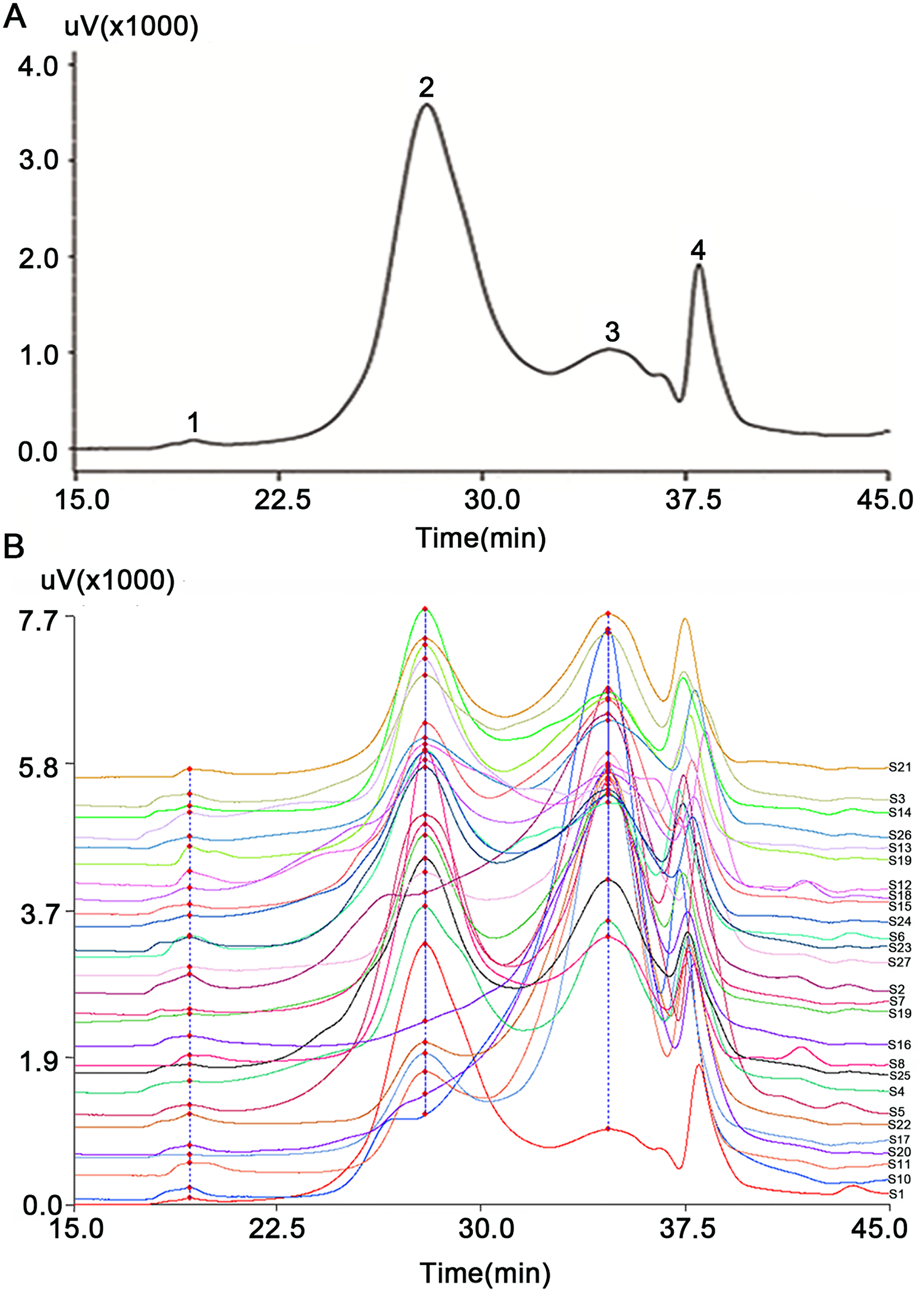
HPGPC fingerprints of L. barbarum fruit arabinogalactan. (A) HPGPC fingerprint of S1; (B) HPGPC fingerprints of 27 batches of L. barbarum fruit arabinogalactan.
HCA Analysis of HPGPC Fingerprints
To further quantify the similarity between samples, the HPGPC fingerprints of 27 samples were analyzed using HCA. As shown in Figure 4, the HCA dendrogram could be classified into two clusters, with cluster I being divided further into subclasses A and B. Subclasses A was composed of S3, S4, S5, S6, S7, S8, S9, S12, S14, S15, S18, S19, S21, S23, S24, S25, and S26, and were similar to S1. Subclasses A mainly included the NQ1 variety and its cultivars (NQ5, NQ7, and NQ9). They were planted in the optimal planting areas for L. barbarum, including Zhongning and Yinchuan in Ningxia, most of Xinjiang, and parts of Inner Mongolia. 24 The result showed that molecular weight was correlated with the producing area and variety. S1 and S13 in Subclasses B were NQ1 and its developed varieties NQ5, indicating molecular weight was associated with varieties. Cluster II was divided further into subclasses C and D. Subclass C included S11, S17, S22, and S27, dissimilar to S1. S11 and S17 were from Guazhou (Gansu Province) and Nomuhong farm (Qinghai Province); S22 and S27 were the summer fruits of Xinjiang Nitraria and Korean wolfberry varieties, indicating molecular weight was related to the producing area. Subclass D comprised S2, S10, S20, and S16 and were dissimilar to S1. S10 and S16 were from Guazhou (Gansu Province) and Nomuhong farm (Qinghai Province), indicating molecular weight was related to the producing area. In conclusion, the molecular weight was associated with the producing area and harvest season.

HCA analysis of HPGPC fingerprints.
HPGPC fingerprints similarity analysis showed that S4, S6, S8, S9, S12, S13, S14, S19, S23, S24, and S25 were similar to S1. When the similarity of HPGPC fingerprints was evaluated using the Chinese Medicine Chromatographic Fingerprint Similarity Evaluation System, the proportion of relative content was higher than that of molecular weight. In these samples similar to S1, the relative percentages of the main active ingredient and the third ingredient were higher than 60% and lower than 40%, respectively. Although the relative percentages of the main active components of S7, S21, and S26 were 58.6%, 51.1%, and 55.2%, with similarity values (R) of 0.88, 0.804, and 0.854, the molecular weights of the main active ingredients were 89.1, 90, and 90.1 kDa, respectively. Therefore, the molecular weights of S7, S21, and S26 were similar to S1 (WM 100.9 kDa). Combined with the results of HCA analysis of HPGPC fingerprints, S4, S6, S7, S8, S9, S12, S13, S14, S19, S21, S23, S24, S25, S26 and S1 were similar.
Carbohydrate content is often applied in the quality control analysis of polysaccharides. Polysaccharide yields are the sample yields multiplied by the carbohydrate content. The quality of S6 and S25 samples was lower than that of S1, while S3, S11, S15, S16, S17, S22, and S27 were comparable to S1. However, the monosaccharide composition and molecular weight analysis showed that S6 and S25 were similar to S1, whereas S3, S11, S15, S16, S17, S22, and S27 were different from S1. Based on these results, the carbohydrate content and polysaccharide yields cannot effectively distinguish the quality difference of polysaccharides.
Combined with monosaccharide composition analysis and molecular weight analysis, the summer fruits S4 (NQ7, Zhongning, Ningxia), S6 (NQ9, Zhongning, Ningxia), S8 (NQ1, Nanliang farm, Yinchuan, Ningxia), S12 (NQ1, Jingyuan, Gansu), S14 (NQ9, Jingyuan, Gansu), S9 (NQ1, Guazhou, Gansu), S24 (NQ1, Xianfeng, Inner Mongolia), and S25 (MQ1, Xianfeng, Inner Mongolia) as well as the autumn fruits of S19 (NQ1), S21 (NQ7), and S23 (Xiao Jian Jiao) from Jinghe in Xinjiang were similar to S1, suggesting that these samples were suitable as functional food and traditional medicine ingredients. Moreover, the optimal planting areas for L. barbarum in China were Zhongning and Yinchuan in Ningxia, Jingyuan in Gansu, Jinghe in Xinjiang, and Xianfeng in Inner Mongolia. Yu et al used remote sensing and GIS technology to determine that Zhongning, Yinchuan, and Wuzhong in Ningxia, Jingyuan in Gansu, the majority area of Xinjiang, and parts of Inner Mongolia were the suitable distribution areas of L. barbarum. 20 Our research result was consistent with that of Yu et al., and found that the Jingyuan in Gansu was also suitable for L. barbarum growth, indicating that the quality of L. barbarum could be differentiated via the main active ingredient of its polysaccharides.
L. barbarum autumn fruits mature from September to October. Compared with the summer fruits, the autumn fruits are round and small, have more seeds, and are bad tasting. So, the L. barbarum autumn fruits are often abandoned by farmers and are generally not sold separately. Our results showed that the autumn fruits (S16, S19, S21, and S23) from Jinghe, Xinjiang were similar to S1, suggesting that the autumn fruits should also be harvested as raw materials for functional food and traditional medicine ingredients.
Conclusion
In our study, the monosaccharide composition and molecular weight fingerprints of 27 L. barbarum fruit arabinogalactans from different locations, varieties, and harvests seasons were evaluated by chemometrics analysis, using the summer fruit of variety NQ1 from Zhongning (Ningxia) as a reference. Altogether, four main findings of our research were as follows. First, the quality of L. barbarum fruit was related to locations and varieties. Second, Zhongning and Yinchuan in Ningxia, Jingyuan in Gansu, Jinghe in Xinjiang, and Xianfeng in Inner Mongolia were the optimal planting areas of L. barbarum in China. Third, the preparation of the main active ingredient was the key for quality control analysis of polysaccharides. Fourth, the quality control analysis of polysaccharides could not only depend on the polysaccharide yield and need to combine the monosaccharide composition and molecular weight analysis. Thus, this study provides a theoretical basis for the quality control of LBP and other functional polysaccharides.
Materials and Methods
Materials and Chemicals
Twenty-seven batches (S1-S27) of L. barbarum were obtained from different regions in China (Table 1). Dextrans and monosaccharide standards for arabinose (Ara), xylose (Xyl), fucose (Fuc), rhamnose (Rha), galactose (Gal), glucose (Glc), mannose (Man), galacturonic acid (GalA), and glucuronic acid (GlcA) were purchased from Sigma-Aldrich. Anhydrous ethanol, sodium hydroxide, and other reagents were of analytical grade.
Preparation of L. barbarum Fruit Arabinogalactan Fraction
L. barbarum fruits were crushed and the contents were extracted twice using hot water (80 °C) at a ratio of 1:5 (w/v) for 2 h. The supernatant was concentrated, precipitated with ethanol (1/2 V) for 12 h, and collected via centrifugation. Next, ethanol was added in the supernatant to two volumes to precipitate for 12 h. The precipitate was collected via centrifugation, dialyzed with a membrane of 8-14 kDa cut-off, and L. barbarum fruit arabinogalactan retentate was collected.
Polysaccharide Yields of L. barbarum Fruit Arabinogalactan
The carbohydrate content of the samples was determined using the phenol sulfuric acid method. 25 Polysaccharide yields of 27 batches L. barbarum fruit arabinogalactans are sample yields multiplied by the carbohydrate content.
Monosaccharide Composition Fingerprints of L. barbarum Fruit Arabinogalactan
Neutral sugars and uronic acids were simultaneously detected using GC, as described previously. 26 L. barbarum fruit arabinogalactans (5 mg) from 27 batches were hydrolyzed in 2 M trifluoroacetic acid at 121 °C for 2 h and then derivatized using alditol–acetate.
Molecular Weight Fingerprints of L. barbarum Fruit Arabinogalactan
The average molecular weights of L. barbarum fruit arabinogalactans from batches S1 to S27 were determined using HPGPC with TSK-gel G 4000 PWXL columns (7.8 × 300 nm). 12 Elution was performed with 0.02 M phosphate buffer (pH 6.0) at a flow rate of 0.40 mL/min and 20 μL of sample per run.
Chemometric Analysis
L. barbarum fruit arabinogalactans from batches S1 to S27 were evaluated using the Similarity Evaluation System for Chromatographic Fingerprint of Traditional Chinese Medicine version 2004A (Chinese Pharmacopoeia Committee), which was applied specifically to analyze the similarity of fingerprints and had been recommended by the State Food and Drug Administration of China.27,28 In addition, the GC and HPGPC fingerprints of arabinogalactans were distinguished by hierarchical cluster analysis (HCA) (SPSS 21.0; IBM), and plotted as a dendrogram to intuitively show the similarity or difference between samples. PCA was conducted using SIMCA 14.1 (Umetrics AB).
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231168467 - Supplemental material for Fingerprint and Chemometric Analysis of Arabinogalactan Derived From Lycium barbarum Fruit
Supplemental material, sj-docx-1-npx-10.1177_1934578X231168467 for Fingerprint and Chemometric Analysis of Arabinogalactan Derived From Lycium barbarum Fruit by Wenqi Huang, Jing Sun, Tingting Liu, Tong Zhao, Xinling Dong, Xiaojing Xu, Yu Lu, Xiaoliang Lin and Guiping Gong in Natural Product Communications
Footnotes
Abbreviations
Acknowledgements
This study was supported by the National Natural Science Foundation of China (No. 31901694), Shaanxi Province Innovation Capability Support Plan-Science and Technology Innovation Team (2020TD-044), and Infinitus (China) Company Ltd (HPG/2016/04/0725).
Authors’ Contribution
Wenqi Huang and Jing Sun contributed to the study conception and design. Tingting Liu and Tong Zhao performed the data collection. Xinling Dong analyzed the data. Xiaojing Xu and Yu Lu performed the statistical analysis. Xiaoliang Lin and Guiping Gong drafted the manuscript. Guiping Gong revised the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
