Abstract
Background
Intestinal injury caused by severe acute pancreatitis (SAP) can induce peripancreatic and systemic infection, and aggravate systemic inflammation. Emodin has demonstrated efficacy in mitigating SAP-associated intestinal injury. Although metabolites in tissues cause histopathophysiological changes, data on the mechanisms of emodin on metabolic processes remain scant.
Methods
The SAP-related intestinal injury rat model was induced by injection of 3.5% sodium taurocholate solution through the biliopancreatic duct. The protective effect of emodin on intestinal injury was evaluated by histologic analyses. On the other hand, we assessed the effect of emodin on metabolic remodeling in intestinal tissues using untargeted metabolomics.
Results
Out of the analyzed 1187 metabolites, untargeted metabolomics identified 99 differential metabolites in the intestinal tissues. Emodin significantly alleviated the inflammatory injury in the pancreas and intestines. Emodin treatment led to significant changes in bile acid metabolism, amino acid metabolism, intestinal microbiota related metabolism, and glycerol phospholipid metabolism in the intestinal tissues. In addition, using the weighted gene co-expression network analysis, we constructed emodin related metabolite–metabolite interaction network and showed that intestinal microbiota related metabolites and glycerol phospholipid metabolism were associated with emodin treatment. Glycine, LPC (0:0/22:6), Spermidine, 11β-hydroxyprogesterone, and N1-methyl-2-Pyridone-5-carboxamide may be efficient molecules after emodin treatment.
Conclusion
Taken together, our data demonstrated that intestinal injury caused by SAP induces an obvious metabolic disorder. Emodin exerts its therapeutic effects through metabolic remodeling.
Introduction
Acute pancreatitis (AP) is caused by abnormal activation of pancreatin, which leads to local inflammation of the pancreas, accompanied by systemic inflammation, and organ dysfunction in severe acute pancreatitis (SAP).1,2 AP is typically a self-limiting disease, however, around 20% of AP cases are associated with organ dysfunction, difficulty in treating, and high mortality. There are 2 main reasons of death from AP: systemic hyperinflammatory response and peripancreatic infection. 3 Intestinal dysfunction caused by intestinal epithelial cell injury is a typical characteristic of SAP. Previous research has demonstrated that intestinal failure is a crucial organ dysfunction and an independent negative prognostic indicator in AP. It is mainly caused by impaired intestinal function, which results in abnormal intestinal flora metabolism and enterogenic infection caused by intestinal flora migration.4,5 Local inflammation driven by the gut may lead to distant organ failure. Treatment targeting intestinal functions significantly improves the prognosis of AP, especially SAP. 6 To develop a new drug with fewer side effects based on the mechanism of SAP intestinal dysfunction, there is a need to study biological processes of AP, search for new biomarkers, and define its pathogenesis.
Rhubarb is an important Chinese herbal, which has long been used as the main ingredient in many Chinese medicines such as Dachengqi decoction used to treat acute abdomen, including AP. 7 Emodin is the main component in rhubarb, and has anti-inflammatory and immune regulation effects as well as improvement of intestinal functions.8,9 A previous study demonstrated that emodin improves the inflammatory response of SAP by regulating the activation of inflammasome. 10 Several studies showed that emodin can improve intestinal injury by enhancing apoptosis and immune regulation of intestinal mucosal cells in SAP.11-13 Meanwhile, the mechanism by which emodin improves intestinal function in SAP is still worthy of further investigation and discussion.
Metabolomics involves the analysis of metabolites in tissues or bodily fluids, which offers insights into the biochemical processes driven by environmental or genetic factors within an organism. Numerous studies have demonstrated that metabolic profiling serves as a novel approach in disease diagnosis and the investigation of pathophysiological mechanisms.14,15 Metabolic disorders are closely associated with the occurrence and development of AP. Patients with AP experience significant metabolic remodeling, which include amino acid metabolism, lipid metabolism, and intestinal microflora metabolism. 16 Indolepropionic acid, an important metabolite of intestinal flora, regulates intestinal mucosal immune response by acting on aryl hydrocarbon receptor signaling. 17 In a SAP mouse model, GSK180, Kynurenine-3-Monooxygenase, a key enzyme of Tryptophan was shown to significantly prevent damage of extrapancreas. 15 However, whether emodin plays a role in regulating the metabolism of intestinal mucosal cells remains unclear.
In order to explore the molecular mechanism by which emodin improves intestinal dysfunction in SAP, this study established a SAP-induced rat model of intestinal injury and utilized untargeted metabolomics to analyze the changes in metabolites in the intestine. This aimed to clarify the pathological process of intestinal impairment in SAP. In addition, using metabolomics, this study explored the possible reversal effect and therapeutic mechanisms of emodin in SAP.
Results and Discussion
Study Design
The flow chart demonstrating the study design is as shown in Figure 1. Tissue samples from the pancreas and ileum were collected from a total of 22 rats, which included 7 sham operation group (SO), 8 SAP group, and 7 emodin intervention group (EMO). To confirm establishment of the model, the pancreas tissue was stained with hematoxylin and eosin. The ileum tissue was used for untargeted metabolomics. After univariate and multivariate analyses of the identified metabolites, biological annotations and weighted gene co-expression network analysis (WGCNA) were performed. Finally, the potential metabolites and pathways affected by emodin in the ileum tissue were explored.

Global design and schematic representation of the study.
Histopathological Analysis of the Pancreas and Ileum
We used hematoxylin and eosin staining to perform histopathological analysis of the pancreas and ileum tissues, to determine successful establishment of the SAP rat model. Compared with the SO group (Figure 2A), a large number of acinar cells were stained pink or edematous in the SAP group (Figure 2B), which suggested that there was hemorrhage, inflammation, and necrosis in the pancreas. In addition, compared with the SAP group, there was recovery of cellular edema and intercellular gaps in the EMO group (Figure 2C). These results demonstrated successful establishment of the SAP rat models. Besides, emodin has a potential to prevent the pancreas from inflammation.
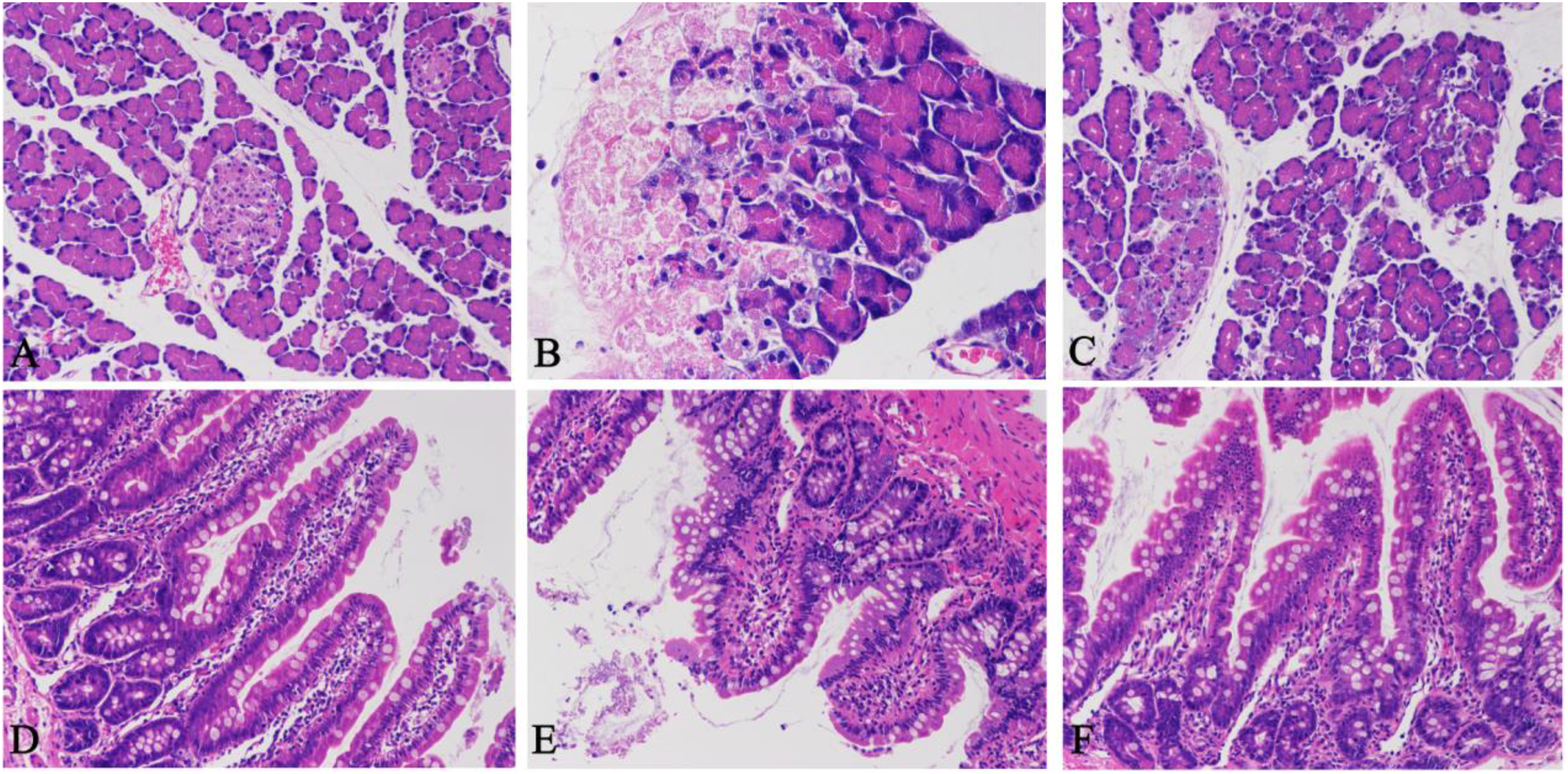
Histological analysis of the effect of emodin on pancreatitis. HE staining of the pancreas (original magnification ×200) in SO (A), SAP (B), and EMO (C) groups. HE staining of the ileum (original magnification ×200) in SO (D), SAP (E), and EMO (F) groups.
Compared with normal ileum tissues in the SO group (Figure 2D), edema of intestinal epithelial cells, shedding of epithelia villi, damage of goblet cells, and neutrophil infiltration were observed in the SAP group (Figure 2E). In addition, the EMO group had better histopathological performance (Figure 2F) compared to the SAP group. These results suggested that there was obvious damage of the ileum tissues in systemic inflammation caused by SAP, and emodin has a protective effect on intestinal barrier.
Polar Metabolites and Lipid Profiles
To obtain global view of the ileal metabolism in rats, we profiled polar metabolites and lipids using untargeted metabolomics platform (original data were listed in Supplemental Table). We evaluated the total ion chromatography (TIC) of different detection methods as shown in Supplemental Figure 1. The TIC data showed that the chromatographic characteristics of different platforms were unique, which can complement each other and expand the identification coverage. After excluding repetitive and inaccurate compounds in the metabolomic analysis, a total of 359 polar metabolites and 828 lipids were identified. As shown in Figure 3A, most lipids, especially strong hydrophobic lipids such as triglycerides (TG) which usually have a longer retention time, had high molecular weight. To evaluate the quality of data and the repeatability of measurement, the coefficient of variation (CV or RSD) between the quality control (QC) samples was divided into different stages and expressed as a Pareto diagram (Supplemental Figure 2). 18
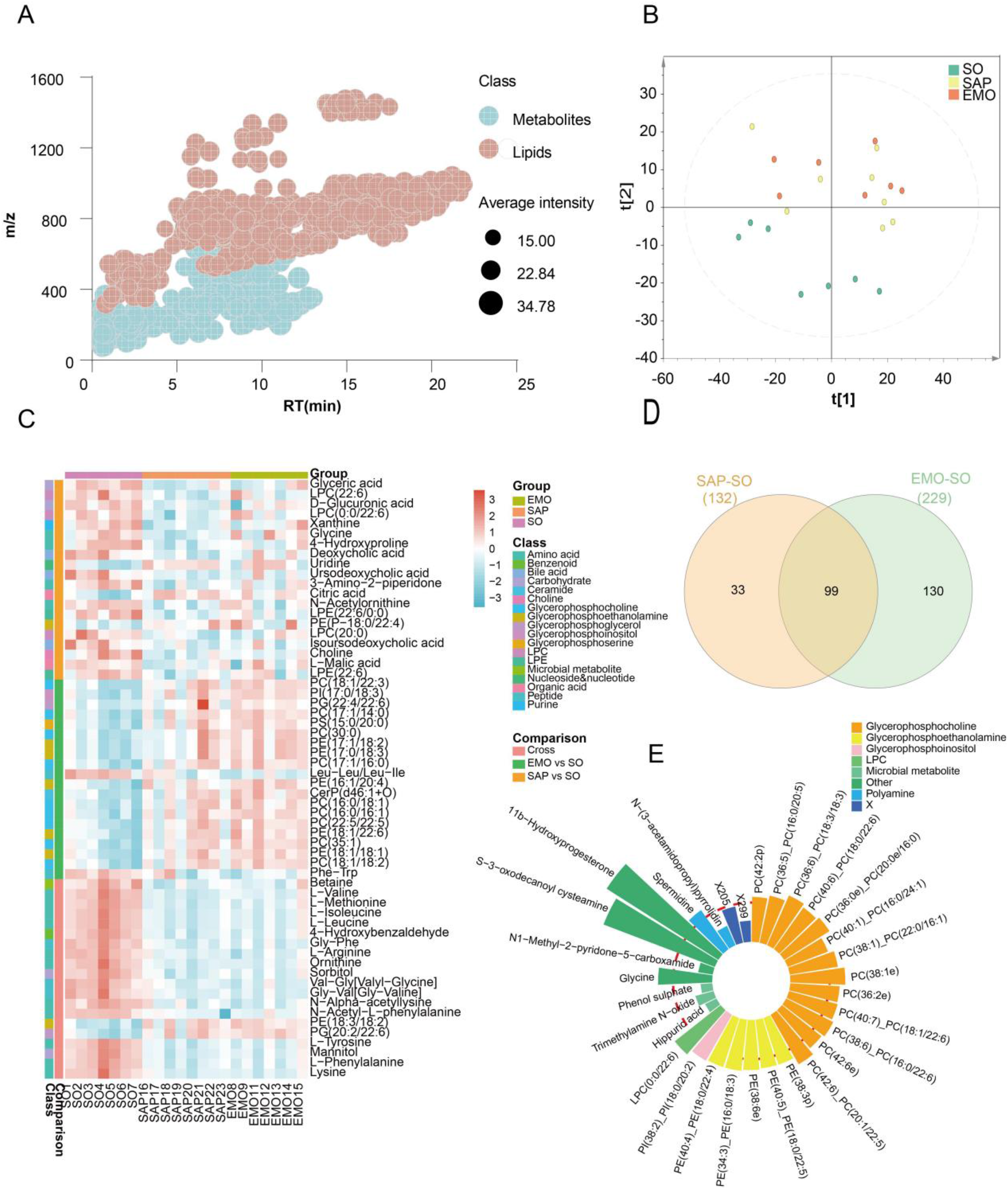
Changes of metabolites in different groups. (A) Cloud plot demonstrating molecular characteristics of the metabolites. (B) PCA score plots of polar metabolites and lipids. (C) Heat map of the top 60 deferential metabolites. (D) Venn diagram of deferential metabolites in the different groups. (E) Relative concentrations of top metabolites in the SO and SAP groups.
Analysis of Differential Metabolites
We use the principal component analysis (PCA) method to characterize the differential metabolites. The points in the plots represent the tested samples, and the distance between them represented the similarity or difference in the samples. The samples were divided into SO group, SAP group, or EMO group, and then the PCA score plot was drawn (Figure 3B). The data showed that there was an obvious boundary between the SO and the SAP groups, but there was more overlap between the SAP and the EMO groups. Thus, there are many similarities in ileum metabolites between 2 groups. Although there was no obvious difference between the metabolites in the SAP and EMO groups, there was a significant difference in few metabolites between the 2 groups, which needed further evaluation.
We then analyzed the metabolites that changed significantly after emodin treatment using a heat-map (Figure 3C). The different types of differential metabolites included organic amino acids such as glycine, and organic acids such as hippuric acid. The lipids which were significantly increased emodin treatment include phosphatidylethanolamine (PE) and phosphatidylcholine (PC). There were 132 metabolites with differences in the SAP and SO group, and 229 metabolites with differences in the EMO and SO group. Among them, 99 metabolites are at the intersection region of these 2 groups (Figure 3D). Out of the 99 metabolites with common differences, we selected the ones with highest statistical significance. The wind rose plot was drawn to visualize the difference between the EMO and SAP group before and after emodin treatment. Among them, the concentration of polar metabolites was lower in the EMO group. Meanwhile, most of lipids were higher in the EMO group. The data showed that except for some microbial related metabolites, there was increase in most lipids such as PC and PE in the EMO group (Figure 3E).
Weighted Gene Co-expression Network Analysis
Previous analyses showed some metabolites with significant differences in the 2 groups. However, data on which metabolites changed and recovered after emodin treatment in the SAP group was not clear. We employed the WGCNA to explore metabolites with similar trends. Our data showed that there were no significant outlier samples after clustering (Figure 4A). Thereafter, we used 18 as a soft threshold (Figure 4B) to get the clustering tree of the WGCNA model (Figure 4C, D). A total of 9 modules were clustered. Using the Pearson correlation analysis of modules and the trend between the groups (Figure 4E), the absolute value of the correlation coefficient greater than 0.4 was set as the candidate module. A total of 3 candidate modules, blue, yellow, and brown, were selected. Blue and yellow modules were significantly negatively correlated with the development of SAP, while the brown module had positive correlation (Figure 4F). The high weight compounds in the brown module were amino acids such as arginine, methionine, threonine, and serine (Figure 5A). However, the high weight compounds in the blue and yellow modules were mainly PC and PE (Figure 5B). These WGCNA outcomes were in sync with the significant differential metabolites, which further validated our results.

WGCNA showing key modules associated with emodin treatment. (A) Sample clustering to detect outliers. (B) Scale-free topology model (left) and mean connectivity (right) for setting soft-threshold powers. (C) Cluster dendrogram and module assignment of the metabolites. (D) Clustering of all modules. (E) Module-module relationships heat map. (F) Module-group relationships heat map.

Metabolite-metabolite interaction (MMI) network of brown (A) and yellow + blue (B).
Biological Function Analysis
To explore the metabolic effects caused by the differential metabolites, we analyzed the metabolic pathway enrichment analysis based on the KEGG database (Figure 6A). We analyzed both the impact factor and significance of the pathway, and showed that emodin had a significant effect on glutathione, glycerophospholipid, and linoleic acid metabolism.

Metabolic remodeling characteristics associated with emodin treatment. (A) KEGG enrichment analysis of the deferential metabolites. (B) Biopan analysis showing dysregulation of lipid metabolism in SAP group. Glycine (C), LPC (0:0/22:6) (D), Spermidine (E), 11b-Hydroxyprogesterone (F), and N1-Methyl-2-pyridone-5-carboxamide (G) were representative metabolites associated with emodin treatment.
Notably, unlike polar metabolites, the functional annotation of lipid pathway in KEGG was incomplete. Hence, we used BioPAN, a newly released Bioinformatics tool, to explore the metabolism of free fatty acids and the transformation between lipid sub-classes (Figure 6B). 19 The results showed that there was significant upregulation of phosphatidylserine (PS) to PE transformation after emodin treatment, but the transformation from PE to PS was inhibited. However, the transformation from PE to PC was not significant. This interplay led to the accumulation of PE. Similar to PE, the transformation of PC to Cardiolipin (CL) and PS pathway was inhibited, and the PC's source had no significant change, which led to increase in the relative concentration of PC.
The relative concentration of glycine was significantly lower in the SAP group than in SO group, which increased after emodin treatment. The trend of LPC (0:0/22:6) and 11B-hydroxyprogesterone was similar to that of glycine. However, N1-methyl-2-Pyridone-5-carboxamide decreased in the SAP group and returned to a state similar to the SO group after emodin treatment. In addition, spermidine also showed an upward trend in the EMO group (Figure 6C-G). Based on the combination of polar metabolites and lipid differential compounds, the relative concentration of most metabolites increased after emodin treatment, while some of the suppressed metabolites were related to intestinal microorganisms.
Discussion
Rhubarb is a key component in traditional Chinese medicine for the treatment of inflammatory diseases of the digestive system. Emodin is the most prominent active ingredient in rhubarb. This study investigated and demonstrated the therapeutic effect of emodin on intestinal injury in a rat model of AP. Using untargeted metabolomics, we showed that there was significant change in bile acid metabolism, amino acid metabolism, intestinal flora-related metabolism, and glycerol phospholipid metabolism in intestinal tissues after emodin treatment. The WGCNA showed the metabolic network related to intestinal inflammation, and demonstrated potential therapeutic targets.
All the patients with AP suffered from intestinal dysfunction in different degrees. 20 The change of microbial microenvironment in the intestinal tract leads to significant changes in metabolic profiles in the intestinal tract and circulation pool. 21 Metabolic disorders of intestinal flora in patients with AP may be involved in systemic inflammation. Besides, metabolites also play an important role in regulating intestinal mucosal barrier and immune barrier. 22 The damage of the intestinal barrier in SAP leads to the displacement of gut flora and contributes to the peripancreatic and abdominal cavity infections, further intensifying systemic inflammation. In this study, there was damage of the intestinal barrier of SAP rat, and occurrence of obvious metabolic remodeling in intestinal tissues, which include bile acid metabolism, amino acid metabolism, as well as dipeptide and glycerol phospholipid metabolism. Bile acid is an important signal regulating molecule, which participates in the intestinal-hepatic circulation of the body. It plays an important role in regulating systemic inflammatory response by modulating activation of inflammatory bodies in inflammatory diseases such as sepsis. 23 In addition, bile acid regulates the homeostasis of glucose metabolism by acting on bile acid receptors in intestinal tract. 24 Bile acids, aromatic amino acids, and dipeptides are mainly metabolized by intestinal flora, and their significant changes indicate that intestinal flora metabolism is closely associated with the occurrence and development of AP.22,25 Glycerophospholipid metabolism is mainly characterized by PC and PE metabolism disorder, and the PC and PE fatty acid chains with these differences are mainly long-chain unsaturated fatty acids which, as precursors of inflammatory mediators, participate in the occurrence of intestinal inflammation.
To assess the mechanism of emodin in treating AP-related intestinal injury, we employed WGCNA algorithm, a method for analyzing the relationship between high-dimensional data and pathological features, to establish a network of metabolite-metabolite interactions. The WGCNA results re-emphasized that the shifts in intestinal related metabolites and glycerophosphate metabolism were related to emodin treatment. We also showed that the content of 4 small metabolic molecules: Glycine, LPC (0:0/22:6), Spermidine, and 11b-Hydroxyprogesterone, decreased in the SAP group. However, intervention with emodin led to increase in their concentrations, which was comparable to that of the sham operation group. In contrast, N1-methyl-2-pyridone-5-carboxamide exerted opposite effects. Spermidine can bind and stabilize RAN and DNA, regulate the function of cellular enzymes, and play an antioxidant role. 26 In addition, spermidine plays an anti-inflammatory role by inhibiting TNF-α and NF-κB in osteoarthritis. Cortisol has anti-inflammatory, antioxidant, and cell membrane stabilizing effects, while 11b-Hydroxyprogesterone is an intermediate of glucocorticoid synthesis, which reflects the activity of cholesterol to cortisol pathway. 27 Emodin treatment led to increase in the contents of the 4 metabolites, and played an anti-inflammatory role. N1-methyl-2-pyridone-5-carboxamide is a degradation product of serotonin. Serotonin plays an important role in regulating intestinal vasoconstriction. Previous studies have shown that 5-HT can regulate acinar dedifferentiation, amylase secretion, and acinar cell injury after pancreatitis. 28 5-HT is necessary for pancreatic inflammation. 29 Therefore, these metabolic molecules or their catalytic enzymes may be the target molecules for emodin in exerting anti-inflammatory effects.
Conclusions
In summary, we descripted the disorders of intestinal metabolism associated with SAP. Emodin exerts its therapeutic effects through metabolic remodeling. However, the specific molecular mechanism of emodin protects against SAP related intestinal injury by metabolic remodeling remains unclear and needs further analysis.
Materials and Methods
Animal Sample Preparation
A total of 24 Male Sprague-Dawley rats weighing 230 to 260 g were obtained from the Experimental Animal Center at Dalian Medical University. The rats were kept at a controlled temperature between 22 °C and 25 °C with a 12-h light/dark cycle and had free access to diet. The animal experiments were approved by the Animal Care and Use Committee at Dalian Medical University (No. AEE18072). The rats were randomly divided into 3 groups: operation group (SO), acute pancreatitis modeling group (SAP) and emodin treatment group (EMO), containing 8 rats each. Before the operation, the rats underwent fasting for 12 h followed by intraperitoneal injection of sodium pentobarbital (30 mg/kg) for anesthesia. Rats in the SAP and EMO groups were injected with 0.1 mL/100 g of 3.5% sodium taurocholate solution through the biliopancreatic duct to induce AP. The rats in the EMO group received 1 mL/100 g of emodin (6.0 mg/mL) by gavage, at 0 and 8 h after modeling. 30 In the SO group, the abdomen was opened and closed in the same place as the other 2 groups. During the process, one rat in the SO group and one in the EMO group died.
Collection of Tissue Samples and Histological Analysis
The distal ileum and pancreas were separated and some of their tissues were quenched in liquid nitrogen before storage at −80 °C. The remaining tissues were fixed in 4% paraformaldehyde, and then embedded in paraffin. The embedded tissues were sliced, and then stained with hematoxylin-eosin (HE, Servicebio, China). The sections were then analyzed under an optical microscope (Olympus BX53, Japan) at 200× and 400× magnification.
Sample Pretreatment
We weighed 20 mg of the ileum sample and controlled any error within 1 mg. Then 80% methanol-water was added to the grinding tube (Jingxin, China) containing the ileum at a volume of 30 μL/mg. Three steel balls were put into the tube and homogenized at a frequency of 70 Hz for 180 s. About 300 μL of the homogenate was transferred to another 2.0 mL EP tube. About 900 μL methyl tert-butyl ether (Sigma-Aldrich, USA) and 250 μL Milli-Q (Merck KGaA, Germany) pure water were added into the homogenate and then vortexed for 180 s, before incubation for 10 min in a rolling mixer. Thereafter, the mixture was keep at room temperature for 10 min, followed by centrifugation at 13 000g for 10 min. About 300 μL of the upper layer containing lipid extract and 250 μL of the lower layer with polar metabolites were transferred into 2 different EP tubes. Immediately after, the samples were concentrated and dried by vacuum centrifugation. The remaining samples were mixed and centrifuged, and the upper and lower layers were used as QC samples.31,32
Metabolomics Analysis
Chromatographic Separation Conditions of Polar Metabolites
Polar metabolites analysis was performed on an Ultimate 3000 ultra-high performance liquid chromatograph. For more details please refer to the supplemental materials and our previous study. 33
For positive ionization, the metabolites were separated using an Excel 2 C18-PFP column (ACE Co., UK, 3.0 μm, 2.1 × 100 mm). The elution process was conducted using a linear gradient ramp, starting with 2% of the mobile phase B (acetonitrile) and increasing to 98% in 10 min, while the mobile phase A consisted of 0.1% formate/water.
Negative ionization utilized an Acquity HSS C18 column (Waters Co., USA, 1.8 μm, 2.1 × 100 mm). The mobile phases comprised of water (A) and acetonitrile/methanol (B), both containing ammonium bicarbonate buffer salt, and were used to elute the separated metabolites. The gradient ramp was initiated with 2% of the mobile phase B and progressed to 100% over a 10-min period, followed by 5 min of column washing and equilibration.
The flow rate, injection volume, and column temperature were kept constant throughout the analysis, set at 0.4 mL/min, 5 μL, and 50 °C, respectively.
Mass Spectrometry Detection Conditions of Polar Metabolites
The detection of polar metabolites was conducted utilizing a Q-Orbitrap mass spectrometer, operating under standard and consistent ionization parameters. The instrument was configured with a heated electrospray ionization source, utilizing a sheath gas of 45 arbitrary units, an auxiliary gas of 10 arbitrary units, a heater temperature of 355 °C, a capillary temperature of 320 °C, and an S-Lens radio frequency level of 55%.
The metabolome extracts were analyzed in full scan mode with a resolution of 70 000 full width at half maximum (FWHM) and an acquisition of 70 000 FWHM resolution, using an automatic gain control (AGC) setting of 1E6 and a maximum injection time of 200 ms. The scan range was defined as 70 to 1000 m/z. Full MS/MS data acquisition was performed with a resolution of 17 500 FWHM.
Chromatographic Separation Conditions for Lipids
The chromatographic separation of lipids was executed in both positive and negative ionization modes using an Accucore C30 core-shell column (Thermo Scientific, USA, 2.6 μm, 2.1 × 100 mm) to separate lipid molecules at a temperature of 50 °C.
The mobile phase employed in the separation process comprised of 2 components, namely 60% acetonitrile in water (A) and 10% acetonitrile in isopropanol (B), both containing 10 millimoles per liter of ammonium formate and 0.1% formic acid. A gradual and optimized separation gradient was employed, starting with 10% B, ramping up to 50% in 5 min, and further increasing to 100% over a period of 23 min. The remaining 7 min were dedicated to column washing and equilibration. The flow rate employed during the separation process was 0.3 mL/min.
Mass Spectrometry Detection Conditions for Lipids
The detection of ionized lipid molecules was performed utilizing the same parameters as previously described for metabolites. The lipid extracts were profiled within a mass range of 300 to 2000 m/z, using the same instrument settings as previously outlined for metabolites.
The key parameters employed during the analysis included a full scan resolution of 70 000 FWHM, an MS/MS resolution of 17 500 FWHM, a loop count of 10, an AGC target of 3E6, a maximum injection time of 200 ms for full scan and 80 ms for MS/MS, and dynamic exclusion with a duration of 8 s.
Statistical Analysis
The relative quantitated information of polar metabolites and lipids were merged and those detected with multiple methods were excluded to guarantee uniqueness, and underwent Log2 transformation for final statistical analysis. 34 Multivariate analysis was conducted with SIMCA-P software (Umetrics, Sweden). 35 On the other hand, univariate analysis including independent sample t-test and p-value FDR adjust, 36 as well as heat-map and metabolic pathway analysis was conduct using the MetaboAnalyst website (http://www.metaboanalyst.ca).37,38 Violin plots were generated with GraphPad Prism 8.0 (GraphPad Software, USA), while Cytoscape (Cytoscape 3.8.0, USA) was used to construct biological networks and perform identification of functional modules. 39 We used the weighted gene coexpression network analysis (WGCNA) package in R Version 4.0.3 (R Core Team, 2020). 40
Supplemental Material
sj-xlsx-3-npx-10.1177_1934578X231163995 - Supplemental material for Emodin Exerts its Therapeutic Effects Through Metabolic Remodeling in Severe Acute Pancreatitis-Related Intestinal Injury
Supplemental material, sj-xlsx-3-npx-10.1177_1934578X231163995 for Emodin Exerts its Therapeutic Effects Through Metabolic Remodeling in Severe Acute Pancreatitis-Related Intestinal Injury by Minjie Wang, Chen Pan, Dawei Deng, Mingzheng Xie and Yongqing Cao in Natural Product Communications
Supplemental Material
sj-docx-4-npx-10.1177_1934578X231163995 - Supplemental material for Emodin Exerts its Therapeutic Effects Through Metabolic Remodeling in Severe Acute Pancreatitis-Related Intestinal Injury
Supplemental material, sj-docx-4-npx-10.1177_1934578X231163995 for Emodin Exerts its Therapeutic Effects Through Metabolic Remodeling in Severe Acute Pancreatitis-Related Intestinal Injury by Minjie Wang, Chen Pan, Dawei Deng, Mingzheng Xie and Yongqing Cao in Natural Product Communications
Supplemental Material
sj-docx-5-npx-10.1177_1934578X231163995 - Supplemental material for Emodin Exerts its Therapeutic Effects Through Metabolic Remodeling in Severe Acute Pancreatitis-Related Intestinal Injury
Supplemental material, sj-docx-5-npx-10.1177_1934578X231163995 for Emodin Exerts its Therapeutic Effects Through Metabolic Remodeling in Severe Acute Pancreatitis-Related Intestinal Injury by Minjie Wang, Chen Pan, Dawei Deng, Mingzheng Xie and Yongqing Cao in Natural Product Communications
Footnotes
Acknowledgements
The authors also thank Prof. Peiyuan Yin and Zeming Wu for establishing the LC-MS method and directing the analysis.
Author Contributions
MinJie Wang: data curation, formal analysis, writing—original draft, writing—review & editing. Chen Pan: formal analysis, investigation, software, methodology, writing—original draft. Dawei Deng: formal analysis, investigation, methodology. Mingzheng Xie: formal analysis, resources, validation. Yongqing Cao: conceptualization, validation, supervision, writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval of this study was obtained from the Animal Care and Use Committee at Dalian Medical University (No. AEE18072).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Animal Care and Use Committee at Dalian Medical University (No. AEE18072) approved protocols. Besides, this study does not contain any studies with human subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
