Abstract
Objectives
Isolation, structure determination, and antimicrobial activities evaluation of compounds from the methanol extract of the marine microalga Thraustochytrium pachydermum TSL10.
Methods
Using chromatographic methods to isolate compounds from the methanol extract of the T pachydermum. The chemical structures were elucidated by analyses of HR-ESI-MS and NMR spectral data. The antimicrobial activities of compounds were evaluated using the dilution turbidimetric broth method as the standard protocol published by the Clinical and Laboratory Standard Institute.
Results
One new sterol, (24R)-ergosta-7,22-diene-3β,5α,9α-triol (
Conclusions
The results suggested sterols
This is a visual representation of the abstract.
Introduction
Microalgae have been known as one of the largest and most diverse photosynthetic organisms. They are potential sources of bioactive compounds such as fatty acids, carotenoids, chlorophylls, phycobiliproteins, and vitamins. The compounds from microalgae have exhibited antibacterial, antifungal, antiviral, antitumor, and anti-inflammatory effects. 1
Thraustochytrium species (Thraustochytriaceae family) are suitable for fermentation technology and produce fatty acids and other value-added biocomponents.2,3 The marine microalga Thraustochytrium pachydermum was isolated from Truong Sa Island, Truong Sa Archipelago, Vietnam in 2021 and is available in GenBank (accession number OL721615). 4 The chemical constituents and biological activities of T pachydermum have not been studied so far. Herein, we report one new sterol and 17 known metabolites from the marine microalgae T pachydermum TSL10 and their antibacterial activities.
Results and Discussion
Compound
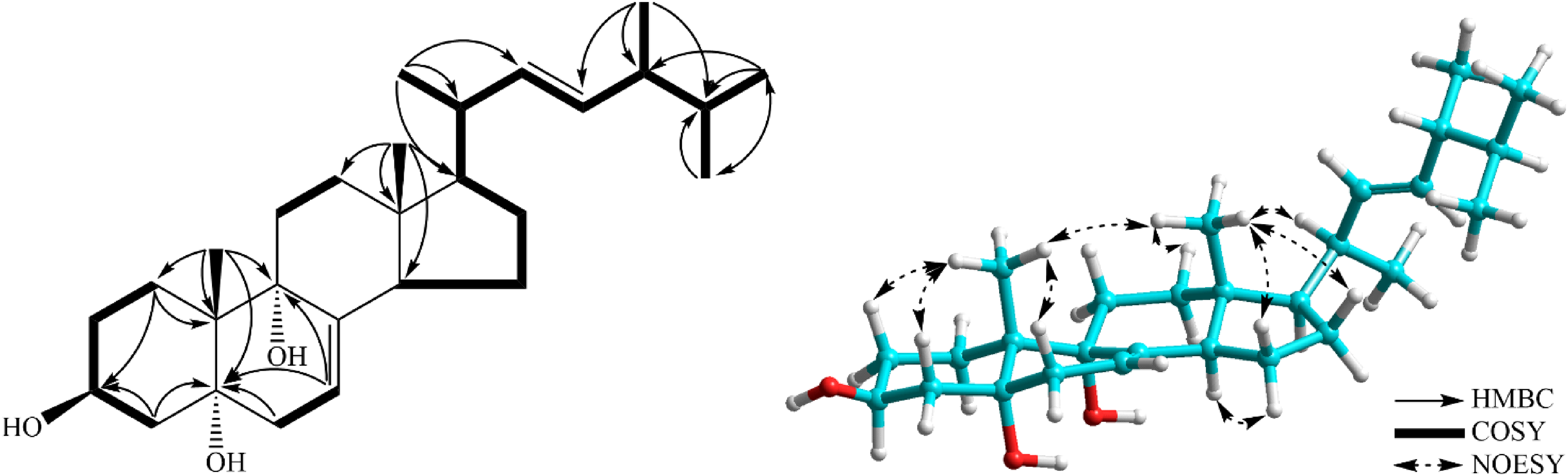
The key HMBC, COSY, and NOESY correlations of compound
1H- and 13C-NMR Spectral Data for

Abbreviation: ND, not determined.
*Overlapped signals.
The known compounds were determined to be ergosta-7,22-diene-3β,5α-diol (

Chemical structures of compounds
All isolated compounds were evaluated for their antimicrobial activities against microorganisms, Gram-( + ) bacteria (Enterococcus faecalis ATCC29212, Staphylococcus aureus ATCC25923, and Bacillus cereus ATCC14579), Gram-(-) bacteria (Escherichia coli ATCC25922, Pseudomonas aeruginosa ATCC27853, and Salmonella enterica ATCC13076), and yeast (Candida albicans ATCC10231) using dilution turbidimetric broth method as the standard protocols published by the Clinical and Laboratory Standard Institute (Table 2). Streptomycin and cycloheximide were used as positive controls. Streptomycin inhibited Gram-( + ) bacteria (E faecalis, S aureus, and B cereus), Gram-(-) bacteria (E coli, P aeruginosa, and S enterica) with MIC values ranging from 32 to 256 µg/mL, respectively. Cycloheximide inhibited the yeast C albicans with MIC value of 32 µg/mL. All isolated sterols
Antimicrobial Activities of Compounds
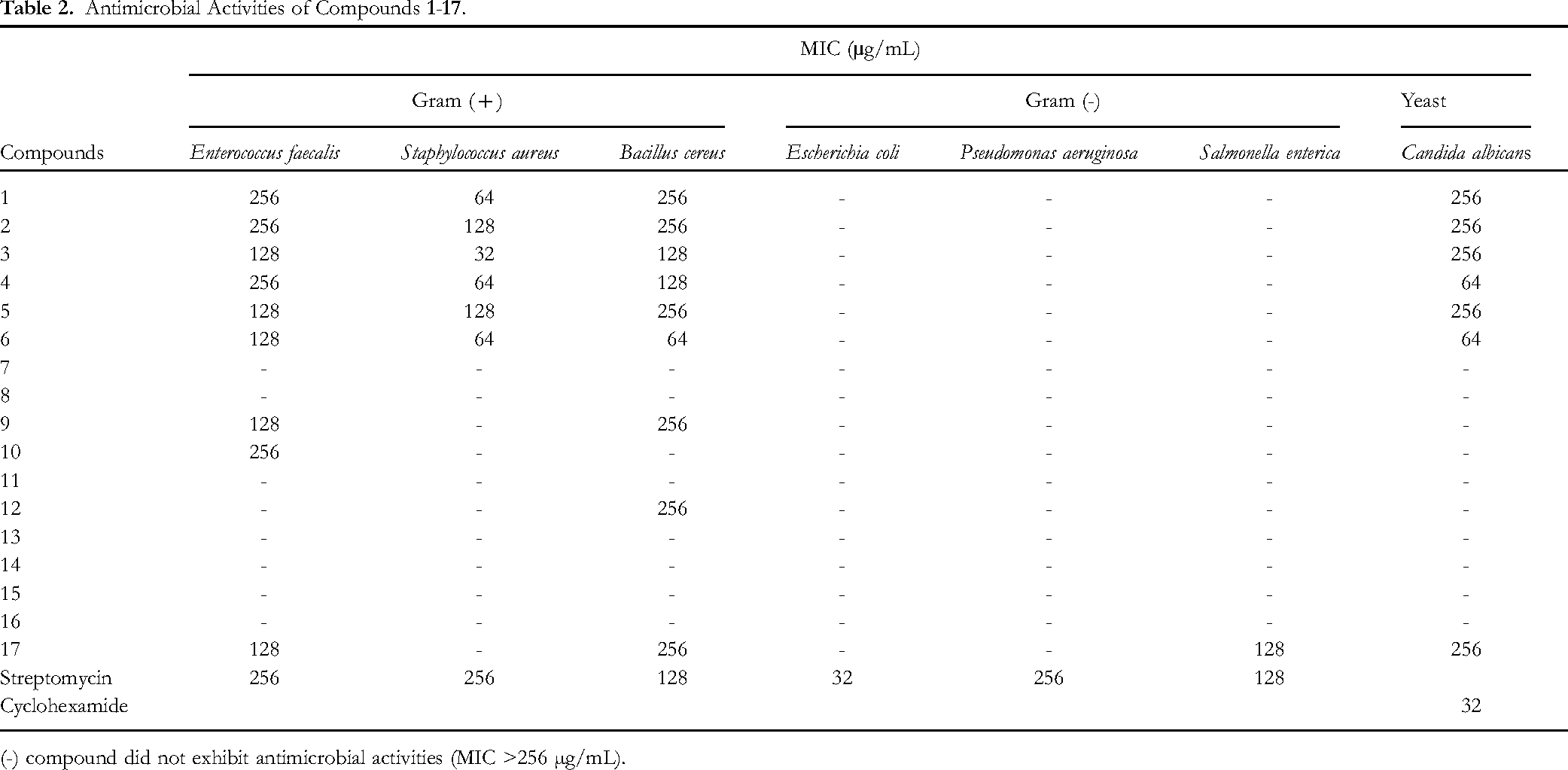
(-) compound did not exhibit antimicrobial activities (MIC >256 µg/mL).
Material and Methods
General See Supplemental Material.
Microalgae Samples
See Supplemental Material.
Biomass Cultivation
An aeration rate when growing biomass was always maintained at 0.5 L air/L/min after the air was filtered through a 0.2 mm filter (Advantec, JP020AN). Biomass cultivation has changed on a large scale of 30 L fermentor: while the dissolved oxygen was maintained above 10% with the manual stirring speed from 300 to a maximum of 400 rpm, at 32 °C÷37 °C. Glucose concentrations of 9%, and 1% industrial yeast extract were used to grow the strain T pachydermum TSL10. After 120 h of cultivation, biomass was filtered and dried.
Extraction and Purification of Compounds
Thraustochytrium pachydermum (2.2 kg) was ultrasonically extracted with methanol (MeOH) at 40 °C 3 times (each 8.8 L, 3 h). The MeOH layer was evaporated in vacuo to give a dark solid residue (420 g). This crude extract was suspended with water (2.0 L) and partitioned with CH2Cl2 to obtain CH2Cl2 soluble fraction (TPD, 102 g) and water layer. The TPD fraction was roughly separated on a silica gel column chromatography (CC), eluting with CH2Cl2/MeOH (100:0, 40:1, 20:1, 10:1, 5:1, vol/vol) to give 5 fractions, TP1A (21.8 g), TP1B (15.7 g), TP1C (8.4 g), TP1D (25.9 g), and TP1E (20.3 g). The TP1B fraction (15.7 g) was chromatographed on a silica gel CC using CH2Cl2/MeOH/water (4/1/0.05, vol/vol/vol) as eluent to give 5 fractions, TP2A (1.3 g), TP2B (6.7 g), TP2C (0.6 g), TP2D (2.8 g), and TP2E (2.5 g).
TP2A (1.3 g) fraction was chromatographed on a silica gel CC and an eluent of CH2Cl2/acetone (7/1, vol/vol). to yield compounds
The TP1C fraction (8.4g) was repeatedly chromatographed on a silica gel CC, eluting with n-hexane/CH2Cl2/acetone (3/1/0.1, vol/vol/vol) to obtain 3 fractions TP3A (2.6 g), TP3B (1.5 g), and TP3C (1.8 g). Compound
Ergosta-7,22-diene-3β,5α,9α-triol (1 )
White amorphous powder;
Antimicrobial Assays
See Supplemental Material.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231157145 - Supplemental material for Chemical Constituents From the Marine Microalgae Thraustochytrium pachydermum
Supplemental material, sj-docx-1-npx-10.1177_1934578X231157145 for Chemical Constituents From the Marine Microalgae Thraustochytrium pachydermum by Nguyen Thi Thu Thuy, Hoang Thi Minh Hien, Nguyen Cam Ha and Le Thi Thom, Dang Diem Hong, Nguyen Van Thinh, Nguyen Trong Dan, Nguyen Dang Hoi, Vu Thi Loan, Hoang Duc Quang, Dan Thi Thuy Hang, Phan Van Kiem, Nguyen Tien Dat, Nguyen Xuan Nhiem in Natural Product Communications
Footnotes
Acknowledgments
This work was supported by the Project KCB-TS-07.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the KCB-TS-07.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
